Introduction
Acute ischemic stroke (AIS) is the leading cause of
death in China and one of the primary mortality-associated factors
worldwide (1). At present,
intravenous thrombolysis and endovascular treatment (EVT) are the
only methods available for the treatment of AIS. As intravenous
thrombolysis must be administered within a strict 4.5-h time-window
and has a relatively low recanalization rate, it is only beneficial
in a proportion of patients (2).
However, EVT has a longer time-window that was extended to 24 h in
2018 (3), and has markedly improved
the application scope of EVT. Once contraindications have been
excluded, the majority of AIS patients may be subjected to EVT
within the 24-h time frame (3).
However, despite advances in surgical instruments in the last 10
years, the overall probability of post-operative complications and
ineffectiveness of treatment for EVT remains relatively high
(4,5). Therefore, there is an urgent
requirement for novel predictive indicators of patient outcome
following EVT for AIS.
A number of biochemical, clinical and imaging
factors have been identified to be associated with the prognosis of
patients with AIS. In terms of biochemical factors, the levels of
glucose on admission (AG), high-sensitivity C-reactive protein
(hs-CRP) and N-terminal pro-brain natriuretic peptide (NT-proBNP)
on admission all associated with the outcome of patients with AIS
(6–8). However, these biochemical indicators
reflect only a limited number of the aspects associated with AIS
(and are not used in combination with symptomatic and medical
imaging data); thus, their predictive accuracy is relatively
low.
Of the imaging characteristics, diffusion-weighted
imaging (DWI) lesion volume, cerebral blood flow, cerebral blood
volume and mean transit time have been associated with patient
outcome (9–11). Due to the requirement for prompt
recanalization and the lack of medical resources in certain
locations, most patients do not undergo a pre-operative perfusion
scan and only receive a conventional magnetic resonance imaging
(MRI) sequence scan, which has limited the popularization of most
MRI modal features. In terms of clinical factors, the National
Institutes of Health Stroke Scale (NIHSS) score, modified Rankin
Scale (mRS) score, age and onset-to-reperfusion time (ORT) have
been indicated to be associated with the outcome of patients with
AIS (12–14). Clinical factors also only reflect the
functional state of the central nervous system and partially
reflect the mechanisms of AIS; therefore, on their own, they are
unable to accurately predict the outcome of patients with AIS.
At present, the number of studies predicting the
outcome of EVT for AIS is relatively low, and the outcomes for
patients who received EVT have only been predicted using single
factors belonging to a single category; they have not been
comprehensively predicted using a combination of biochemical,
imaging features and clinical indicators. The present study aimed
to use widely recognized prognostic indictors, including
biochemical, imaging and clinical factors to predict the outcome of
EVT, and to provide a reference for the establishment of
personalized treatment plans.
Patients and methods
Patients
A retrospective analysis was performed using the
clinical and imaging data of 169 patients with anterior circulation
acute ischemic stroke (ACAIS) who received EVT between October 2014
and August 2018 at the Second Clinical College of Guangzhou
University of Chinese Medicine (Guangzhou, China). All patients
accepted mechanical thrombectomy using a solitaire stent retriever;
if the vessel remained narrow or the distal circulation remained
poor following thrombectomy, angioplasty was subsequently
performed. A total of 10 patients with malignant tumors or
neurological diseases were excluded, 12 patients were excluded due
to a history of stroke, 6 due to having received unknown
pre-treatment at another hospital and 23 patients dropped out
during the follow-up period. Ultimately, 118 patients were included
in the present study. The clinicopathological and demographic data
of all patients were obtained from the hospital information
management system and image examination center, and included age,
sex, risk factors, DWI images, AG, hs-CRP, high-density lipoprotein
(HDL), low-density lipoprotein (LDL) and total cholesterol (TC)
levels, in addition to systolic blood pressure (SBP), diastolic
blood pressure (DBP), ORT, NIHSS score on admission and clinical
outcome (Tables I and II). The patients were followed up for
three months following EVT, and the NIHSS and mRS scores were used
to assess each patient's state on admission and three months after
EVT. The NIHSS and mRS scores were independently evaluated by two
experienced neurosurgeons; discrepancies were resolved through
discussion or determined by more senior neurosurgeons.
 | Table I.Demographic and baseline data of the
patients (categorical variables). |
Table I.
Demographic and baseline data of the
patients (categorical variables).
| Item | Poor
outcomea | Favorable
outcomea | Sum | χ2 | P-value |
|---|
| Sex |
|
|
|
|
|
|
Males | 37 (52.1) | 35 (74.5) | 72 | 5.94 | 0.02 |
|
Females | 34 (47.9) | 12 (25.5) | 46 |
|
|
| Smoking |
|
|
|
|
|
|
Yes | 20 (28.2) | 19 (40.4) | 39 | 1.92 | 0.23 |
| No | 51 (71.8) | 28 (59.6) | 79 |
|
|
| Drinking |
|
|
|
|
|
|
Yes | 7 (9.9) | 9
(19.1) | 16 | 2.08 | 0.18 |
| No | 64 (90.1) | 38 (80.9) | 102 |
|
|
| Hypertension |
|
|
|
|
|
|
Yes | 45 (63.4) | 29 (61.7) | 74 | 0.03 | 1.00 |
| No | 26 (36.6) | 18 (38.3) | 44 |
|
|
| Diabetes |
|
|
|
|
|
|
Yes | 18 (25.4) | 7
(14.9) | 25 | 1.85 | 0.25 |
| No | 53 (74.6) | 40 (85.1) | 93 |
|
|
 | Table II.Patient data in association with the
outcome. |
Table II.
