Introduction
Hepatitis C virus (HCV) infection has been detected
in ~170 million patients worldwide, and chronic hepatitis C (CHC)
infection is a major risk factor for cirrhosis and hepatocellular
carcinoma, which are associated with high morbidity and mortality
(1,2). The traditional standard of care,
interferon (IFN)-based therapies, may achieve sustained virological
response (SVR) rates of ~50%, and have serious side effects
(3). Direct-acting anti-virals
(DAAs) are replacing IFN-based therapy as the standard of care,
with SVR rates of >90% (4).
However, the recurrence rate in DAA-treated patients remains
significant (5,6).
Approximately two-thirds of HCV patients experience
extra-hepatic manifestations in the cardiovascular, renal and
central nervous systems, including mixed essential
cryoglobulinemia, non-Hodgkin's lymphoma and glomerulonephritis
(7–9). Almost 415 million individuals worldwide
have diabetes mellitus. According to statistics, one patient
succumbs to its complications every six seconds (10). CHC patients also have a higher
prevalence of insulin resistance (IR) and type 2 diabetes mellitus
(T2DM), and IR improvement can increase the positive response to
antiviral therapy regardless of HCV genotype (11–14). CHC
patients with IR have higher rates of liver fibrosis (15) and 20% lower rates of SVR compared to
patients without IR (16). Of note,
patients with T2DM have an elevated risk of acquiring HCV infection
(17). This may be due to lower
immunity in patients with T2DM. Therefore, for patients with T2DM
patients, avoiding the need to share needles can reduce the risk of
HCV infection.
IR in HCV patients has been reported to be mediated
by the HCV core protein, which interferes with glucose metabolism
and insulin signaling by inhibiting the expression of insulin
receptor substrate (IRS)1 and IRS2 (13,18–20).
HCV-induced IR in the tissue of HCV-infected patients and animal
models has also been indicated to be mediated via a number of
cytokines, including tumor necrosis factor-α (21).
Viral clearance in HCV patients treated with
pegylated interferon α-2a and ribavirin has been associated with
reduced IR [measured as a reduction in homeostasis model assessment
(HOMA) scores after 24 weeks of therapy] and reduced fasting serum
insulin and serum glucose levels (20,22,23). By
contrast, elevated HOMA-IR scores were indicated to be associated
with a 3-fold increase in the risk of failure to achieve SVR in HCV
patients treated with pegylated interferon plus ribavirin (24). SVR was associated with a reduction in
HOMA-IR in patients with HCV genotype 1 infection, but not in
patients infected with genotypes 2 or 3 (25), suggesting that viral factors have an
important role in modulating the insulin signaling pathway.
However, non-diabetic, non-cirrhotic patients with HCV genotype 1
infection treated with pegylated (Peg-)IFN plus ribavirin exhibited
a reduction in IR, independent of the virological outcome (26). A recent meta-analysis reported that
achieving SVR was associated with reduced IR at the follow-up
visit, and also protected against the incidence of diabetes
(27). However, this study was
limited by the lack of randomized data comparing SVR to non-SVR
patients.
Despite the discrepancies arising from differences
in study design and variables evaluated, these results, along with
the result that SVR was associated with a significant reduction in
mean fasting glucose levels and fasting serum insulin levels
(22,28), suggest that SVR is linked to improved
glycemic control in HCV patients. The present meta-analysis aimed
to evaluate the effect of HCV treatment response on IR by comparing
the reduction in IR among HCV patients who achieved SVR and those
who did not (non-SVR group) after receiving IFN-based therapy.
Materials and methods
Literature search strategy
The PRISMA guidelines were followed for the
processes and reporting in the present systematic review and
meta-analysis (29). To identify
relevant studies for inclusion in the present meta-analysis, the
PubMed, Cochrane and Embase databases were searched using
combinations of the following search terms: ‘HCV’, ‘hepatitis C’,
‘interferon’, ‘antiviral’, ‘treatment response’ and ‘insulin
resistance’. The search strategy for PubMed included the terms
‘insulin resistance’ AND (‘hepatitis C’ OR ‘HCV’) AND (‘antiviral’
OR ‘interferon’); the search filters were as follows: Abstract
available; English language. The search included studies published
in the time window from inception until December 19, 2017.
Selection of studies
The inclusion criteria for the meta-analysis were as
follows: i) Prospective and retrospective studies; ii) studies on
HCV patients who received IFN-based therapy; iii) studies in which
IR was evaluated in SVR patients and non-SVR patients; and iv)
studies reporting quantitative outcomes. Exclusion criteria were as
follows: i) Publication type: Reviews, letters, commentaries,
editorials, case reports, personal communications; ii) studies in
which patients received other interventions; and iii) studies which
did not report quantitative primary outcomes.
A 2-step screening process was used to identify
studies that would be included in the meta-analysis. The first step
involved an evaluation of the title and abstract of each article,
and citations not meeting the inclusion criteria and meeting the
exclusion criteria were discarded. The second step involved the
evaluation of full-text copies of the remaining citations to
identify those that met all of the inclusion criteria and none of
the exclusion criteria. The screening process and identification of
eligible studies were performed by two independent reviewers JHH
and NJL. A third reviewer, MLC, was consulted if there were any
uncertainties regarding eligibility. The reference lists of the
relevant studies were hand-searched to identify further studies
that met the inclusion criteria.
Data extraction and analysis
Data were extracted from the eligible studies by two
independent reviewers. Regarding the presentation of patient
characteristics in the studies selected, parameters including age
were expressed as the mean ± standard deviation (SD) and
information regarding gender as the percentage of males in the
given groups. For the clinical outcomes, the incidence of IR and
scores/levels of HOMA-IR, HOMA-β, fasting glucose and fasting
insulin were summarized in terms of basal values and values at the
end of treatment (EOT) for each of group, if available. Data were
summarized as the percentage of affected patients regarding the
incidence of IR and as the mean ± standard deviation for other
categorical data. The effect size was set as the reduction of the
incidence rate of IR, HOMA-IR scores and HOMA-β scores from the
basal value to the value after EOT for the SVR and non-SVR groups
from each study. For studies reporting on non-responders and
relapsers, these data were combined into the non-SVR group. The
reduction in the incidence rate of IR was defined as the difference
in the reduction in the incidence rate of IR with a 95% confidence
interval (95%CI; lower limit, upper limit) and P-value for
comparison between the SVR and non-SVR groups; the reduction of
HOMA-IR and HOMA-β scores was defined as the difference in
reduction of the mean HOMA-IR and HOMA-β scores from the basal
value to the value after EOT with 95%CI and P-value for comparison
between the SVR and non-SVR groups; a combined effect was
calculated for those studies with completed measurements.
For the effect size of reduction in the incidence
rate of IR, a difference in the reduction of the IR incidence rate
>0 indicated that the SVR group had a greater reduction in the
IR incidence rate than the non-SVR group; reduction in the IR
incidence rate <0 indicated that the SVR group had a smaller
reduction in the IR incidence rate than the non-SVR group;
reduction of IR incidence rate=0 indicated that the SVR and non-SVR
groups had similar reduction rates. For the effect size of
reduction in HOMA-IR and HOMA-β scores, an effect size of outcomes
<0 indicated that the SVR group had a greater reduction than the
non-SVR group; an effect size of outcomes >0 indicated that the
non-SVR group had a greater reduction than the SVR group; and an
effect size of outcomes=0 indicated that the SVR and non-SVR groups
had a similar reduction in outcomes.
