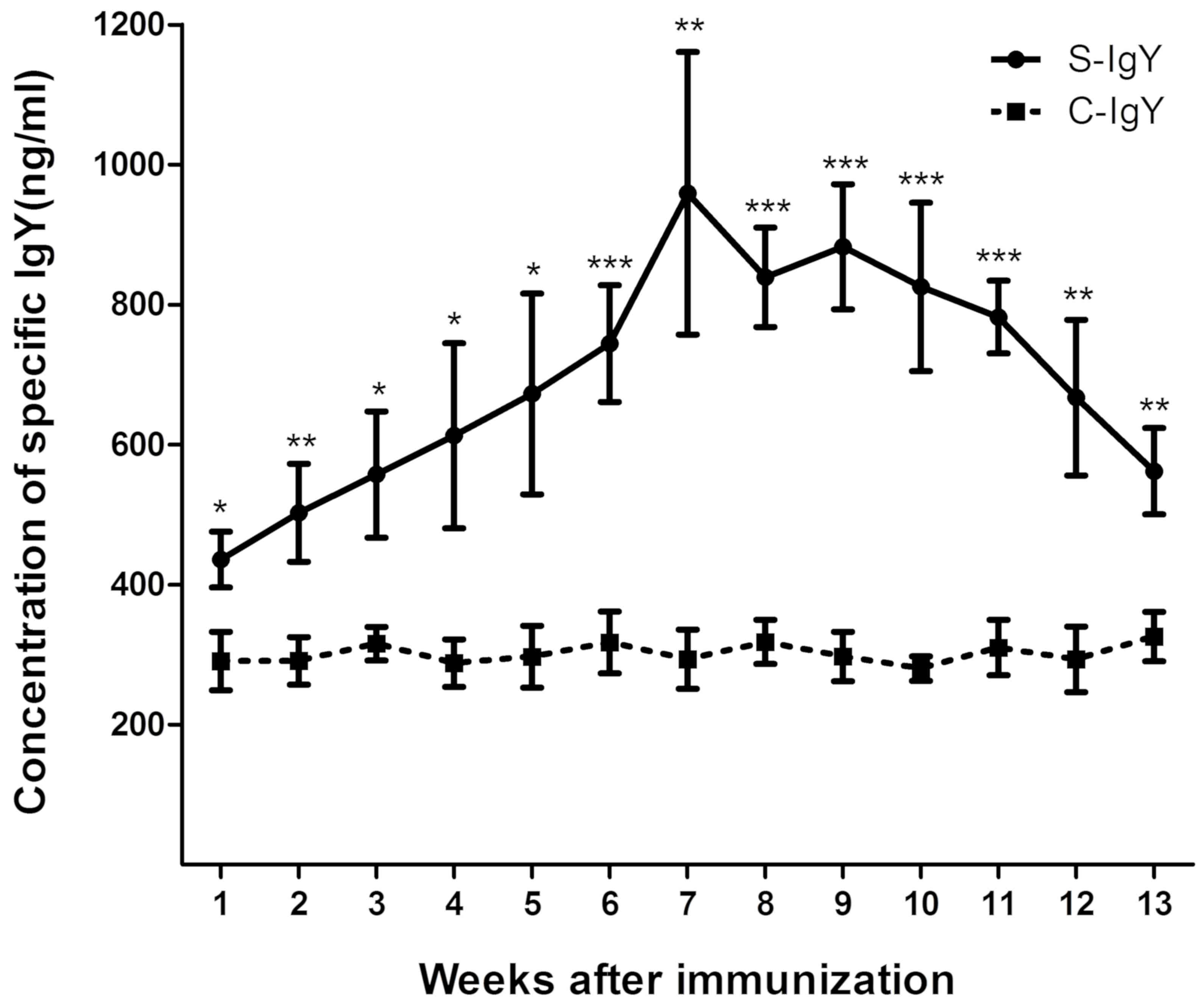
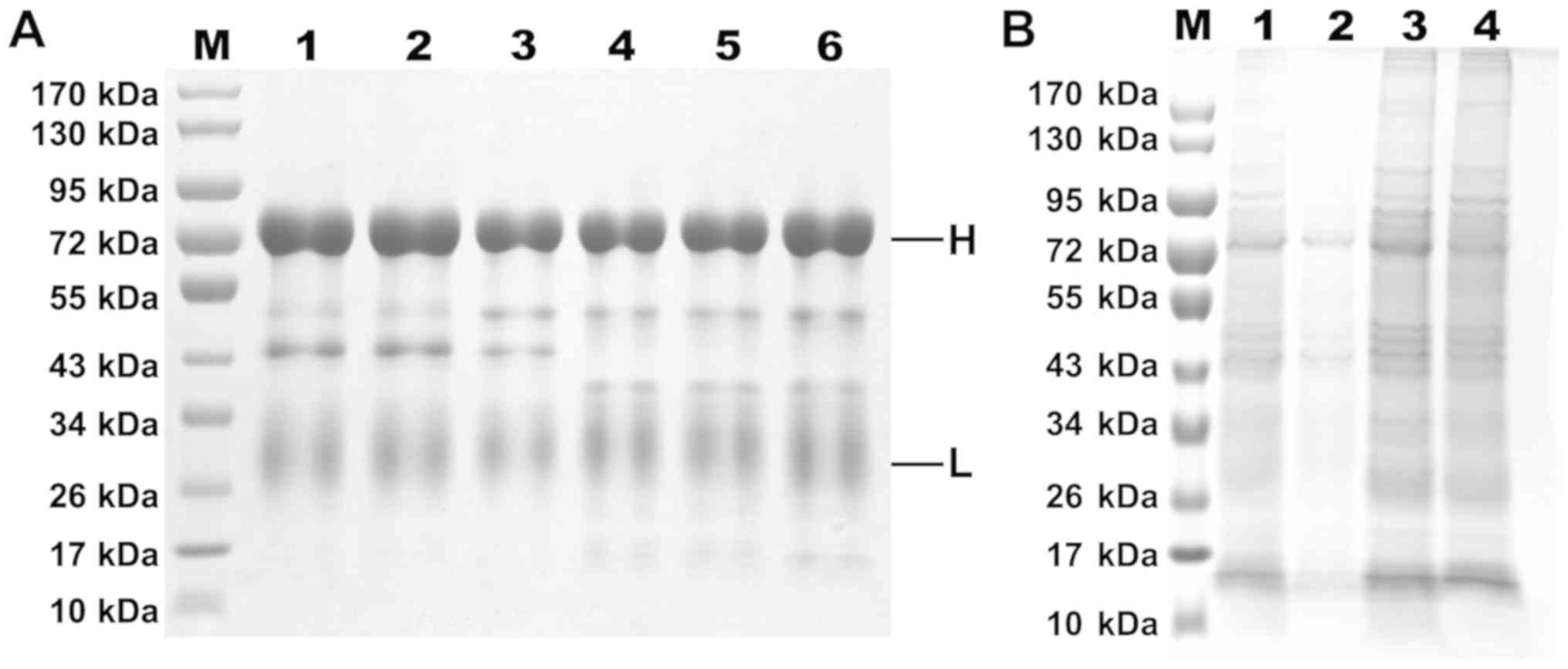
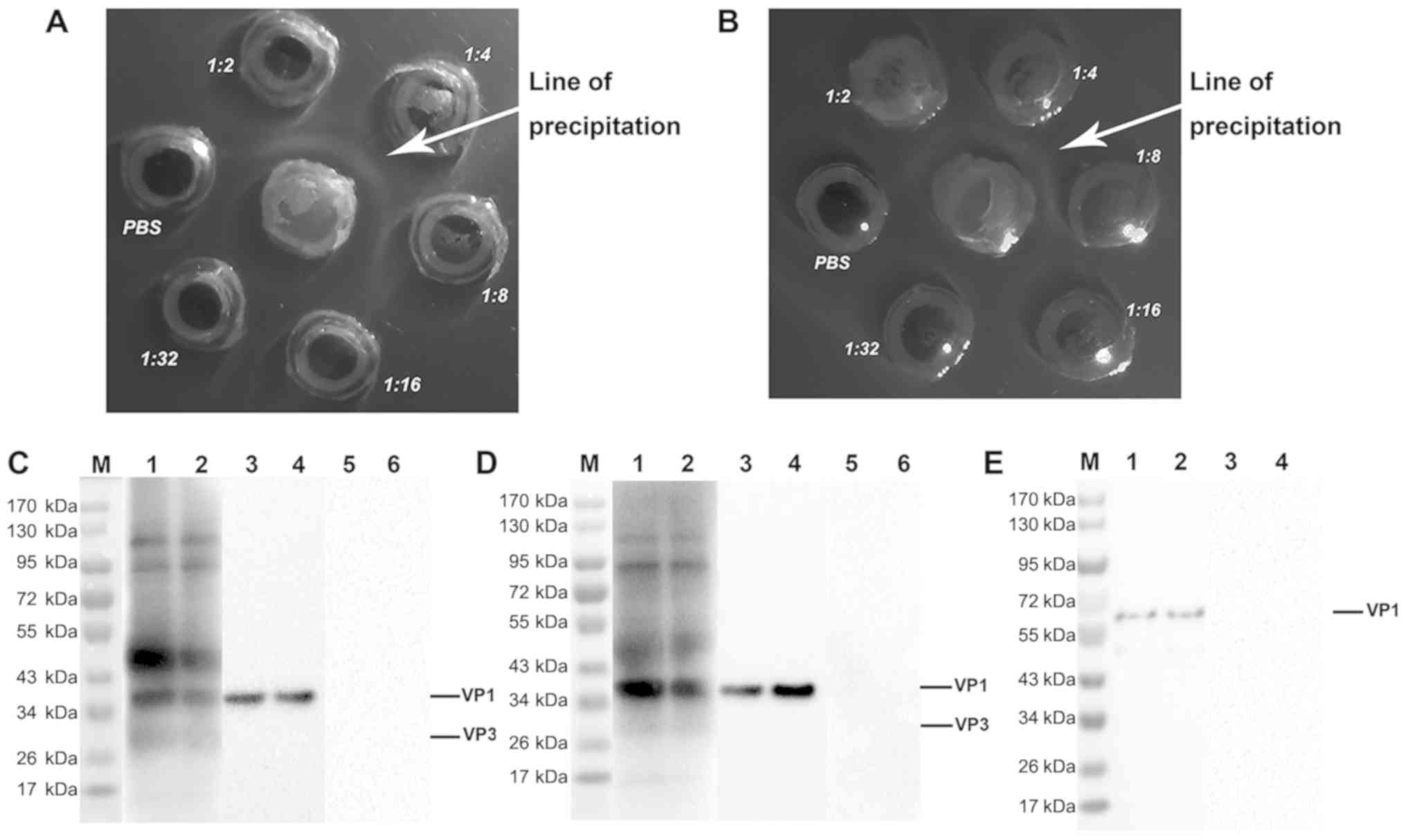
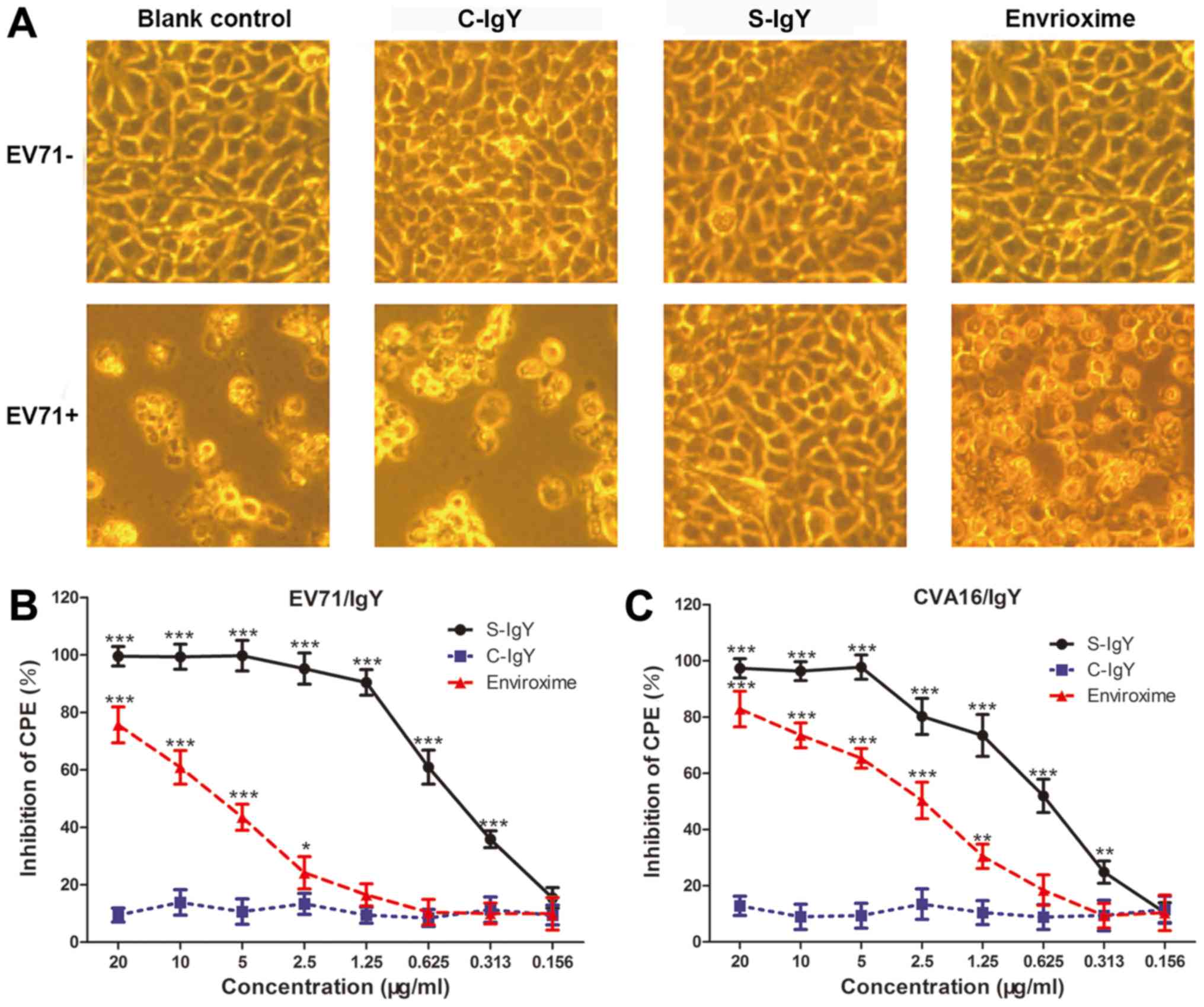
|
1
|
Solomon T, Lewthwaite P, Perera D, Cardosa
MJ, McMinn P and Ooi MH: Virology, epidemiology, pathogenesis, and
control of enterovirus 71. Lancet Infect Dis. 10:778–790. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Yip CC, Lau SK, Woo PC and Yuen KY: Human
enterovirus 71 epidemics: What's next? Emerg Health Threats J.
6:197802013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu SL, Pan H, Liu P, Amer S, Chan TC,
Zhan J, Huo X, Liu Y, Teng Z, Wang L and Zhuang H: Comparative
epidemiology and virology of fatal and nonfatal cases of hand, foot
and mouth disease in mainland China from 2008 to 2014. Rev Med
Virol. 25:115–128. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
4
|
Anastasina M, Domanska A, Palm K and
Butcher S: Human picornaviruses associated with neurological
diseases and their neutralization by antibodies. J Gen Virol.