Patient data in association with the
outcome.
|
| Poor outcome
(n=71)a | Favorable outcome
(n=47)a |
|
|
|---|
|
|
|
|
|
|
|---|
| Item | Mean (SD) | Median
(P75-P25) | Mean (SD) | Median
(P75-P25) | t/Z | P-value |
|---|
| Age (years) | 70.5 (10.2) | 72 (14) | 63.49 (11.21) | 62.0 (16.0) | 3.53 | <0.01 |
| DWI lesion volume
(mm3) | 30,914.6
(41489.7) | 12,921.9
(34015.8) | 7,724.8
(7606.2) | 5,840.3
(6852.0) | 3.60 |
<0.01b |
| AG (mmol/l) | 9.6 (4.6) | 8.1 (3.2) | 7.1 (1.9) | 6.6 (2.3) | 3.93 |
<0.01b |
| NIHSS score | 15.0 (5.8) | 14.0 (7.0) | 9.4 (3.4) | 9.0 (5.0) | 5.40 |
<0.01b |
| ORT (min) | 376.4 (232.2) | 300.0 (225.0) | 301.4 (141.4) | 270.0 (177.0) | 1.61 |
0.11b |
| hs-CRP (mg/l) | 52.0 (36.3) | 45.2 (32.0) | 39.0 (25.8) | 33.1 (33.5) | 2.13 |
0.03b |
| Operation time
(min) | 142.1 (56.4) | 135 (80) | 133.6 (56.3) | 130 (46) | 0.82 |
0.41b |
| SBP (mmHg) | 152.4 (25.1) | 152 (34) | 153.7 (24.9) | 149.0 (31.0) | 0.07 |
0.94b |
| DBP (mmHg) | 84.9 (15.0) | 81 (25) | 88.8 (15.7) | 85 (21) | 1.33 |
0.18b |
| HDL (mmol/l) | 1.2 (0.3) | 1.1 (0.2) | 1.1 (0.3) | 1.1 (0.3) | 1.04 |
0.30b |
| LDL (mmol/l) | 2.9 (1.0) | 2.8 (1.2) | 2.9 (0.7) | 2.9 (1.1) | 0.26 |
0.79b |
| TC (mmol/l) | 4.6 (1.1) | 4.4 (1.5) | 4.3 (0.8) | 4.2 (1.1) | 1.31 |
0.19 |
All patients met the following inclusion criteria:
i) Diagnosed with AIS according to the 2018 Guidelines for the
Early Management of Patients with Acute Ischemic Stroke (15); ii) for patients with ACAIS, the
occlusions were located in the intracranial internal carotid or
middle cerebral artery; iii) an ORT at <24 h; and iv) an imaging
examination performed no more than 1.5 h prior to surgery. The
exclusion criteria included the following: i) The occurrence of
intracranial diseases, including tumors, infection or any
neurological or psychiatric disorders that may have affected
neurological function; ii) an NIHSS score <7 on admission; and
iii) patients with posterior circulation AIS. The present study was
approved by the Ethics Committee of The Second Clinical College of
Guangzhou University of Chinese Medicine (Guangzhou, China).
Clinical evaluation
The baseline assessments were performed within 1.5 h
of admission and included evaluations of the NIHSS score, AG, blood
pressure, demographic data, history of smoking or drinking and
history of diabetes mellitus or hypertension. A history of smoking
and drinking was defined as the patient admitting that they had
previously smoked or consumed alcohol. A history of high blood
pressure was defined as a systolic blood pressure >140 mmHg or
diastolic blood pressure >90 mmHg at any point prior to
admission. Diabetes was defined as a blood glucose level >11.1
mmol/l. A fasting venous blood sample was used to detect blood
lipids and hs-CRP levels were determined in the early morning and
within 24 h of admission. Neurological assessments were performed
by evaluating the NIHSS and mRS scores at admission and during the
follow-up examination (3 months after surgery). The 3-month mRS
score was used to assess patient outcome; a score of 0–2 was
considered as a favorable outcome and a score of 3–6 was considered
to indicate a poor outcome (16,17).
