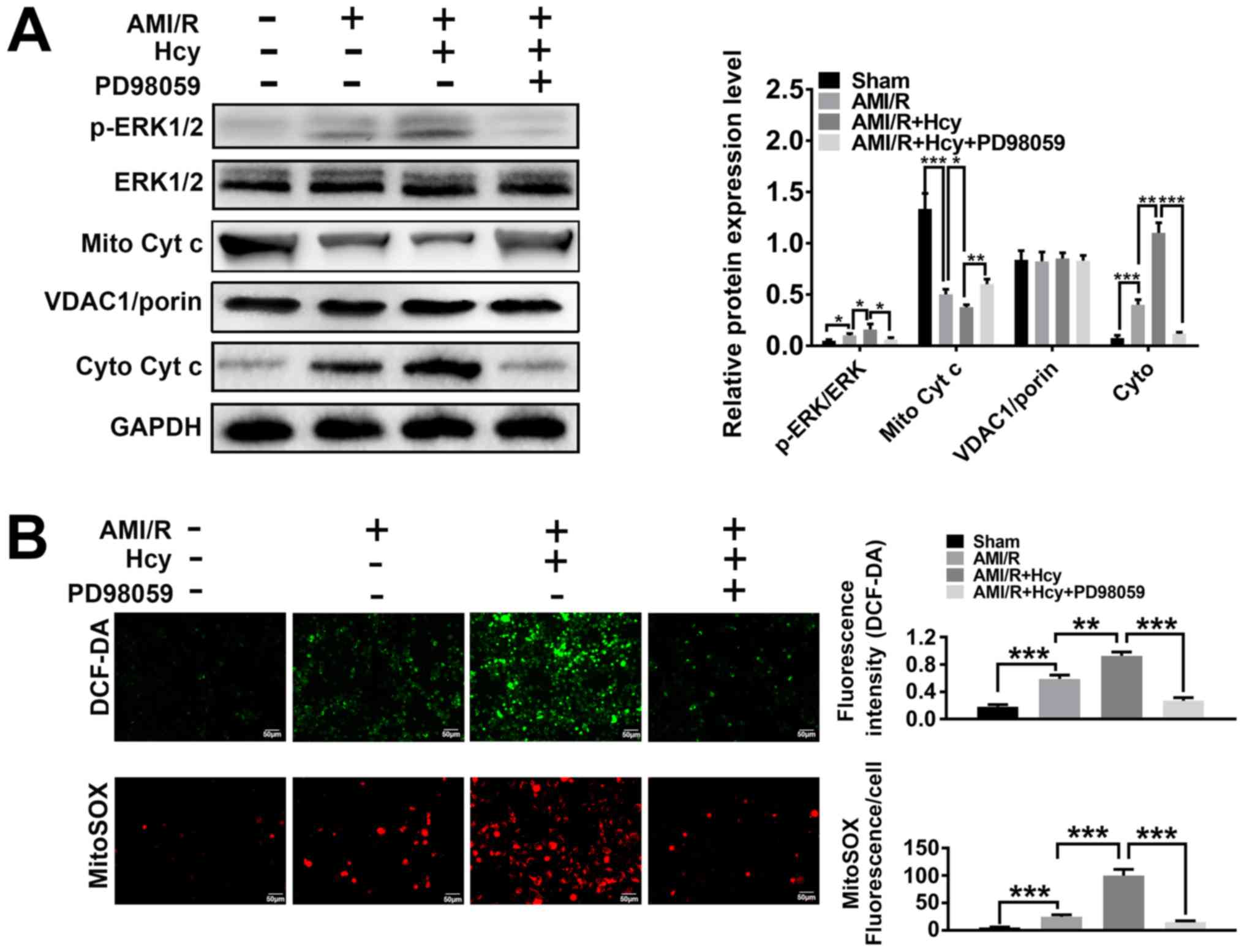
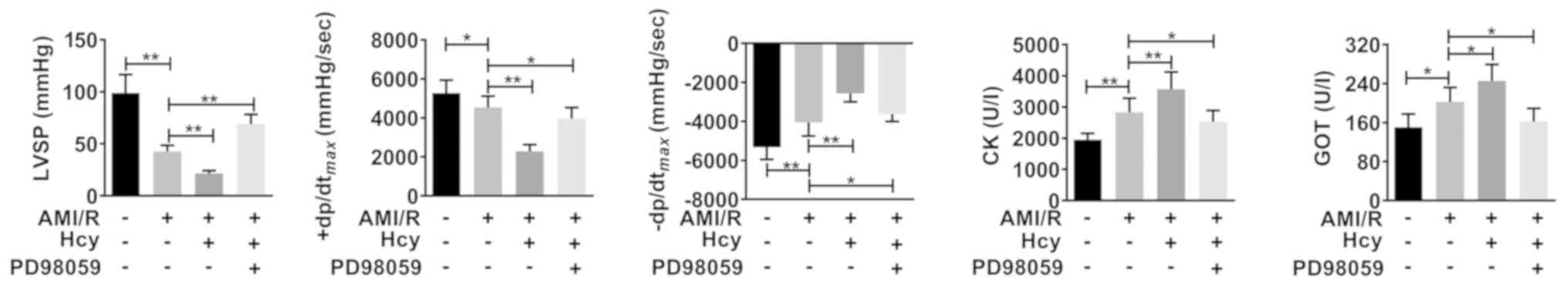
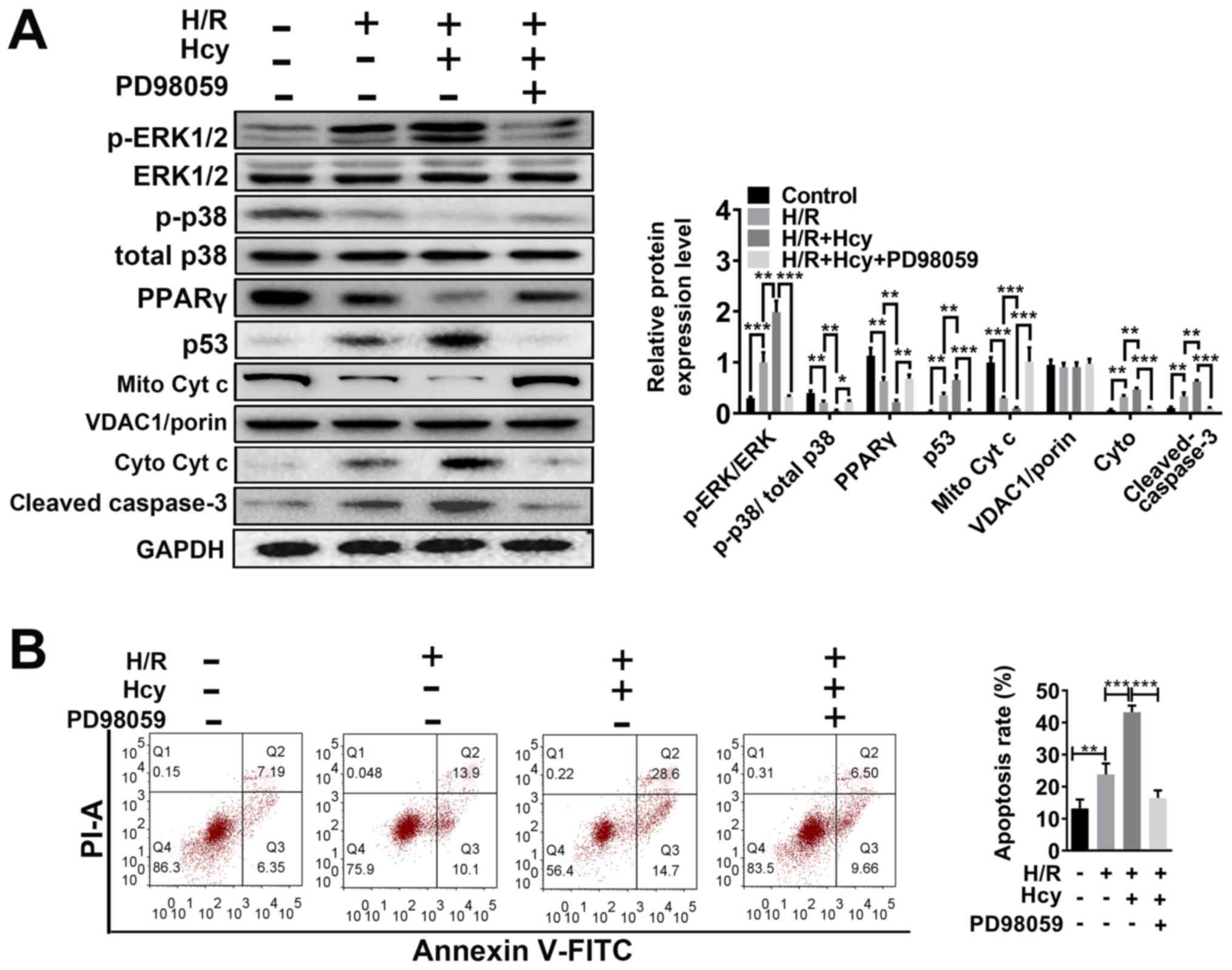
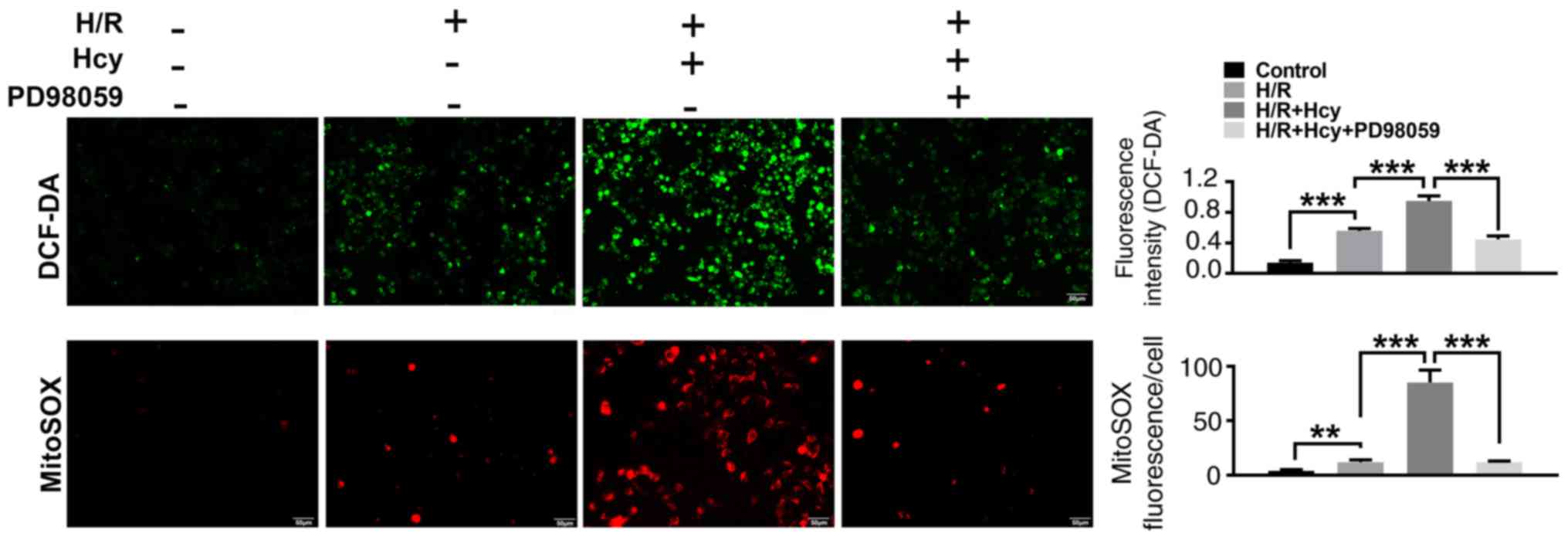
|
1
|
Thygesen K, Alpert JS, Jaffe AS, Simoons
ML, Chaitman BR and White HD: Joint ESC/ACCF/AHA/WHF Task Force for
Universal Definition of Myocardial Infarction; Authors/Task Force
Members Chairpersons, Thygesen K, Alpert JS, et al. Third
universal definition of myocardial infarction. J Am Coll Cardiol.
