Lupus nephritis (LN) is one of the most severe
complications of SLE and its incidence rate reaches up to 60% in
patients with SLE worldwide. Among patients with SLE, 50-80% are
cases of pediatric-onset SLE (7-10).
As comprehensively reviewed (10),
without pharmacotherapy, long-term LN may induce irreversible renal
injury and subsequently develops into end-stage renal disease.
Traditional treatments of LN involve a combination therapy of
cyclophosphamide with glucocorticoids, which have been demonstrated
to improve the long-term prognosis. However, its usage is limited
by severe adverse effects, including hemorrhagic cystitis,
amenorrhea, malignancy and sepsis. Novel immunosuppressants,
including tacrolimus, cyclosporine and mycophenolate mofetil are
required to inhibit the side effects of traditional treatments.
Tacrolimus has been reported to be a safe and effective agent for
treating patients with LN (10).
However, considerable intra- and inter-individual
pharmacokinetic variability makes it difficult to establish
individualized tacrolimus dosage regimens. Of note, population
pharmacokinetics is able to differentiate in terms of
pharmacokinetic variability and has a higher statistical power to
verify the effect of multiple factors on the pharmacokinetic
behaviour of tacrolimus compared to traditional pharmacokinetic
analysis and makes it possible to formulate an optimal dosage
schedule (11,12). Hence, the present study aimed to
establish a population pharmacokinetics model of tacrolimus and
further optimize the initial dosage regimen for tacrolimus in
pediatric and adolescent patients with LN.
The clinical information of pediatric and adolescent
patients with LN treated between August 2014 and September 2019 at
the Children's Hospital of Fudan University (Shanghai, China) was
retrospectively collected. The clinical information was collected
from the hospital's information system and tacrolimus whole-blood
levels were acquired from a therapeutic drug detection system.
Partial basic clinical information data with partial overlap were
collected from certain patients in previous studies (13,14). The
present study was approved by the Research Ethics Committee of the
Children's Hospital of Fudan University [ethical approval code:
(2019)020]. The present study was a retrospective study and was
approved by the ethics committee of this hospital without the
requirement for written informed consent. A previous study have
used lower patient numbers (15).
The population pharmacokinetics model was
established using Nonlinear Mixed Effects Modeling software
(NONMEM®; version VII; ICON Development Solutions Ltd.)
by the first-order conditional estimation method with interaction.
The pharmacokinetics parameters included apparent oral clearance
(CL/F), volume of distribution (V/F) and absorption rate constant
(Ka), where the value of Ka was fixed at 4.48 per hour (13,16-18).
Inter-individual variabilities were estimated by equation (i):
A=T(A) x exp(ηi). A and T(A) represent the individual
parameter value and the typical individual parameter value,
respectively. ηi represented a symmetrical distribution
(0, ω2), which was a random term with zero mean and
variance ω squared (ω2).