Image acquisition and DWI lesion
volume calculation
MRI was performed using a MAGNETOM Verio 3.0T
scanner (Siemens AG). The MRI protocol included DWI, apparent
diffusion coefficient, T1-weighted imaging, T2-weighted imaging,
fluid-attenuated inversion recovery imaging and MR angiography. The
DWI scan parameters were as follows: B value, 0 and 1,000; echo
time, 100 msec; repetition time, 6,600 msec; phrase encoding
direction, AàP; field of view (FOV) read, 220 mm; FOV phase, 100%;
slice thickness, 5.0 mm; slice spacing, 1.0 mm; slices, 20;
bandwidth, 1,002 Hz/Pixel; echo spacing, 1.08 msec; fat saturation
mode, weak; concatenation, 1; and scan time, 74 sec. Within the DWI
images, the lesions were manually delineated using ImageJ Software
(version 1.8.0; National Institutes of Health) by three associate
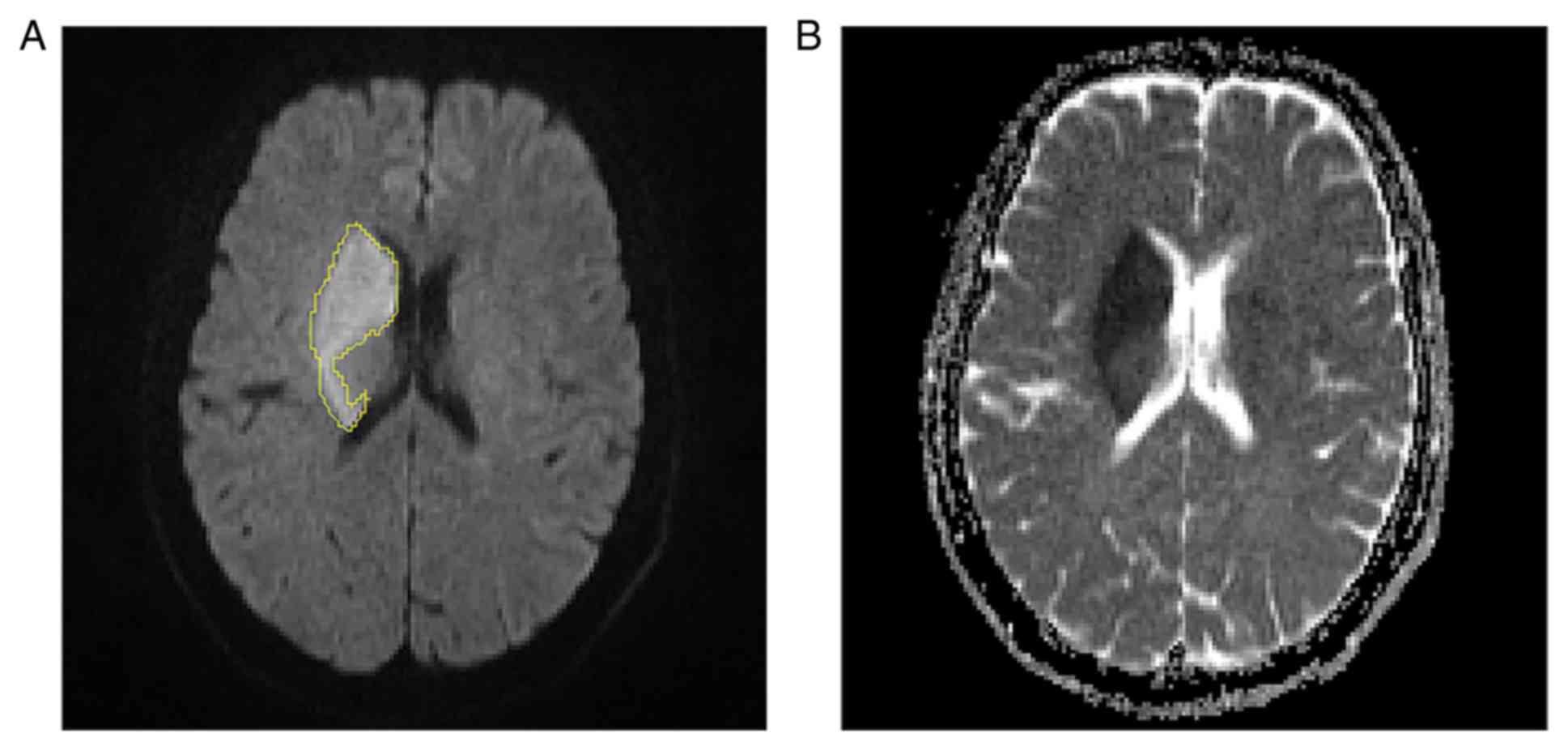
chief physicians (initials, BL, GL and WZ) (Fig. 1); the area occupied by the lesion on
each slice was then measured and the total of these areas was
multiplied by the slice thickness.
Statistical analysis
Continuous or numerical variables were expressed as
the mean ± standard deviation and median (interquartile range).
Shapiro-Wilk test was used to test the normality of variances and
Levene test was used to test the homogeneity of variances. Normally
distributed variables with assumed homogeneity of variance were
compared using the independent-samples t-test, and non-normally
distributed variables or those without homogeneity of variance were
compared using the Mann-Whitney U-test. Categorical variables were
expressed as the composition ratio or rate and comparisons were
performed using Pearson's χ2 or Fisher's exact test.
Non-conditional logistic stepwise regression was used to identify
factors that were significantly associated with patient outcome,
which can establish the optimal statistical model. The specificity
and sensitivity of the threshold value for each predictive factor
were analyzed by receiver operating characteristic (ROC) curve
analysis. Statistical analyses were performed using SPSS 19.0 for
Windows (IBM Corp.) and P<0.05 was considered to indicate a
statistically significant difference.
Results
Predictors of patient outcome at 3
months post-surgery
Patient outcome was predicted using non-conditional
logistic stepwise regression analysis, and the follow-up result at
3 months after EVT was used as the dependent variable (Favorable
outcome, Y=0; Poor outcome, Y=1). The results indicated that the
model was able to effectively predict patient outcome
(χ2=78.916; P<0.001) and that the coincidence rate
(accuracy) of the model was calculated to be 83.1% (Tables III and IV), which was higher compared with that
obtained using only 1 or 2 factors. Factors affecting patient
outcome were sex, age, DWI lesion volume, AG, NIHSS score and ORT.