98:1145–1158. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhu FC, Meng FY, Li JX, Li XL, Mao QY, Tao
H, Zhang YT, Yao X, Chu K, Chen QH, et al: Efficacy, safety, and
immunology of an inactivated alum-adjuvant enterovirus 71 vaccine
in children in China: A multicentre, randomised, double-blind,
placebo-controlled, phase 3 trial. Lancet. 381:2024–2032. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wright HT Jr, Landing BH, Lennette EH and
McAllister RM: Fatal infection in an infant associated with
Coxsackie virus group A, type 16. N Engl J Med. 268:1041–1044.
1963. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Legay F, Leveque N, Gacouin A, Tattevin P,
Bouet J, Thomas R and Chomelt JJ: Fatal coxsackievirus A-16
pneumonitis in adult. Emerg Infect Dis. 13:1084–1086. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Goto K, Sanefuji M, Kusuhara K, Nishimura
Y, Shimizu H, Kira R, Torisu H and Hara T: Rhombencephalitis and
coxsackievirus A16. Emerg Infect Dis. 15:1689–1691. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Wang CY, Li Lu F, Wu MH, Lee CY and Huang
LM: Fatal coxsackievirus A16 infection. Pediatr Infect Dis J.
23:275–276. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen WJ, Arnold JC, Fairchok MP, Danaher
PJ, McDonough EA, Blair PJ, Garcia J, Halsey ES, Schofield C,
Ottolini M, et al: Epidemiologic, clinical, and virologic
characteristics of human rhinovirus infection among otherwise
healthy children and adults: Rhinovirus among adults and children.
J Clin Virol. 64:74–82. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li R, Liu L, Mo Z, Wang X, Xia J, Liang Z,
Zhang Y, Li Y, Mao Q, Wang J, et al: An inactivated enterovirus 71
vaccine in healthy children. N Engl J Med. 370:829–837. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Tung WS, Bakar SA, Sekawi Z and Rosli R:
DNA vaccine constructs against enterovirus 71 elicit immune
response in mice. Genet Vaccines Ther. 5:62007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Foo DG, Alonso S, Chow VT and Poh CL:
Passive protection against lethal enterovirus 71 infection in
newborn mice by neutralizing antibodies elicited by a synthetic
peptide. Microbes Infect. 9:1299–1306. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kirk K, Poh CL, Fecondo J, Pourianfar H,
Shaw J and Grollo L: Cross-reactive neutralizing antibody epitopes
against Enterovirus 71 identified by an in silico approach.
Vaccine. 30:7105–7110. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Foo DG, Alonso S, Phoon MC, Ramachandran
NP, Chow VT and Poh CL: Identification of neutralizing linear
epitopes from the VP1 capsid protein of Enterovirus 71 using
synthetic peptides. Virus Res. 125:61–68. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhou H, Wang G, Li XF, Li Y, Zhu SY, Qin
CF and Tang R: Alumina-encapsulated vaccine formulation with
improved thermostability and immunogenicity. Chem Commun (Camb).
52:6447–6450. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cao R, Han J, Deng Y, Yu M, Qin E and Qin
C: Presence of high-titer neutralizing antibodies against
enterovirus 71 in intravenous immunoglobulin manufactured from
Chinese donors. Clin Infect Dis. 50:125–126. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zhang G, Zhou F, Gu B, Ding C, Feng D, Xie
F, Wang J, Zhang C, Cao Q, Deng Y, et al: In vitro and in vivo
evaluation of ribavirin and pleconaril antiviral activity against
enterovirus 71 infection. Arch Virol. 157:669–679. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chong P, Hsieh SY, Liu CC, Chou AH, Chang
JY, Wu SC, Liu SJ, Chow YH, Su IJ and Klein M: Production of EV71
vaccine candidates. Hum Vaccin Immunother. 8:1775–1783. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Reed Z and Cardosa MJ: Status of research
and development of vaccines for enterovirus 71. Vaccine.
