Introduction
MicroRNAs (miRNAs/miRs), which are composed of 19-23
nucleotides, are a class of small, non-coding RNAs that serve a key
role in regulating various biological processes, including tumor
formation. miRNAs also modulate a variety of cellular processes,
including the cell cycle and proliferation by attaching to the
corresponding target binding sites at the 3' untranslated region on
their respective mRNA (1-4).
Moreover, miR-375 can regulate insulin secretion and is regarded as
a pancreatic islet-specific miRNA (5). Previous studies have also demonstrated
that the expression of miR-375 is enriched in different organs,
while its expression is downregulated in numerous malignant cancer
types, including gastric and esophageal cancer, hepatocellular
carcinoma and head and neck cancer (6-9);
hence, miR-375 is known to be an important cancer-related miRNA
(10,11).
Avian leukemia is a general term used to describe
various types of tumor diseases in poultry that are caused by the
avian leukemia virus (ALV) and avian sarcoma virus (12). Avian leukemia was first identified in
1998 and can cause several infectious benign and malignant tumors,
thus leading to large economic loss in the poultry industry
(13-15).
Until recently, there has been no effective vaccine to treat the
disease. ALVs are classified into seven subgroups, referred to as
A-E, J and K (16). Among these
seven subgroups, ALV-J is the most prevalent and causes more damage
to chickens compared with the other subgroups (17). Furthermore, ALV-J infection causes
immunosuppression and various forms of myeloid leukosis and, to a
lesser extent, solid tumors, such as hemangiomas and histiocytic
sarcomas (12). Tumor hepatocyte
gene chip screening in a previous study revealed that the
expression of gga-miR-375 was significantly decreased in
ALV-J-induced liver tumors in chickens (18).
The Hippo signaling pathway is a tumor suppressor
pathway that regulates cell cycle distribution, cell proliferation,
stem cell functions and organ size (18,19). The
effectors of the Hippo signaling pathway, Yes-associated protein
(YAP) and transcriptional co-activator with PDZ-binding motif (TAZ)
serve a key role in tumorigenesis, and recent preclinical data
support the development of novel Hippo pathway-based targeted
therapies to prevent and treat malignancies using pharmacological
inhibition of YAP and TAZ activity (20,21).
YAP1 was originally identified as a factor that can control tissue
growth and organ size in Drosophila melanogaster, and it is
a key component of the Hippo signaling pathway and functions as an
important oncogene in mammals (22,23).
Furthermore, YAP1, a 65 kDa proline rich phosphoprotein, is the
primary downstream effector of the Hippo signaling pathway and acts
as a transcriptional co-activator, which can bind to transcription-
and cell cycle-associated genes, including cyclin D1 and cyclin E,
and therefore regulates cell proliferation (24). A previous study revealed that
overexpression of YAP1 can promote tumorigenesis in most, but not
all, tumor types evaluated (25,26). For
example, as the core protein in the Hippo pathway, YAP1 can induce
malignant mesothelioma cell proliferation (27). In addition, the abnormal expression
of YAP1 could promote cell proliferation in vitro (28). It has also been shown that
gga-miR-375 is significantly downregulated, while YAP1 is
upregulated in liver tumors in chickens infected with ALV-J, and
also that YAP1 is the target gene of gga-miR-375(29). Furthermore, previous studies have
revealed that the cell cycle and cell proliferation have a close
association with tumor formation (30-33).
Considering that gga-miR-375 and YAP1 play a key role in
tumorigenesis in ALV-J-infected chickens (29), the present study aimed to investigate
whether gga-miR-375 and YAP1 affected the cell cycle and
proliferation in DF-1 cells to further determine the novel function
of gga-miR-375 and YAP1.
The present results suggested that ALV-J infection
may promote the cell cycle by promoting cell transition from
G1 to S/G2 phase and may increase cell
proliferation in DF-1 cells. Furthermore, it was identified that
gga-miR-375 inhibited the cell cycle by maintaining DF-1 cells
within the G1 phase and decreased cell proliferation,
while YAP1 promoted DF-1 cell transition from G1 to
S/G2 phase. It was further demonstrated that the
knockdown of gga-miR-375 expression promoted the cell cycle and
cell proliferation by targeting YAP1. Therefore, the present
results provide novel insights on the cell cycle and cell
proliferation regulated by gga-miR-375. Furthermore, the critical
role of gga-miR-375 and YAP1 in the cell cycle and cell
proliferation was identified in DF-1 cells.
Materials and methods
Cell culture
DF-1 cells, an immortalized chicken embryo
fibroblast cell line, were purchased from the American Type Culture
Collection. Cells were cultured in DMEM supplemented (Gibco; Thermo
Fisher Scientific, Inc.) with 10% FBS (Gibco; Thermo Fisher
Scientific, Inc.) and 1% penicillin/streptomycin (Gibco; Thermo
Fisher Scientific, Inc.) in a humidified incubator at 37˚C with 5%
CO2.The DF-1 cell line is a classic model for in
vitro research (34-36).
Virus and plasmid
The GD1109 strain of ALV-J and the PRK5-Flag were
gifted from Professor Wencheng Lin, South China Agricultural
University. The PRK5-Flag-YAP1 plasmid has been previously
constructed and preserved (29). The
cDNA of DF-1 cells was used to amplify YAP1 gene which was cloned
into the pRK5-FLAG vector. The primers for YAP1 amplification are
as follows: 5'-ATGGATCCCGGGCAGCCTCA-3' and 5'-AGTTTTCTTGGTTATAG-3'.
The groups of pRK5-FLAG vector and mock were the control groups.
For each transfection, 2 µg of plasmid were respectively
transfected in a 6-well plate for 48 h using
Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) in accordance with the manufacturer's protocol.
Western blot analysis was used to detect the efficiency of cell
transfection. 48 h after cell transfection, subsequent experiments
were performed.
RNA oligoribonucleotides and cell
transfection
gga-miR-375 mimic and its negative control miRNA
(gga-miR-NC) or anti-gga-miR-375 and anti-gga-miR-con inhibitor or
siRNA-YAP1 and its negative control siRNA-scramble (siRNA-Scra)
were purchased from Shanghai GenePharma Co., Ltd. The RNA
oligoribonucleotides are listed in Table
I. To prove YAP1 is a mediator of gga-miR-375, the following
groups were set up: Anti-gga-miR-375; anti-gga-miR-375 +
siRNA-YAP1; and anti-gga-miR-con + siRNA-YAP1. DF-1 cells
(5x104 cells per well) were cultured in 6-well plates
overnight at 37˚C prior to transfection. Transfection was performed
using X-tremeGENE small interfering (si)RNA transfection reagent
(Roche Diagnostics GmbH) in a 6-well plate according to the
manufacturer's protocol, and used at the concentration of 40 nM for
each transfection (29). Following
48 h of transfection, reverse transcription-quantitative PCR
(RT-qPCR) and western blotting were conducted.
 | Table IRNA oligoribonucleotides used for
cell transfection. |
Table I
RNA oligoribonucleotides used for
cell transfection.
| Gene | Sequence
(5'-3') |
|---|
| gga-miR-375 | Sense:
UUUGUUCGUUCGGCUCGCGUUA |
| Antisense:
UAACGCGAGCCGAACGAACAAA |
| gga-miR-NC | Sense:
UUGUACUACACAAAAGUACUG |
| Antisense:
GUACUUUUGUGUAGUACAAUU |
|
Anti-gga-miR-con | Sense:
CAGUACUUUUGUGUAGUACAA |
|
Anti-gga-miR-375 | Sense:
UAACGCGAGCCGAACGAACAAA |
| siRNA-YAP1 | Sense:
GGACAGAGAUACUUCCUUATT |
| Antisense:
UAAGGAAGUAUCUCUGUCCTT |
| siRNA-Scra | Sense:
UUCUCCGAACGUGUCACGUTT |
| Antisense:
ACGUGACACGUUCGGAGAATT |
Infection experiments
ALV-J infection experiments included the
experimental and the mock infected control group, which were
performed in 6-well plates. 2 ml DMEM with 1.0x105 cells
were added into each well. Once the density of the cells reached
70-80%, the experimental group was infected with a multiplicity of
infection of 1 for ALV-J at 37˚C and viral infection was detected
at 24, 48 and 72 h.
RNA isolation and RT-qPCR
TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) was used to extract total RNA from DF-1
cells. RNA was reverse transcribed into cDNA using the PrimeScript
RT reagent kit (Takara Bio, Inc.) according to the manufacturer's
protocol. The following RT conditions were used: Initial
denaturation at 37˚C for 15 min, followed by 85˚C for 5 sec and 4˚C
for 5 min. The relative expression levels of miRNAs were determined
using TaqMan miRNA assay (Thermo Fisher Scientific, Inc.) on an
CFX96 Touch™ RT PCR detection system (Bio-Rad Laboratories, Inc.).