The results also suggested that female patients exhibited a poorer
outcome compared with male patients (odds ratio=3.39; 95%
confidence interval, 1.022–11.24; Table
V).
 | Table III.Omnibus tests of model
coefficients. |
Table III.
Omnibus tests of model
coefficients.
| Step 6 | χ2 | Degree of
freedom | P-value |
|---|
| Step |
4.134 | 1 |
0.042 |
| Block | 78.916 | 6 | <0.001 |
| Model | 78.916 | 6 | <0.001 |
 | Table IV.Classification Table. |
Table IV.
Classification Table.
|
| Predicted |
|---|
|
|
|
|---|
|
| Groups |
|
|---|
|
|
|
|
|---|
| Observed | Favorable
outcome | Poor outcome | Percentage
correct |
|---|
| Step 6 |
|
|
|
|
Groups |
|
|
|
|
Favorable
outcome | 37 | 10 | 78.7 |
|
Poor outcome | 10 | 61 | 85.9 |
| Overall
percentage |
|
| 83.1 |
 | Table V.The role of each factor for the
prediction of AIS in the non-conditional logistic stepwise
regression modela. |
Table V.
The role of each factor for the
prediction of AIS in the non-conditional logistic stepwise
regression modela.
|
|
|
|
|
|
| 95% EXP (β) |
|---|
|
|
|
|
|
|
|
|
|---|
| Variable | β | S.E. | Wald | P-value | Exp (β) | Lower limit | Upper limit |
|---|
| Sex |
1.221 | 0.611 |
3.984 | 0.046 | 3.389 | 1.022 | 11.235 |
| Age |
0.090 | 0.031 |
8.537 | 0.003 | 1.094 | 1.030 |
1.163 |
| DWI lesion
volume | <0.001 | 0.001 | 11.208 | 0.001 | 1.000 | 1.000 |
1.000 |
| AG |
0.421 | 0.153 |
7.602 | 0.006 | 1.524 | 1.129 |
2.055 |
| NIHSS score |
0.193 | 0.072 |
7.108 | 0.008 | 1.213 | 1.052 |
1.397 |
| ORT |
0.004 | 0.002 |
4.548 | 0.033 | 1.004 | 1.000 |
1.007 |
ROC curve analysis of the predictors
of patient outcome
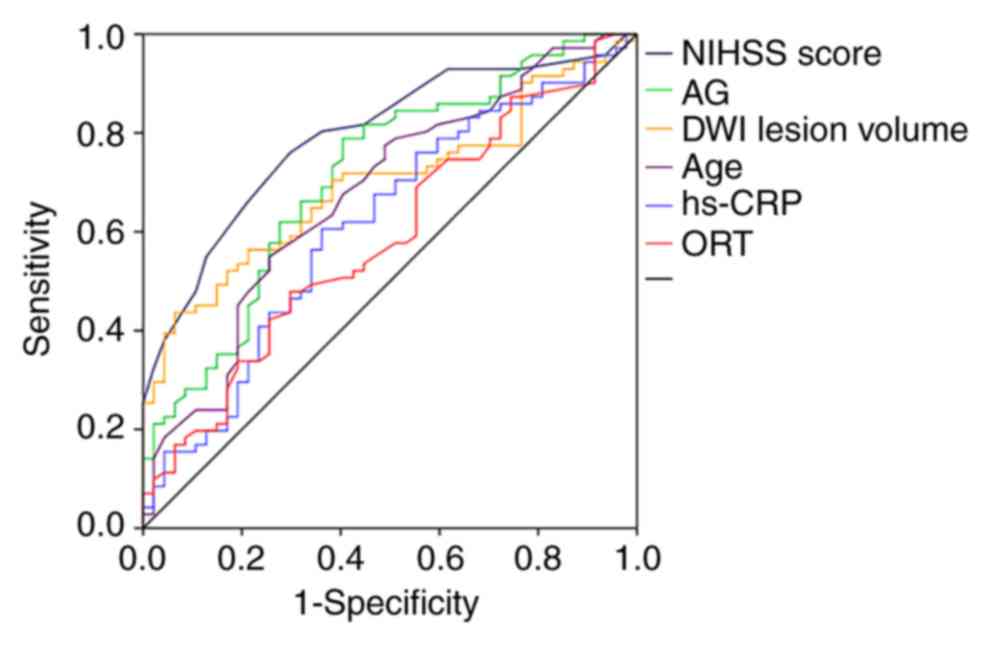
ROC curve analyses showed that the Area Under the
ROC Curve (AUC) of the statistical model was calculated to be 0.823
(Table VI). In addition, age,
lesion volume, AG, NIHSS score on admission and hs-CRP were all
found to be associated with the outcome of patients following EVT
(P<0.05); however, the AUC calculated for each of the
aforementioned single factors was not large (Fig. 2), ranging from 0.616 to 0.794
(Table VII). After taking
specificity and sensitivity into account, the cut-off value of each
factor is presented in Table VII.
The specificity of the significant factors ranged from 60.6 to
76.1%, and the sensitivity ranged from 61.7 to 70.2%. No
significant influence on patient outcome was determined for ORT,
HDL, LDL, TC, SBP and DBP (P>0.05). Regarding the predictive
value of each factor, the order was NIHSS score, AG, lesion volume,
age and hs-CRP.
 | Table VI.Receiver operating characteristic
curve analysis of statistical model for the outcome of patients
with anterior circulation acute ischemic strokea. |
Table VI.