34:2967–2970. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li Y, Bao H, Zhang X, Zhai M, Bao X, Wang
D and Zhang S: Epidemiological and genetic analysis concerning the
non-enterovirus 71 and non-coxsackievirus A16 causative agents
related to hand, foot and mouth disease in Anyang city, Henan
Province, China, from 2011 to 2015. J Med Virol. 89:1749–1758.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mizuta K, Aoki Y, Suto A, Ootani K,
Katsushima N, Itagaki T, Ohmi A, Okamoto M, Nishimura H, Matsuzaki
Y, et al: Cross-antigenicity among EV71 strains from different
genogroups isolated in Yamagata, Japan, between 1990 and 2007.
Vaccine. 27:3153–3158. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Zhang Y, Zhu Z, Yang W, Ren J, Tan X, Wang
Y, Mao N, Xu S, Zhu S, Cui A, et al: An emerging recombinant human
enterovirus 71 responsible for the 2008 outbreak of hand foot and
mouth disease in Fuyang city of China. Virol J. 7:942010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Jolles S, Sewell WA and Misbah SA:
Clinical uses of intravenous immunoglobulin. Clin Exp Immunol.
142:1–11. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ameratunga R, Sinclair J and Kolbe J:
Increased risk of adverse events when changing intravenous
immunoglobulin preparations. Clin Exp Immunol. 136:111–113. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Han JF, Cao RY, Deng YQ, Tian X, Jiang T,
Qin ED and Qin CF: Antibody dependent enhancement infection of
enterovirus 71 in vitro and in vivo. Virol J. 8:1062011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Wang W, Wang EQ and Balthasar JP:
Monoclonal antibody pharmacokinetics and pharmacodynamics. Clin
Pharmacol Ther. 84:548–558. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sgro C: Side-effects of a monoclonal
antibody, muromonab CD3/orthoclone OKT3: Bibliographic review.
Toxicology. 105:23–29. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tini M, Jewell UR, Camenisch G, Chilov D
and Gassmann M: Generation and application of chicken egg-yolk
antibodies. Comp Biochem Physiol A Mol Integr Physiol. 131:569–574.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Lee J, Kang HE and Woo HJ: Stability of
orally administered immunoglobulin in the gastrointestinal tract. J
Immunol Methods. 384:143–147. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Nilsson E, Stålberg J and Larsson A: IgY
stability in eggs stored at room temperature or at + 4°C. Br Poult
Sci. 53:42–46. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schade R, Staak C, Erhard M, Hugl H, Koch
G and Hendriksen C: The production of avian (Egg Yolk) Antibodies:
IgY. ATLA. 24:925–934. 1997.
|
|
33
|
Ulmer-Franco AM, Cherian G, Quezada N,
Fasenko GM and McMullen LM: Hatching egg and newly hatched chick
yolk sac total IgY content at 3 broiler breeder flock ages. Poult
Sci. 91:758–764. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhang WW: The use of gene-specific IgY
antibodies for drug target discovery. Drug Discov Today. 8:364–371.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yang YH, Park D, Yang G, Lee SH, Bae DK,
Kyung J, Kim D, Choi EK, Son JC, Hwang SY and Kim YB:
Anti-Helicobacter pylori effects of IgY from egg york of
immunized hens. Lab Anim Res. 28:55–60. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Akbari MR, Ahmadi A, Mirkalantari S and
Salimian J: Anti-Vibriocholerae IgY antibody inhibits
mortality in suckling mice model. J Natl Med Assoc. 110:84–87.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Thu HM, Myat TW, Win MM, Thant KZ, Rahman
S, Umeda K, Nguyen SV, Icatlo FC Jr, Higo-Moriguchi K, Taniguchi K,
et al: Chicken Egg Yolk antibodies (IgY) for prophylaxis and
treatment of rotavirus diarrhea in human and animal neonates: A
concise review. Korean J Food Sci Anim Resour. 37:1–9. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Fu CY, Huang H, Wang XM, Liu YG, Wang ZG,
Cui SJ, Gao HL, Li Z, Li JP and Kong XG: Preparation and evaluation
of anti-SARS coronavirus IgY from yolks of immunized SPF chickens.