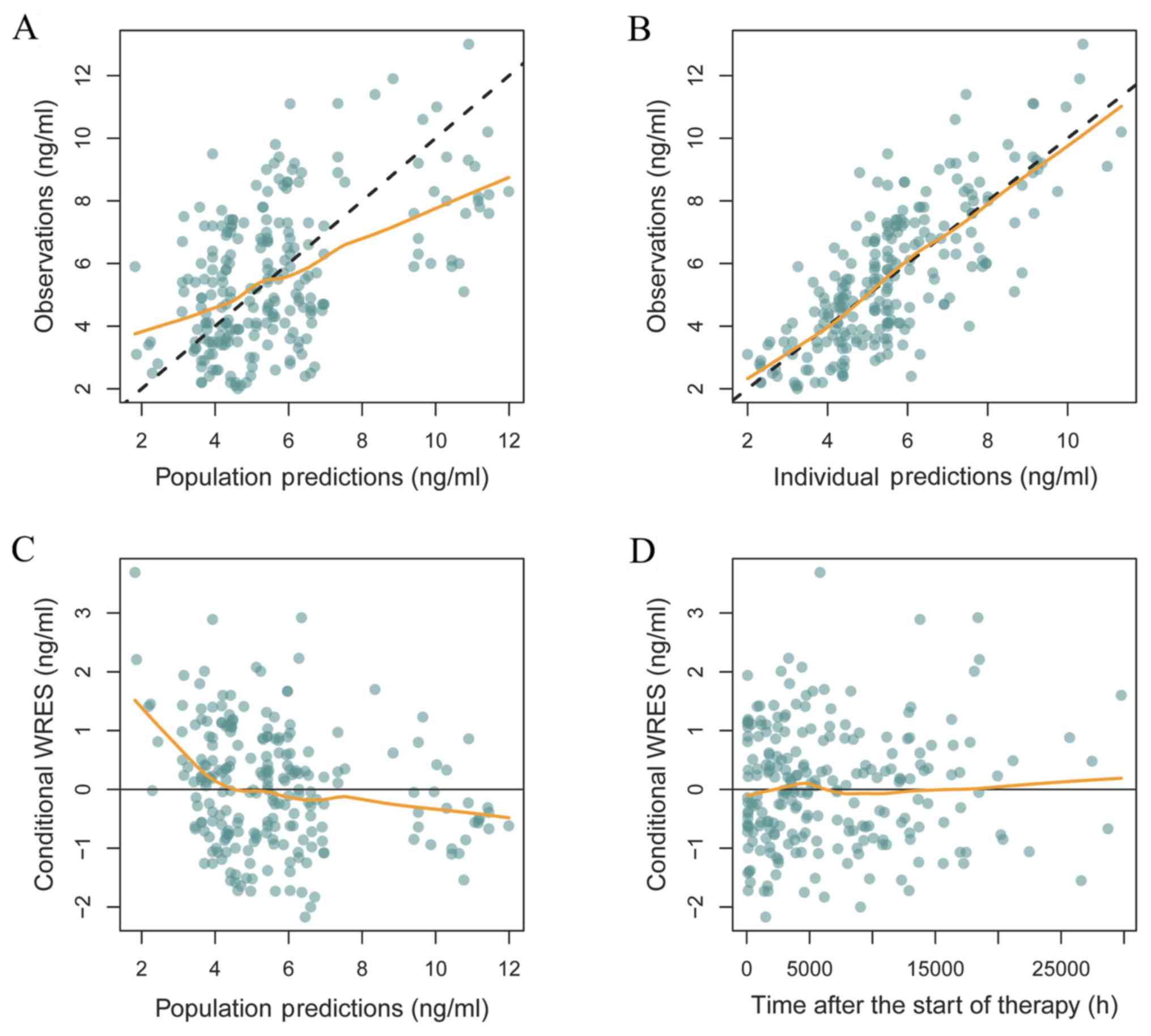
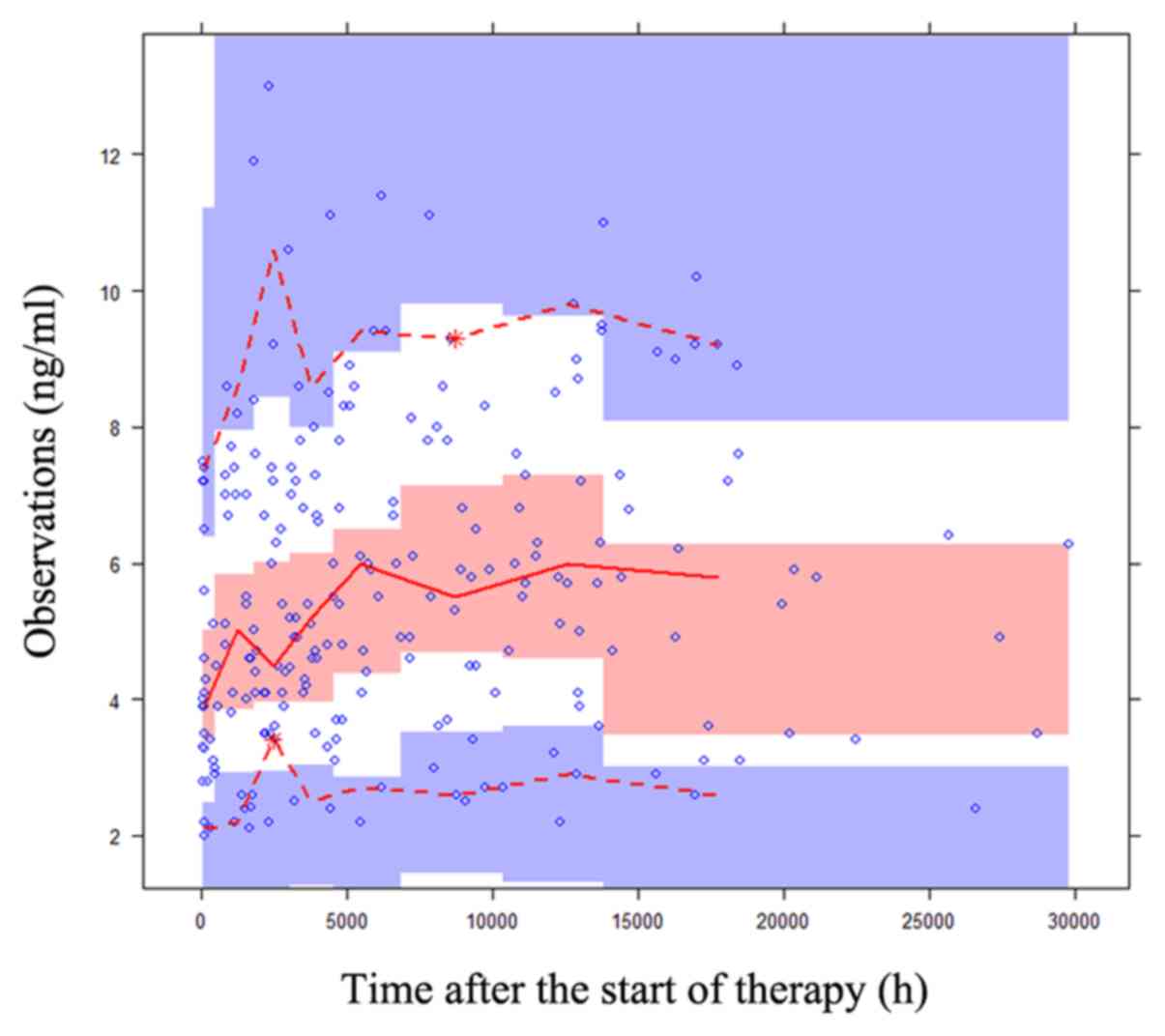
The reliability and stability of the final
parameters were assessed by bootstrap (n=1,000), which was
performed using the NONMEM® software (version VII; ICON
Development Solutions Ltd), goodness of fit plots and
prediction-corrected visual predictive check (VPC) plots. The Monte
Carlo method was used for the simulation of the optimal initial
dose, including six weight groups (10, 20, 30, 40, 50 and 60 kg)
and seven initial dosing regimens (0.01, 0.05, 0.10, 0.15, 0.20,
0.25 and 0.30 mg/kg daily) split into two doses. Based on previous
publications, the therapeutic window of tacrolimus treatment in LN
is between 5 and 15 ng/ml; thus, this was used in the present study
(10).
The clinical information of 32 pediatric and
adolescent patients with LN (5 males and 27 females) was collected
for the present study and was used for population modelling. The
clinical information of certain patients was collected in previous
studies (13,14). Table I
presents patient characteristics and drug combinations.
