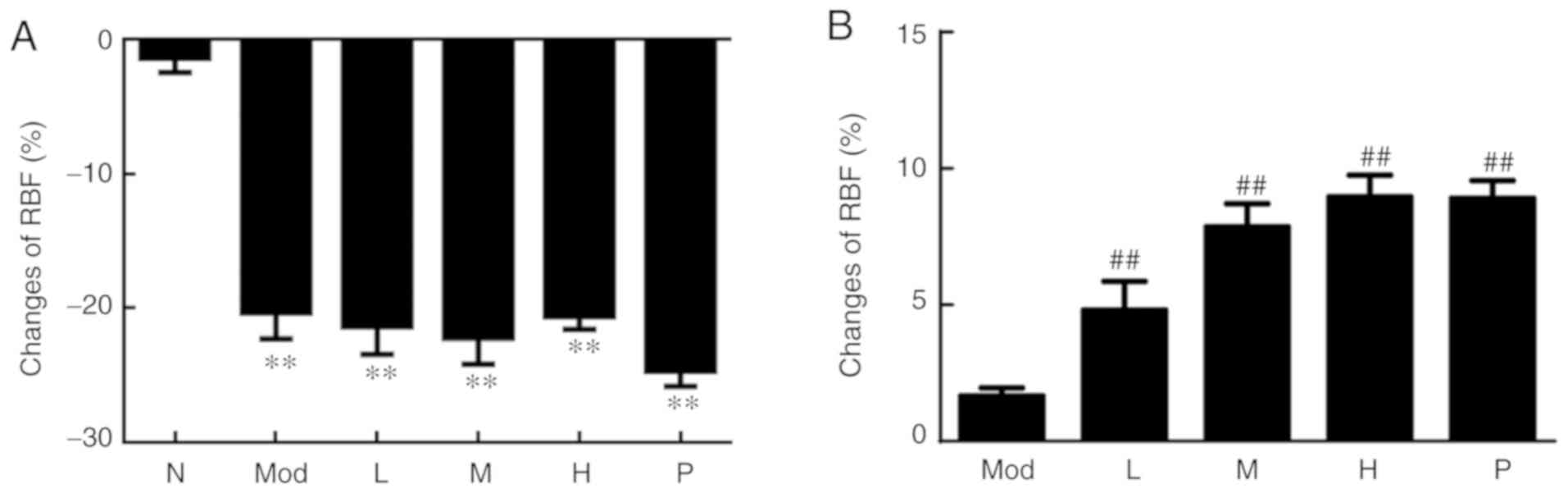
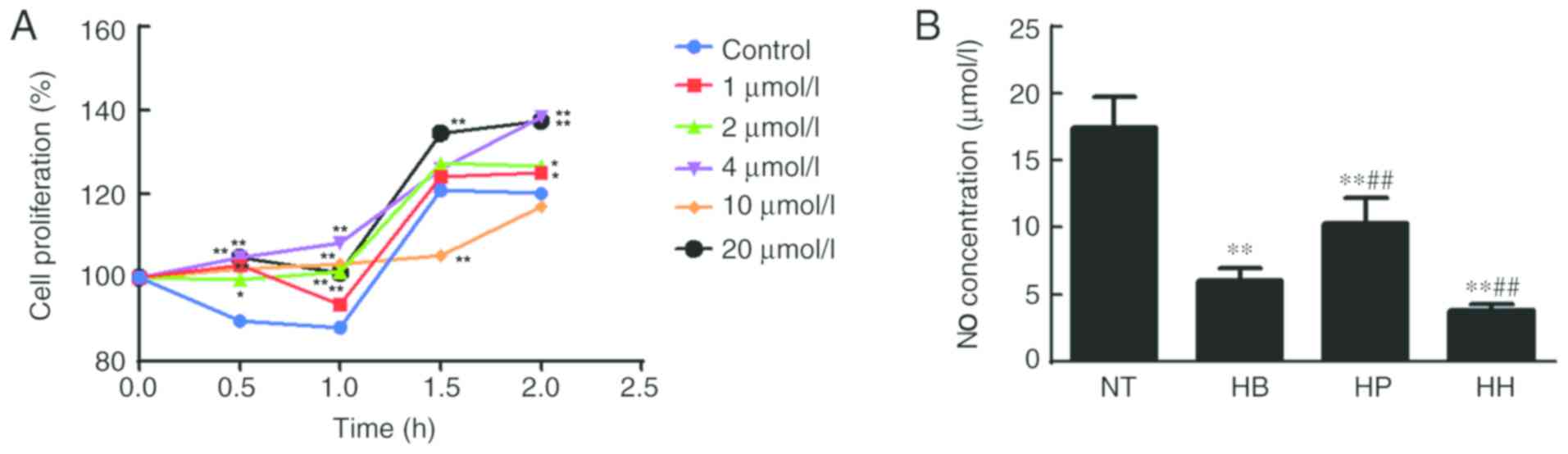