Study heterogeneity was evaluated using
χ2-based Cochran's Q and I2 statistics. A
random-effects model (DerSimonian-Laird method) was considered for
the meta-analysis if Q statistics with P<0.10 or
I2>50% were obtained; otherwise, a fixed-effects
model (Mantel-Haenszel method) was employed for the meta-analysis.
Regarding the Q statistics, P<0.10 was considered to indicate
statistical significance regarding heterogeneity. For the
I2 statistics, heterogeneity was assessed as follows:
I2=0–25% indicated no heterogeneity,
I2=25–50% moderate heterogeneity, I2=50–75%
high heterogeneity and I2=75–100% very high
heterogeneity. A two-sided P<0.05 was considered to indicate
statistical significance. Sensitivity analysis was performed using
a leave-one-out approach. According to Sterne et al
(30) study, when the number of
studies in the meta-analysis is <10, funnel plot asymmetry
should not be used. Therefore, in the current study, publication
bias was not assessed due to the limitation in the number of
studies. All statistical analyses were performed using the
statistical software Comprehensive Meta-Analysis, version 2.0
(Biostat).
Quality assessment
The Quality In Prognosis Studies tool was used to
assess the quality of included studies (31). The tool comprises six areas:
Participation, attrition, measurement of prognostic factors,
measurement of and controlling for confounding variables,
measurement of outcomes and analytic approaches. The quality of
included studies was independently appraised by two reviewers JHH
and NJL. Any discrepancies were resolved by a third reviewer
MLC.
Results
Study selection
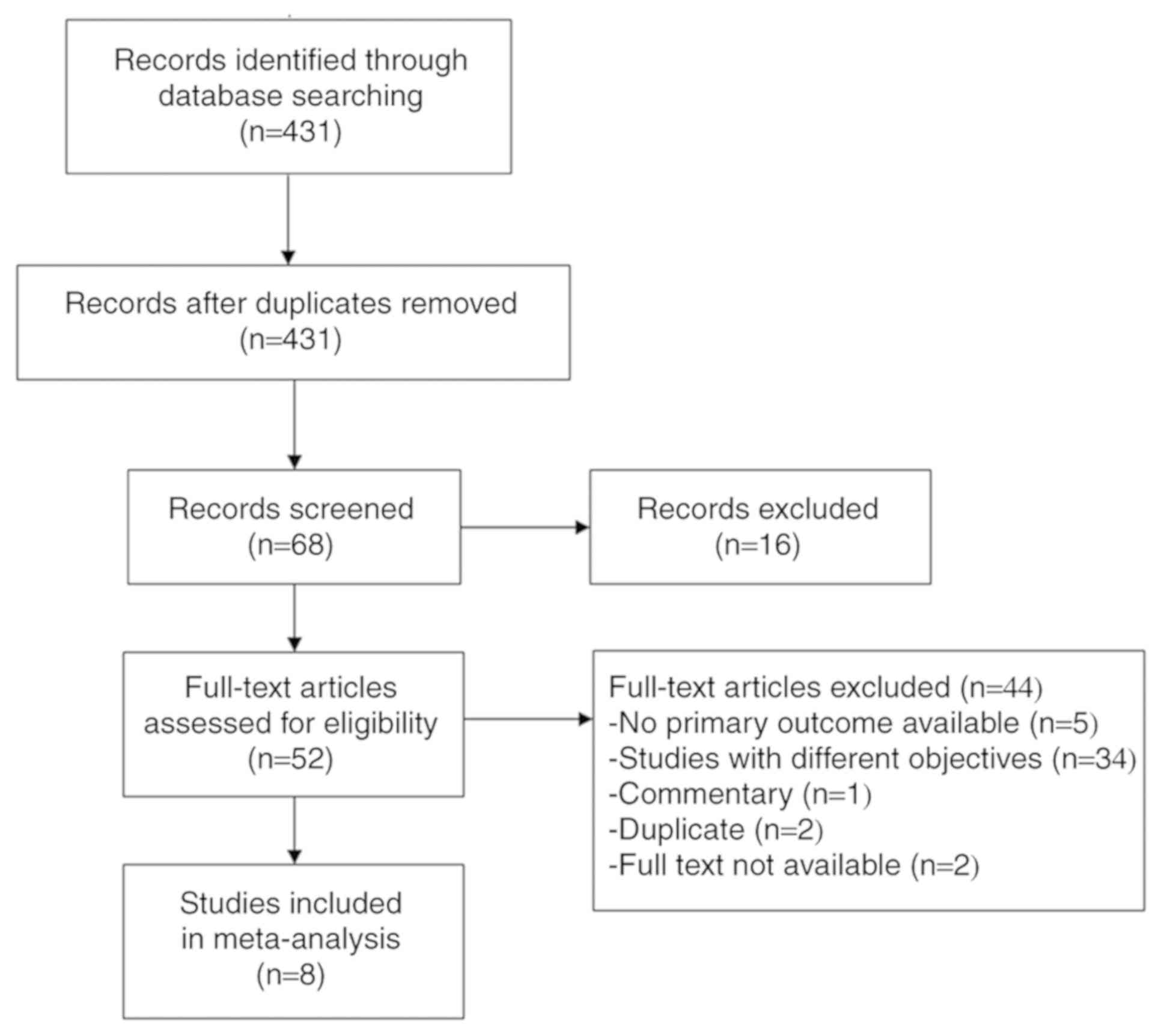
A flow chart of the study selection process is
presented in Fig. 1. A total of 431
studies were initially identified, of which 52 were evaluated for
selection for the meta-analysis. After excluding 44 studies (5
studies did not report primary outcomes; 34 had different
objectives; 1 was a commentary; 2 were duplicates; and 2 did not
have full texts available), a total of 8 studies were included in
the final analysis (19,20,22,26,32–35).
Study characteristics
The present meta-analysis included 5 prospective
studies, 2 retrospective studies and 1 randomized controlled trial
(RCT). The analysis included a total of 533 subjects in the SVR
group and 346 subjects in the non-SVR group. The age of study
subjects ranged from 40.4 to 63.2 years, and the percentage of
males ranged from 45.5 to 85.2%. The follow-up time was 6 months
after EOT in 7 studies, while one study had a follow-up time of 24
months after EOT (Table I). A
comparison of the incidence rate of IR, as well as HOMA-IR scores,
HOMA-β scores, fasting glucose levels and fasting insulin levels
between the baseline and EOT is provided in Table II.
 | Table I.Summary of study characteristics of
the eight studies included in the meta-analysis. |
Table I.