The following thermocycling conditions were used for this qPCR:
Initial denaturation at 95˚C for 15 min, followed by 40 cycles at
95˚C for 10 sec and at 60˚C for 60 sec. The relative expression
levels of mRNA were quantified using the SYBR Premix Ex Taq (Takara
Bio, Inc.) according to the manufacturer's protocol. The following
thermocycling conditions were used for this qPCR: Initial
denaturation at 95˚C for 3 min, followed by 40 cycles at 95˚C for 5
sec and at 60˚C for 30 sec. β-actin and U6 were used for mRNA and
miRNA normalization, respectively. Specific primers were purchased
from Sangon Biotech Co., Ltd. and are listed in Table II. The relative mRNA expression was
calculated using the 2-ΔΔCq method
(37).
 | Table IIPrimers used for reverse
transcription-quantitative PCR. |
Table II
Primers used for reverse
transcription-quantitative PCR.
| Gene | Primers
(5'-3') |
|---|
| YAP1 | F:
GAACTCAGCATCAGCCATGA |
| R:
CTACGGAGAGCCAATTCCTG |
| gga-miR-375 |
TGTTCGTTCGGCTCGCGTTA |
| β-actin | F:
CTGGCACCTAGCACAATGAA |
| R:
CTGCTTGCTGATCCACATCT |
| U6 | F:
GCCTGGACTGAGTAAGAGCG |
| R:
GCCCCTTTTTGTCCGTTTCC |
Cell proliferation and the cell cycle
assay
DF-1 cells (4,000 cells/well) were cultured in
96-well plates and when the cell density reached 70-80%,
transfection was performed. A total of 10 µl Cell Counting Kit-8
(CCK-8; Dojindo Molecular Technologies, Inc.) reagent was added to
each well 24, 48 and 72 h after transfection and then incubated at
37˚C for 4 h according to the manufacturer's instructions. The
absorbance was recorded at 450 nm.
The cell cycle assay was performed in 6-well plates
and when the cell density reached 70-80%, transfection was
performed. Flow cytometry was used to evaluate the distribution of
cells in different phases of the cell cycle, basing the evaluation
on the DNA content of 500 µl 1% propidium iodide (PI; Thermo Fisher
Scientific, Inc.)-stained nuclei. After transfection for 48 h,
cells were trypsinized and resuspended in 75% ethanol at a density
of 1x106 cells/ml, overnight at 4˚C, resuspended in PBS
and dyed using the cell cycle dye solution (Cell Cycle and
Apoptosis Analysis kit; Nanjing KeyGen Biotech Co., Ltd.) for 30
min at 37˚C in the dark. A flow cytometry (BD FACScalibur; BD
Bioscience) was used to evaluate the cell cycle. Each group
contained three replicate wells and all of the experiments were
repeated independently three times. The results were analyzed using
CellQuest Pro 4.0.2 (BD Biosciences).
Western blot analysis
gga-miR-375 mimic, pRK5-Flag-YAP1, small interfering
(si)RNA-YAP1 and gga-miR-375 inhibitor were transfected into DF-1
cells and subsequently subjected to western blot analysis. Proteins
were homogenized or lysed in ice-cold RIPA lysis buffer (Santa Cruz
Biotechnology, Inc.). The concentration of the protein was examined
using Bradford protein assay kit (Bio-Rad Laboratories, Inc.).
Protein samples (40 µg/lane) were separated by 12% SDS-PAGE and
transferred to PVDF membranes (EMD Millipore). The membranes were
blocked in Tris-buffered saline with 5% non-fat milk and 0.5% BSA
for 1 h at room temperature. Membranes with corresponding proteins
were incubated with the polyclonal rabbit anti-YAP1 primary
antibody (cat. no. 13584-1-AP; 1:1,000; ProteinTech Group, Inc.),
polyclonal rabbit anti-phospho-YAP1 primary antibody (cat. no.
13008; 1:11,000; Cell Signaling Technology, Inc.), polyclonal
rabbit anti-cyclin D1 primary antibody (cat. no. 55506; 1:1,000;
Cell Signaling Technology, Inc.), polyclonal mouse anti-p53 primary
antibody (cat. no. 2524; 1:11,000; Cell Signaling Technology,
Inc.), polyclonal rabbit anti-cyclin E primary antibody (cat. no.
4129; 1:1,000; Cell Signaling Technology, Inc.), monoclonal mouse
anti-p27 primary antibody (the antibody was generated and
generously provided by Professor Wencheng Lin; 1:1,000), polyclonal
rabbit anti-MST1 primary antibody (cat. no. 3682; 1:1,000; Cell
Signaling Technology, Inc.), polyclonal rabbit anti-SAV1 primary
antibody (cat. no. 3507; 1:1,000; Cell Signaling Technology, Inc.),
polyclonal rabbit anti-MOB1 primary antibody (cat. no. 3863;
1:1,000; Cell Signaling Technology, Inc.), polyclonal rabbit
anti-LATS1 primary antibody (cat. no. 9153; 1:1,000; Cell Signaling
Technology, Inc.), monoclonal rabbit anti-GAPDH primary antibody
(cat. no. 2118; 1:1,000; Cell Signaling Technology, Inc.) and
polyclonal mouse anti-β-actin primary antibody (cat. no. 33308;
1:1,000; Bioss Antibodies, Inc.) overnight at 4˚C. The membranes
were then incubated with a horseradish peroxidase (HRP)-conjugated
mouse polyclonal anti-rabbit IgG (H+L)-(cat. no. 0293R; 1:500;
Bioss Antibodies, Inc) and goat polyclonal anti-mouse IgG-HRP (cat.
no. 7076; 1:11,000; Cell Signaling Technology, Inc.) for 1.5 h at
RT and washed four times using 0.1% Tween-20 solution. Blots were
visualized with SuperSignal™ West Femto Maximum Sensitivity
Substrate (Thermo Fisher Scientific, Inc.) and bands were
quantified using Image-Pro Plus v6.0 (Media Cybernetics, Inc.).
Luciferase reporter assay
The putative target genes of gga-miR-375 were
predicted using public available algorithms, including TargetScan
(http://targetscan.org) and miRDB (http://www.mirdb.org). The predicted gga-miR-375
binding sites in the wild-type (WT) 3'UTR of YAP1 and the
corresponding mutant type (MUT) gga-miR-375 binding sites were
cloned into a pGL3 vector (Promega Corporation). For the luciferase
reporter assay, DF-1 cells (1x105 cells/well) were
seeded in 96-well plates and co-transfected with 300 ng WT-YAP1 or
MUT-YAP1, and 100 nM of gga-miR-375 mimic or gga-miR-NC using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) in accordance with the manufacturer's protocol.
After 48 h transfection, the relative luciferase activities were
measured using the dual-luciferase reporter assay system (Promega
Corporation). Renilla luciferase activity was used for
normalization.
Statistical analysis
Statistical analysis was performed using SPSS 13.0
statistical software (SPSS, Inc.). A one-way or two-way ANOVA
followed by the Tukey's post hoc tests were used to compare
differences between two groups. A Student's t-test was used to
compare differences between two groups. P<0.05 was considered to
indicate a statistically significant difference. For each separate
set of experiments, three independent replicates were evaluated.
Data are presented as the mean ± SD.
Results
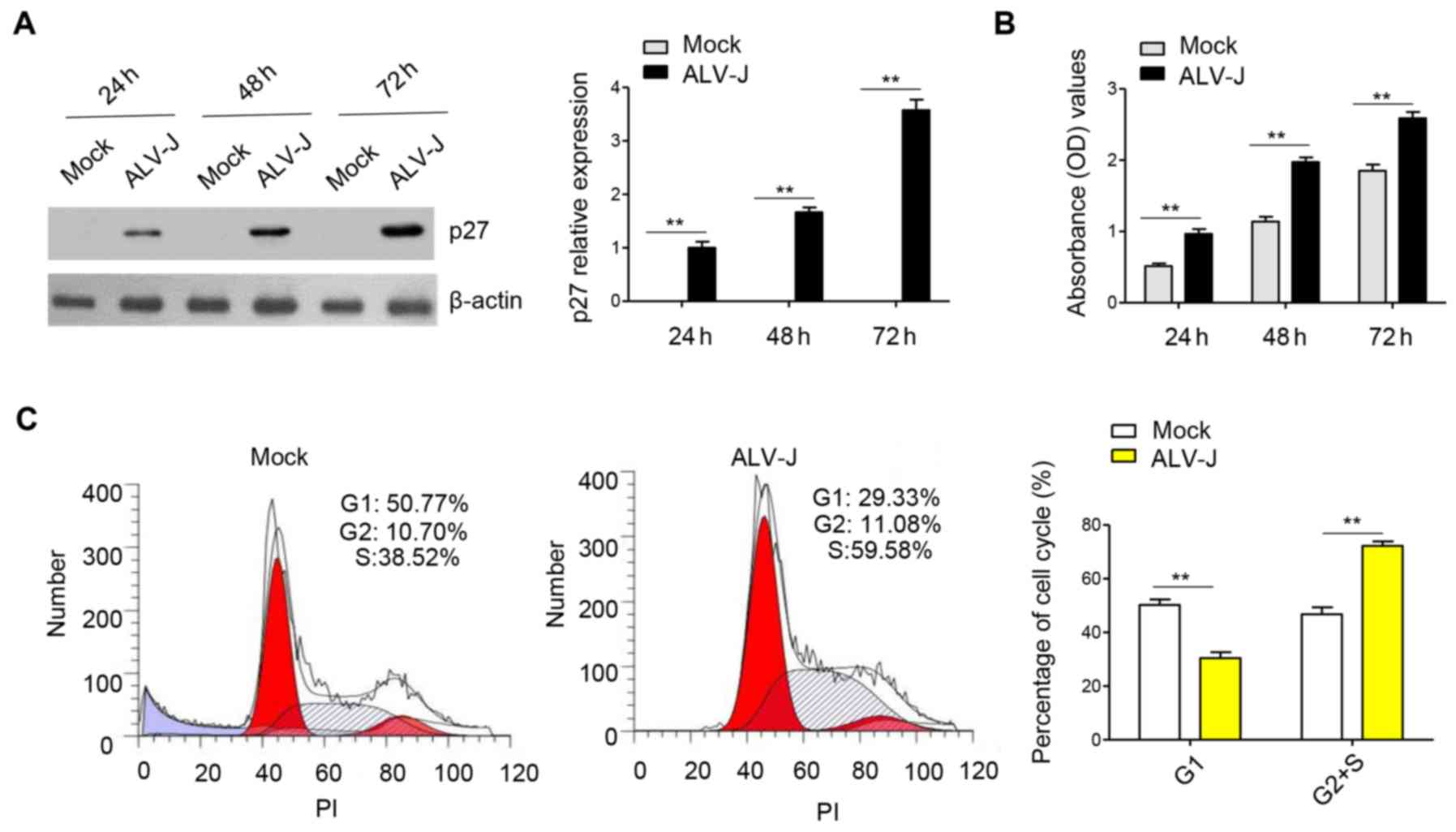
Infection with ALV-J promotes the cell
cycle and cell proliferation in DF-1 cells
In order to investigate the effect of ALV-J
infection on cell proliferation, DF-1 cells were infected with
ALV-J and the capsid protein p27 of ALV-J was used to track the
viral replication (Fig. 1A). CCK-8
is a widely used method for cell proliferation based on water
soluble tetrazolium-8; therefore, a CCK-8 assay was used to
determine proliferation of DF-1 cells. It was identified that ALV-J
infection could significantly promote cell proliferation compared
with the mock group (Fig. 1B).