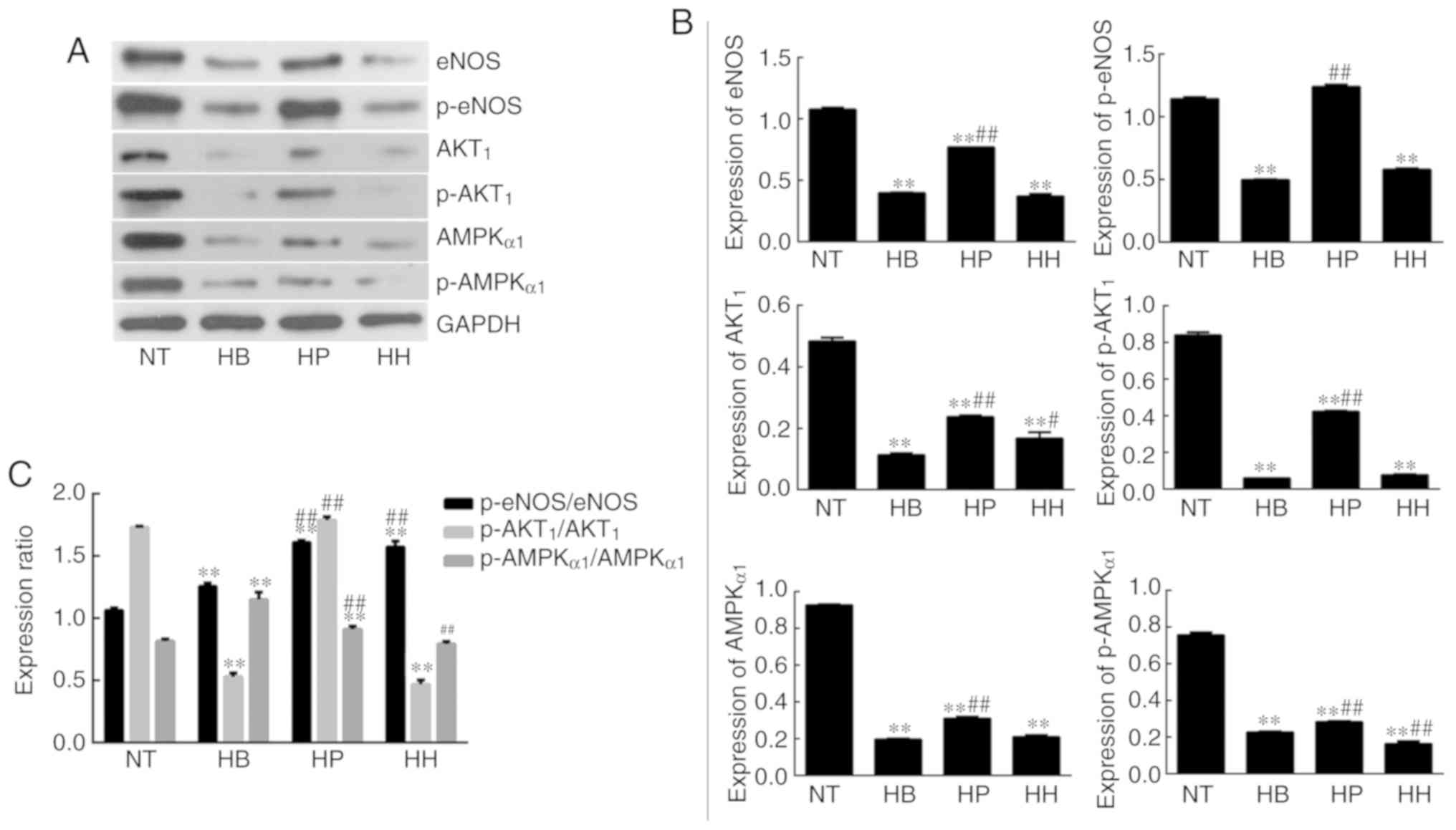
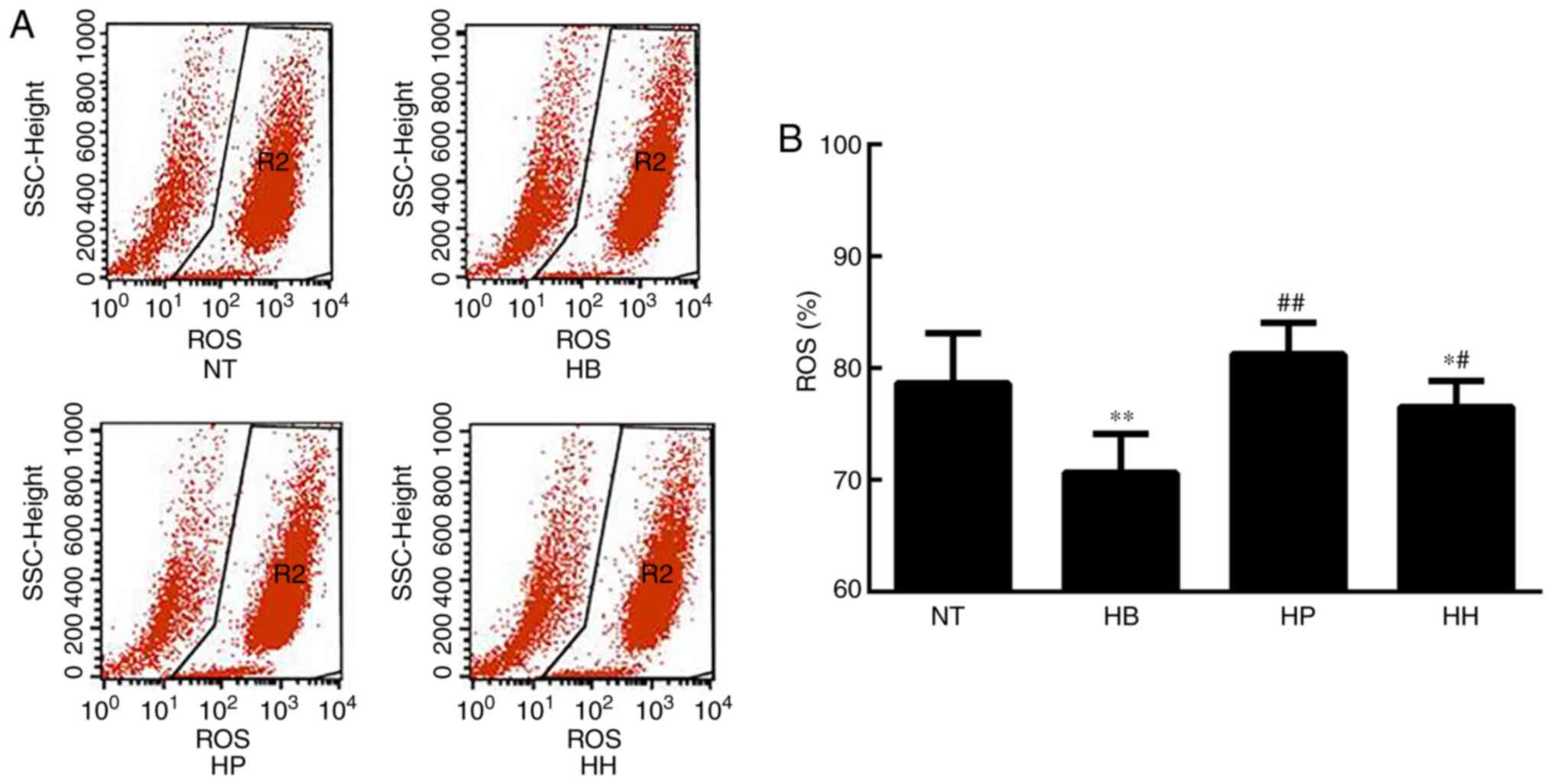
|
1
|
al-Awami M, Schillinger M and Minar E:
Vasospasm of the scrotum-a manifestation of Raynaud's phenomenon?