Summary of study characteristics of
the eight studies included in the meta-analysis.
| 1st author
(year) | Study design | Study period of
patients | Number | Compared
groups | Treatment protocol
(dose, duration) | Age (years) | Males (%) | Genotype (n) | Country | Follow-up time | (Refs.) |
|---|
| Jung (2014) | Retrospective | Jan 5 2005 to Dec
2011 | 49 | SVR | n.s. | 48±11.2 | n.s | 1/non-1: 28/21 | Korea | 6 months after
EOT | (32) |
|
|
|
| 11 | Non-SVR |
| 56.6±9.9 |
| 1/non-1: 8/3 |
|
|
|
| Chan (2013) | Prospective | 2003–2007 | 48 | SVR | All but five
patients received PEG-IFN-2a and weight-based RBV therapy (1,000 mg
for patients with BW <75 kg or 1,200 mg daily for patients with
a BW >75 kg). The remaining 5 received PEG-IFN-2b and weight-
based RBV therapy. Treatment duration was genotype-dependent (12
months for genotype 1 and 6 months for genotypes 2 and 3) | 42±10 | 24 (50) | 1/2/3: 24/6/17 | Australia | 6 months after
EOT | (33) |
|
|
|
| 38 | Non-SVR |
|
| 21 (55) | 1/2/3: 20/4/13 |
|
|
|
| Aghemo (2012) | RCT | MIST sub-study | 230 | SVR | Combination of
PEG-IFN-2a (180 g/week) and RBV (800–1,200 mg/day) or a combination
of PEG-IFN-2b (1.5 g/kg/week) and daily RBV (800–1,200 mg) for a
standard duration based on the HCV genotype. |
|
| 1/4: 78 2/3:
152 | Italy | 24 months after
EOT | (19) |
|
|
|
| 124 | Non-SVR |
| n.s. | n.s. | 1/4: 94 2/3:
30 |
|
|
|
| Khattab (2012) | Prospective | Feb 2008 to Jan
2011 | 61 | SVR | Peg-IFNα-2a (180
g/week) or a combination of Peg-IFNα-2b (1.5 g/kg/week) and RBV
(1,000 mg or 1,200 mg/day for BW≤ or >75 kg, respectively)
during the study period for 48 weeks independent of the rapid
virological response. | 40.4±5.8 | 52 (85.2) | 4: 107 | Egypt | 6 months after
EOT | (34) |
|
|
|
| 46 | Non-SVR |
| 42.8±5.1 | 34 (73.9) |
|
|
|
|
| Kim (2009) | Prospective | May 2004 to Nov
2006 | 22 | Sustained
Responders | Combination of
Peg-IFNα-2a (180 µg) and RBV (800–1,000 mg/day, according to the
genotype of HCV) | 57.6±8.9 | 10 (45.5) | 1b/2a: 9/13 | Korea | 6 months after
EOT | (22) |
|
|
|
| 6 | Non-responder |
| 50.8±9.6 | 4 (66.7) | 1b/2a: 6/0 |
|
|
|
| Kawaguchi
(2009) | Retrospective | Jan 2002 to Aug
2007 | 48 | SVR | Patients with a
high viral load or past history of IFN treatment received 6 or 10
MU of IFN-2b three times a week for 24 weeks plus RBV 600–1,000
mg/day and were treated with Peg-IFN-2b 1.5 mg/kg/week plus RBV
daily for 48 weeks (genotype 1) or 24 weeks (genotype 2). Patients
with a low viral load received 5-10MU of natural IFN three times a
week for 24 weeks or 6 MU of IFN daily for 8 weeks and were treated
with Peg-IFNα-2a 180 mg/week for 24–48 weeks or the above-mentioned
IFN regimen. | 52.3±12.1 | 35 (72.9) | 1/2: 31/17 | Japan | 6 months after
EOT | (35) |
|
|
|
| 24 | Non-SVR |
| 56.1±8.1 | 13 (54.2) | 1/2: 21/3 |
|
|
|
| Petta (2009) | Prospective | May 2005 to Nov
2006 | 46 | SVR | n.s. | 47.0±12.6 | 30 (65.2) | 1: 83 | Italy | 6 months after
EOT | (26) |
|
|
|
| 20 | Relapsers |
| 54.7±11.9 | 17 (45.9) |
|
|
|
|
|
|
|
| 17 | Non-responders |
|
|
|
|
|
|
|
| Kawaguchi
(2007) | Prospective | n.s. | 29 | SVR | n.s. | 58.5±8.6 | 19 (65.5) | 1/2: 11/18 | Japan | 6 months after
EOT | (20) |
|
|
|
| 48 | Non-responders |
| 61.7±7.7 | 27 (56.2) | 1/2: 42/6 |
|
|
|
|
|
|
| 12 | Relapsers |
| 63.2±6.1 | 8 (1.5) | 1/2: 10/2 |
|
|
|
 | Table II.Summary of the clinical outcomes at
the first visit and after EOT for all included studies. |
Table II.
Summary of the clinical outcomes at
the first visit and after EOT for all included studies.
| 1st author
(year) | Compared
groups | Basal incidence of
IR (%) | Incidence of IR
after EOT (%) | Basal HOMA-IR | HOMA-IR after
EOT | Basal HOMA- | HOMA- after
EOT | Basal FPG
(mg/dl) | FPG after EOT
(mg/dl) | Basal fasting
insulin (µU/ml) | Fasting insulin
