Introduction
Cardiogenic shock is the most severe form of acute
heart disease (1). Optimal
revascularization strategies for patients with cardiogenic shock
are complex (2) as the majority of
patients with cardiogenic shock have multi-vessel disease (3). Echocardiographic and angiographic
assessments aid the diagnosis and guide revascularization
strategies for patients with cardiogenic shock associated with
myocardial infarction. For such patients, coronary angiography or
echocardiography are used as diagnostic techniques (4). The severity of mitral annular
calcification (5), aortic valve
sclerosis (6), the ejection fraction
of the left ventricle (7) evaluated
by echocardiography (4), and the
ejection fraction of the left ventricle and severity of disease
evaluated by angiography are predictors of survival (4). To the best of our knowledge, no
previously published studies have compared transthoracic
echocardiography and coronary angiography assessments in
cardiogenic shock-associated myocardial infarction in a large
population.
The primary objective of the current prospective
cohort study was to assess correlations between the diagnostic
parameters (derived by coronary angiography and acute transthoracic
echocardiography) and the estimated ejection fraction in patients
with cardiogenic shock. The secondary objective of the study was to
provide further insight into the pathogenesis of acute myocardial
infarction in the management of cardiogenic shock in a Chinese
setting.
Materials and methods
Patient selection
A total of 3,908 patients aged ≥18 years with acute
myocardial infarction were admitted to the Department of Cardiology
at PLA General Hospital, Chongqing Traditional Chinese Medicine
Hospital (Chongqing, China) and the Hospital of Beijing the Hui
People (Beijing, China) between February 2014 and July 2018 due to
medical emergency (i.e. requiring immediate medical attention) were
subjected to a 12-lead electrocardiogram (ECG) diagnosis (8) prior to revascularization. The male to
female ratio was 2.5:1 and mean age of patients was 64.55±10.45
years (age range, 55–82 years). Patients with acute myocardial
infarction (diagnosed as a new left bundle branch block, new Q
waves and ST elevations on the ECG) who exhibited signs of
end-organ hypoperfusion (not due to sustained ventricular
arrhythmia and/or bradycardia), including chest pain, back pain,
dyspnea, warm and/or flushed skin, raised respiratory rate,
confusion and hypotension (including signs of dizziness,
lightheadedness, blurred vision, nausea, fainting, syncope,
fatigue, and/or lack of concentration) were included in the current
study. Patients with a history of revascularization, systemic
illness (for example, anoxic brain damage may adversely affect the
outcome of coronary bypass surgery), heparin allergy, anaphylaxis,
sepsis, hypovolemia and/or those receiving the revascularization
treatment were excluded from the study. Additionally, patients who
had not provided consent or were not willing to undergo diagnosis
were excluded.
Coronary angiography
Coronary angiograms were digitally recorded by seven
cardiologists with three years' experience prior to
revascularization, and coronary parameters were analyzed per the
Thrombolysis In Myocardial Infarction Criteria (3). Based on the ejection of contrast media
into the left atrium, mitral regurgitation was graded as follows:
i) 0, absent (no ejection of contrast media); ii) 1, mild (contrast
refluxes into the left atrium but cleared on each beat); iii) 2,
moderate (left atrial contrast density gradually increased but did
not equal left ventricle density); iv) 3, severe (the density of
contrast in the atrium and ventricle equalized after several
beats); and v) 4, extreme (the left atrium had become as dense as
the left ventricle on the first beat and contrast was seen
refluxing into the pulmonary veins) (4).
Image analysis for angiography
The type of lesion was graded per the American Heart
Association/American College of Cardiology guidelines (9) as follows: i) A, <10 mm in size,
concentric, readily accessible, discrete, <45° smooth contours,
absent or little calcification, no involvement of side branches,
not completely occluded, not ostial and no thrombus; ii) B1, one
characteristic from the following: 10–20 mm in size, eccentric,
proximal segment had moderate tortuosity, irregular contour,
thrombus present, moderate or heavy calcification, total occlusion
was <3 months old and an ostial/bifurcation lesion which
required two guide wires; iii) B2, ≥2 of the characteristics
described for the B1 type; and iv) C, >20 mm, diffuse, proximal
segment had excessive tortuosity, total occlusion was >3 months
old, side branches were involved, friable lesions and a degenerated
vein graft (10). The culprit vessel
collateral score was graded per the Rentrop classification
(11) as follows: i) 0, collateral
flow was absent; ii) 1, collateral flow was present in secondary
branches only; iii) 2, collateral flow was present in the major
vessels but had not reached the culprit lesion; and iv) 3,
collateral flow reached the infarct-associated artery and the
culprit lesion. The culprit lesion thrombus score was graded as
follows: i) 0, no thrombus; ii) 1, possible thrombus; iii) 2, the
dimension of the thrombus was less than one-half of the diameter of
the culprit vessel diameter; iv) 3, the dimension of the thrombus
was greater than one-half but less than two-thirds of the culprit
vessel diameter; v) 4, the dimension of the thrombus was greater
than two-thirds of the culprit vessel diameter but did not result
in total culprit vessel occlusion; and vi) 5, total culprit vessel
occlusion (12).
Transthoracic echocardiography
All patients were subjected to standard 2D
transthoracic echocardiography using a Sonimage HS1 ultrasound
system (Konica Minolta Medical & Graphic Imaging Europe B.V.)
and S4-2 phased transducers performed by cardiac ultra-sonographers
(with 3 years' experience) prior to revascularization. Images were
blindly analyzed by cardiothoracic surgeons (with 3 years'
experience) at the Chongqing Traditional Chinese Medicine Hospital
(Chongqing, China) (5).
Image analysis for
echocardiography
Mitral regurgitation was calculated according to the
following formula for the regurgitant fraction of the mitral valve:
Regurgitant fraction (%)=(mitral valve regurgitant
volume)/(trans-mitral volume)×100. Grading was performed as
follows: i) 0, absent (regurgitant fraction, 0); ii) 1, mild
(regurgitant fraction, <30%); iii) 2, moderate (regurgitant
fraction, 30–39%); iv) 3, severe (regurgitant fraction, 40–49%);
and v) 4, extreme (regurgitant fraction, ≥50%) (4). The relative wall thickness and
shortening fraction were measured from the short-axis view using
3-MHz transducers. The four-chambered view was taken for the mitral
flow velocity and was calculated according to the following
equation: Mitral flow velocity=(peak early diastolic flow
velocity)/(peak atrial flow velocity) (13). The left ventricular diastolic
dysfunction was graded as follows: i) I, isolated early relaxation
abnormality (initial stage); ii) II, increased filling pressure in
the atrium (moderate condition); and iii) III, restrictive filling
of the heart (severe condition) (14).