Receiver operating characteristic
curve analysis of statistical model for the outcome of patients
with anterior circulation acute ischemic strokea.
|
|
|
| Asymptotic 95%
CI |
|---|
|
|
|
|
|
|---|
| AUC | S.E. | P-value | Lower limit | Upper limit |
|---|
| 0.823 | 0.042 | <0.001 | 0.740 | 0.906 |
 | Table VII.Receiver operating characteristic
curve analysis of predictive factors for the outcome of patients
with anterior circulation acute ischemic stroke. |
Table VII.
Receiver operating characteristic
curve analysis of predictive factors for the outcome of patients
with anterior circulation acute ischemic stroke.
|
|
|
|
| Asymptotic 95%
CI |
|
|
|---|
|
|
|
|
|
|
|
|
|---|
| Outcome
measure | AUC | Cut-off value | P-value | Lower limit | Upper limit | Specificity
(%) | Sensitivity
(%) |
|---|
| Age | 0.677 | 68.5 |
0.001 | 0.578 | 0.776 | 63.4 | 61.7 |
| DWI lesion
volume | 0.696 | 6494.5 | <0.001 | 0.603 | 0.790 | 70.4 | 61.7 |
| AG | 0.714 | 7.2 | <0.001 | 0.620 | 0.809 | 73.2 | 61.7 |
| NIHSS score | 0.794 | 11.5 | <0.001 | 0.713 | 0.874 | 76.1 | 70.2 |
| hs-CRP | 0.616 | 39.5 |
0.034 | 0.512 | 0.720 | 60.6 | 63.8 |
| ORT | 0.588 | 272.5 |
0.108 | 0.483 | 0.692 | 53.5 | 55.3 |
Discussion
EVT is one of the main methods to treating AIS at
present. In order to determine the treatment plan after EVT,
clinicians need to predict the outcome of patients with AIS who
underwent EVT. However, as the outcome of AIS is under the
influence of a variety of factors, the accuracy of in predicting
the outcome of EVT by using only one or two factors is relatively
low. In the present study, non-conditional logistic stepwise
regression was used to identify the factors significantly
associated with patient outcome, including biochemical, clinical
and imaging parameters, which were then applied to establish an
optimal predictive statistical model. In addition, multivariate
logistic regression model is convenient and stable, and is commonly
applied in many outcome prediction models of some diseases
(18,19). The ROC curve also showed that the
statistical model in the present study exhibited higher AUC and
verified sex, age, DWI lesion volume, AG, hs-CRP and NIHSS score on
admission to be significantly associated with patient outcome.
Previous studies have predicted the outcome of AIS
patients using only one or two factors, particularly the NIHSS
score and age, which have been recognized as the most direct
contributors to patient outcome (20). The NIHSS score indicates the degree
of neurological function damage and is frequently used to determine
the severity of the patient's condition on admission (21). The majority of studies have used the
NIHSS score as an important predictive factor for patient outcome
following AIS; however, the outcome threshold frequently differed
among these studies (21). The
present study also indicated that the NIHSS score on admission was
the strongest factor associated with patient outcome, which was
consistent with the conclusions of previous studies.
Age is a clinical indicator of the aging of cells of
the nervous system. Aging results in molecular damage, organelle
dysfunction and cellular injury within the neurovascular unit,
which leads to structural and functional impairments (22). Furthermore, the acceleration of aging
may contribute to an increased risk of cerebrovascular pathologies
and limit the recovery capacity or neuroplasticity of the nervous
system. It has been indicated that older patients are more prone to
stroke and exhibit more severe post-stroke neurological and
behavioral deficits compared with younger patients (23). The present study supported the
association between age and outcome of patients with AIS. In
previous studies on outcome prediction, age was frequently combined
with other factors to predict patient outcome; a study by Möbius
et al (24) achieved an
accuracy of 73% using the NIH Stroke Scale index, the SPAN-100
index, which relied primarily on age and NIHSS score to predict the
outcome of intravenous thrombolysis following AIS.
Since brain tissues have strict requirements
regarding recanalization time, the outcome of patients with AIS has
been indicated to correlate with the ORT (25). Todo et al (26) revealed that ORT combined with the
NIHSS score was able to predict the outcome of patients with AIS
with an accuracy of 77.1%. The ROC curve in the present study did
not indicate significant correlation between ORT and patient
outcome, which was in line with a study by Koizumi et al
(27), which suggested that the ORT
was not an important outcome predictor of mechanical thrombectomy
in elderly patients. It was speculated that this may have been due
to the short ORT of all patients (within the 24-h time window, with
most patients treated within 12 h).
In addition, the present study indicated that males
had a more favorable outcome compared with females; this supported
the results of a study by White et al (28), which predicted more severe strokes
and poorer outcomes in females. This may have been caused by the
between-sex differences in androgen levels. Indeed, further studies
indicated that decreased testosterone levels were associated with
insulin resistance, blood lipid metabolism and mortality following
cardiovascular disease (29). Kim
and Vemuganti (23) also revealed
that estrogen and progesterone serve important neuroprotective
roles following ischemia, while the estrogen receptor density in
the brain was increased only in males after ischemia, and was kept
unchanged in females. As the age of the majority of female AIS
patients is between middle and advanced age, which is generally
after the menopause and faced the decline of estrogen levels and
estrogen receptor expression, this further explains why males may
have better outcomes compared with females following AIS.