VASA. 33:87–88. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Barr WG and Fahey PJ: Reduction of
pulmonary capillary blood volume following cold exposure in
patients with Raynaud's phenomenon. Chest. 94:1195–1199. 1988.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Belch JJ, Land D, Park RH, McKillop JH and
MacKenzie JF: Decreased oesophageal blood flow in patients with
Raynaud's phenomenon. Br J Rheumatol. 27:426–430. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Brouwer RM, Wenting GJ and Schalekamp MA:
Acute effects and mechanism of action of ketanserin in patients
with primary Raynaud's phenomenon. J Cardiovasc Pharmacol.
15:868–876. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brown S: Diagnosis and management of
patients with Raynaud's phenomenon. Nurs Stand. 26:41–46. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Engelhart M and Seibold JR: The effect of
local temperature versus sympathetic tone on digital perfusion in
Raynaud's phenomenon. Angiology. 41:715–723. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fardoun MM, Nassif J, Issa K, Baydoun E
and Eid AH: Raynaud's Phenomenon: A brief review of the underlying
mechanisms. Front Pharmacol. 7:4382016. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hughes M and Herrick AL: Raynaud's
phenomenon. Best Pract Res Clin Rheumatol. 30:112–132. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kuryliszyn-Moskal A, Kita J and Hryniewicz
A: Raynaud's phenomenon: New aspects of pathogenesis and the role
of nailfold videocapillaroscopy. Reumatologia. 53:87–93. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
An X, Long C, Deng X, Tang A, Xie J, Chen
L and Wang Z: Higenamine inhibits apoptosis and maintains survival
of gastric smooth muscle cells in diabetic gastroparesis rat model
via activating the beta2-AR/PI3K/AKT pathway. Biomed Pharmacother.
95:1710–1717. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bai G, Yang Y, Shi Q, Liu Z, Zhang Q and
Zhu YY: Identification of higenamine in Radix Aconiti Lateralis
Preparata as a beta2-adrenergic receptor agonist1. Acta Pharmacol
Sin. 29:1187–1194. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bao YX, Yu GR, Xu JM, Xu YQ, Bian YT and
Zheng DS: Effect of acute higenamine administration on
bradyarrhythmias and HIS bundle. A clinical study of 14 cases and
animal experiment on dogs. Chin Med J (Engl). 95:781–784.
1982.PubMed/NCBI
|
|
13
|
Chen YL, Zhuang XD, Xu ZW, Lu LH, Guo HL,
Wu WK and Liao XX: Higenamine combined with [6]-gingerol suppresses
doxorubicin-triggered oxidative stress and apoptosis in
cardiomyocytes via upregulation of PI3K/Akt pathway. Evid Based
Complement Alternat Med. 2013:9704902013.PubMed/NCBI
|
|
14
|
Zhang N, Lian Z, Peng X, Li Z and Zhu H:
Applications of Higenamine in pharmacology and medicine. J
Ethnopharmacol. 196:242–252. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Stajić A, Anđelković M, Dikić N, Rašić J,
Vukašinović-Vesić M, Ivanović D and Jančić-Stojanović B:
Determination of higenamine in dietary supplements by UHPLC/MS/MS
method. J Pharm Biomed Anal. 146:48–52. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhang N, Qu K, Wang M, Yin Q, Wang W, Xue
L, Fu H, Zhu H and Li Z: Identification of higenamine as a novel
alpha1-adrenergic receptor antagonist. Phytother Res. 33:708–717.