Statistical analysis
Categorical data are presented as frequency
(percentage) whilst continuous data are presented as mean ±
standard deviation. InStat (version 3.1; GraphPad Software, Inc.)
was used to analyze the data. Categorical data were analyzed by the
χ2 test and continuous data were analyzed by the
Wilcoxon test (15). The Spearman's
correlation test was used to analyze correlations between the
diagnostic parameters (echocardiographically- and
angiographically-measured data) and estimated ejection fractions. A
Spearman's correlation coefficient (r) of 0.123–0.994 was
considered significant (4).
P<0.05 was considered to indicate a statistically significant
difference.
Results
Patient demographic characteristics
and medical history
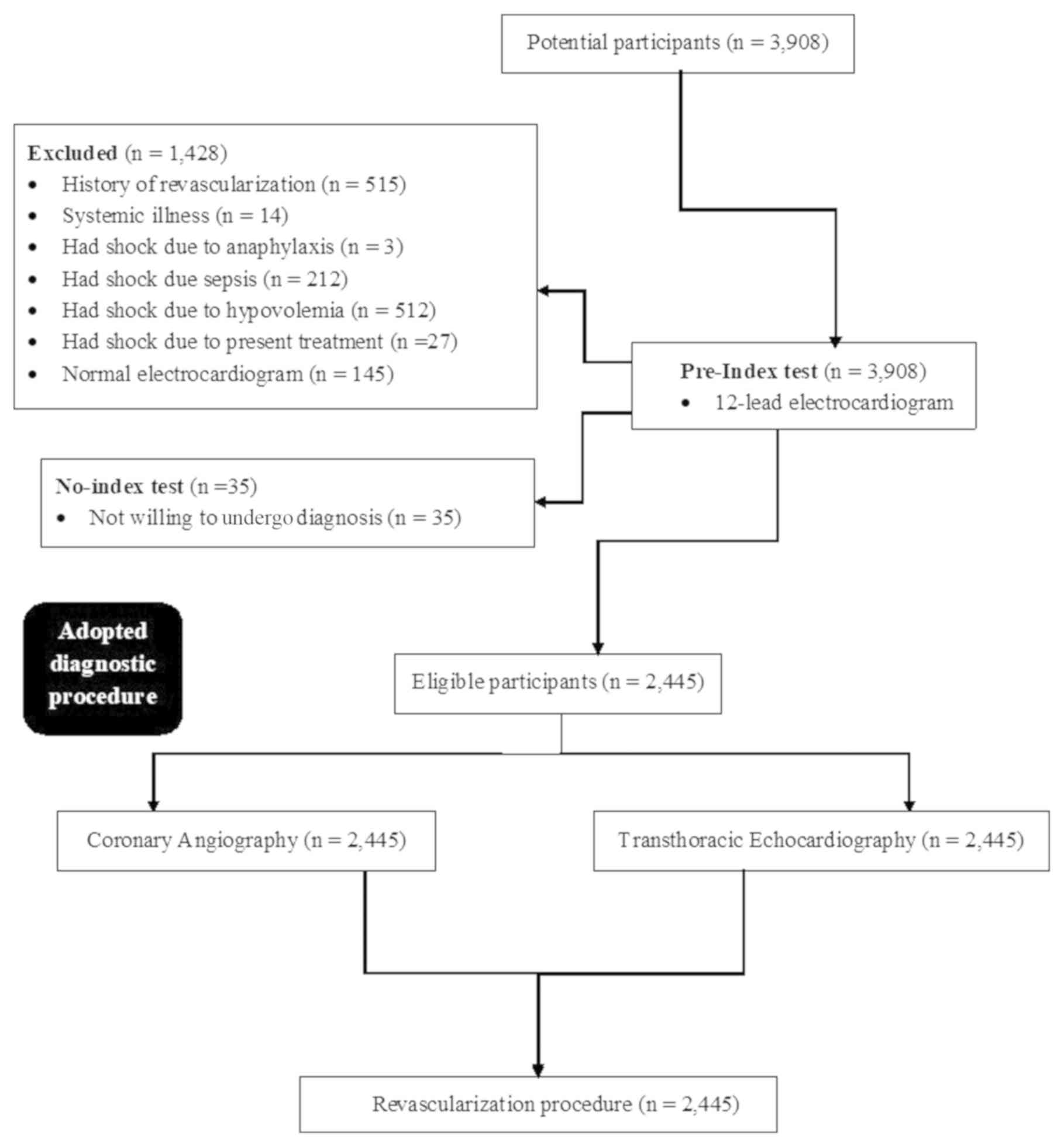
Among the enrolled patients, 515 had undergone
previous revascularization, 14 had systemic illness, 212 had septic
shock, 512 had hypovolemic shock, 27 had were admitted to hospital
due to a side effect (shock) of the medication they were
administered, 145 had a normal ECG and 35 were not willing to
undergo further diagnostic testing following admission to the
emergency department or the cardiology department at the Chongqing
Traditional Chinese Medicine Hospital, (Chongqing, China), the
Fourth Medical Center of PLA General Hospital (Beijing, China) and
the Hospital of Beijing The Hui People (Beijing, China) where three
patients were afflicted with shock due to anaphylaxis. Therefore,
they were excluded from the analysis. A total of 2,445 patients
with cardiogenic shock were included in the current prospective
cohort study, where 165 patients died in the emergency department
and during follow-up period (main cause of death was cardiogenic
shock). A schematic representation of the study design is presented
in Fig. 1.
Among the enrolled patients, 115 had end-organ
hypoperfusion, 629 had a new left bundle branch block on the ECG,
715 patients had new Q waves on the ECG, 986 patients had ST
elevations on the ECG and 101 patients had a history of coronary
artery bypass graft. All the patients received oral treatment
(including β-blockers, angiotensinogen converting enzyme
inhibitors, angiotensin receptor blocker, calcium channel blocker,
and statins) for cardiogenic shock. The other demographic
characteristics of the enrolled patients are presented in Table I.
 | Table I.Demographic characteristics and
medical history of the enrolled patients. |
Table I.