60:1581–1598. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Hausenloy DJ and Yellon DM: Myocardial
ischemia-reperfusion injury: A neglected therapeutic target. J Clin
Invest. 123:92–100. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
3
|
Eltzschig HK and Eckle T: Ischemia and
reperfusion-from mechanism to translation. Nat Med.
17(1391)2011.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Yellon DM and Hausenloy DJ: Myocardial
reperfusion injury. N Engl J Med. 357:1121–1135. 2007.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Heusch G, Libby P, Gersh B, Yellon D, Böhm
M, Lopaschuk G and Opie L: Cardiovascular in coronary artery
disease and heart failure. Lancet. 383:1933–1943. 2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ganote C, Worstell J and Kaltenbach J:
Oxygen-induced enzyme release after irreversible myocardial injury.
Effects of cyanide in perfused rat hearts. Am J Pathol. 84:327–350.
1976.PubMed/NCBI
|
|
7
|
Zweier JL, Flaherty JT and Weisfeldt ML:
Direct measurement of free radical generation following reperfusion
of ischemic myocardium. Proc Natl Acad Sci USA. 84:1404–1407.
1987.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Loor G, Kondapalli J, Iwase H, Chandel NS,
Waypa GB, Guzy RD, Vanden Hoek TL and Schumacker PT: Mitochondrial
oxidant stress triggers cell death in simulated
ischemia-reperfusion. Biochim Biophys Acta. 1813:1382–1394.
2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Murphy E and Steenbergen C: Mechanisms
underlying acute protection from cardiac ischemia-reperfusion
injury. Physiol Rev. 88:581–609. 2008.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Di Lisa F and Bernardi P: Modulation of
mitochondrial permeability transition in ischemia-reperfusion
injury of the heart. Advantages and limitations. Curr Med Chem.
22:2480–2487. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Paradies G, Petrosillo G, Pistolese M and
Ruggiero FM: Reactive oxygen species affect mitochondrial electron
transport complex I activity through oxidative cardiolipin damage.
Gene. 286:135–141. 2002.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Abe Ji, Baines CP and Berk BC: Role of
mitogen-activated protein kinases in ischemia and reperfusion
injury: The good and the bad. Circ Res. 86:607–609. 2000.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Das A, Salloum FN, Xi L, Rao YJ and
Kukreja RC: ERK phosphorylation mediates sildenafil-induced
myocardial protection against ischemia-reperfusion injury in mice.
Am J Physiol Heart Circ Physiol. 296:H1236–H1243. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yue TL, Wang C, Gu JL, Ma XL, Kumar S, Lee
JC, Feuerstein GZ, Thomas H, Maleeff B and Ohlstein EH: Inhibition
of extracellular signal-regulated kinase enhances
ischemia/reoxygenation-induced apoptosis in cultured cardiac
myocytes and exaggerates reperfusion injury in isolated perfused
heart. Circ Res. 86:692–699. 2000.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Di Cristo G, Berardi N, Cancedda L,
Pizzorusso T, Putignano E, Ratto GM and Maffei L: Requirement of
ERK activation for visual cortical plasticity. Science.
292:2337–2340. 2001.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wald DS, Law M and Morris JK: Homocysteine
and cardiovascular disease: Evidence on causality from a
meta-analysis. BMJ. 325(1202)2002.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ganguly P and Alam SF: Role of
homocysteine in the development of cardiovascular disease. Nutri J.
14(6)2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Malinow M, Levenson J, Giral P, Nieto F,
Razavian M, Segond P and Simon A: Role of blood pressure, uric
acid, and hemorheological parameters on plasma homocyst (e)ine
concentration. Atherosclerosis. 114:175–183. 1995.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Hoogeveen EK, Kostense PJ, Beks PJ,
Mackaay AJ, Jakobs C, Bouter LM, Heine RJ and Stehouwer CD:
Hyperhomocysteinemia is associated with an increased risk of
cardiovascular disease, especially in non-insulin-dependent
diabetes mellitus: A population-based study. Arterioscler Thromb
Vasc Biol. 18:133–138. 1998.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Shao L, Wu D, Zhang P, Li W, Wang J, Su G,
Liao Y, Wang Z and Liu K: The significance of microthrombosis and
fgl2 in no-reflow phenomenon of rats with acute myocardial
ischemia/reperfusion. Clin Appl Thromb Hemost. 19:19–28.