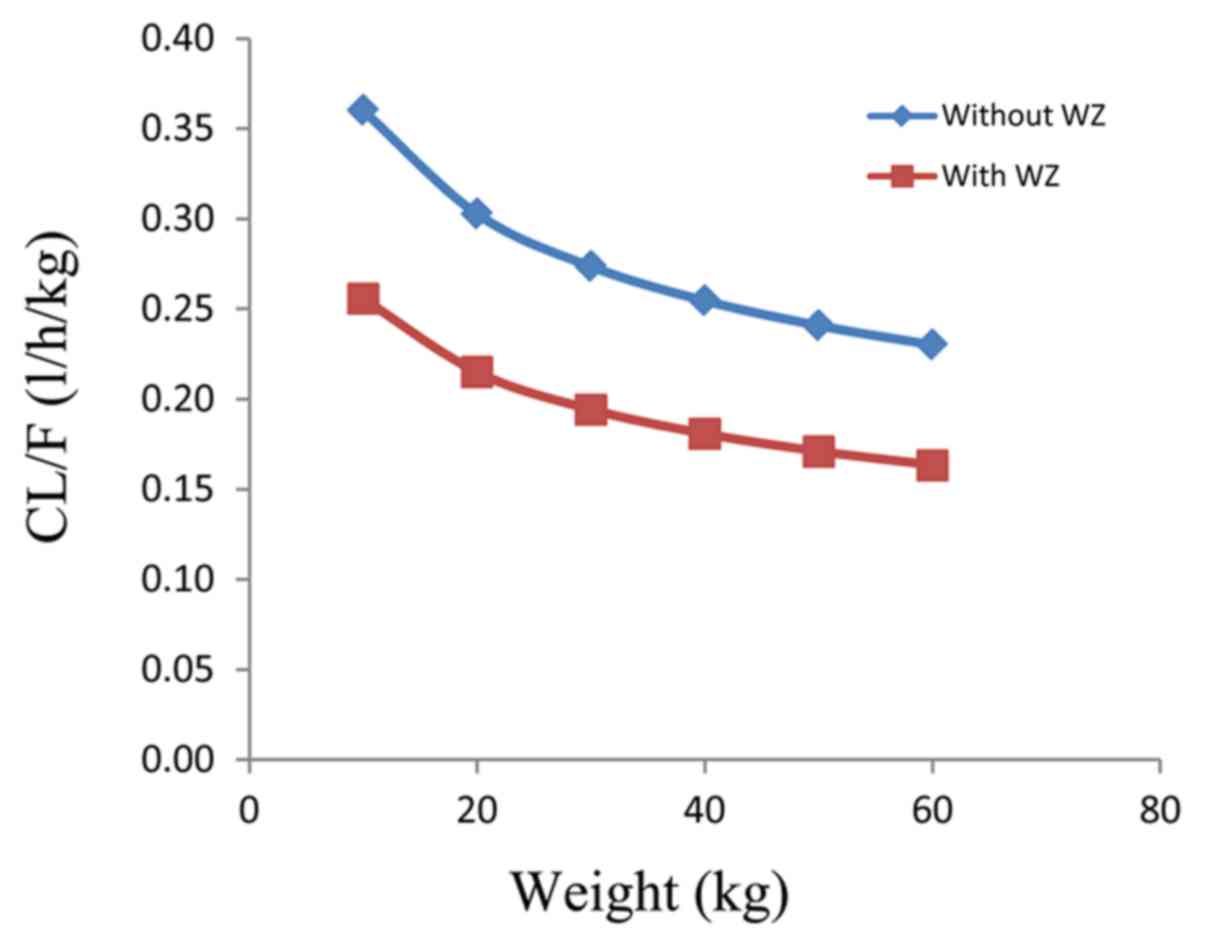
The final covariate model was described by equations
(vi) and (vii), respectively:
CL/F=θCL/F x
(WT/70)0.75 x (1 + WZ x θWZ) and
V/F=θV/F x (WT/70).
θCL/F and
θV/F are the typical population
values of CL/F and V/F, respectively. θWZ is the
coefficient of wuzhi capsule. When patients were co-administered
wuzhi capsule and tacrolimus, WZ=1 was used; otherwise, WZ=0 was
applied.
Body weight and co-administration of wuzhi capsule
affected tacrolimus clearance in pediatric and adolescent patients
with LN, and for the same body weight, the rate of tacrolimus
clearance in patients who were not administered wuzhi capsule vs.
those who were administered wuzhi capsule was 1:0.71 (Fig. 3). Fig.