Demographic characteristics and
medical history of the enrolled patients.
| Demographic
characteristic | No. patients
(n=2,445) |
|---|
| Age |
|
| Range,
years | 55–82 |
| Mean ±
SD, years | 64.55±10.45 |
| Sex |
|
| Male | 1,741 (71) |
|
Female | 704 (29) |
| Ethnicity |
|
|
Chinese | 2,439 (99.75) |
|
China-based American (Han
Chinese) | 5 (0.2) |
|
Non-Chinese | 1 (0.05) |
| Diabetes | 729 (30) |
| End-organ
hypoperfusion | 115 (5) |
| New left bundle
branch block in ECG | 629 (26) |
| New Q waves in
ECG | 715 (29) |
| ST elevations in
ECG | 986 (40) |
| History of coronary
artery bypass graft | 101 (4) |
| Heart rate,
beats/min | 98.15±21.15 |
| Diastolic blood
pressure, mmHg | 89.11±19.15 |
| Systolic blood
pressure, mmHg | 131.15±18.45 |
| Cardiac index
(l/min/m2) | 1.95±0.73 |
| Treatment
received |
|
|
β-blockers | 452 (18) |
|
Angiotensinogen converting
enzyme inhibitors | 345 (14) |
|
Angiotensin receptor
blocker | 868 (36) |
| Calcium
channel blocker | 645 (26) |
|
Statins | 112 (5) |
|
Others | 23 (1) |
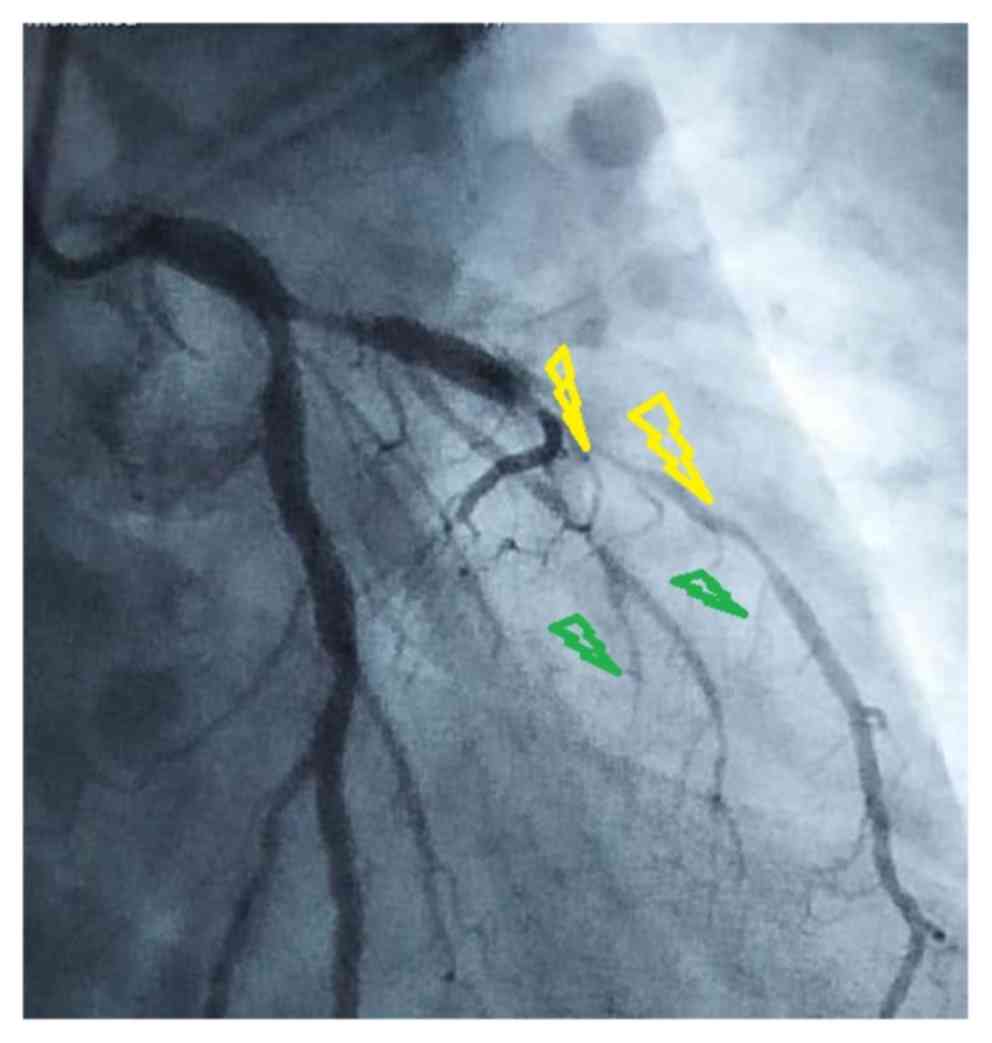
Coronary angiographic evaluation
Coronary angiography-derived mitral regurgitation
grade had a significant correlation with the number of culprit
vessels (r=0.907; P=0.034). The coronary angiography-derived number
of culprit vessels had no significant correlation with the location
of the culprit vessels (r=−0.21; P=0.735). The coronary angiography
derived culprit lesion type had no significant correlation with the
culprit vessel collateral score (r=0.054; P=0.947) and culprit
lesion thrombus score (r=−0.406; P=0.594). The other coronary
angiography assessments of the enrolled patients are presented in
Table II.
 | Table II.Angiographic findings of the enrolled
patients. |
Table II.
Angiographic findings of the enrolled
patients.
|
|
| Spearman's
correlation |
|---|
|
|
|
|
|---|
| Characteristic | No. patients
(n=2,445) | P-value | r-value |
|---|
| Mitral
regurgitationa |
|
|
|
| 0 | 5 (0.2) | 0.034 | 0.907 |
| 1 | 885 (36.3) |
|
|
| 2 | 745 (30.5) |
|
|
| 3 | 468 (19) |
|
|
| 4 | 342 (14) |
|
|
| Number of culprit
vessel |
|
|
|
| 0 | 5 (0.2) |
P-valuee |
r-valuee |
| 1 | 1,358 (55.5) |
|
|
| 2 | 758 (31) |
|
|
| 3 | 311 (12.7) |
|
|
| 4 | 13 (0.6) |
|
|
|
Total | 3,859 |
|
|
| Location of the
culprit vessels |
|
|
|
| Right
coronary artery | 1,485 (38) | 0.735 | 0.21 |
| Left
coronary artery | 641 (17) |
|
|
| Left
anterior descending artery | 815 (21) |
|
|
| Left
circumflex artery | 573 (15) |
|
|
|
Saphenous vein graft | 345 (9) |
|
|
| Culprit lesion
typeb |
|
|
|
| A | 556 (14) |
P-valuef |
r-valuef |
| B1 | 1,341 (35) |
|
|
| B2 | 1,152 (30) |
|
|
| C | 810 (21) |
|
|
| Culprit vessel
collateral scorec |
|
|
|
| 0 | 1,313 (34) | 0.947 |
|
| 1 | 1,245 (32) |
|
|
| 2 | 854 (22) |
| 0.054 |
| 3 | 447 (12) |
|
|
| Culprit lesion
thrombus scored |
|
|
|
| 0 | 1,341 (36) | 0.594 |
|
| 1 | 845 (22) |
|
|
| 2 | 741 (19) |
| 0.406 |
| 3 | 441 (11) |
|
|
| 4 | 289 (7) |
|
|
| 5 | 202 (5) |
|
|
Transthoracic echocardiographic
evaluation
The echocardiographically-derived mitral
regurgitation had a significant strong correlation with the number
of culprit vessel (r=0.896; P=0.04). The
echocardiographically-derived number of culprit vessels had no
significant correlation with the degree of left ventricular
diastolic dysfunction (r=−0.693; P=0.513). The other
echocardiographic assessments of the enrolled patients are
presented in Table III.
 | Table III.Echocardiographic findings of the
enrolled patients. |
Table III.