2019. View
Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wang Y, Geng J, Jiang M, Li C, Han Y and
Jiang J: The cardiac electrophysiology effects of higenamine in
guinea pig heart. Biomed Pharmacother. 109:2348–2356. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cohen PA, Travis JC, Keizers PHJ, Boyer FE
and Venhuis BJ: The stimulant higenamine in weight loss and sports
supplements. Clin Toxicol (Phila). 57:125–130. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wu MP, Zhang YS, Zhou QM, Xiong J, Dong YR
and Yan C: Higenamine protects ischemia/reperfusion induced cardiac
injury and myocyte apoptosis through activation of β2-AR/PI3K/AKT
signaling pathway. Pharmacol Res. 104:115–123. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Duan W, Chen J, Wu Y, Zhang Y and Xu Y:
Protective effect of higenamine ameliorates collagen-induced
arthritis through heme oxygenase-1 and PI3K/Akt/Nrf-2 signaling
pathways. Exp Ther Med. 12:3107–3112. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liu C, Zhu C, Wang G, Xu R and Zhu Y:
Higenamine regulates Nrf2-HO-1-Hmgb1 axis and attenuates intestinal
ischemia-reperfusion injury in mice. Inflamm Res. 64:395–403. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Lee YS, Kang YJ, Kim HJ, Park MK, Seo HG,
Lee JH, Yun-Choi HS and Chang KC: Higenamine reduces apoptotic cell
death by induction of heme oxygenase-1 in rat myocardial
ischemia-reperfusion injury. Apoptosis. 11:1091–1100. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Chang KC, Chong WS and Lee IJ: Different
pharmacological characteristics of structurally similar
benzylisoquinoline analogs, papaverine, higenamine, and GS 389, on
isolated rat aorta and heart. Can J Physiol Pharmacol. 72:327–334.
1994. View
Article : Google Scholar : PubMed/NCBI
|
|
24
|
Luo D, Xu Z, Hu X, Zhang F, Bian H, Li N,
Wang Q, Lu Y, Zheng Q and Gu J: URI prevents potassium
dichromate-induced oxidative stress and cell death in gastric
cancer cells. Am J Transl Res. 8:5399–5409. 2016.PubMed/NCBI
|
|
25
|
Chang KC, Lim JK and Park CW:
Pharmacological evaluation of GS-389, a novel
tetrahydroisoquinoline analog related to higenamine, on vascular
smooth muscle. Life Sci. 51:67–74. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Hao EW, Deng JG, Du ZC, Yan K, Zheng ZW,
Wang Q, Huang LZ, Bao CH, Deng XQ, Lu XY and Tang ZL: Experimental
study on two-way application of traditional Chinese medicines
capable of promoting blood circulation and removing blood stasis
with neutral property in cold and hot blood stasis syndrome I.
Zhongguo Zhong Yao Za Zhi. 37:3302–3306. 2012.(In Chinese).
PubMed/NCBI
|
|
27
|
Ryan TJ and Copeman PW: Microvascular
pattern and blood stasis in skin disease. Br J Dermatol.
81:563–573. 1969. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ning SY, Jiang BP, Xu L, Fang TH and Wu
MH: Effect of Liangxuehuayu Recipe on hemorheology in rats with
blood stasis syndrome. Asian Pac J Trop Med. 5:935–938. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Kimura I, Makino M, Takamura Y, Islam MA
and Kimura M: Positive chronotropic and inotropic effects of
higenamine and its enhancing action on the aconitine-induced
tachyarrhythmia in isolated murine atria. Jpn J Pharmacol.