Furthermore, the cell cycle was assessed using flow cytometry and
the results indicated that the percentage of cells in G1
phase was decreased, while the percentage of cells in S and
G2 phase was significantly increased in the ALV-J
infected group compared with the mock group (Fig. 1C). Thus, the results indicated that
ALV-J infection in vitro promoted proliferation and the cell
cycle by accelerating the transition from G1 phase to
S/G2 phase.
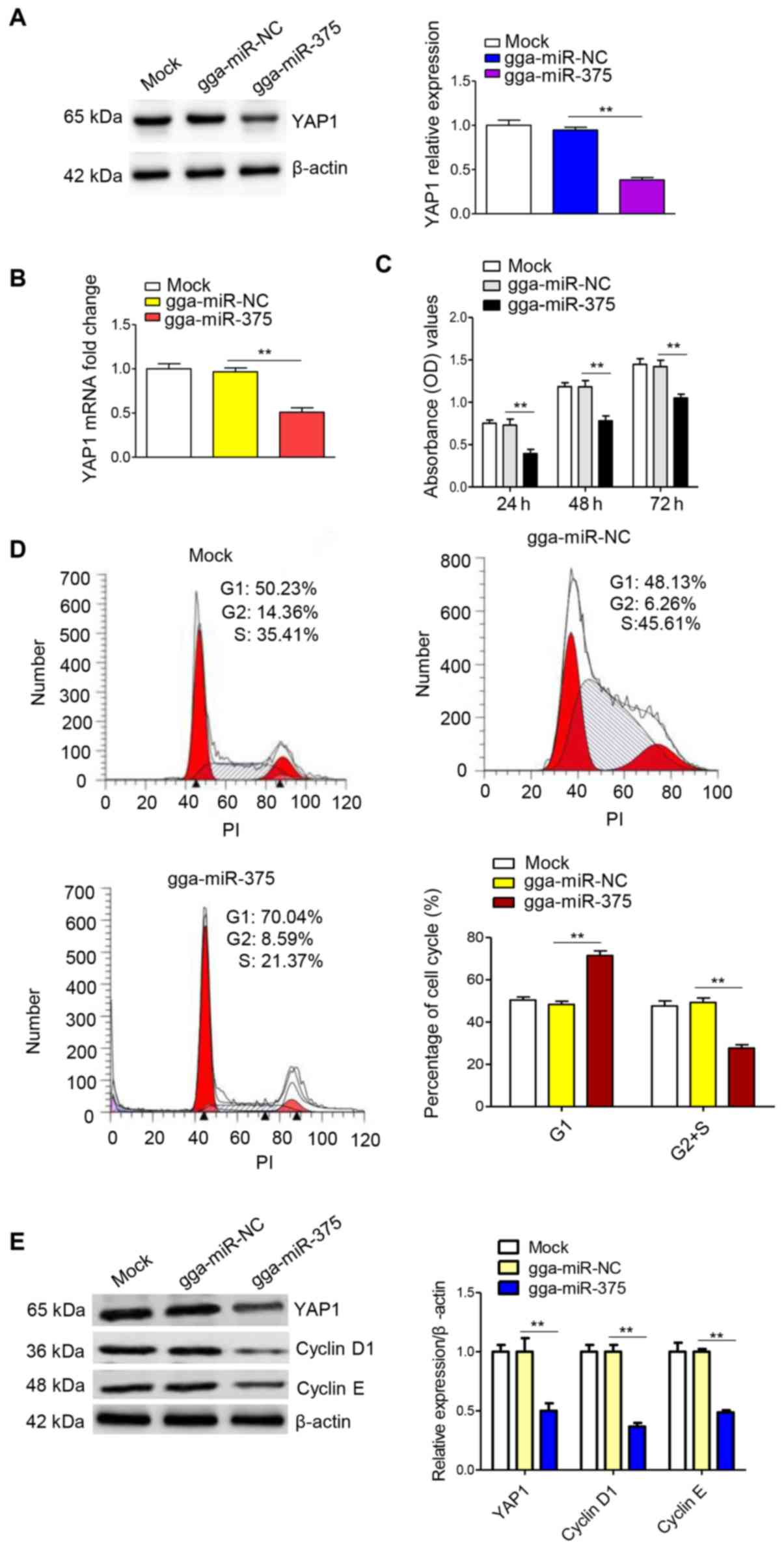
Overexpression of gga-miR-375 inhibits
the cell cycle and proliferation of DF-1 cells
A previous study showed that ALV-J infection could
downregulate gga-miR-375, while upregulating YAP1 expression
(18). In addition, YAP1 was
confirmed to be a target gene of gga-miR-375 in a previous study
(29). In the present study, the
effect of ALV-J infection on gga-miR-375 and YAP1 expression levels
was analyzed, and a luciferase reporter gene assay experiment was
performed to identify whether YAP1 is a target gene of gga-miR-375
(Fig. S1).
Next, the effect of gga-miR-375 overexpression on
YAP1, cell proliferation and the cell cycle was assessed. Western
blot analysis and RT-qPCR results suggested that gga-miR-375
overexpression significantly decreased YAP1 protein (Fig. 2A) and mRNA expression (Fig. 2B) levels. To determine the effect of
gga-miR-375 overexpression on cell proliferation, a CCK-8 assay was
performed and the results indicated that overexpression of
gga-miR-375 could significantly inhibit cell proliferation compared
with the NC group (Fig. 2C). In
addition, flow cytometry was conducted to investigate the effect of
gga-miR-375 on the cell cycle, 48 h after transfection in DF-1
cells. It was identified that the overexpression of gga-miR-375
could significantly increase the percentage of cells in
G1 phase, while the percentage of cells in
S/G2 phase was significantly decreased compared with the
NC group (Fig. 2D), which was
consistent with the cell proliferation results. In addition,
overexpression of gga-miR-375 downregulated the protein expression
levels of YAP1, cyclin D1 and cyclin E (Fig. 2E). Furthermore, overexpression of
gga-miR-375 upregulated the protein expression of p53 (Fig. S2A). Collectively, the results
demonstrated that overexpression of gga-miR-375 downregulated cell
proliferation and the cell cycle by sequestering the cells in
G1 phase.
Knockdown of gga-miR-375 promotes the
cell cycle and cell proliferation in DF-1 cells
The expression of gga-miR-375 was knocked down using
a gga-miR-375 inhibitor, and the effect on cell proliferation and
cell cycle was subsequently investigated. DF-1 cells were
transfected with either gga-miR-375 inhibitor or an inhibitor
control, and a mock group was used as the control group. The
RT-qPCR results demonstrated that the gga-miR-375 inhibitor could
significantly downregulate the mRNA expression of gga-miR-375
compared with the control group (Fig.
3A). In addition, western blot analysis and RT-qPCR results
suggested that knockdown of gga-miR-375 significantly upregulated
the protein and mRNA expression levels of YAP1 (Fig. 3B and C). Cell proliferation was determined using
a CCK-8 assay, and the results indicated that knockdown of
gga-miR-375 could significantly promote proliferation compared with
the control group (Fig. 3D).
Furthermore, flow cytometry was performed to assess the effect of
gga-miR-375 knockdown on the cell cycle, 48 h after transfection in
DF-1 cells, and it was identified that knockdown of gga-miR-375
significantly decreased the number of cells in G1 phase,
but increased the number of cells in S/G2 phase
(Fig. 3E). In addition, knockdown of
gga-miR-375 in DF-1 cells upregulated the total protein expression
levels of YAP1, cyclin D1 and cyclin E (Fig. 3F). It was also identified that the
knockdown of gga-miR-375 downregulated the protein expression of
p53 (Fig. S2B). Therefore, the
results suggested that knockdown of gga-miR-375 could promote cell
proliferation and cell cycle progression by increasing the number
of DF-1 cells from G1 to S/G2 phase.