Echocardiographic findings of the
enrolled patients.
|
|
| Spearman's
Correlation |
|---|
|
|
|
|
|---|
| Characteristic | No. patients
(n=2,445) | P-value | r-value |
|---|
| Mitral
regurgitationa |
| 0.04 |
|
| 0 | 1 (0.05) |
|
|
| 1 | 833 (34) |
|
|
| 2 | 825 (33.95) |
| 0.896 |
| 3 | 444 (18) |
|
|
| 4 | 342 (14) |
|
|
| Number of culprit
vessels |
|
P-valuec |
r-valuec |
| 0 | 1 (0.05) |
|
|
| 1 | 1,283 (52) |
|
|
| 2 | 799 (33) |
|
|
| 3 | 341 (13.95) |
|
|
| 4 | 21 (1) |
|
|
|
Total | 3988 |
|
|
| Left ventricular
end diastolic dimension, mm | 51.12±5.45 | N/A |
|
| Left ventricular
end systolic dimension, mm | 35.15±4.12 | N/A |
|
| Left ventricular
end diastolic volume, ml | 96.15±15.14 | N/A |
|
| Left ventricular
end systolic volume, ml | 43.42±18.12 | N/A |
|
| Left ventricular
mass index, g/m2 | 125.15±21.11 | N/A |
|
| % Left ventricular
ejection fraction | 31.91±17.15 | N/A |
|
| Wall motion score
index | 1.89±0.22 | N/A |
|
| % Shortening
fraction | 21.12±8.11 | N/A |
|
| Relative wall
thickness, cm | 0.46±0.09 | N/A |
|
| E wave of
trans-mitral flow, cm/sec | 65±21 | N/A |
|
| A wave of
trans-mitral flow, cm/sec | 87±22 | N/A |
|
| Left ventricular
diastolic dysfunctionb |
| 0.513 | −0.693 |
| Grade
I | 1,122 (46) |
|
|
| Grade
II | 781 (32) |
|
|
| Grade
III | 542 (22) |
|
|
Correlations between imaging
modalities for diagnostic parameters
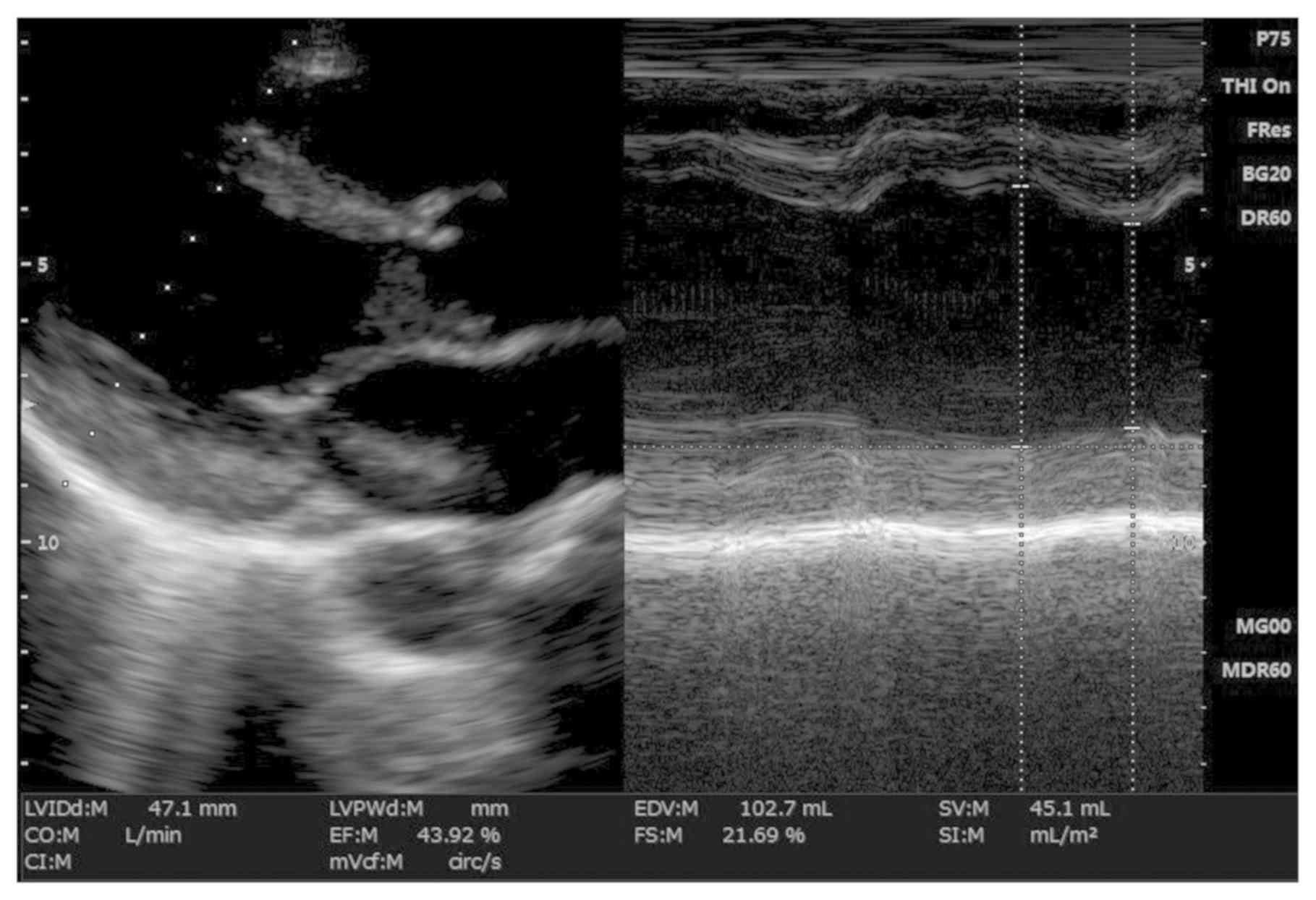
The left ventricular ejection fractions derived by
coronary angiography (31.12±12.45%; 28% as the lowest and 42% as
the highest ejection fractions detected; Fig. 2) were similar to values obtained by
transthoracic echocardiography (31.91±17.15%; lower limit, 29%;
upper limit, 46%; Fig. 3). There was
a significant positive correlation between angiographically- and
echocardiographically-measured left ventricular ejection fractions
(r=0.356; P=0.045). There was a significant difference between
angiographically and echocardiographically measured low ejection
fractions (P=0.041; Fig. 3). Using a
cut-off of <36%, the sensitivity of echocardiography was found
to be 75% (mean range, 55–95%) and specificity was found to be 68%
(mean range, 35–95%).