after EOT (µU/ml) | (Refs.) |
|---|
| Jung (2014) | SVR | n.s. | n.s. | 3.4±2.69 | 2.5±0.2 | 149.6±117 | 104.3±6.5 | n.s. | n.s. | n.s. | n.s. | (32) |
|
| Non-SVR | n.s. | n.s. | 3.4±2.31 | 3.1±0.2 | 111.1±65.6 | 102.2±5.3 | n.s. | n.s. | n.s. | n.s. |
|
| Chan (2013) | SVR | n.s. | n.s. | 3.0±1.0 | 2.3±0.7 | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | (33) |
|
| Non-SVR | n.s. | n.s. | 3.1±0.7 | 3.1±0.9 | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
|
| Aghemo (2012) | SVR | HOMA score >2:
12% | 10% | 1.11±0.8 | 1.18±1.1 | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. | (19) |
|
| Non-SVR | HOMA score >2:
11% | 19% | 1.20±0.85 | 1.49±1.3 | n.s. | n.s. | n.s. | n.s. | n.s. | n.s. |
|
| Khattab (2012) | SVR | n.s. | n.s. | 2.03±0.6 | 1.2±0.3 | n.s. | n.s. | 4.5±0.6
(mmol/l) | n.s. | 13.7±5.1 | n.s. | (34) |
|
| Non-SVR | n.s. | n.s. | 4.05±0.8 | Non-responders:
3.77±0.6 Relapsers: 2±0.4 | n.s. | n.s. |
| n.s. |
| n.s. |
|
| Kim (2009) | SVR | HOMA-IR >3: 14
(63.6) |
| 5.28±4.55 | 2.62±1.07 | n.s. | n.s. | 112.6±33.0 | 99.9±14.9 | 18.13±14.24 | 10.40±3.12 | (22) |
|
| Non-SVR | HOMA-IR >3: 4
(66.7) |
| 3.34±1.24 | 3.4 5± 1.28 | n.s. | n.s. | 105.5±15.2 | 107.0±14.6 | 12.50±3.16 | 13.06±4.02 |
|
| Kawaguchi
(2009) | SVR | n.s. | n.s. | 1.86±1.08 | 1.67±0.93 | 174±211 | 105±60 | n.s. | n.s. | n.s. | n.s. | (35) |
|
| Non-SVR | n.s. | n.s. | 2.50±1.13 | 2.50±1.31 | 193±145 | 157±126 | n.s. | n.s. | n.s. | n.s. |
|
| Petta (2009) | SVR | HOMA ≤2.7: 30% | 8.80% | 2.64±1.85 | 1.53±0.92 | n.s. | n.s. | n.s. | n.s. | 11.9±7.3 | n.s. | (26) |
|
| Relapsers | HOMA ≤2.7: 30% | 19.30% | 2.64±1.16 | 1.85±0.80 | n.s. | n.s. | n.s. | n.s. | 12.5±5.8 | n.s. |
|
|
| Non-responders | HOMA ≤2.7: | 17.50% 52% | 3.10±1.80 | 2.05±0.84 | n.s. | n.s. | n.s. | n.s. |
| n.s. |
|
| Kawaguchi
(2007) | SVR | n.s. | n.s. | 3.07±0.95 | 2.2±0.7 | 113.7±21.3 | 92.6±14.0 | n.s. | n.s. | n.s. | n.s. | (20) |
|
| Non-responders | n.s. | n.s. | 3.95±1.69 | 3.6±1.2 | 120.0±26.1 | 112.4±24.1 | n.s. | n.s. | n.s. | n.s. |
|
|
| Relapsers | n.s. | n.s. | 3.73±1.21 | 3.6±1.2 | 121.5±13.3 | 117.4±17.4 | n.s. | n.s. | n.s. | n.s. |
|
Meta-analysis
Reduction in incidence of IR
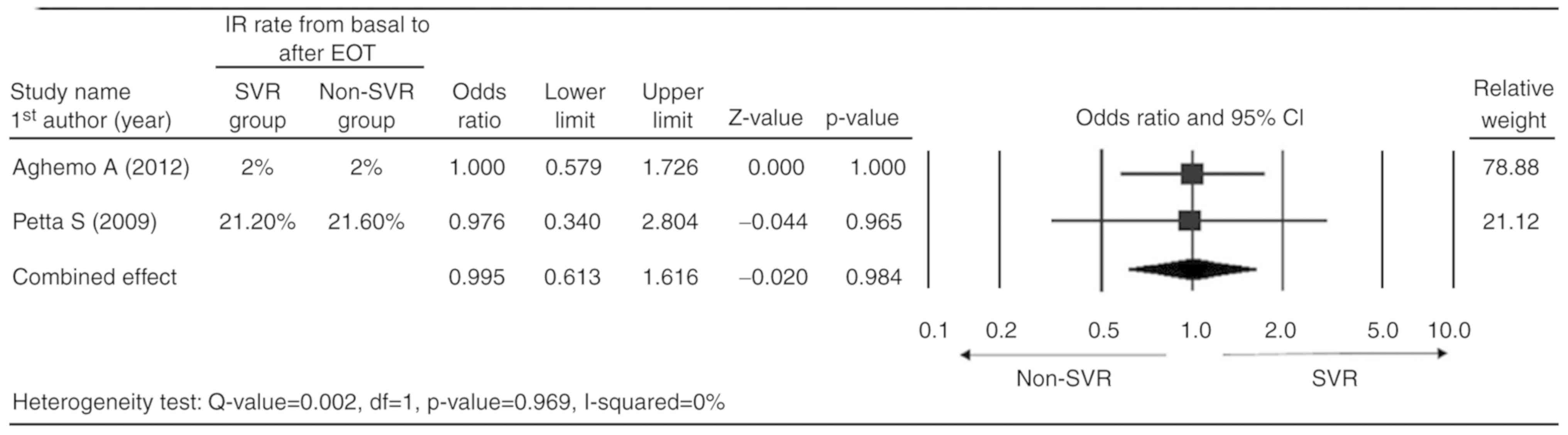
Only the two studies which reported complete data
for the IR rate (basal value and value after EOT) were considered
for the meta-analysis (19,26). A fixed-effect model was applied
according to the heterogeneity test [Q-value=0.002; degrees of
freedom (df)=1; P=0.969; I2=0%]. The combined analysis
indicated that the SVR and non-SVR groups had a similar reduction
in IR rate (odds ratio=0.995; 95%CI=0.613–1.616; P=0.984; Fig. 2).
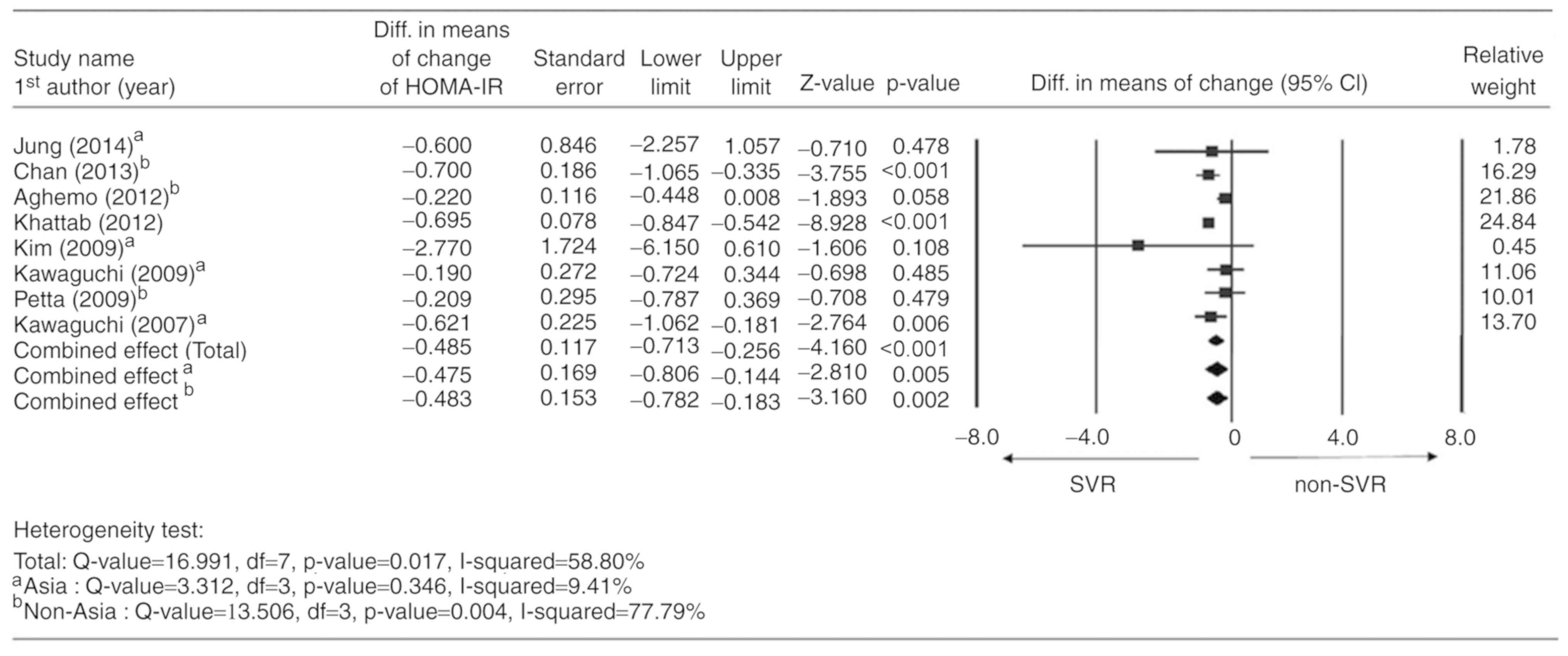
Reduction in mean HOMA-IR
All of the 8 studies reported a change in HOMA-IR
between the basal value and the value after EOT, and were therefore
considered for meta-analysis. A random-effects model was applied
according to the heterogeneity test (Q-value=16.991; df=7; P=0.017;
I2=58.80%). The combined analysis indicated that the SVR
group had a significantly higher mean reduction in HOMA-IR compared
with the non-SVR group (difference in means=−0.485; 95%CI=−0.713 to
−0.256; P<0.001). In addition, subgroup analysis indicated
similar changes in Asian and non-Asian studies (Asian: Difference
in means=−0.475; 95%CI=−0.806 to −0.144; P=0.005; non-Asian:
Difference in means=−0.483; 95%CI=−0.782 to −0.183; P=0.002;
Fig. 3). Table SI summarizes the meta-analysis for
studies with genotypes 1/2. The results indicated that the SVR
group had a higher mean reduction in HOMA-IR as compared with the
non-SVR group (difference in means=−0.403; P=0.007). However, the
change in HOMA-IR was similar between the SVR and non-SVR groups
after removing the study by Kawaguchi et al (20) from 2007 (difference in means=−0.233,
P=0.241; Table SI).