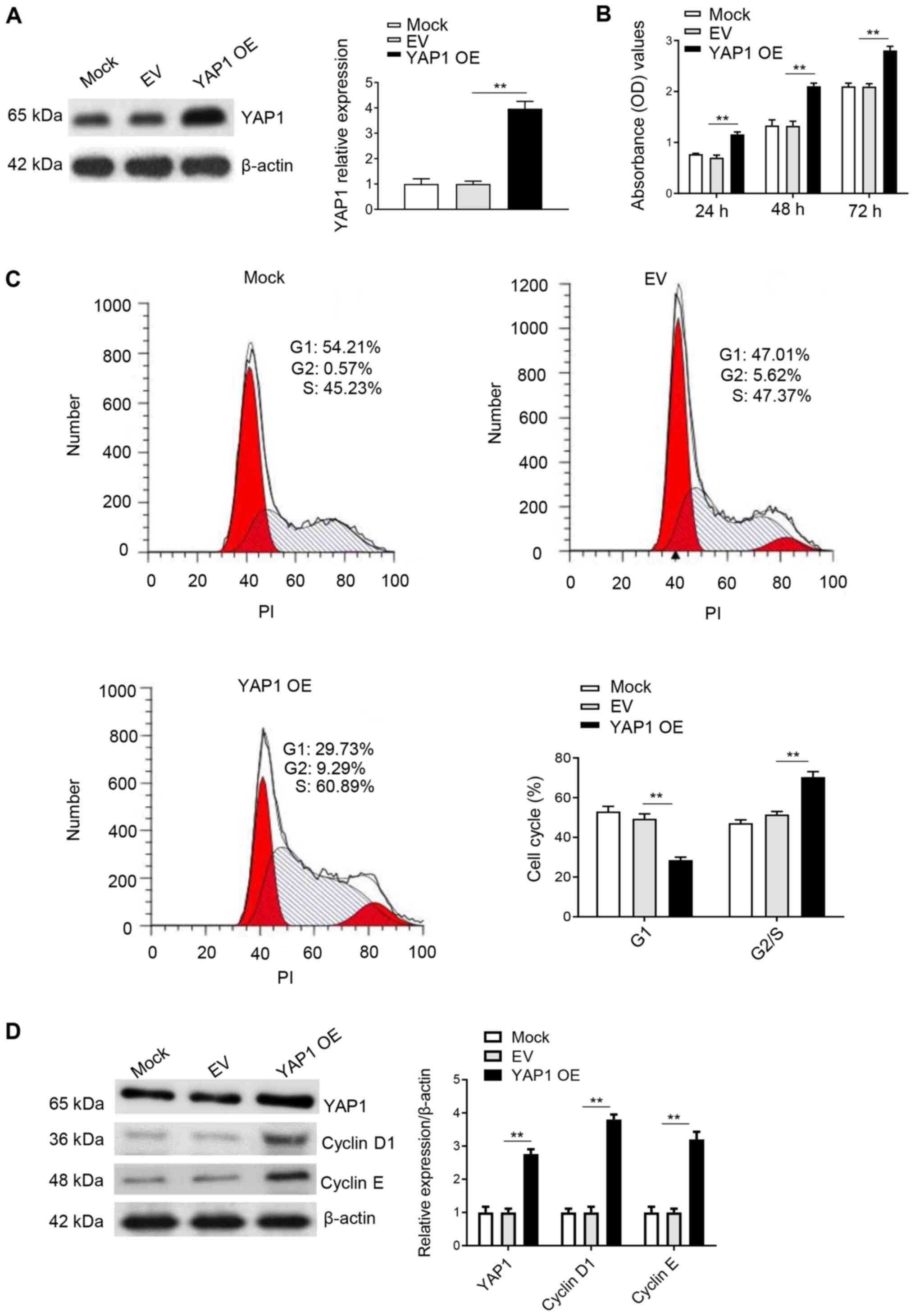
Overexpression of YAP1 promotes the
cell cycle progression and proliferation in DF-1 cells
DF-1 cells were cultured in 6-well plates,
transfected with pRK5-Flag-YAP1 overexpression plasmid and
collected 48 h after transfection. Western blot analysis results
indicated that YAP1 was successfully overexpressed in DF-1 cells
(Fig. 4A). Moreover, the CCK-8 assay
results identified that the transfection of YAP1 for 24, 48 and 72
h could significantly promote cell proliferation compared with the
control groups (Fig. 4B).
Furthermore, the effect of YAP1 on the cell cycle was determined
using flow cytometry 48 h after transfection. It was identified
that overexpression of YAP1 in DF-1 cells significantly decreased
the percentage of cells in G1 phase, while the
percentage of cells in S and G2 phase was significantly
increased compared with the control group (Fig. 4C). In addition, overexpression of
YAP1 increased the protein expression levels of cyclin D1 and
cyclin E (Fig. 4D). Thus, it was
speculated that the overexpression of YAP1 promoted cell
proliferation and the cell cycle by accelerating the transition of
DF-1 cells from G1 to G2/S phase.
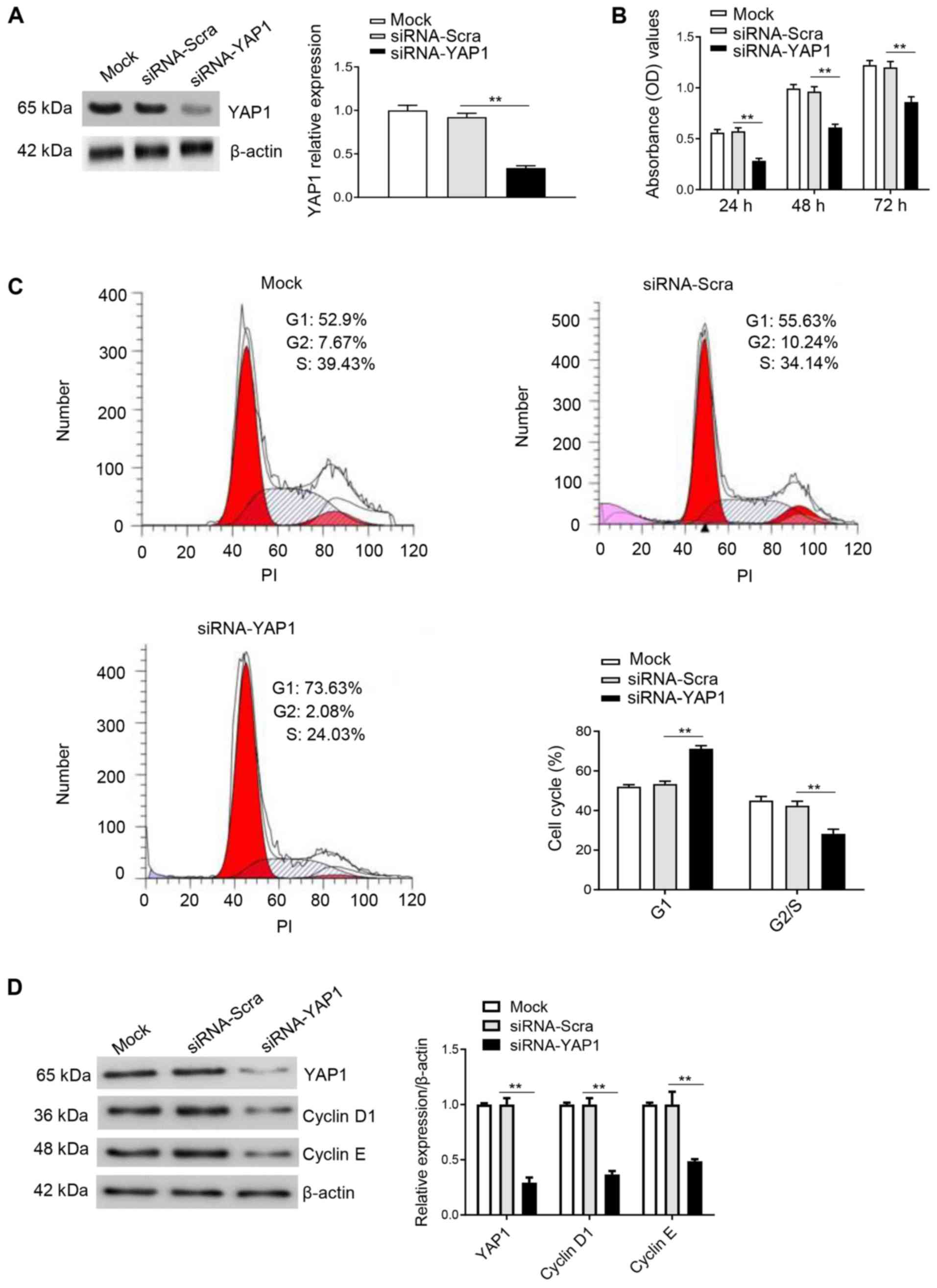
Knockdown of YAP1 inhibits cell cycle
progression and proliferation in DF-1 cells
YAP1 was knocked down using siRNA, and its effect on
cell proliferation and the cell cycle was evaluated. Either
siRNA-YAP1 or siRNA-Scra control were transfected into DF-1 cells.
Western blot analysis results indicated that YAP1 expression was
significantly decreased in the siRNA-YAP1 transfection group
(Fig. 5A). Moreover, the CCK8 assay
results identified that knockdown of YAP1 could significantly
inhibit cell proliferation compared with the control group
(Fig. 5B). Furthermore, the effect
of YAP1 knockdown on the cell cycle was observed using flow
cytometry 48 h after transfection in DF-1 cells. The results
suggested that knockdown of YAP1 in DF-1 cells significantly
upregulated the number of cells in G1 phase and
downregulated the number of cells in S/G2 phase
(Fig. 5C). Furthermore, knockdown of
YAP1 inhibited the protein expression levels of cyclin D1 and
cyclin E (Fig. 5D). Collectively,
the results suggested that knockdown of YAP1 inhibited
proliferation and the cell cycle by maintaining DF-1 cells in the
G1 phase.