2013.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Leary S, Underwood W, Anthony R, Cartner
S, Corey D, Grandin T, Greenacre C, Gwaltney-Brant S, McCrackin MA,
Meyer R, et al: AVMA guidelines for the euthanasia of animals: 2013
edition.
|
|
22
|
Yin Y, Guan Y, Duan J, Wei G, Zhu Y, Quan
W, Guo C, Zhou D, Wang Y, Xi M and Wen A: Cardioprotective effect
of Danshensu against myocardial ischemia/reperfusion injury and
inhibits apoptosis of H9c2 cardiomyocytes via Akt and ERK1/2
phosphorylation. Eur J Pharmacol. 699:219–226. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Feigin VL, Forouzanfar MH, Krishnamurthi
R, Mensah GA, Connor M, Bennett DA, Moran AE, Sacco RL, Anderson L,
Truelsen T, et al: Global and regional burden of stroke during
1990-2010: Findings from the Global Burden of Disease Study 2010.
Lancet. 383:245–255. 2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Scarabelli TM, Stephanou A, Pasini E,
Comini L, Raddino R, Knight RA and Latchman DS: Different signaling
pathways induce apoptosis in endothelial cells and cardiac myocytes
during ischemia/reperfusion injury. Circ Res. 90:745–748.
2002.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Xu Q, Li X, Lu Y, Shen L, Zhang J, Cao S,
Huang X, Bin J and Liao Y: Pharmacological modulation of autophagy
to protect cardiomyocytes according to the time windows of
ischaemia/reperfusion. Br J Pharmacol. 172:3072–3085.
2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Kim SJ, Yoo KY, Jeong CW, Kim WM, Lee HK,
Bae HB, Kwak SH, Li M and Lee J: Urinary trypsin inhibitors afford
cardioprotective effects through activation of PI3K-Akt and ERK
signal transduction and inhibition of p38 MAPK and JNK. Cardiology.
114:264–270. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hassan A, Hunt BJ, O'sullivan M, Bell R,
D'souza R, Jeffery S, Bamford JM and Markus HS: Homocysteine is a
risk factor for cerebral small vessel disease, acting via
endothelial dysfunction. Brain. 127:212–219. 2004.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Misra HP: Generation of superoxide free
radical during the autoxidation of thiols. J Biol Chem.
249:2151–2155. 1974.PubMed/NCBI
|
|
29
|
Majors A, Ehrhart LA and Pezacka EH:
Homocysteine as a risk factor for vascular disease. Enhanced
collagen production and accumulation by smooth muscle cells.
Arterioscler Thromb Vasc Biol. 17:2074–2081. 1997.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Basu A, Jenkins AJ, Stoner JA, Thorpe SR,
Klein RL, Lopes-Virella MF, Garvey WT and Lyons TJ: DCCT/EDIC
Research Group. Plasma total homocysteine and carotid intima-media
thickness in type 1 diabetes: A prospective study. Atherosclerosis.
236:188–195. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhang S, Bai YY, Luo LM, Xiao WK, Wu HM
and Ye P: Association between serum homocysteine and arterial
stiffness in elderly: A community-based study. J Geriatr Cardiol.
11:32–38. 2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Balakumar P, Singh AP and Singh M: Rodent
models of heart failure. J Pharmacol Toxicol Methods. 56:1–10.
2007.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Purcell NH, Wilkins BJ, York A,
Saba-El-Leil MK, Meloche S, Robbins J and Molkentin JD: Genetic
inhibition of cardiac ERK1/2 promotes stress-induced apoptosis and
heart failure but has no effect on hypertrophy in vivo. Proc Natl
Acad Sci USA. 104:14074–14079. 2007.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Bueno OF, De Windt LJ, Tymitz KM, Witt SA,
Kimball TR, Klevitsky R, Hewett TE, Jones SP, Lefer DJ, Peng CF, et
al: The MEK1-ERK1/2 signaling pathway promotes compensated cardiac
hypertrophy in transgenic mice. EMBO J. 19:6341–6350.
2000.PubMed/NCBI View Article : Google Scholar
|