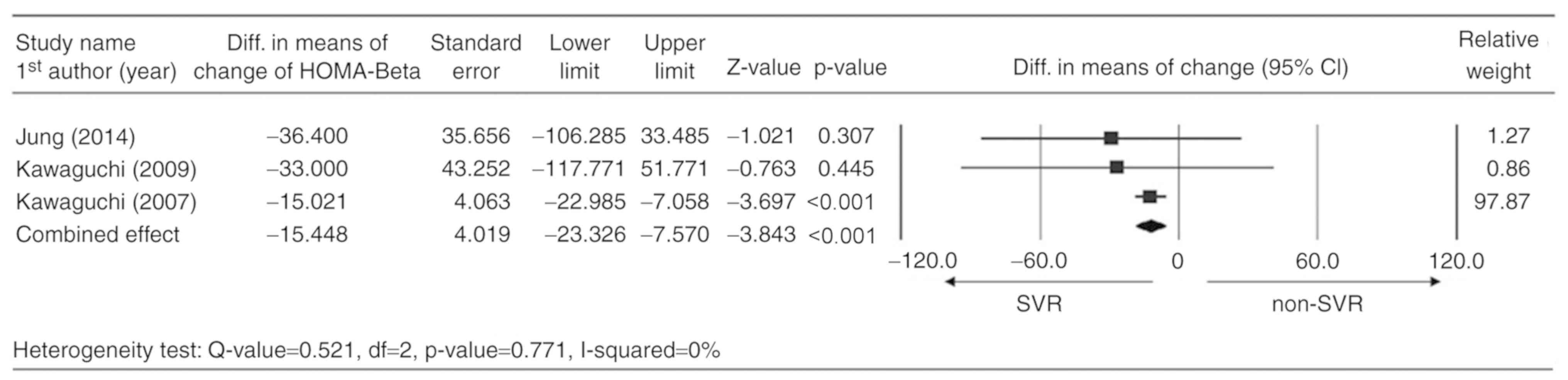
Reduction in mean HOMA-β
Only three studies reported the complete data for
the change in HOMA-β scores between the basal value and the value
after EOT and were considered for meta-analysis (20,32,35). A
fixed-effects model was used according to the heterogeneity test
(Q-value=0.521; df=2; P=0.771; I2=0%). The combined
analysis indicated that the SVR group had a significantly higher
mean reduction in HOMA-β scores compared with that in the non-SVR
group (difference in means=−15.448; 95%CI=−23.326 to −7.570;
P<0.001; Fig. 4). Table SI summarizes the results of a
subgroup analysis for genotypes 1/2. The results suggested that the
SVR group had a higher mean reduction in HOMA-β scores than the
non-SVR group (difference in means=−15.179; P<0.001). However,
the change in HOMA-β scores was similar between the SVR and non-SVR
groups after excluding the study by Kawaguchi et al
(20) (difference in means=−33.0;
P=0.445; Table SI).
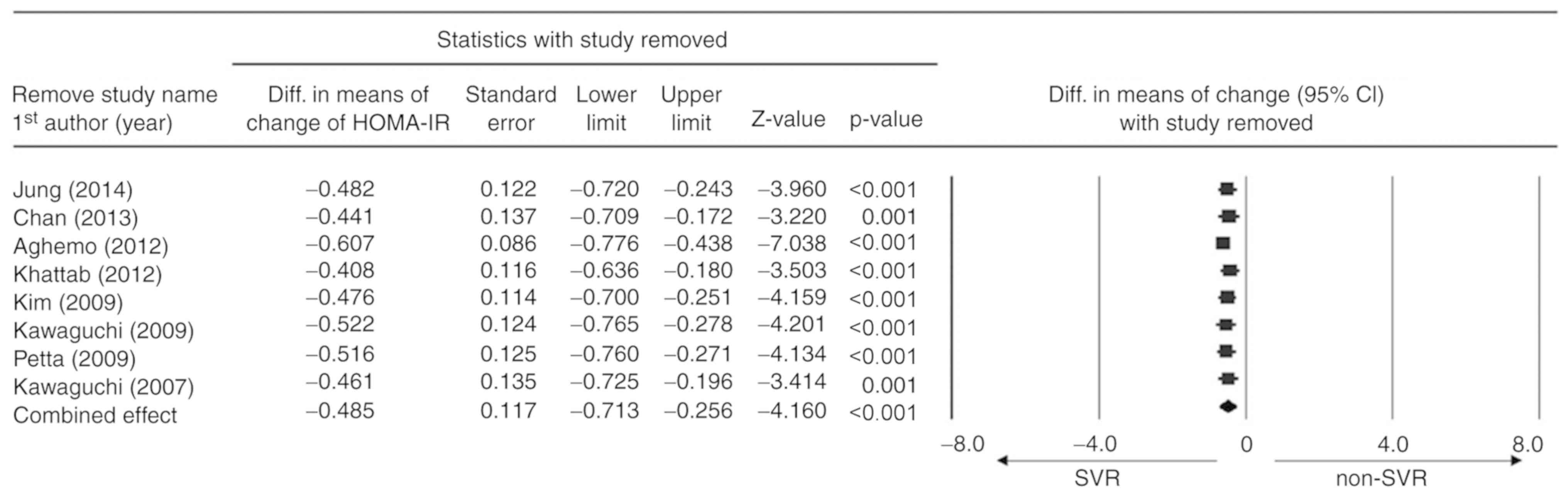
Sensitivity analysis
Sensitivity analyses were performed for HOMA-IR
reduction using a leave-one-out approach (Fig. 5). There was no significant difference
in the direction and magnitude of the combined estimates with the
removal of any one study, indicating that the meta-analysis had
good reliability and that the data were not overly influenced by
any given study.