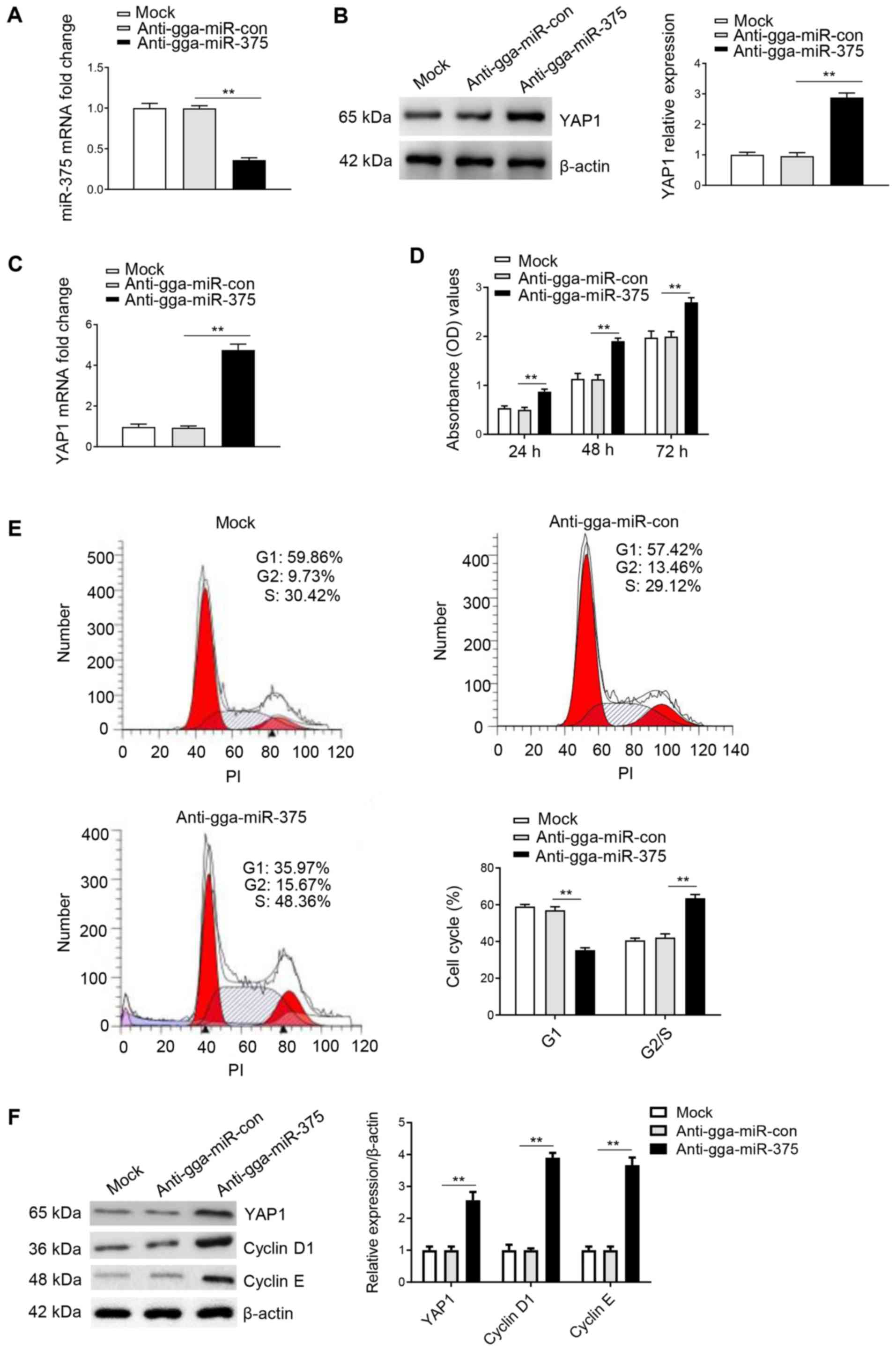
Knockdown of gga-miR-375 expression
promotes the cell cycle and cell proliferation by targeting
YAP1
To further assess whether YAP1 is a mediator of
gga-miR-375, the following groups were used: Anti-gga-miR-375;
anti-gga-miR-375 + siRNA-YAP1; and anti-gga-miR-con + siRNA-YAP1.
In addition, anti-gga-miR-con, anti-gga-miR-375 + siRNA-Scra and
anti-gga-miR-con + siRNA-Scra were used as the control groups,
while the mock group was used as a negative control. It was
identified that gga-miR-375 was successfully silenced by
transfection of anti-gga-miR-375 (Fig.
6A), and western blot analysis results indicated that
inhibiting the expression of gga-miR-375 through transfection of
anti-gga-miR-375 could significantly upregulate the protein
expression of YAP1 compared with the anti-gga-miR-con group
(Fig. 6B). The results also
indicated that transfection of anti-gga-miR-375 could significantly
increase the protein expression of YAP1 compared with the
anti-gga-miR-con group (Fig. 6C).
Moreover, the CCK-8 assay results identified that the
anti-gga-miR-375 only group or anti-gga-miR-375 + siRNA-Scra group
could significantly promote cell proliferation from 24-72 h
(Fig. 6D). However, proliferation
was decreased after co-transfection with anti-gga-miR-375 +
siRNA-YAP1 compared with the anti-gga-miR-375 + siRNA-Scra group
(Fig. 6D), thus indicating that YAP1
may be a mediator of gga-miR-375 in regulating cell
proliferation.
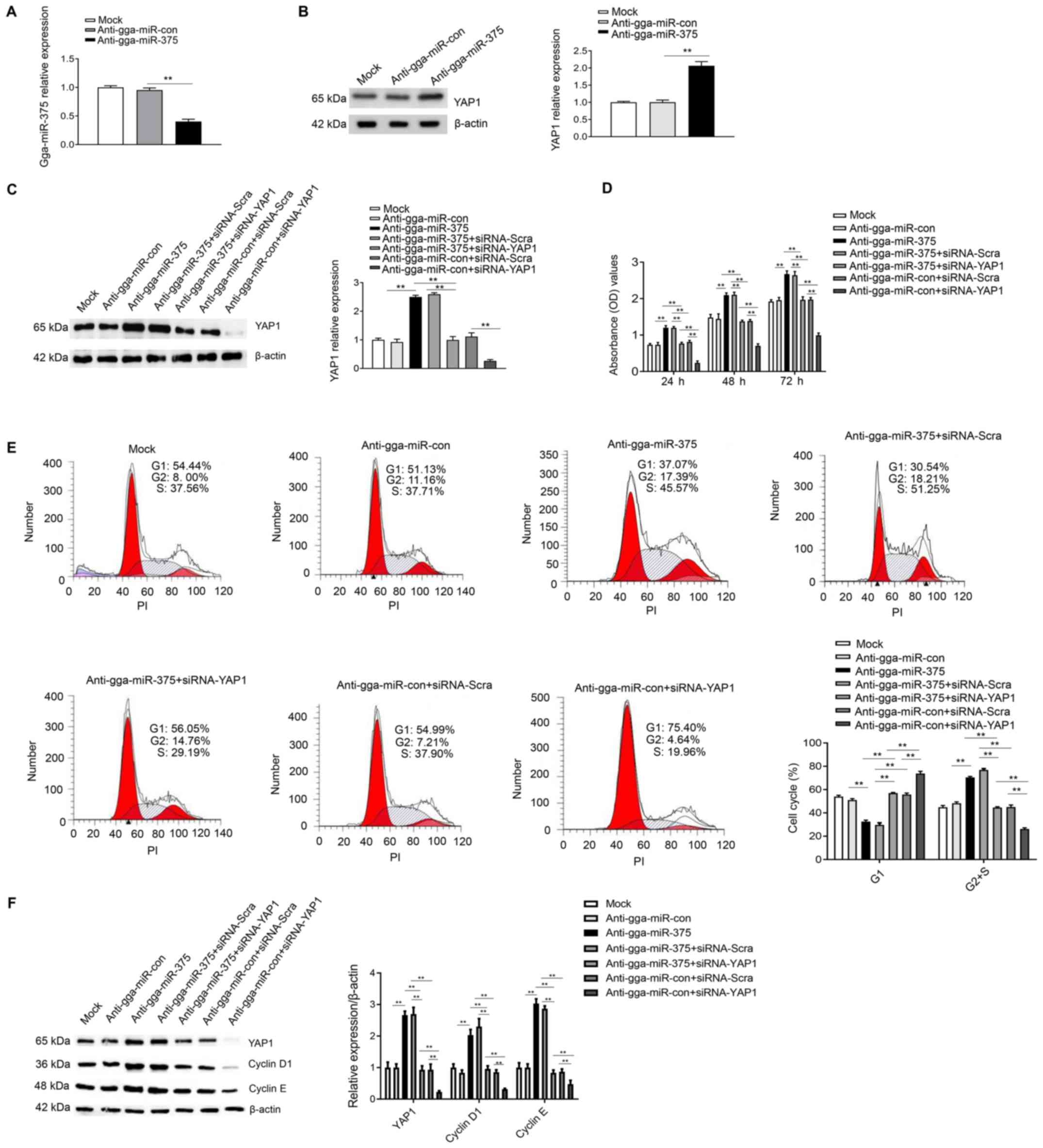 | Figure 6Knockdown of gga-miR-375 expression
promotes the cell cycle and cell proliferation by targeting YAP1.