Echocardiographically-measured left ventricular
function was correlated with angiographically-calculated disease
severity (r=0.656; P=0.012). Echocardiographically-measured
ejection fractions were significantly negatively correlated with
angiographic jeopardy score (r=−0.290; P=0.032). The left
ventricular main obstruction measured by coronary angiography was
correlated with left ventricular ejection fraction measured
echocardiographically (27.12±2.12% vs. 27.31±5.05%; r=0.71;
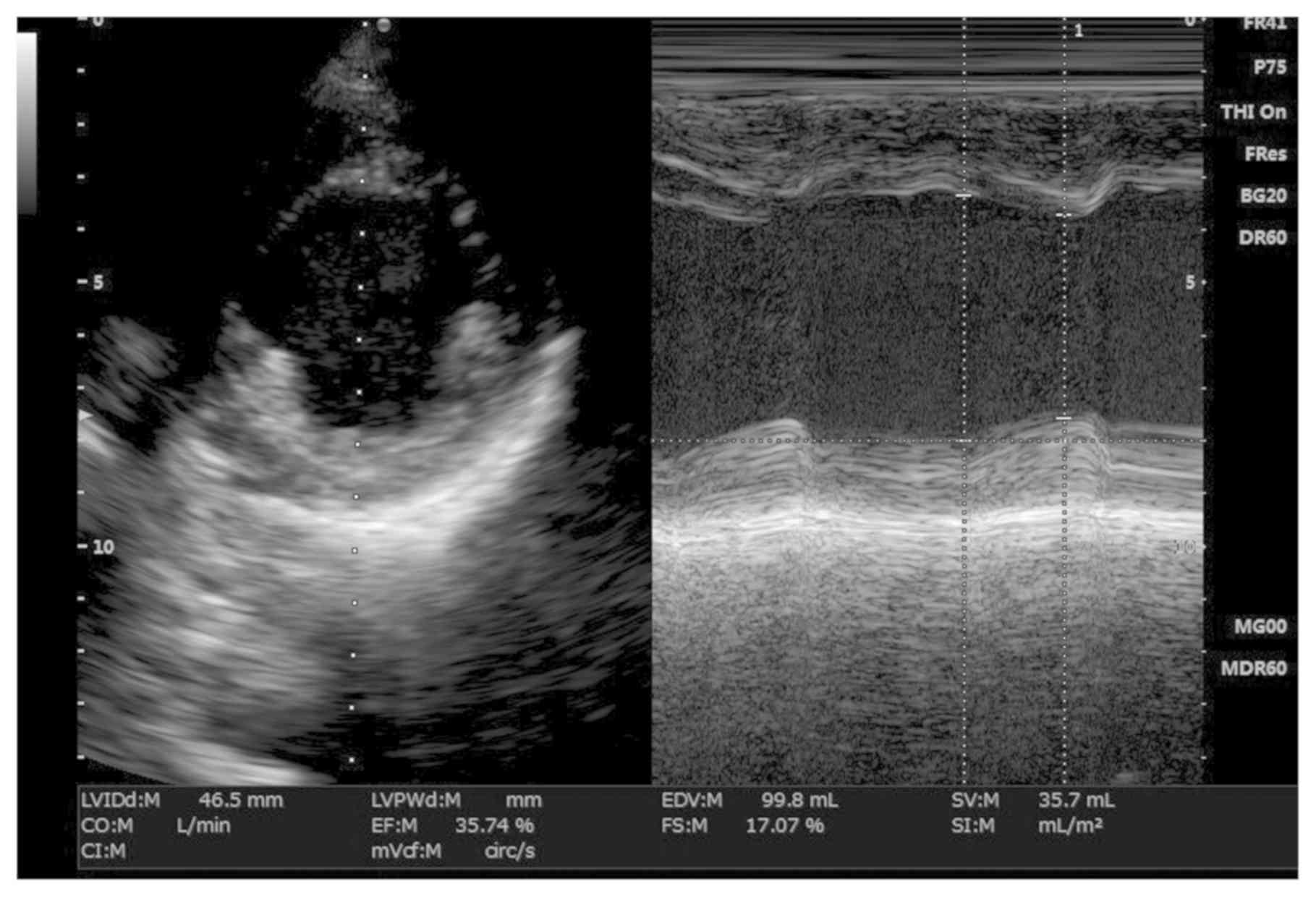
P=0.016). The lower median left ventricular end-systolic volumes
measured echocardiographically were correlated with those measured
angiographically (r=0.73; P=0.029; Fig.
4). There were no correlations between the number of diseased
vessels, collateral flow to the culprit vessel derived
angiographically and worst left ventricular function evaluated
echocardiographically (P>0.05 for all comparisons; Table IV). There was no significant
difference between the number of culprit vessels derived
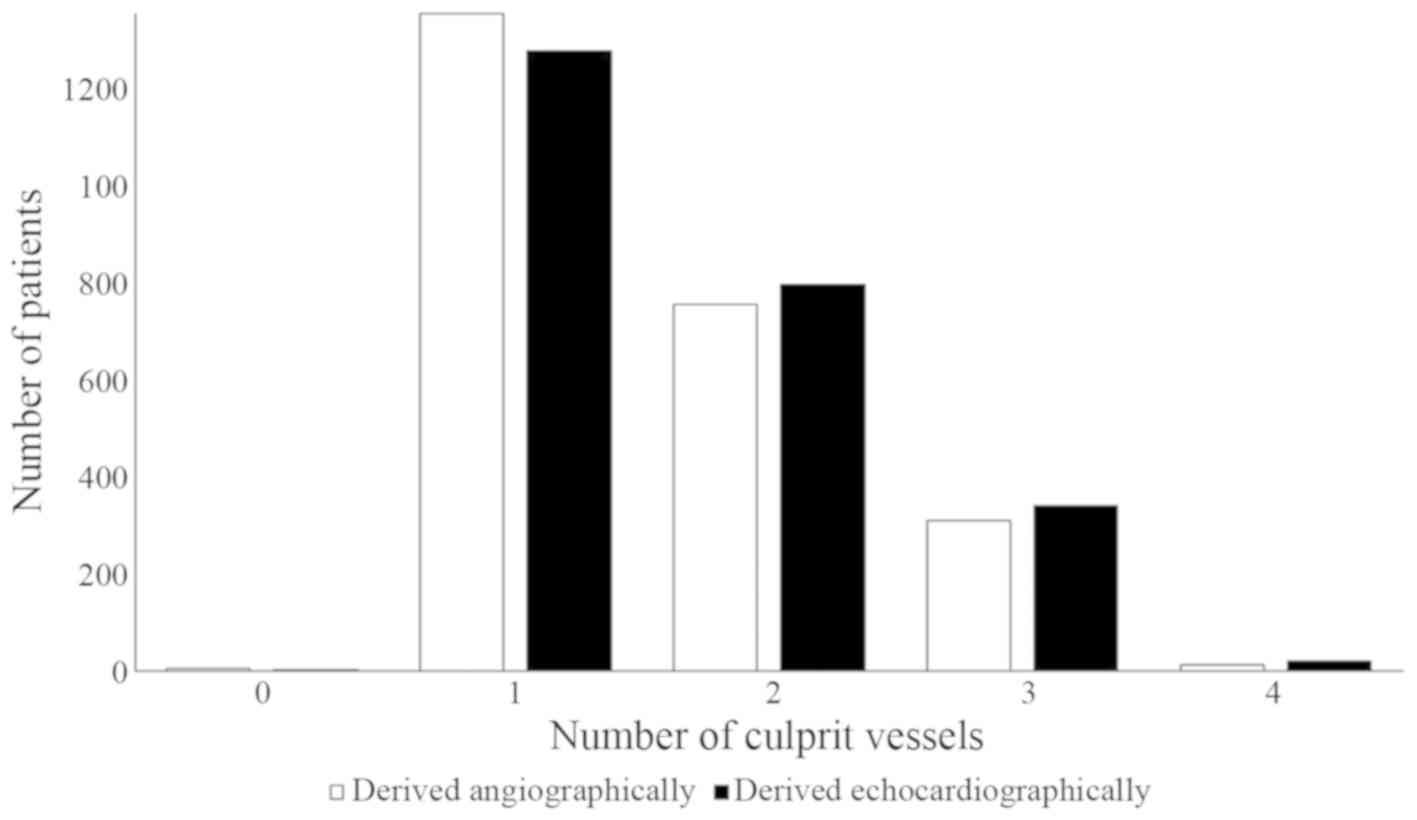
angiographically and echocardiographically (P=0.058; Fig. 5). Echocardiographically-derived
mitral regurgitation on the same day as that derived
angiographically. The r and P-values indicated that there was
strong correlation between mitral regurgitation derived
echocardiographically and mitral regurgitation derived
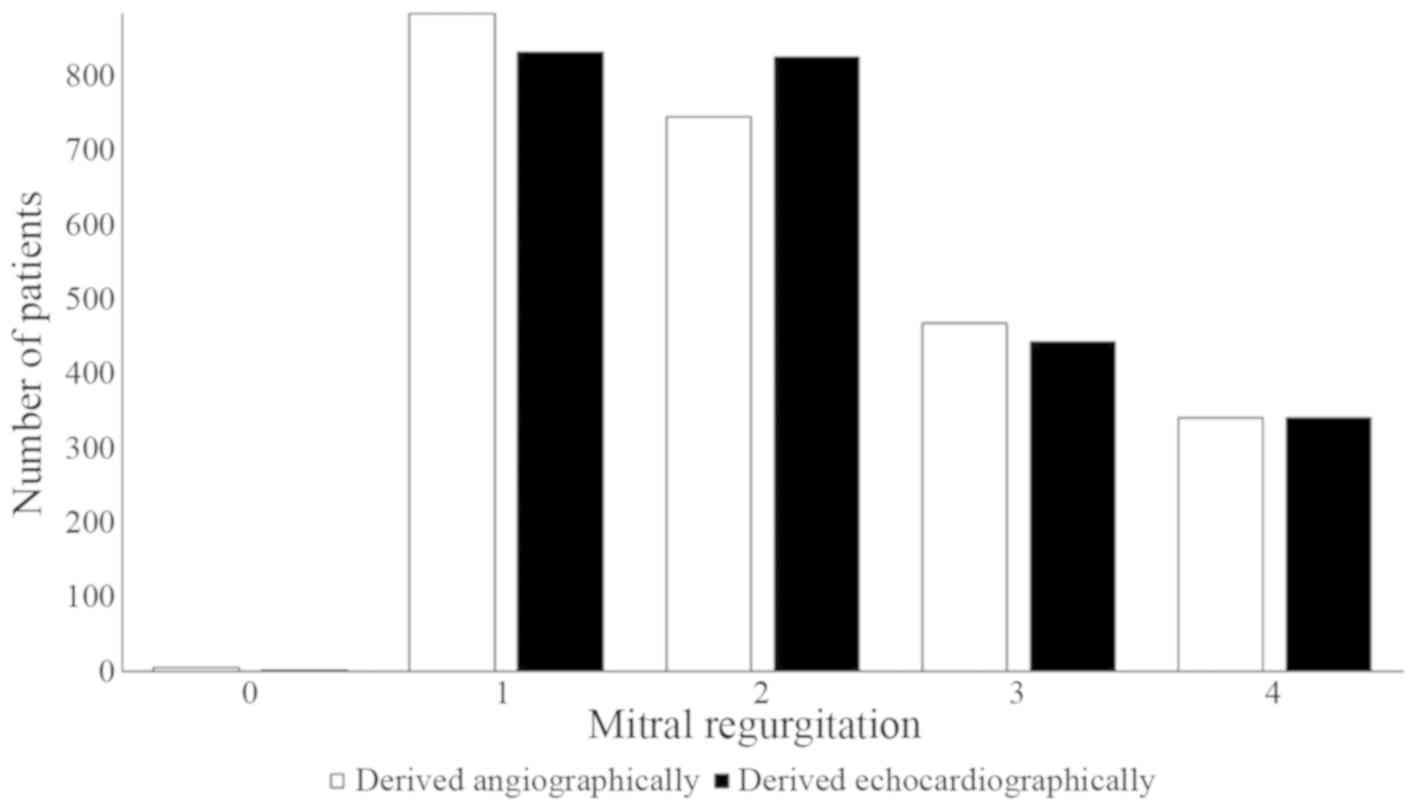