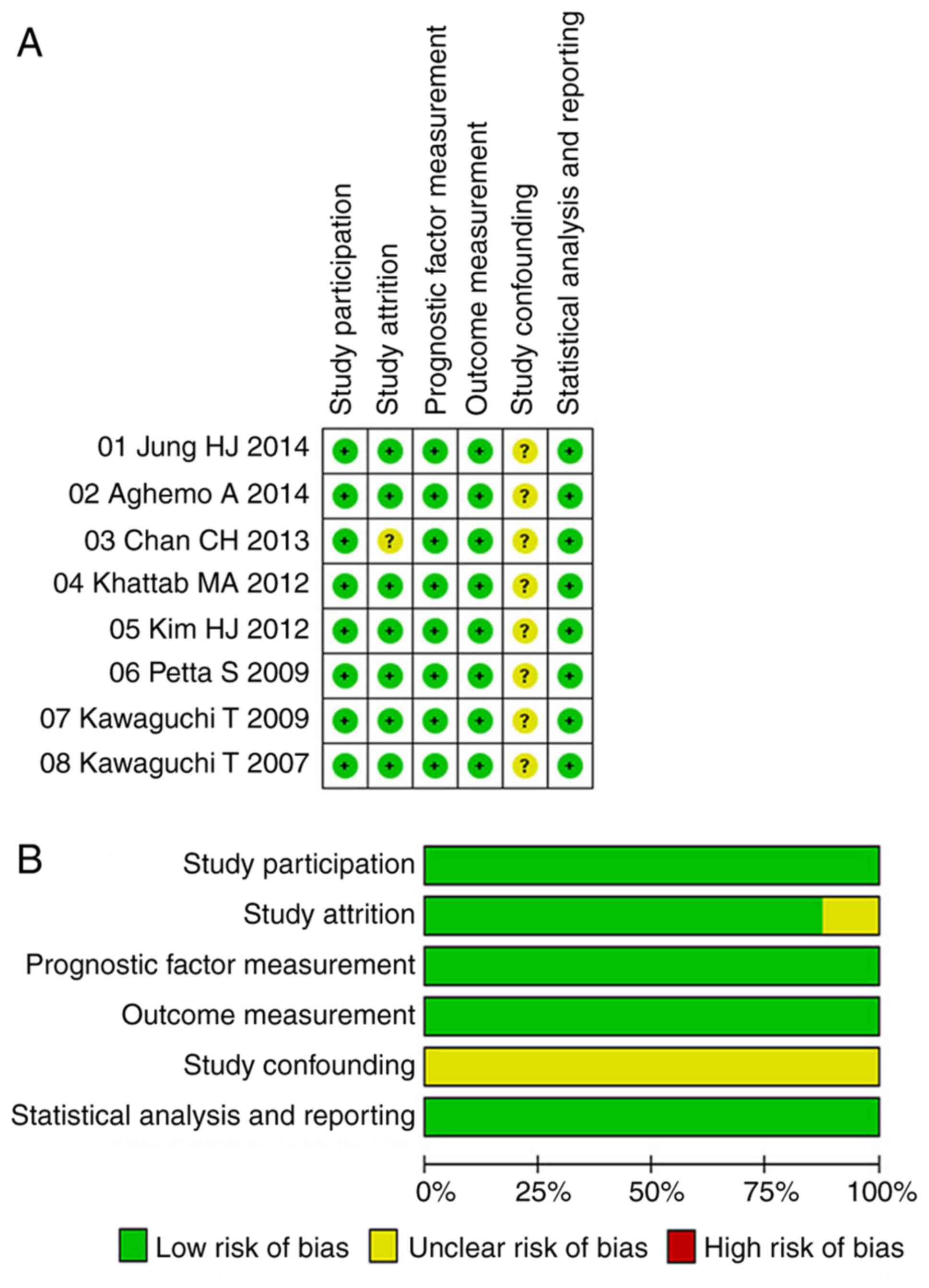
Quality assessment
In general, the quality of the analysis was good,
with a low risk of bias in terms of study participation, study
attrition, measurement of prognostic factors, measurement of
outcomes and analytic approaches (Fig.
6). The risk of bias regarding confounding was unclear in all
included studies.
Discussion
The present meta-analysis evaluated data from 8
studies comparing the reduction in IR between SVR and non-SVR
groups of HCV patients treated with IFN-based therapy. The pooled
analysis indicated no significant difference in the reduction of IR
between the SVR and non-SVR groups. However, patients who achieved
SVR had a significantly higher mean reduction in HOMA-IR and HOMA-β
scores compared to patients in the non-SVR group.
The HOMA-IR score has been indicated to be a
predictor of liver fibrosis and may be used as a surrogate marker
for SVR at EOT (33). A previous
meta-analysis of 13 studies reported that elevated HOMA-IR
(regardless of cut-off value, as well as with the cut-off value set
at >2) was associated with lower SVR rates after IFN-based
therapy in patients infected with HCV of the genotype 1, 2 or 3
(36). Another study indicated that
SVR was associated with a reduction in the mean HOMA-IR in patients
with genotype 1 of HCV, but not in those with genotypes 2 or 3
(25). In addition, HOMA-IR
continued to decrease at the end of the follow-up in patients with
genotype 4 HCV who achieved SVR, but remained unchanged in non-SVR
patients (34). In the present
study, a subgroup analysis was only performed for genotypes 1/2.
The mean reduction of HOMA-IR and HOMA-β scores was higher in
patients who achieved SVR. However, the results became
insignificant after removing the study by Kawaguchi et al
(20) from 2007. To further confirm
and clarify the present results, further studies are required. A
more complete understanding of the role of the genotype of HCV in
the interaction between IR and SVR is needed, since the genotype
impacts SVR rates (37).
The baseline HOMA-IR score is considered an
important determinant of viral response, since it was demonstrated
to be lower in responders than in non-responders (16,32). HCV
patients who failed to achieve SVR exhibited an increase in mean
post-treatment HOMA-IR scores compared to the baseline value
(19). In addition, the mean
pre-treatment HOMA-IR score was indicated to be similar in the SVR
and non-SVR groups, although the HOMA-IR scores significantly
improved at the EOT only in the SVR group (33). It was recently reported that viral
clearance improved insulin sensitivity in CHC patients with
baseline IR, while it increased the HOMA-IR in patients without
baseline IR (38). One explanation
may be that baseline IR is due to HCV infection, which is known to
impair the host's glucose metabolism (39).