The DWI infarct lesion volume indicates the size of
the area of damaged brain tissue. Numerous studies have indicated
that the DWI infarct lesion volume was associated with the outcome
of patients with AIS and may serve as an independent predictor for
outcome (9,10). In fact, a number of studies suggested
that the efficacy of any treatment in patients with a DWI lesion
volume of >10 cm3 is limited (30,31). A
study by Liggins et al (32)
determined that the predictive accuracy of the DWI infarct volume
and age regarding poor outcome of EVT was 82%. The present study
also indicated a relatively strong correlation between lesion
volume and the outcome of EVT.
Among the biochemical factors used to predict
patient outcome, AG is the most representative. It is generally
accepted that AG represents an independent predictor of patient
outcome following AIS (33).
Abnormalities in the endocrine system caused by stress after a
stroke lead to an increase in cortisol, adrenaline and glucagon
expression levels, which subsequently leads to a rise in blood
glucose levels. Not only does hyperglycemia have a toxic effect on
the nervous system, but it may also inhibit the fibrinolytic system
and further aggravate the patient's condition (34). The present study verified the
correlation between AG and the outcome following EVT. hs-CRP is a
marker of cardiovascular or cerebrovascular inflammatory responses,
which is involved in cerebrovascular injury and pathological
alterations, and indicates the severity of inflammation in brain
tissue (35). hs-CRP may activate
the complement system, increase the production of plasminogen
inhibitors that damage vascular endothelial cells, and ultimately
promote multiple-organ and microcirculatory dysfunction (33). Cai et al (7) revealed that hs-CRP predicted the
outcome of patients with AIS, and in the present study, ROC curve
analysis only indicated a weak correlation between hs-CRP and
patient outcome, with the AUC calculated to be 0.616. In contrast,
non-conditional logistic stepwise regression analysis disregarded
hs-CRP as a predictor of patient outcome, which may have been due
to the non-conditional logistic stepwise regression comprehensively
considered the relationship between multiple variables and
outcomes, whilst ROC curve analysis only consider the association
between hs-CRP and outcome. In addition, ROC analysis also showed
the lesser effect of hs-CRP on the outcome of AIS, since the AUC,
specificity and sensitivity of hs-CRP were relatively lower
compared with other significant factors.
In addition, various studies have indicated that LDL
and blood pressure may be considered to be risk factors for AIS,
which may be associated with the prognosis of AIS (36,37).
However, no previous study has used these factors to directly
predict the outcome of AIS and achieve high accuracy. The present
study did not reveal any significant differences in LDL and blood
pressure between patients with good or poor outcomes. It may be
hypothesized that this is due to these factors not being associated
with the stress reaction after AIS, and that patients may have
received anti-hypertensive and/or hypolipidemic therapy prior to or
after the onset of AIS.
As clinical, imaging and biochemical factors
indicate the severity of AIS from different aspects (and the
abnormalities of these factors may occur at different times), the
accuracy of predicting the outcome of AIS by combining three
different types of factor may be higher than that achieved with
single factors alone. Regarding the prediction of the outcome of
EVT using a combination of clinical, biochemical and imaging
factors, the present study achieved an accuracy of 83.1%, which is
higher than that obtained with any single factor alone (as
determined by ROC curve analysis) in the present and in previous
studies (26,32). Furthermore, unlike the texture
features of the medical image (used in radiomics) that are easily
affected by variations in instrument model or manufacturer, the
factors selected in the present study have universally applicable
standard values, increasing the research practicability. Future
studies will encompass the collection and integration of additional
factors associated with the outcome of patients with AIS and
establish more accurate predictive models for the generation of
more precise auxiliary information.
The present study had several limitations. First,
due to the limitation of the condition, not all of the indicators
associated with patient outcome were investigated (e.g.,
homocysteine and NT-proBNP levels) and abnormal alterations to
these variables may still cause injury to the brain. Investigation
of additional factors in future studies may further enhance the
predictive accuracy. Furthermore, the present study had a
relatively small sample size and was a single-center study lacking
the validation of multicenter results.
In conclusion, a predictive model based on
non-conditional logistic stepwise regression analysis was able to
predict the outcome of EVT following AIS. Patients belonging to
different outcome groups displayed significant differences in sex,
age, DWI lesion volume, hs-CRP, NIHSS score on admission and/or AG.
The present study provided novel insight into the predictive
capacity of these factors for patients with AIS.