(A) DF-1 cells transfected with anti-gga-miR-375 had decreased
gga-miR-375 mRNA expression at 48 h as assessed by reverse
transcription-quantitative PCR. Anti-gga-miR-con and mock groups
are the controls. (B) Protein expression of YAP1 was measure after
DF-1 cells were transfected for 48 h with anti-gga-miR-375. (C)
Protein expression levels of YAP1 after 48 h transfection with
anti-gga-miR-375 or co-transfected with anti-gga-miR-375 +
siRNA-YAP1. (D) Knockdown of gga-miR-375 promoted cell
proliferation by targeting YAP1, as identified by Cell Counting
Kit-8 assay results after 24, 48 and 72 h. (E) Flow cytometry
results indicated that knockdown of gga-miR-375 expression promoted
the cell cycle by targeting YAP1. The cell cycle assay was
performed after knockdown of gga-miR-375 or gga-miR-375 + YAP1 in
DF-1 cells and evaluated with a FACSCalibur flow cytometer 48 h
after transfection. (F) DF-1 cells transfected with
anti-gga-miR-375 or anti-gga-miR-375 + siRNA-YAP1 were harvested 48
h after transfection for western blot analysis with antibodies
against YAP1, cyclin D1, cyclin E and β-actin. Data are presented
as the mean ± SD of three independent experiments.
**P<0.01. YAP1, yes-associated protein 1;
miR, microRNA; siRNA, small interfering RNA; PI, propidium iodide;
OD, optical density; Scra, scramble. |
The cell cycle analysis results demonstrated that
co-transfection of anti-gga-miR-375 + siRNA-YAP1 could inhibit the
cell cycle by maintaining cells in G1 phase compared
with the anti-gga-miR-375 + siRNA-Scra group (Fig. 6E), therefore suggesting that YAP1 is
a mediator of gga-miR-375 in regulating cell cycle. Furthermore,
western blot analysis results identified that co-transfection of
anti-gga-miR-375 + siRNA-YAP1 could decrease the protein expression
levels of YAP1, cyclin D1 and cyclin E compared with the
anti-gga-miR-375 + siRNA-Scra group (Fig. 6F). Collectively, the results
indicated that knockdown of gga-miR-375 expression promoted the
cell cycle and cell proliferation by targeting YAP1.
It has been shown that serine phosphorylation of
YAP1 leads to YAP1 downregulation by cytoplasm sequestration and/or
degradation, while non-phosphorylated (p)-YAP1 is activated and
enters the nucleus (38). Moreover,
YAP1 acts as a transcriptional activator and can bind to numerous
transcription factors to regulate the expression levels of various
genes (23,39). Therefore, the present study detected
p-YAP1 expression after ALV-J infection, knockdown of gga-miR-375
or overexpression of gga-miR-375. It was found that ALV-J infection
or knockdown of gga-miR-375 could decrease p-YAP1 expression, while
the overexpression of gga-miR-375 could increase p-YAP1 expression
(Fig. S3). The results also
demonstrated that after ALV-J infection or knockdown of
gga-miR-375, p-YAP1 expression was downregulated, thus indicating
that YAP1 is not degraded in the cytoplasm and this leads to an
increase of YAP1. Since YAP1 is a member of the Hippo signaling
pathway, the present study detected the expression levels of other
members of this signaling pathway, including macrophage stimulating
1 (MST1), MOB kinase activator 1 (MOB1) and large tumor suppressor
kinase 1 (LATS1), after overexpression gga-miR-375. It was
demonstrated that the overexpression of gga-miR-375 had no effect
on the protein expression levels of MST1, salvador family WW domain
containing protein 1, MOB1 and LATS1 (Fig. S4). Therefore, the results suggested
that gga-miR-375 directly affected YAP1, which in turn affected the
cell cycle and cell proliferation, and ultimately tumor
formation.
Discussion
ALV-J is a retrovirus that commonly causes natural
infection in chickens, and possibly neoplastic disease and other
production problems (40). The cell
cycle is regulated by a large number of transcription factors and
signaling molecules, and affects cell proliferation (41); endless proliferation of cells is a
hallmark of cancer (42).
Furthermore, tumors are a type of disease characterized by
uncontrolled cell proliferation. The cell cycle is a continuous and
precise process, and it serves a key role in monitoring and
regulating the development of tumor formation. Moreover,
abnormality of cell cycle regulation is closely associated with the
occurrence and development of tumors (43). Therefore, an in-depth study of cell
cycle regulation would help to further the understanding of the
mechanism involved in tumor formation. As ALV-J infection causes
tumors in chickens, it was hypothesized that ALV-J infection could
affect cell proliferation and the cell cycle. Therefore, the
present study investigated the effect of ALV-J infection on these
cellular processes in the DF-1 cell line. The results demonstrated
that ALV-J infection promoted cell proliferation and the cell cycle
by downregulating G1 phase, but upregulating
S/G2 phase.
miRNAs serve an important role in numerous
biological processes, including cell development, differentiation
and proliferation, and may target oncogenes or acts as a tumor
suppressor in the cell cycle (44).
In the process of cell proliferation, miRNA can serve as a tumor
suppressor or oncogene regulator, depending on the tissue and
cellular environment (45). A
previous study revealed that gga-miR-375 is significantly
downregulated in ALV-J-induced tumors in the livers of chickens
(29). Considering that in the
present study, ALV-J infection was identified to promote cell
proliferation and the cell cycle, it was speculated that
gga-miR-375 may affect these cellular processes and could regulate
the target gene YAP1, which may serve a key role in controlling
this effect. Moreover, p53 is a tumor suppressor gene and its
primary role is to arrest the cell cycle (46). In the present study, overexpression
of gga-miR-375 was also identified to increase p53 protein
expression, while knockdown of gga-miR-375 decreased p53 protein
expression. Therefore, the results indicated that the
downregulation of gga-miR-375 by ALV-J infection decreased the
protein expression of p53, which affected the arrest of the cell
cycle.
The present study also examined the effect of
gga-miR-375 and YAP1 on cell proliferation and the cell cycle. It
was identified that overexpression of gga-miR-375 inhibited cell
proliferation and the cell cycle by reducing cell transformation
from G1 phase to S/G2 phase, while knockdown
of gga-miR-375 promoted these cellular processes by increasing
cellular transition from G1 phase to S/G2
phase. YAP1 is a target gene of gga-miR-375, and gga-miR-375 serves
a suppressive role in the regulation of YAP1 (18,47).
Furthermore, YAP1 has been reported to be a core protein in the
Hippo pathway that affects the cell cycle (48-50).
In relation to the target gene of gga-miR-375, YAP1, the flow
cytometry cell cycle analysis results demonstrated that YAP1
overexpression could increase the transition of DF-1 cells from
G1 to S/G2 phase. Moreover, the CCK-8 assay
results suggested that the overexpression of YAP1 could increase
cell proliferation from 24-72 h. However, knockdown of YAP1
decreased cell proliferation and inhibited the cell cycle by
maintaining DF-1 cells at G1 phase.
Cyclin D1 and cyclin E play a key role in regulating
the cell cycle, and the downregulation of these factors results in
the arrest of the cell cycle (51,52). The
present results suggested that gga-miR-375 overexpression in DF-1
cells downregulated the total protein expression levels of YAP1,
cyclin D1 and cyclin E. However, knockdown of gga-miR-375 in DF-1
cells upregulated the protein expression of YAP1, and also
increased the protein expression levels of cyclin D1 and cyclin E.
Moreover, overexpression of YAP1 increased the expression levels of
cyclin D1 and cyclin E, while knockdown of YAP1 inhibited the
expression levels of these factors. Therefore, the present results
suggested that downregulation of gga-miR-375 and upregulation of
YAP1 by ALV-J infection affected the cell cycle by altering the
protein expression levels of cyclin D1 and cyclin E. Numerous
studies have shown that cyclin D1 and cyclin E can mediate the
development of cancer (53,54); therefore, as gga-miR-375 and YAP1 can
affect the expression levels of these two proteins, this may be the
mechanism through which gga-miR-375 targets YAP1 to promote tumor
formation.
The present results identified that YAP1 may be a
mediator of gga-miR-375, and that gga-miR-375 could affect cell
proliferation and the cell cycle by targeting YAP1. Thus, the
results provided evidence that the cell cycle and cell
proliferation may be affected by gga-miR-375 targeting YAP1 in DF-1
cells. In addition, it was demonstrated that after ALV-J infection
or knockdown of gga-miR-375, p-YAP1 expression was downregulated,
thus indicating that YAP1 is not degraded in the cytoplasm and this
leads to an increase of YAP1. It was further found that the
overexpression of gga-miR-375 had no effect on the protein
expression levels of MST1, salvador family WW domain containing
protein 1, MOB1 and LATS1. Therefore, the results suggested that
gga-miR-375 directly affected YAP1, which in turn affected the cell
cycle and cell proliferation, and ultimately tumor formation.