Of note, although a recent meta-analysis of 17
studies concluded that IR was associated with a 3-fold higher risk
of failure to achieve SVR, regardless of viral genotype, certain
studies included failed to demonstrate an association between IR
and SVR (24). This result was
attributed to a number of factors, including differences between
the studies in calculating and interpreting HOMA-IR scores,
inclusion of easy-to-cure cohorts and the presence of other
variables strongly associated with IR (24). The present meta-analysis indicated no
significant difference in the reduction of IR rates between the SVR
and non-SVR groups, although the SVR group had a significantly
higher mean reduction in HOMA-IR scores compared to the non-SVR
group. It is important to note that only 2 studies included in the
present meta-analysis provided complete data for IR rates (19,26). In
addition, each study used a different HOMA-IR cut-off value. The
present results suggest that it may be necessary to evaluate
baseline IR in order to elucidate the impact of SVR on IR. It may
also be useful to analyze the rates of de-novo IR
development, since it has been previously reported that
non-diabetic CHC patients who achieved SVR after IFN-based therapy
had lower rates of de-novo IR compared to non-SVR patients
(19).
The present meta-analysis included three studies
reporting on the HOMA-β score in the SVR and non-SVR groups
(20,32,35).
These studies all indicated that patients who achieved SVR had a
significant reduction in their HOMA-β values compared with the
non-SVR group. The results of the pooled analysis (combined effect)
were consistent with the individual analysis results of the
individual studies. This suggested that HOMA-β, a measure of
insulin secretion in the pancreas, had a significantly higher mean
reduction after EOT vs. baseline in the SVR group compared with
that in the non-SVR group. These results further support that SVR
is not merely a surrogate marker of therapeutic efficacy, but an
actual aim to pursue in most HCV-infected patients. The present
results also suggest that clinical management of IR and serum
glucose levels may be an important way to impact the therapeutic
response in HCV patients. Furthermore, L-arginine supplementation
was reported to improve insulin sensitivity in rats fed a high-fat
diet, and might be beneficial in the treatment of HCV patients in
the future (40).
The present study has several strengths and
limitations. Of note, it is the first systematic review and
meta-analysis to indicate that HCV patients who achieved SVR after
IFN therapy had improved HOMA-IR and HOMA-β scores. Furthermore,
the present study followed the PRISMA guidelines, which enables
transparent and complete reporting (29). Regarding the limitations, only one
RCT was included. In addition, the study did not include any
subgroup analysis for HCV genotypes 3/4 or different HOMA-IR
cut-off scores. Furthermore, other factors correlated with IR, e.g.
insulin-like growth factor-1, were not evaluated (41). Finally, the number of studies
analyzing IR rates and HOMA-β was small.
In conclusion, the present study revealed that HCV
patients who achieved SVR after IFN-based therapy exhibited
improvement in HOMA-IR and HOMA-β scores. However, since no
reduction in IR rates was determined, it is important to interpret
the results with caution. Further studies are required to evaluate
the complex association between SVR and IR.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets analyzed during the present study are
available from the corresponding author on reasonable request.
Authors' contributions
JHH made substantial contributions to research
concept, screening process, identification of eligible studies and
manuscript preparation. MLC performed data analysis and prepared
the manuscript. NJL screened, identified eligible studies, and
analyzed the data. CTY carried out data extraction and data quality
evaluation. TJH conceived the research study and supervised the
other authors to ensure integrity of the analysis. All authors
reviewed the manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
CHC
|
chronic hepatitis C
|
|
CI
|
confidence interval
|
|
DAA
|
direct-acting antiviral
|
|
EOT
|
end of treatment
|
|
HCV
|
hepatitis C virus
|
|
HOMA
|
homeostasis model assessment
|
|
IR
|
insulin resistance
|
|
RCT
|
randomized controlled trial
|
|
SD
|
standard deviation
|
|
SVR
|
sustained virological response
|
|
T2DM
|
type 2 diabetes mellitus
|
References
|
1
|
Lavanchy D: The global burden of Hepatitis
C. Liver Int. 29 (Suppl 1):S74–S81. 2009. View Article : Google Scholar
|
|
2
|
Axley P, Ahmed Z, Ravi S and Singal AK:
Hepatitis C virus and hepatocellular carcinoma: A narrative review.
J Clin Transl Hepatol. 6:79–84. 2018.PubMed/NCBI
|
|
3
|
Pawlotsky JM: New Hepatitis C therapies:
The toolbox, strategies, and challenges. Gastroenterology.
146:1176–1192. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Crouchet E, Wrensch F, Schuster C, Zeisel
MB and Baumert TF: Host-targeting therapies for Hepatitis C virus
infection: Current developments and future applications. Therap Adv
Gastroenterol. 11:17562848187594832018. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Averhoff FM, Glass N and Holtzman D:
Global burden of Hepatitis C: Considerations for healthcare
providers in the United States. Clin Infect Dis. 55 (Suppl
1):S10–S15. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Dhingra A, Kapoor S and Alqahtani SA:
Recent advances in the treatment of Hepatitis C. Discov Med.
18:203–208. 2014.PubMed/NCBI
|
|
7
|
Zignego AL, Ferri C, Pileri SA, Caini P
and Bianchi FB; Italian Association of the study of liver
commission on Extrahepatic manifestations of HCV infection, :
Extrahepatic manifestations of Hepatitis C Virus infection: A
general overview and guidelines for a clinical approach. Dig Liver
Dis. 39:2–17. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Cacoub P, Gragnani L, Comarmond C and
Zignego AL: Extrahepatic manifestations of chronic Hepatitis C
virus infection. Dig Liver Dis. 46 (Suppl 5):S165–S173. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
White DL, Ratziu V and El-Serag HB:
Hepatitis C infection and risk of diabetes: A systematic review and
meta-analysis. J Hepatol. 49:831–844. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nadolnik K, Skrypnik D, Skrypnik K and
Bogdański P: Diabetic nephropathy in the elderly-clinical practice.
Rocz Panstw Zakl Hig. 69:327–334. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Moucari R, Asselah T, Cazals-Hatem D,
Voitot H, Boyer N, Ripault MP, Sobesky R, Martinot-Peignoux M,
Maylin S, Nicolas-Chanoine MH, et al: Insulin resistance in chronic
Hepatitis C: Association with genotypes 1 and 4, serum HCV RNA
level, and liver fibrosis. Gastroenterology. 134:416–423. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang CS, Wang ST, Yao WJ, Chang TT and
Chou P: Hepatitis C virus infection and the development of type 2
diabetes in a community-based longitudinal study. Am J Epidemiol.
166:196–203. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kralj D, Virović Jukić L, Stojsavljević S,
Duvnjak M, Smolić M and Čurčić IB: Hepatitis C virus, insulin
resistance, and steatosis. J Clin Transl Hepatol. 4:66–75. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
El-Zayadi AR and Anis M: Hepatitis C virus
induced insulin resistance impairs response to anti viral therapy.