Acknowledgements
We would like to acknowledge the valuable
cooperation of The Department of Neurosurgery, The Second Clinical
College of Guangzhou University of Chinese Medicine (Guangzhou,
China) in evaluating patient NIHSS score and mRS as part of their
daily clinical work.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
BL and XW conceived and designed the study. XW, XL,
YW, SZ and WL collected and organized the data. BL, GL and WZ
manually delineated the DWI infarct lesion data. XW and AO analyzed
and interpreted the data, and XW wrote the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of The Second Clinical College of Guangzhou University of
Chinese Medicine (Guangzhou, China). The present study is a
retrospective clinical analysis without any additional intervention
for the patients. The requirement for informed consent was waived
due to the retrospective nature of this analysis.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Glossary
Abbreviations
Abbreviations:
|
ACAIS
|
anterior circulation acute ischemic
stroke
|
|
AG
|
glucose on admission
|
|
AIS
|
acute ischemic stroke
|
|
BNP
|
brain natriuretic peptide
|
|
DWI
|
diffusion-weighted imaging
|
|
EVT
|
endovascular treatment
|
|
HDL
|
high-density lipoprotein
|
|
hs-CRP
|
high-sensitivity C-reactive
protein
|
|
LDL
|
low-density lipoprotein
|
|
mRS
|
modified Rankin Scale
|
|
NIHSS
|
National Institutes of Health Stroke
Scale
|
|
ORT
|
onset-to-reperfusion time
|
|
ROC
|
receiver operating characteristic
|
References
|
1
|
Wufuer A, Wubuli A, Mijiti P, Zhou J,
Tuerxun S, Cai J, Ma J and Zhang X: Impact of collateral
circulation status on favorable outcomes in thrombolysis treatment:
A systematic review and meta-analysis. Exp Ther Med. 15:707–718.
2018.PubMed/NCBI
|
|
2
|
Hacke W: Interventional thrombectomy for
major stroke-a step in the right direction. N Engl J Med.
372:76–77. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Nogueira RG, Jadhav AP, Haussen DC, Bonafe
A, Budzik RF, Bhuva P, Yavagal DR, Ribo M, Cognard C, Hanel RA, et
al: Thrombectomy 6 to 24 h after stroke with a mismatch between
deficit and infarct. N Engl J Med. 378:11–21. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bishop BM: Endovascular interventions for
acute ischemic stroke: A review of recent trials. Ann Pharmacother.
50:219–228. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Pfaff J, Rohde S, Engelhorn T, Doerfler A,
Bendszus M and Möhlenbruch MA: Mechanical thrombectomy using the
new solitaire™ platinum stent-retriever: Reperfusion results,
complication rates and early neurological outcome. Clin
Neuroradiol. 29:311–319. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang L, Zhou Z, Tian X, Wang H, Yang D,
Hao Y, Shi Z, Lin M, Wang Z, Zheng D, et al: Impact of relative
blood glucose changes on mortality risk of patient with acute
ischemic stroke and treated with mechanical thrombectomy. J Stroke
Cerebrovasc Dis. 28:213–219. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cai Z, He W, Zhuang FJ and Chen Y: The
role of high high-sensitivity C-reactive protein levels at
admission on poor prognosis after acute ischemic stroke. Int J
Neurosci. 129:423–429. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bunevicius A, Kazlauskas H, Raskauskiene
N, Mickuviene N, Ndreu R, Corsano E and Bunevicius R: Role of
N-terminal pro-B-type natriuretic peptide, high-sensitivity
C-reactive protein, and inteleukin-6 in predicting a poor outcome
after a stroke. Neuroimmunomodulation. 22:365–372. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Han M, Choi JW, Rim NJ, Kim SY, Suh HI,
Lee KS, Hong JM and Lee JS: Cerebral infarct volume measurements to
improve patient selection for endovascular treatment. Medicine
(Baltimore). 95:e47022016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Padroni M, Bernardoni A, Tamborino C,
Roversi G, Borrelli M, Saletti A, De Vito A, Azzini C, Borgatti L,
Marcello O, et al: Cerebral blood volume aspects is the best
predictor of clinical outcome in acute ischemic stroke: A
retrospective, combined semi-quantitative and quantitative
assessment. PLoS One. 11:e01479102016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Hirano T: Searching for salvageable brain:
The detection of ischemic penumbra using various imaging
modalities? J Stroke Cerebrovasc Dis. 23:795–798. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Jeong HG, Kim BJ, Choi JC, Hong KS, Yang
MH, Jung C, Han MK and Bae HJ: Posttreatment national institutes of
health stroke scale is superior to the initial score or
thrombolysis in cerebral ischemia for 3-month outcome. Stroke.
49:938–944. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kim DH, Kim B, Jung C, Nam HS, Lee JS, Kim
JW, Lee WJ, Seo WK, Heo JH, Baik SK, et al: Consensus statements by
Korean society of interventional neuroradiology and Korean stroke
society: Hyperacute endovascular treatment workflow to reduce
door-to-reperfusion time. Korean J Radiol. 19:838–848. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Beumer D, Rozeman AD, Lycklama À, Nijeholt
GJ, Brouwer PA, Jenniskens SF, Algra A, Boiten J, Schonewille W,
van Oostenbrugge RJ, Dippel DW, et al: The effect of age on outcome
after intra-arterial treatment in acute ischemic stroke: A MR CLEAN
pretrial study. BMC Neurol. 16:682016. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Powers WJ, Rabinstein AA, Ackerson T,
Adeoye OM, Bambakidis NC, Becker K, Biller J, Brown M, Demaerschalk
BM, Hoh B, et al: 2018 Guidelines for the Early management of
patients with acute ischemic stroke: A guideline for healthcare
professionals from the American Heart Association/American Stroke
Association. Stroke. 49:e46–e110. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Peng JW, Liu Y, Meng G, Zhang JY and Yu
LF: Effects of salvianolic acid on cerebral perfusion in patients
after acute stroke: A single-center randomized controlled trial.
Exp Ther Med. 16:2600–2614. 2018.PubMed/NCBI
|
|
17
|
Luo G, Mo D, Tong X, Liebeskind DS, Song
L, Ma N, Gao F, Sun X, Zhang X, Wang B, et al: Factors associated
with 90-day outcomes of patients with acute posterior circulation
stroke treated by mechanical thrombectomy. World Neurosurg.