In conclusion, it was identified that gga-miR-375
inhibited proliferation and the cell cycle by maintaining DF-1
cells in G1 phase. Moreover, YAP1 could promote cell
proliferation and the cell cycle by increasing cellular transition
from G1 to S/G2 phase. The results also
indicated that gga-miR-375 inhibited these cellular processes by
targeting YAP1. Furthermore, downregulation of gga-miR-375 and
upregulation of YAP1 by ALV-J infection affected the cell cycle by
altering the protein expression levels of cyclin D1 and cyclin E,
which may be the possible mechanism through which gga-miR-375
targets YAP1 to promote tumor formation. Therefore, the results of
the present study provided further evidence of the role between
gga-miR-375 and YAP1 in proliferation and the cell cycle in DF-1
cells, thus suggesting that these cellular processes are affected
by gga-miR-375 targeting of YAP1, which may be an important
regulator of tumorigenesis.
Supplementary Material
Figure S1. Identification of YAP1 as a
target gene of gga-miR-375 and the expression levels of gga-miR-375
and YAP1 in DF-1 cells after ALV-J infection. (A) Seed region
complementary to gga-miR-375 and YAP1. (B) gga-miR-375 is important
for the relative luciferase activity of YAP1 (C) gga-miR-375 was
successfully overexpressed after transfection with gga-miR-375
mimic in DF-1 cells. (D) Reverse transcription-quantitative PCR
results identified that gga-miR-375 overexpression could
downregulate the mRNA expression of YAP1 48 h after transfection.
(E) Overexpression of gga-miR-375 could downregulate the protein
expression of YAP1 48 h after transfection using western blot
analysis. (F) ALV-J infection decreased gga-miR-375 expression 48 h
after transfection in DF-1 cells. ALV-J infection increases YAP1
(G) mRNA and (H) protein expression levels 48 h after transfection
in DF-1 cells. Data are presented as the mean ± SD of three
independent experiments. **P<0.01. ALV-j, avian
leukosis virus subgroup J; YAP1, Yes-associated protein 1; miR,
microRNA; wt, wild-type; mut, mutant; NC, negative control; 3'UTR,
3'untranslated region.
Figure S2. gga-miR-375 affects the
protein expression of p53 in DF-1 cells. (A) Overexpression of
gga-miR-375 upregulated the protein expression of p53. Cells were
transfected with gga-miR-375 mimic, gga-miR-NC or mock for 48 h and
western blot analysis with antibodies against p53 and β-actin was
performed. gga-miR-NC and mock groups were used as the control
groups. (B) Knockdown of gga-miR-375 downregulated the protein
expression of p53. DF-1 cells were transfected with gga-miR-375
inhibitor and harvested 48 h after transfection for western blot
analysis. Anti-gga-miR-con group and mock group were the control
groups. Data are presented as the mean ± SD of three independent
experiments. **P<0.01. miR, microRNA; NC, negative
control; con, control.
Figure S3. Effect of ALV-J infection,
gga-miR-375 overexpression or silencing gga-miR-375 on YAP1 and
p-YAP1. (A) ALV-J infection increased the expression of YAP1, but
decreased p-YAP1 at 48 h after infection in DF-1 cells. (B) Cells
were transfected with gga-miR-375 inhibitor and harvested 48 h
after transfection for western blot analysis with antibodies
against YAP1, p-YAP1 and β-actin. Anti-gga-miR-con and mock groups
were used as the control groups. (C) Cells were transfected with
gga-miR-375 and harvested 48 h after transfection for western blot
analysis with antibodies against YAP1, p-YAP1 and β-actin.
gga-miR-con group and mock group were the control groups. Data are
presented as the mean ± SD of three independent experiments.
**P<0.01. p, phosphorylated; miR, microRNA; con,
control; YAP1, Yes-associated protein 1.
Figure S4. Effect of gga-miR-375 on
the expression levels of MST1, SAV1, MOB1 and LATS1. Cells were
transfected with gga-miR-375 and harvested 48 h after transfection
for western blot analysis with antibodies against MST1, SAV1, MOB1,
LATS1 and GAPDH. gga-miR-NC group was the control group. Data are
presented as the mean ± SD of three independent experiments. n.s.,
not significant; NC, negative control; miR, microRNA; MST1,
Macrophage stimulating 1; MOB1, MOB kinase activator 1; LATS1,
large tumor suppressor kinase 1; SAV1, salvador family WW domain
containing protein 1.
Acknowledgements
Not applicable.
Funding
This study was supported by the National Natural
Science Foundation of China (grant nos. 31902252, 31972659,
31802185, 31672564 and 31472217), the China Postdoctoral Science
Foundation (grant no. 2019M652922), the International Science and
Technology Cooperation Project of Guangdong Province (grant no.
2016A050502042), the Construction Project of Modern Agricultural
Science and Technology Innovation Alliance in Guangdong Province
(grant no. 2018LM1112) and the Natural Science Foundation of
Guangdong Province (grant nos. 2019A1515012006, 2018B030315009 and
S2013030013313).
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
ZL, YY, YW, SC and QX conceived and designed the
original study. XZ conducted the statistical analysis and drafted
the manuscript. ZL, YY, YW, SC and QX contributed to the
acquisition and interpretation of data. SL and FC contributed to
the data interpretation and analysis. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Indiková I, Much P, Stipkovits L,
Siebert-Gulle K, Szostak MP, Rosengarten R and Citti C: Role of the
GapA and CrmA cytadhesins of mycoplasma gallisepticum in promoting
virulence and host colonization. Infect Immun. 81:1618–1624.
2013.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Jenkins C, Geary SJ, Gladd M and
Djordjevic SP: The mycoplasma gallisepticum OsmC-like protein
MG1142 resides on the cell surface and binds heparin. Microbiology.
153:1455–1463. 2007.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Kato M and Slack FJ: microRNAs: Small
molecules with big roles-C. elegans to human cancer. Biol Cell.
100:71–81. 2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Zhao Y, Wang Z, Hou Y, Zhang K and Peng X:
gga-miR-99a targets SMARCA5 to regulate mycoplasma gallisepticum
(HS strain) infection by depressing cell proliferation in chicken.
Gene. 627:239–247. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Poy MN, Eliasson L, Krutzfeldt J, Kuwajima
S, Ma X, Macdonald PE, Pfeffer S, Tuschl T, Rajewsky N, Rorsman P
and Stoffel M: A pancreatic islet-specific microRNA regulates
insulin secretion. Nature. 432:226–230. 2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ding L, Xu Y, Zhang W, Deng Y, Si M, Du Y,
Yao H, Liu X, Ke Y, Si J and Zhou T: MiR-375 frequently
downregulated in gastric cancer inhibits cell proliferation by
targeting JAK2. Cell Res. 20:784–793. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wang P, Xu LL, Li L, Ren S, Tang J, Zhang
M and Xu M: The microRNA-375 as a potentially promising biomarker
to predict the prognosis of patients with head and neck or
esophageal squamous cell carcinoma: A meta-analysis. Eur Arch
Otorhinolaryngol. 276:957–968. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Winther M, Alsner J, Tramm T, Baeksgaard
L, Holtved E and Nordsmark M: Evaluation of miR-21 and miR-375 as
prognostic biomarkers in esophageal cancer. Acta Oncol.
54:1582–1591. 2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Fan YP, Liao JZ, Lu YQ, Tian DA, Ye F,
Zhao PX, Xiang GY, Tang WX and He XX: MiR-375 and doxorubicin
co-delivered by liposomes for combination therapy of hepatocellular
carcinoma. Mol Ther-Nucleic Acids. 7:181–189. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
He J, Xie G, Tong J, Peng Y, Huang H, Li
J, Wang N and Liang H: Overexpression of microRNA-122 re-sensitizes
5-FU-resistant colon cancer cells to 5-FU through the inhibition of
PKM2 in vitro and in vivo. Cell Biochem Biophys. 70:1343–1350.
2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Shaker OG, Mohammed SR, Mohammed AM and
Mahmoud Z: Impact of microRNA-375 and its target gene SMAD-7
polymorphism on susceptibility of colorectal cancer. J Clin Lab
Anal. 32:2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Cheng Z, Liu J, Cui Z and Zhang L: Tumors
associated with avian leukosis virus subgroup J in layer hens
during 2007 to 2009 in China. J Vet Med Sci. 72:1027–1033.
2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Payne LN, Howes K, Gillespie AM and Smith
LM: Host range of Rous sarcoma virus pseudotype RSV(HPRS-103) in 12
avian species: Support for a new avian retrovirus envelope
subgroup, designated J. J Gen Virol. 73:2995–2997. 1992.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Meng F, Li Q, Zhang Y, Cui Z, Chang S and
Zhao P: Isolation and characterization of subgroup J Avian Leukosis
virus associated with hemangioma in commercial Hy-Line chickens.
Poult Sci. 97:2667–2674. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Liang X, Gu Y, Chen X, Li T, Gao Y, Wang
X, Fang C, Fang S and Yang Y: Identification and characterization
of a novel natural recombinant avian leucosis virus from Chinese
indigenous chicken flock. Virus Genes. 55:726–733. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Li X, Lin W, Chang S, Zhao P, Zhang X, Liu
Y, Chen W, Li B, Shu D, Zhang H, et al: Isolation, identification
and evolution analysis of a novel subgroup of avian leukosis virus
isolated from a local Chinese yellow broiler in south China. Arch
Virol. 161:2717–2725. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Meng F, Li Q, Zhang Y, Zhang Z, Tian S,
Cui Z, Chang S and Zhao P: Characterization of subgroup J avian
Leukosis virus isolated from Chinese indigenous chickens. Virol J.
15(33)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Li H, Shang H, Shu D, Zhang H, Ji J, Sun
B, Li H and Xie Q: gga-miR-375 plays a key role in tumorigenesis
post subgroup J Avian Leukosis virus infection. PLos One.