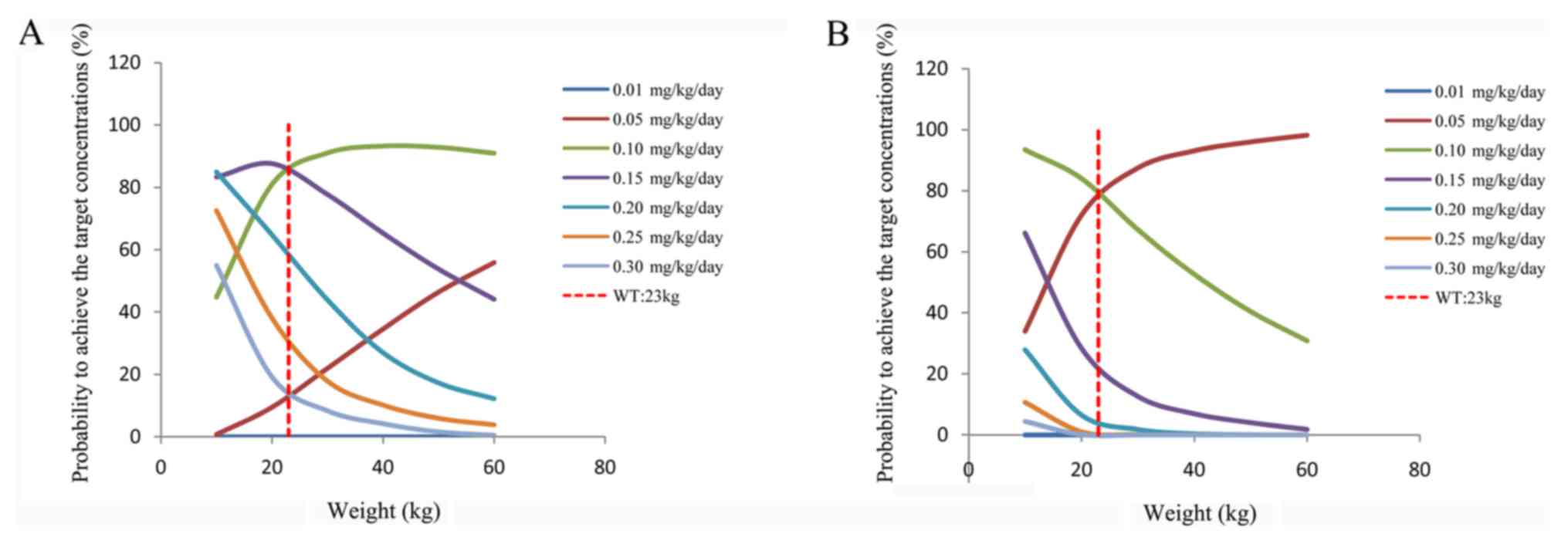
4 presents the probability of achieving the target
concentrations under different initial doses. In addition, in
patients who were not administered wuzhi capsule, the initial
dosage regimen of 0.15 mg/kg/day was recommended for a body weight
of 10-23 kg and 0.10 mg/kg/day for 23-60 kg; in patients who were
co-administered wuzhi capsule and tacrolimus, the initial dosage
regimen of 0.10 mg/kg/day was recommended for a body weight of
10-23 kg and 0.05 mg/kg/day for 23-60 kg, as presented in Table III.
Tacrolimus, also known as FK506, is a 23-membered
lactone ring that is isolated from Streptomyces tsukubaensis
and used as a potent immunosuppressant. It has been reported that
tacrolimus is 100 times stronger than cyclosporine and it may exert
its effects by inhibiting the function of T lymphocytes and
downregulating the expression of interleukin-2 (20,21). In
addition, it has been used as the first-line drug for patients with
liver and renal transplant (16,22-34). Furthermore, it has been
demonstrated that tacrolimus may be used to improve the outcome of
patients who undergo bone marrow (35-42),
lung (43) and heart transplantation
(44).