109:e318–e328. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Stoltzfus JC: Logistic regression: A brief
primer. Acad Emerg Med. 18:1099–1104. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Meurer WJ and Tolles J: Logistic
regression diagnostics: Understanding how well a model predicts
outcomes. JAMA. 317:1068–1069. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Amitrano D, Silva IR, Liberato BB,
Batistella V, Oliveira J and Nascimento OJ: Simple prediction model
for unfavorable outcome in ischemic stroke after intravenous
thrombolytic therapy. Arq Neuropsiquiatr. 74:986–989. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wu Z, Zeng M, Li C, Qiu H, Feng H, Xu X,
Zhang H and Wu J: Time-dependence of NIHSS in predicting functional
outcome of patients with acute ischemic stroke treated with
intravenous thrombolysis. Postgrad Med J. 95:181–186. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Soriano-Tárraga C, Mola-Caminal M,
Giralt-Steinhauer E, Ois A, Rodríguez-Campello A, Cuadrado-Godia E,
Gómez-González A, Vivanco-Hidalgo RM, Fernández-Cadenas I, Cullell
N, et al: Biological age is better than chronological as predictor
of 3-month outcome in ischemic stroke. Neurology. 89:830–836. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Kim TH and Vemuganti R: Effect of sex and
age interactions on functional outcome after stroke. CNS Neurosci
Ther. 21:327–336. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Möbius C, Blinzler C, Schwab S, Köhrmann M
and Breuer L: Re-evaluation of the stroke prognostication using age
and NIH Stroke Scale index (SPAN-100 index) in IVT
patients-the-SPAN 10065 index. BMC Neurol. 18:1292018.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jadhav AP, Desai SM, Kenmuir CL, Rocha M,
Starr MT, Molyneaux BJ, Gross BA, Jankowitz BT and Jovin TG:
Eligibility for endovascular trial enrollment in the 6- to 24-h
time window: Analysis of a single comprehensive stroke center.
Stroke. 49:1015–1017. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Todo K, Sakai N, Kono T, Hoshi T, Imamura
H, Adachi H and Kohara N: National institutes of health stroke
scale-time score predicts outcome after endovascular therapy in
acute ischemic stroke: A retrospective single-center study. J
Stroke Cerebrovasc Dis. 25:1187–1191. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Koizumi S, Ota T, Shigeta K, Amano T, Ueda
M, Matsumaru Y, Shiokawa Y and Hirano T: Onset to reperfusion time
was not important in mechanical thrombectomy for elderly patients:
A retrospective multicenter study in Tama area, Tokyo. Cerebrovasc
Dis. 46:89–96. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
White BM, Magwood GS, Burns SP and Ellis C
Jr: Sex differences in patient-reported poststroke disability. J
Womens Health (Larchmt). 27:518–524. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Oskui PM, French WJ, Herring MJ, Mayeda
GS, Burstein S and Kloner RA: Testosterone and the cardiovascular
system: A comprehensive review of the clinical literature. J Am
Heart Assoc. 2:e0002722013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Tonetti DA, Gross BA, Desai SM, Jadhav AP,
Jankowitz BT and Jovin TG: Final infarct volume of <10
cm3 is a strong predictor of return to home in
nonagenarians undergoing mechanical thrombectomy. World Neurosurg.
119:e941–e946. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kim SM, Kwon SU, Kim JS and Kang DW: Early
infarct growth predicts long-term clinical outcome in ischemic
stroke. J Neurol Sci. 347:205–209. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liggins JT, Yoo AJ, Mishra NK, Wheeler HM,
Straka M, Leslie-Mazwi TM, Chaudhry ZA, Kemp S, Mlynash M, Bammer
R, et al: A score based on age and DWI volume predicts poor outcome
following endovascular treatment for acute ischemic stroke. Int J
Stroke. 10:705–709. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wada S, Yoshimura S, Inoue M, Matsuki T,
Arihiro S, Koga M, Kitazono T, Makino H, Hosoda K, Ihara M and
Toyoda K: Outcome prediction in acute stroke patients by continuous
glucose monitoring. J Am Heart Assoc. 7(pii):
e0087442018.PubMed/NCBI
|
|
34
|
Zhu Z, Yang J, Zhong C, Xu T, Wang A, Bu
X, Peng Y, Peng H, Xu T, Chen CS, et al: Abnormal glucose
regulation, hypoglycemic treatment during hospitalization and
prognosis of acute ischemic stroke. J Neurol Sci. 379:177–182.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Zhang X, Huang WJ and Yu ZG: Relationship
between the hypersensitive c-reactive protein (hs-CRP) level and
the prognosis of acute brainstem infarction. Cell Biochem Biophys.
72:107–110. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Meng X, Wen R and Li X: Values of serum
LDL and PCT levels in evaluating the condition and prognosis of
acute cerebral infarction. Exp Ther Med. 16:3065–3069.
2018.PubMed/NCBI
|
|
37
|
Billington CK, Appleton JP, Berge E,
Sprigg N, Glover M and Bath PMW: Impact of hydration status on
haemodynamics, effects of acute blood pressure-lowering treatment
and prognosis after stroke. Br J Clin Pharmacol. 84:2914–2922.
2018. View Article : Google Scholar : PubMed/NCBI
|