9:2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang D, He J, Dong J, Meyer TF and Xu T:
The HIPPO pathway in gynecological malignancies. Am J Cancer Res.
10:610–629. 2020.PubMed/NCBI
|
|
20
|
Ma Y, Yang Y, Wang F, Wei Q and Qin H:
Hippo-YAP signaling pathway: A new paradigm for cancer therapy. Int
J Cancer. 137:2275–2286. 2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Johnson R and Halder G: The two faces of
Hippo: Targeting the Hippo pathway for regenerative medicine and
cancer treatment. Nat Rev Drug Discov. 13:63–79. 2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Bao YJ, Hata Y, Ikeda M and Withanage K:
Mammalian Hippo pathway: From development to cancer and beyond. J
Biochem. 149:361–379. 2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Machado-Neto JA, Lazarini M, Favaro P,
Franchi-GC Jr, Nowill AE, Saad ST and Traina F: ANKHD1, a novel
component of the Hippo signaling pathway, promotes YAP1 activation
and cell cycle progression in prostate cancer cells. Exp Cell Res.
324:137–145. 2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Edgar BA: From cell structure to
transcription: Hippo forges a new path. Cell. 124:267–273.
2006.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang H, Ramakrishnan SK, Triner D,
Centofanti B, Maitra D, Győrffy B, Sebolt-Leopold JS, Dame MK,
Varani J, Brenner DE, et al: Tumor-selective proteotoxicity of
verteporfin inhibits colon cancer progression independently of
YAP1. Sci Signal. 8(ra98)2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Wang H, Du YC, Zhou XJ, Liu H and Tang SC:
The dual functions of YAP-1 to promote and inhibit cell growth in
human malignancy. Cancer Metastasis Rev. 33:173–181.
2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Mizuno T, Murakami H, Fujii M, Ishiguro F,
Tanaka I, Kondo Y, Akatsuka S, Toyokuni S, Yokoi K, Osada H and
Sekido Y: YAP induces malignant mesothelioma cell proliferation by
upregulating transcription of cell cycle-promoting genes. Oncogene.
31:5117–5122. 2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Overholtzer M, Zhang J, Smolen GA, Muir B,
Li W, Sgroi DC, Deng CX, Brugge JS and Haber DA: Transforming
properties of YAP, a candidate oncogene on the chromosome 11q22
amplicon. Proc Natl Acad Sci USA. 103:12405–12410. 2006.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zhang X, Yan Y, Lin W, Li A, Zhang H, Lei
X, Dai Z, Li X, Li H, Chen W, et al: Circular RNA Vav3 sponges
gga-miR-375 to promote epithelial-mesenchymal transition. RNA Biol.
16:118–132. 2019.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Frampton D, Schwenzer H, Marino G, Butcher
LM, Pollara G, Kriston-Vizi J, Venturini C, Austin R, de-Castro KF,
Ketteler R, et al: Molecular signatures of regression of the canine
transmissible venereal tumor. Cancer Cell.
33(620-633.e6)2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Morgan TM, Mehra R, Tiemeny P, Wolf JS, Wu
S, Sangale Z, Brawer M, Stone S, Wu CL and Feldman AS: A multigene
signature based on cell cycle proliferation improves prediction of
mortality within 5 yr of radical nephrectomy for renal cell
carcinoma. Eur Urol. 73:763–769. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Sun F, Zhang Y, Xu L, Li S, Chen X, Zhang
L, Wu Y and Li J: Proteasome inhibitor MG132 enhances
cisplatin-induced apoptosis in osteosarcoma cells and inhibits
tumor growth. Oncol Res. 26:655–664. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Zhu Y, Gu J, Li Y, Peng C, Shi M, Wang X,
Wei G, Ge O, Wang D, Zhang B, et al: MiR-17-5p enhances pancreatic
cancer proliferation by altering cell cycle profiles via disruption
of RBL2/E2F4-repressing complexes. Cancer Lett. 412:59–68.
2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Liao Z, Dai Z, Cai C, Zhang X, Li A, Zhang
H, Yan Y, Lin W, Wu Y, Li H, et al: Knockout of Atg5 inhibits
proliferation and promotes apoptosis of DF-1 cells. In Vitro Cell
Dev Biol Anim. 55:341–348. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Giotis ES, Ross CS, Robey RC, Nohturfft A,
Goodbourn S and Skinner MA: Constitutively elevated levels of SOCS1
suppress innate responses in DF-1 immortalised chicken fibroblast
cells. Sci Rep. 7(17485)2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Abu-Bonsrah KD, Zhang D and Newgreen DF:
CRISPR/Cas9 targets chicken embryonic somatic cells in vitro and in
vivo and generates phenotypic abnormalities. Sci Rep. 6(34524)2016.
View Article : Google Scholar
|
|
37
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Warren JSA, Xiao YX and Lamar JM: YAP/TAZ
activation as a target for treating metastatic cancer. Cancers.
10:2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chan SW, Lim CJ, Chen L, Chong YF, Huang
C, Song H and Hong W: The Hippo pathway in biological control and
cancer development. J Cell Physiol. 226:928–939. 2011.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Zhang Y, Guan X, Chen Z, Cao D, Kang Z,
Shen Q, Lei Q, Li F, Li H, Leghari MF, et al: The high conserved
cellular receptors of avian leukosis virus subgroup J in Chinese
local chickens contributes to its wide host range. Poult Sci.
97:4187–4192. 2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ross CL, Kaushik S, Valdes-Rodriguez R and
Anvekar R: MicroRNAs in cutaneous melanoma: Role as diagnostic and
prognostic biomarkers. J Cell Physiol. 233:5133–5141.
2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Panagiotakopoulou M, Lendenmann T,
Pramotton FM, Giampietro C, Stefopoulos G, Poulikakos D and Ferrari
A: Cell cycle-dependent force transmission in cancer cells. Mol
Biol Cell. 29:2528–2539. 2018.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Chen BW, Chen W, Liang H, Liu H, Liang C,
Zhi X, Hu LQ, Yu XZ, Wei T, Ma T, et al: Inhibition of mTORC2
induces cell-cycle arrest and enhances the cytotoxicity of
doxorubicin by suppressing MDR1 expression in HCC cells. Mol Cancer
Ther. 14:1805–1815. 2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Li JL, Fu FQ, Wan XC, Huang SS, Wu DL and
Li Y: Up-regulated miR-29c inhibits cell proliferation and
glycolysis by inhibiting SLC2A3 expression in prostate cancer.
Gene. 665:26–34. 2018.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Lim LP, Lau NC, Garrett-Engele P, Grimson
A, Schelter JM, Castle J, Bartel DP, Linsley PS and Johnson JM:
Microarray analysis shows that some microRNAs downregulate large
numbers of target mRNAs. Nature. 433:769–773. 2005.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Chen J: The cell-cycle arrest and
apoptotic functions of p53 in tumor initiation and progression.
Cold Spring Harb Perspect Med. 6(a026104)2016.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Li H, Ji J, Xie Q, Shang H, Zhang H, Xin
X, Chen F, Sun B, Xue C, Ma J and Bi Y: Aberrant expression of
liver microRNA in chickens infected with subgroup J avian leukosis
virus. Virus Res. 169:268–271. 2012. View Article : Google Scholar
|
|
48
|
Schlegelmilch K, Mohseni M, Kirak O,
Pruszak J, Rodriguez JR, Zhou D, Kreger BT, Vasioukhin V, Avruch J,
Brummelkamp TR and Camargo FD: Yap1 acts downstream of α-catenin to
control epidermal proliferation. Cell. 144:782–795. 2011.PubMed/NCBI View Article : Google Scholar
|
|
49
|
von Gise A, Lin ZQ, Schlegelmilch K, Honor
LB, Pan GM, Buck JN, Ma Q, Ishiwata T, Zhou B, Camargo FD and Pu
WT: YAP1, the nuclear target of Hippo signaling, stimulates heart
growth through cardiomyocyte proliferation but not hypertrophy.
Proc Natl Acad Sci USA. 109:2394–2399. 2012.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Yang Z, Zhang M, Xu K, Liu L, Hou WK, Cai
YZ, Xu P and Yao JF: Knockdown of YAP1 inhibits the proliferation
of osteosarcoma cells in vitro and in vivo. Oncol
Rep. 32:1265–1272. 2014.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Oak C, Khalifa AO, Isali I, Bhaskaran N,
Walker E and Shukla S: Diosmetin suppresses human prostate cancer
cell proliferation through the induction of apoptosis and cell
cycle arrest. Int J Oncol. 53:835–843. 2018.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Lu HF, Chen YS, Yang JS, Chen JC, Lu KW,
Chiu TH, Liu KC, Yeh CC, Chen GW, Lin HJ and Chung JG: Gypenosides
induced G0/G1 arrest via inhibition of cyclin E and induction of
apoptosis via activation of caspases-3 and -9 in human lung cancer
A-549 cells. In Vivo. 22:215–221. 2008. View Article : Google Scholar
|
|
53
|
Qie S and Diehl JA: Cyclin D1, cancer
progression, and opportunities in cancer treatment. J Mol Med
(Berl). 94:1313–1326. 2016.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Luhtala S, Staff S, Tanner M and Isola J:
Cyclin E amplification, over-expression, and relapse-free survival
in HER-2-positive primary breast cancer. Tumour Biol. 37:9813–9823.
2016.PubMed/NCBI View Article : Google Scholar
|