angiographically (1–2 vs. 1–2; r=0.99; P=0.001; Fig. 6).
 | Table IV.Correlations between coronary
angiographic diagnostic parameters and transthoracic
echocardiographic evaluations. |
Table IV.
Correlations between coronary
angiographic diagnostic parameters and transthoracic
echocardiographic evaluations.
|
|
| Spearman's
Correlation |
|---|
|
|
|
|
|---|
| Coronary
angiographic diagnostic parameter | Transthoracic
echocardiographic evaluation | P-value |
|---|
| Left ventricular
ejection fractions | Left ventricular
ejection fraction | 0.045a |
| Disease
severity | Left ventricular
function | 0.012a |
| Jeopardy score | Ejection
fraction | 0.032b |
| Left ventricular
main obstruction | Left ventricular
ejection fraction | 0.016a |
| Lower median left
ventricular end-systolic volume | Lower median left
ventricular end-systolic volume | 0.029a |
| Number of diseased
vessels | Worst left
ventricular function | 0.061 |
| Collateral flow to
the culprit vessel | Worst left
ventricular function | 0.082 |
Discussion
The current study investigated Chinese patients with
acute myocardial infarction using angiography and transthoracic
echocardiography. Myocardial infarction may be caused by a number
of factors and several treatment options are available (11). The results obtained in the current
study may aid the selection of optimal revascularization strategies
and the identification of mortality-associated factors in patients
with cardiogenic shock.