World J Gastroenterol. 18:212–224. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Petta S, Cammà C, Di Marco V, Alessi N,
Cabibi D, Caldarella R, Licata A, Massenti F, Tarantino G,
Marchesini G and Craxì A: Insulin resistance and diabetes increase
fibrosis in the liver of patients with genotype 1 HCV infection. Am
J Gastroenterol. 103:1136–1144. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Deltenre P, Louvet A, Lemoine M, Mourad A,
Fartoux L, Moreno C, Henrion J, Mathurin P and Serfaty L: Impact of
insulin resistance on sustained response in HCV patients treated
with pegylated interferon and ribavirin: A meta-analysis. J
Hepatol. 55:1187–1194. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Guo X, Jin M, Yang M, Liu K and Li JW:
Type 2 diabetes mellitus and the risk of hepatitis C virus
infection: A systematic review. Sci Rep. 3:29812013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sheikh MY, Choi J, Qadri I, Friedman JE
and Sanyal AJ: Hepatitis C virus infection: Molecular pathways to
metabolic syndrome. Hepatology. 47:2127–2133. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Aghemo A, Prati GM, Rumi MG, Soffredini R,
D'Ambrosio R, Orsi E, De Nicola S, Degasperi E, Grancini V and
Colombo M: Sustained virological response prevents the development
of insulin resistance in patients with chronic hepatitis C.
Hepatology. 56:1681–1687. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kawaguchi T, Ide T, Taniguchi E, Hirano E,
Itou M, Sumie S, Nagao Y, Yanagimoto C, Hanada S, Koga H and Sata
M: Clearance of HCV improves insulin resistance, beta-cell
function, and hepatic expression of insulin receptor substrate 1
and 2. Am J Gastroenterol. 102:570–576. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Kawaguchi Y and Mizuta T: Interaction
between hepatitis C virus and metabolic factors. World J
Gastroenterol. 20:2888–2901. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Kim HJ, Park JH, Park DI, Cho YK, Sohn CI,
Jeon WK and Kim BI: Clearance of HCV by combination therapy of
pegylated interferon alpha-2a and ribavirin improves insulin
resistance. Gut Liver. 3:108–115. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Delgado-Borrego A, Jordan SH, Negre B,
Healey D, Lin W, Kamegaya Y, Christofi M, Ludwig DA, Lok AS and
Chung RT; Halt-C Trial Group, : Reduction of insulin resistance
with effective clearance of hepatitis C infection: Results from the
HALT-C trial. Clin Gastroenterol Hepatol. 8:458–462. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Eslam M, Aparcero R, Kawaguchi T, Del
Campo JA, Sata M, Khattab MA and Romero-Gomez M: Meta-analysis:
Insulin resistance and sustained virological response in hepatitis
C. Aliment Pharmacol Ther. 34:297–305. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Thompson AJ, Patel K, Chuang WL, Lawitz
EJ, Rodriguez-Torres M, Rustgi VK, Flisiak R, Pianko S, Diago M,
Arora S, et al: Viral clearance is associated with improved insulin
resistance in genotype 1 chronic hepatitis C but not genotype 2/3.
Gut. 61:128–134. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Petta S, Cammà C, Di Marco V, Cabibi D,
Ciminnisi S, Caldarella R, Licata A, Massenti MF, Marchesini G and
Craxì A: Time course of insulin resistance during antiviral therapy
in non-diabetic, non-cirrhotic patients with genotype 1 HCV
infection. Antivir Ther. 14:631–639. 2009.PubMed/NCBI
|
|
27
|
Cacoub P, Desbois AC, Comarmond C and
Saadoun D: Impact of sustained virological response on the
extrahepatic manifestations of chronic hepatitis C: A
meta-analysis. Gut. 67:2025–2034. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Ciancio A, Bosio R, Bo S, Pellegrini M,
Sacco M, Vogliotti E, Fassio G, Bianco Mauthe Degerfeld AGF, Gallo
M, Giordanino C, et al: Significant improvement of glycemic control
in diabetic patients with HCV infection responding to direct-acting
antiviral agents. J Med Virol. 90:320–327. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Liberati A, Altman DG, Tetzlaff J, Mulrow
C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J
and Moher D: The PRISMA statement for reporting systematic reviews
and meta-analyses of studies that evaluate health care
interventions: Explanation and elaboration. J Clin Epidemiol.
62:e1–e34. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Sterne JA, Sutton AJ, Ioannidis JP, Terrin
N, Jones DR, Lau J, Carpenter J, Rucker G, Harbord RM, Schmid CH,
et al: Recommendations for examining and interpreting funnel plot
asymmetry in meta-analyses of randomised controlled trials. BMJ.
343:d40022011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hayden JA, van der Windt DA, Cartwright
JL, Côté P and Bombardier C: Assessing bias in studies of
prognostic factors. Ann Intern Med. 158:280–286. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Jung HJ, Kim YS, Kim SG, Lee YN, Jeong SW,
Jang JY, Lee SH, Kim HS and Kim BS: The impact of pegylated
interferon and ribavirin combination treatment on lipid metabolism
and insulin resistance in chronic hepatitis C patients. Clin Mol
Hepatol. 20:38–46. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Chan CH, Hansen RD, Gilliver RS and Jones
BE: Sustained virological response following chronic hepatitis C
treatment is associated with improvement in insulin resistance.
Intern Med J. 43:656–662. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Khattab MA, Eslam M, Shatat M,
Abd-Aalhalim H, Mousa YI, Samir F, Aly H, Shaker O and Shaker Y:
Changes in adipocytokines and insulin sensitivity during and after
antiviral therapy for hepatitis C genotype 4. J Gastrointestin
Liver Dis. 21:59–65. 2012.PubMed/NCBI
|
|
35
|
Kawaguchi Y, Mizuta T, Oza N, Takahashi H,
Ario K, Yoshimura T, Eguchi Y, Ozaki I, Hisatomi A and Fujimoto K:
Eradication of hepatitis C virus by interferon improves whole-body
insulin resistance and hyperinsulinaemia in patients with chronic
hepatitis C. Liver Int. 29:871–877. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Laurito MP and Parise ER: Association
between insulin resistance and sustained virologic response in
hepatitis C treatment, genotypes 1 versus 2 and 3: Systematic
literature review and meta-analysis. Braz J Infect Dis. 17:555–563.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Fried MW, Jensen DM, Rodriguez-Torres M,
Nyberg LM, Di Bisceglie AM, Morgan TR, Pockros PJ, Lin A, Cupelli
L, Duff F, et al: Improved outcomes in patients with hepatitis C
with difficult-to-treat characteristics: Randomized study of higher
doses of peginterferon alpha-2a and ribavirin. Hepatology.
48:1033–1043. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chang ML, Kuo CJ, Pao LH, Hsu CM and Chiu
CT: The evolving relationship between adiponectin and insulin
sensitivity in hepatitis C patients during viral clearance.
Virulence. 8:1255–1264. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Chang ML: Metabolic alterations and
hepatitis C: From bench to bedside. World J Gastroenterol.
22:1461–1476. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Miczke A, Suliburska J, Pupek-Musialik D,
Ostrowska L, Jabłecka A, Krejpcio Z, Skrypnik D and Bogdański P:
Effect of L-arginine supplementation on insulin resistance and
serum adiponectin concentration in rats with fat diet. Int J Clin
Exp Med. 8:10358–10366. 2015.PubMed/NCBI
|
|
41
|
Kujawska-Luczak M, Szulinska M, Skrypnik
D, Musialik K, Swora-Cwynar E, Kregielska-Narozna M, Markuszewski
L, Grzymislawska M and Bogdanski P: The influence of orlistat,
metformin and diet on serum levels of insulin-like growth factor-1
in obese women with and without insulin resistance. J Physiol
Pharmacol. 69:2018.
|