66:75–80. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang Z, Liu X, Tao Z, Shi R, Zhang X, Yao
Z, Liu Y, Zhu K and Chen B: Effects of higeramine on hemodynamics
and its tolerability and safety, an experimental study. Zhonghua Yi
Xue Za Zhi. 82:352–355. 2002.(In Chinese). PubMed/NCBI
|
|
31
|
Michiels C: Endothelial cell functions. J
Cell Physiol. 196:430–443. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Brandes RP, Schmitz-Winnenthal FH, Félétou
M, Gödecke A, Huang PL, Vanhoutte PM, Fleming I and Busse R: An
endothelium-derived hyperpolarizing factor distinct from NO and
prostacyclin is a major endothelium-dependent vasodilator in
resistance vessels of wild-type and endothelial NO synthase
knockout mice. Proc Natl Acad Sci USA. 97:9747–9752. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Tang PC, Ng YF, Ho S, Gyda M and Chan SW:
Resveratrol and cardiovascular health-promising therapeutic or
hopeless illusion? Pharmacol Res. 90:88–115. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fu J, Han Y, Wang J, Liu Y, Zheng S, Zhou
L, Jose PA and Zeng C: Irisin lowers blood pressure by improvement
of endothelial dysfunction via AMPK-Akt-eNOS-NO pathway in the
spontaneously hypertensive rat. J Am Heart Assoc. 5:e0034332016.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yamawaki H, Kuramoto J, Kameshima S, Usui
T, Okada M and Hara Y: Omentin, a novel adipocytokine inhibits
TNF-induced vascular inflammation in human endothelial cells.
Biochem Biophys Res Commun. 408:339–343. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Fujimura N, Jitsuiki D, Maruhashi T,
Mikami S, Iwamoto Y, Kajikawa M, Chayama K, Kihara Y, Noma K, Goto
C and Higashi Y: Geranylgeranylacetone, heat shock protein
90/AMP-activated protein kinase/endothelial nitric oxide
synthase/nitric oxide pathway, and endothelial function in humans.
Arterioscler Thromb Vasc Biol. 32:153–160. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Thors B, Halldórsson H and Thorgeirsson G:
Thrombin and histamine stimulate endothelial nitric-oxide synthase
phosphorylation at Ser1177 via an AMPK mediated pathway independent
of PI3K-Akt. FEBS Lett. 573:175–180. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Liu W, Liu JY, Yin ZY, Long CL and Wang H:
The characteristics of vascular endothelial injuries induced by
extreme environmental factors. Zhongguo Ying Yong Sheng Li Xue Za
Zhi. 29:494–500. 2013.PubMed/NCBI
|
|
39
|
Turton EPL, Kent PJ and Kester RC:
VASCULAR REVIEW: The aetiology of Raynaud's phenomenon. Cardiovasc
Surgery. 6:431–440. 1998. View Article : Google Scholar
|
|
40
|
Matucci-Cerinic M, Kahaleh B and Wigley
FM: Review: Evidence that systemic sclerosis is a vascular disease.
Arthritis Rheum. 65:1953–1962. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Cao Y, Ruan Y, Shen T, Huang X, Li M, Yu
W, Zhu Y, Man Y, Wang S and Li J: Astragalus polysaccharide
suppresses doxorubicin-induced cardiotoxicity by regulating the
PI3k/Akt and p38MAPK pathways. Oxid Med Cell Longev.
2014:6742192014. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Awad EM, Khan SY, Sokolikova B, Brunner
PM, Olcaydu D, Wojta J, Breuss JM and Uhrin P: Cold induces
reactive oxygen species production and activation of the NF-kappa B
response in endothelial cells and inflammation in vivo. J Thromb
Haemost. 11:1716–1726. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Maeng J, Sheverdin V, Shin H, Ha I, Bae
SS, Yang-Yen HF and Lee K: Up-regulation of Rhoa/Rho kinase pathway
by translationally controlled tumor protein in vascular smooth
muscle cells. Int J Mol Sci. 15:10365–10376. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Jeyaraj SC, Unger NT, Eid AH, Mitra S,
Paul El-Dahdah N, Quilliam LA, Flavahan NA and Chotani MA: Cyclic
AMP-Rap1A signaling activates RhoA to induce α(2c)-adrenoceptor
translocation to the cell surface of microvascular smooth muscle
cells. Am J Physiol Cell Physiol. 303:C499–C511. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Bailey SR, Eid AH, Mitra S, Flavahan S and
Flavahan NA: Rho kinase mediates cold-induced constriction of
cutaneous arteries: Role of alpha2C-adrenoceptor translocation.