The current prospective cohort study suggested that
transthoracic echocardiography and coronary angiography findings in
patients with cardiogenic shock prior to revascularization were
correlated with each other. The left ventricular ejection fractions
and mitral regurgitation grades were positively correlated among
the enrolled patients. The results of the present study were in
line with those of the SHOCK trial performed in Caucasian patients
(4,15). However, certain differences in
echocardiographically-derived results compared with coronary
angiographically-derived results were observed in the present
study. This is because echocardiograms are technically more
difficult to perform in critically ill patients (including those
requiring mechanical ventilation) compared with angiography
(4). Perfusion imaging with contrast
echocardiography (16) may improve
accuracy and reduce the variability of echocardiography images.
The present study demonstrated that mitral
regurgitation quantified echocardiographically was correlated with
mitral regurgitation quantified angiographically. This result was
similar to results obtained in the SHOCK trial (4,15).
However, the present results differed from those of previously
published studies (1,17–19)
suggesting that mitral regurgitation is an independent factor for
mortality in patients (derived from institutional records) with
cardiogenic shock. Collectively, the present study suggests that
mitral regurgitation is a marker of worse prognosis for patients
with cardiogenic shock.
The numbers of culprit vessels derived
angiographically and echocardiographically were correlated with the
severity of mitral regurgitation. The results obtained in the
current study were in line with those of the SHOCK trial (4,15), the
SHORTWAVE study (7) and the STEMI
study (18) performed in Caucasian
patients. However, the aforementioned trials enrolled a limited
number of the patients (127, 386 and 147, respectively). Mitral
regurgitation is affected by presumed distortion in left
ventricular geometry (4,20) and left ventricular dysfunction
(21). A previous study (9) suggested that mitral regurgitation
should be evaluated in patients with cardiogenic shock to assess
cardiac risk.
In the present study, angiographic findings revealed
that 1,082 patients had >1 culprit vessel while
echocardiographic results showed that 1,161 patients had >1
culprit vessel. A possible reason for this could be plaque ruptures
leading to thrombotic occlusion in multiple coronary arteries
(22).
The present study had a number of limitations. As
the current study enrolled a large population for angiography,
echocardiography and revascularization procedures, different
cardiologists, ultra-sonographers and medical and non-medical staff
were involved and inter-and intra-evaluator reliabilities were not
evaluated.
In addition, several aspects of the present study
were not in line with those of the SHOCK trial (15). In particular, coronary angiography
failed to derive a correlation between the number of culprit
vessels and the location of the culprit vessels, between the
culprit lesion type and the culprit vessel collateral score and
between the culprit lesion type and culprit lesion thrombus score.
Echocardiography failed to derive a correlation between the number
of culprit vessels and degree of left ventricular diastolic
dysfunction. A possible explanation is that the ejection fraction
of the left ventricle is decreased in patients with cardiogenic
shock (23). Therefore, a broad
spectrum of left ventricular function is not available.
Furthermore, excessive nitric oxide production has been reported in
patients with cardiogenic shock and this inhibits myocardial
contractility (24). Compensatory
hyperkinesis is a normal manifestation of patients with cardiogenic
shock (25) and this affects the
ejection fraction of the heart. A further randomized trial is
required to investigate such correlations.
Important parameters, including vitamin D, calcium,
parathormone and phosphorous levels, were not evaluated in the
present study. The degree of deposition of calcium on the mitral
valve may help to predict the pathophysiology of patients with
cardiogenic shock (26) as calcium
deposition may cause elevation of the mitral leaflets (5). Exercise-induced electrocardiography has
a better prediction of risk stratification of mitral regurgitation
compared with electrocardiography (27) but was not performed in the current
study. The present study did not evaluate epicardial fat thickness
by electrocardiography and did not derive a correlation with mitral
regurgitation. A possible justification is that, unlike magnetic
resonance imaging and computed tomography, electrocardiography is
not an exact method for the quantification of epicardial fat
thickness (28). Follow-up data
after percutaneous coronary intervention regarding the event-free
survival and overall survival were not discussed.
In conclusion, the worst coronary artery outcomes
are associated with more severe mitral regurgitation. Transthoracic
echocardiography and coronary angiography results were correlated
with the estimated ejection fractions in the current large
population study, despite the vast operational difference between
these two techniques in patients with cardiogenic shock.
Acknowledgements
Not applicable.
Funding
The present study was supported by the Science and
Technology Innovation Nursery Fund Project of PLA General Hospital,
Beijing, China (grant no. 14KMM26).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
JC and YaH contributed equally to the
conceptualization, literature review, data curation, funding and
analysis of the study. YuH administrated the study, contributed to
data curation, analysis, the literature review and methodology
development. XY provided software and contributed to the literature
review and methodology development. GZ contributed to resources,
formal analysis and literature review of the study. JZ provided
software and contributed to the literature review, data curation
and formal analysis of the study. YY provided software, contributed
to the literature review and conceptualization of the study and
drafted, reviewed and edited the manuscript for intellectual
content. All authors read and approved the manuscript for
publication.
Ethics approval and consent to
participate
The present study is registered in Research Registry
(www.researchregistry.com; UID no.
researchregistry4512; dated February 15, 2014). The protocol
(CTH/CL/2/14; dated February 11, 2014) was approved by the
Chongqing Traditional Chinese Medicine Hospital review board. All
patients provided informed consent prior to enrollment in the
study.
Patient consent for publication
All patients provided informed consent for
publication prior to enrollment in the study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Rudiger A: Understanding cardiogenic
shock. Eur J Heart Fail. 17:466–467. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Aissaoui N, Puymirat E, Tabone X,
Charbonnier B, Schiele F, Lefèvre T, Durand E, Blanchard D, Simon
T, Cambou JP and Danchin N: Improved outcome of cardiogenic shock
at the acute stage of myocardial infarction: A report from the USIK
1995, USIC 2000, and FAST-MI French nationwide registries. Eur
Heart J. 33:2535–2543. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Thiele H, Desch S, Piek JJ, Stepinska J,
Oldroyd K, Serpytis P, Montalescot G, Noc M, Huber K, Fuernau G, et
al: CULPRIT-SHOCK Investigators. Multivessel versus culprit lesion
only percutaneous revascularization plus potential staged
revascularization in patients with acute myocardial infarction
complicated by cardiogenic shock: Design and rationale of
CULPRIT-SHOCK trial. Am Heart J. 172:160–169. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Berkowitz MJ, Picard MH, Harkness S,
Sanborn TA, Hochman JS and Slater JN: Echocardiographic and
angiographic correlations in patients with cardiogenic shock
secondary to acute myocardial infarction. Am J Cardiol.