J Virol Methods. 133:112–115. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Xiao Y, Gao X, Maragh S, Telford WG and
Tona A: Cell lines as candidate reference materials for quality
control of ERBB2 amplification and expression assays in breast
cancer. Clin Chem. 55:1307–1315. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lee MS, Cohen B, Hand J and Nokes DJ: A
simplified and standardized neutralization enzyme immunoassay for
the quantification of measles neutralizing antibody. J Virol
Method. 78:209–217. 1999. View Article : Google Scholar
|
|
41
|
Feng Q, He Y and Lu J: Virus-like
particles produced in pichia pastoris induce protective immune
responses against coxsackievirus A16 in mice. Med Sci Monit.
22:3370–3382. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Liu CC, Chou AH, Lien SP, Lin HY, Liu SJ,
Chang JY, Guo MS, Chow YH, Yang WS, Chang KH, et al: Identification
and characterization of a cross-neutralization epitope of
Enterovirus 71. Vaccine. 29:4362–4372. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Ang LW, Koh BK, Chan KP, Chua LT, James L
and Goh KT: Epidemiology and control of hand, foot and mouth
disease in Singapore, 2001–2007. Ann Acad Med Singapore.
38:106–112. 2009.PubMed/NCBI
|
|
44
|
Wang X, Peng W, Ren J, Hu Z, Xu J, Lou Z,
Li X, Yin W, Shen X, Porta C, et al: A sensor-adaptor mechanism for
enterovirus uncoating from structures of EV71. Nat Struct Mol Biol.
19:424–429. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Zhang YX, Huang YM, Li QJ, Li XY, Zhou YD,
Guo F, Zhou JM and Cen S: A highly conserved amino acid in VP1
regulates maturation of enterovirus 71. PLoS Pathog.
13:e10066252017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Curry S, Fry E, Blakemore W, Abu-Ghazaleh
R, Jackson T, King A, Lea S, Newman J and Stuart D: Dissecting the
roles of VP0 cleavage and RNA packaging in picornavirus capsid
stabilization: The structure of empty capsids of foot-and-mouth
disease virus. J Virol. 71:9743–9752. 1997.PubMed/NCBI
|
|
47
|
Kristensen T, Newman J, Guan SH, Tuthill
TJ and Belsham GJ: Cleavages at the three junctions within the
foot-and-mouth disease virus capsid precursor (P1-2A) by the 3C
protease are mutually independent. Virology. 522:260–270. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chong P, Liu CC, Chow YH, Chou AH and
Klein M: Review of enterovirus 71 vaccines. Clin Infect Dis.
60:797–803. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Lim XF, Jia Q, Khong WX, Yan B, Premanand
B, Alonso S, Chow VT and Kwang J: Characterization of an
isotype-dependent monoclonal antibody against linear neutralizing
epitope effective for prophylaxis of enterovirus 71 infection. PLoS
One. 7:e297512012. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Mine Y and Kovacs-Nolan J: Chicken egg
yolk antibodies as therapeutics in enteric infectious disease: A
review. J Med Food. 5:159–169. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Solhi R, Alebouyeh M, Khafri A, Rezaeifard
M and Aminian M: In vitro evaluation of cross-strain inhibitory
effects of IgY polyclonal antibody against H. pylori. Microb
Pathog. 110:682–687. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Müller S, Schubert A, Zajac J, Dyck T and
Oelkrug C: IgY antibodies in human nutrition for disease
prevention. Nutr J. 14:1092015. View Article : Google Scholar : PubMed/NCBI
|