Circ Res. 94:1367–1374. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Eid AH, Chotani MA, Mitra S, Miller TJ and
Flavahan NA: Cyclic AMP acts through Rap1 and JNK signaling to
increase expression of cutaneous smooth muscle
alpha2C-adrenoceptors. Am J Physiol Heart Circ Physiol.
295:H266–H272. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Honda M, Suzuki M, Nakayama K and Ishikawa
T: Role of alpha2C-adrenoceptors in the reduction of skin blood
flow induced by local cooling in mice. Br J Pharmacol. 152:91–100.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jantschak F, Popp AM, Hofmann RA, Villalón
CM, Centurión D and Pertz HH: Postjunctional α2C-adrenoceptors
mediate vasoconstriction in rat tail artery: Influence of
precontraction and temperature on vasoreactivity. Naunyn
Schmiedebergs Arch Pharmacol. 382:487–497. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Furspan PB, Chatterjee S, Mayes MD and
Freedman RR: Cooling-induced contraction and protein tyrosine
kinase activity of isolated arterioles in secondary Raynaud's
phenomenon. Rheumatology (Oxford). 44:488–494. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Leonov Y, Sterz F, Safar P, Radovsky A,
Oku K, Tisherman S and Stezoski SW: Mild cerebral hypothermia
during and after cardiac arrest improves neurologic outcome in
dogs. J Cereb Blood Flow Metab. 10:57–70. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Arbour RB: Brain death: Assessment,
controversy, and confounding factors. Critical Care Nurse.
33:27–46. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Globus MY, Alonso O, Dietrich WD, Busto R
and Ginsberg MD: Glutamate release and free radical production
following brain injury: Effects of posttraumatic hypothermia. J
Neurochem. 65:1704–1711. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Geocadin RG, Koenig MA, Jia X, Stevens RD
and Peberdy MA: Management of brain injury after resuscitation from
cardiac arrest. Neurol Clin. 26487–506. (ix)2008. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Bradley SM, Liu W, McNally B, Vellano K,
Henry TD, Mooney MR, Burke MN, Brilakis ES, Grunwald GK, Adhaduk M,
et al: Temporal trends in the use of therapeutic hypothermia for
out-of-hospital cardiac arrest. JAMA Netw Open. 1:e1845112018.
View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Lee HY, Lee JS, Kim EJ, Han JW, Lee HW,
Kang YJ and Chang KC: Inhibition of lipopolysaccharide-induced
inducible nitric oxide (iNOS) mRNA expression and nitric oxide
production by higenamine in murine peritoneal macrophages. Arch
Pharm Res. 22:55–59. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Kang YJ, Lee YS, Lee GW, Lee DH, Ryu JC,
Yun-Choi HS and Chang KC: Inhibition of activation of nuclear
factor kappaB is responsible for inhibition of inducible nitric
oxide synthase expression by higenamine, an active component of
aconite root. J Pharmacol Exp Ther. 291:314–320. 1999.PubMed/NCBI
|
|
57
|
Park JE, Kang YJ, Park MK, Lee YS, Kim HJ,
Seo HG, Lee JH, Hye Sook YC, Shin JS, Lee HW, et al: Enantiomers of
higenamine inhibit LPS-induced iNOS in a macrophage cell line and
improve the survival of mice with experimental endotoxemia. Int
Immunopharmacol. 6:226–233. 2006. View Article : Google Scholar : PubMed/NCBI
|