98:1004–1008. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zemer WN, Shapira Y, Weisenberg D,
Monakier D, Bental T, Sagie A and Vaturi M: Association between
mitral annular calcium and flail mitral leaflet in degenerative
mitral valve disease. Am J Cardiol. 116:121–124. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Saha SA, Beatty AL, Mishra RK, Whooley MA
and Schiller NB: Usefulness of an echocardiographic composite
cardiac calcium score to predict death in patients with stable
coronary artery disease (from the Heart and Soul Study). Am J
Cardiol. 116:50–58. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Faganello G, Faggiano P, Candido R,
Tarantini L, Di Lenarda A, De Feo S and Cioffi G: The worrisome
liaison between left ventricular systolic dysfunction and mitral
annulus calcification in type 2 diabetes without coronary artery
disease: Data from the SHORTWAVE study. Nutr Metab Cardiovasc Dis.
23:1188–1194. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Newby DE, Williams MC, Flapan AD, Forbes
JF, Hargreaves AD, Leslie SJ, Lewis SC, McKillop G, McLean S, Reid
JH, et al: Role of multidetector computed tomography in the
diagnosis and management of patients attending the rapid access
chest pain clinic, The Scottish computed tomography of the heart
(SCOT-HEART) trial: Study protocol for randomized controlled trial.
Trials. 13:1842012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Nishimura RA, Otto CM, Bonow RO, Carabello
BA, Erwin JP III, Guyton RA, O'Gara PT, Ruiz CE, Skubas NJ, Sorajja
P, et al: 2014 AHA/ACC guideline for the management of patients
with valvular heart disease: Executive summary: A report of the
American College of Cardiology/American Heart Association Task
Force on Practice Guidelines. J Am Coll Cardiol. 63:2438–2488.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hoe J: CT coronary angiography of chronic
total occlusions of the coronary arteries: How to recognize and
evaluate and usefulness for planning percutaneous coronary
interventions. Int J Cardiovasc Imaging. 25:43–54. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Figueras J, Otaegui I, Marti G, Domingo E,
Baneras J, Barrabes JA, Del Blanco BG and Garcia-Dorado D: Area at
risk and collateral circulation in a first acute myocardial
infarction with occluded culprit artery. STEMI vs. non-STEMI
patients. Int J Cardiol. 259:14–19. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Duman H, Cetin M, Durakoglugil ME,
Degirmenci H, Hamur H, Bostan M, Karadag Z and Cicek Y: Relation of
angiographic thrombus burden with severity of coronary artery
disease in patients with ST-segment elevation myocardial
infarction. Med Sci Monit. 17:3540–3556. 2015. View Article : Google Scholar
|
|
13
|
Suzue M, Mori K, Inoue M, Hayabuchi Y,
Nakagawa R and Kagami S: Developmental changes in the left
ventricular diastolic wall strain on M-mode echocardiography. J
Echocardiogr. 12:98–105. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Grant AD, Negishi K, Negishi T, Collier P,
Kapadia SR, Thomas JD, Marwick TH, Griffin BP and Popovic ZB:
Grading diastolic function by echocardiography: Hemodynamic
validation of existing guidelines. Cardiovasc Ultrasound.
13:282015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Sanborn TA, Sleeper LA, Webb JG, French
JK, Bergman G, Parikh M, Wong SC, Boland J, Pfisterer M, Slater JN,
et al: Correlates of one-year survival in patients with cardiogenic
shock complicating acute myocardial infarction: Angiographic
findings from the SHOCK trial. J Am Coll Cardiol. 42:1373–1379.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Eskandari M and Monaghan M: Contrast
echocardiography in daily clinical practice. Herz. 42:271–178.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Song JM, Kang SH, Lee EJ, Shin MJ, Lee JW,
Chung CH, Kim DH, Kang DH and Song JK: Echocardiographic predictors
of left ventricular function and clinical outcomes after successful
mitral valve repair: Conventional two-dimensional versus
speckle-tracking parameters. Ann Thorac Surg. 91:1816–1823. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Engström AE, Vis MM, Bouma BJ, Claessen
BE, Sjauw KD, Baan J Jr, Meuwissen M, Koch KT, de Winter RJ,
Tijssen JG, et al: Mitral regurgitation is an independent predictor
of 1-year mortality in ST-elevation myocardial infarction patients
presenting in cardiogenic shock on admission. Acute Card Care.
12:51–57. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Harjola VP, Lassus J, Sionis A, Kober L,
Tarvasmäki T, Spinar J, Parissis J, Banaszewski M, Silva-Cardoso J,
Carubelli V, et al: Clinical picture and risk prediction of
short-term mortality in cardiogenic shock. Eur J Heart Fail.
17:501–509. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
20
|
Timek TA, Lai DT, Bothe W, Liang D,
Daughters GT, Ingels NB and Miller DC: Geometric perturbations in
multiheaded papillary tip positions associated with acute ovine
ischemic mitral regurgitation. J Thorac Cardiovasc Surg.
150:232–237. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Tu Y, Zeng QC, Huang Y and Li JY:
Percutaneous coronary intervention for acute myocardial infarction
with mitral regurgitation. J Geriatr Cardiol. 13:521–527.
2016.PubMed/NCBI
|
|
22
|
Ananthakrishna R, Wang LJ, Zhao LP and Tan
HC: Double jeopardy in acute ST-segment elevation myocardial
infarction. Singapore Med J. 58:225–227. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rogers PA, Daye J, Huang H, Blaustein A,
Virani S, Alam M, Kumar A, Paniagua D, Kar B, Bozkurt B, et al:
Revascularization improves mortality in elderly patients with acute
myocardial infarction complicated by cardiogenic shock. Int J
Cardiol. 172:239–241. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wong VW and Lerner E: Nitric oxide
inhibition strategies. Future Sci OA. 1(pii): FSO352015.PubMed/NCBI
|
|
25
|
Wu HM and Tzeng BH: Dynamic left
ventricular outflow tract obstruction with cardiogenic shock in
apical ballooning syndrome. Acta Cardiol Sin. 29:370–373.
2013.PubMed/NCBI
|
|
26
|
Pandit A, Mookadam F, Boddu S, Pandit A,
Tandar A, Chaliki H, Cha S and Lee HR: Vitamin D levels and left
ventricular diastolic function. Open Heart. 1:e0000112014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Dulgheru R, Marchetta S, Sugimoto T, Go
YY, Girbea A, Oury C and Lancellotti P: Exercise testing in mitral
regurgitation. Prog Cardiovasc Dis. 60:342–350. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Park JS, Lee YH, Seo KW, Choi BJ, Choi SY,
Yoon MH, Hwang GS, Tahk SJ and Shin JH: Echocardiographic
epicardial fat thickness is a predictor for target vessel
revascularization in patients with ST-elevation myocardial
infarction. Lipids Health Dis. 15:1942016. View Article : Google Scholar : PubMed/NCBI
|