In previous years, clinical experiments have also
indicated that tacrolimus has useful applications in systemic-onset
juvenile idiopathic arthritis (45-48),
nephrotic syndrome (49-55),
SLE (56-65),
myasthenia gravis (66,67), ulcerative colitis (68,69) and
autoimmune hepatitis (70).
Furthermore, according to a previous review article, tacrolimus is
a safe and effective agent for treating patients with LN (10).
It has been reported that the underlying mechanism
of action of tacrolimus in LN is primarily its inhibitory effect on
the dephosphorylation of the nuclear factor of activated T cells,
which thereby reduces the activity of genes encoding interleukin-2
and associated cytokines (71),
leading to the inhibition of T-cell activation. In addition, the
effect of tacrolimus on LN is also the result of its well-known
antiproteinuric effects that have been utilized in the treatment of
a variety of kidney pathologies (72). It has also been reported that in
mouse models of SLE, tacrolimus inhibits the progression of
glomerular hypercellularity, crescent formation and proteinuria
development, and suppresses the increase of anti-double-stranded
DNA antibody serum levels in animal models of spontaneous LN
(73). Therefore, from the above
perspective, the mechanism of action of tacrolimus in treating LN
is well explained.
In the present study, body weight and
co-administration of wuzhi capsule were indicated to influence
tacrolimus clearance in pediatric and adolescent patients with LN.
A previous similar study demonstrated a non-linear association
between drug clearance and body weight in patients (19). In the present study, the rate of
tacrolimus clearance in patients who were not administered wuzhi
capsule and those who were administered wuzhi capsule with the same
body weight was 1:0.71. Wuzhi capsule is a Chinese patent medicine,
which contains the primary active ingredients schisandrin,
schisantherin A and schisandrol B (83). It has been demonstrated that wuzhi
capsule increases the concentration of tacrolimus (84-86)
via inhibition of the enzyme cytochrome P450, family 3 (CYP3A) in
order to inhibit the metabolization of tacrolimus (86,87).
This is able to reduce the dose of tacrolimus required and reduce
medical costs, particularly in patients who require to take
tacrolimus over a long period of time. In addition, the present
study revealed a wide range of hemoglobin levels, which may be due
to differences in various physiological or pathological states
among the pediatric patients. The specific mechanisms remain to be
further explored.
Next, Monte Carlo simulation was used to further
predict the optimal dose. This indicated that in patients who
weren't administered wuzhi capsule, the initial dosage regimen of
0.15 mg/kg/day was recommended for a body weight of 10-23 kg and
0.10 mg/kg/day for 23-60 kg; in patients who were co-administered
wuzhi capsule, the initial dosage regimen of 0.10 mg/kg/day was
recommended for a body weight of 10-23 kg and 0.05 mg/kg/day for
23-60 kg.
However, there are limitations to the present study.
Polymorphisms of CYP3A5 may be associated with tacrolimus required
dose; however, the present study was based on real-world data, in
which pharmacogenetics were not considered in tacrolimus dosing and
therefore, no routine clinical testing was performed with this
regard. Therefore, it should be further investigated whether the
inclusion of genotyping in this model is able to better explain the
variability in the dosage of tacrolimus. In addition, future
studies with more patients are required to verify the results of
the present study.
In conclusion, to the best of our knowledge, the
present study was the first to construct a population
pharmacokinetics model of tacrolimus and optimize the initial
dosage regimen for tacrolimus treatment in pediatric and adolescent
patients with LN.
Not applicable.
This work was supported by the Scientific Research
Project of Science and Technology Commission of Shanghai
Municipality (grant no. 18DZ1910604/19DZ1910703) and the Shanghai
Science and Technology Commission (grant no. 19XD1400900).
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
ZL and HX conceived and designed the study. XC and
DW collected and analyzed the data. XC wrote the manuscript. All
authors read and approved the final manuscript.
The study was approved by the Research Ethics
Committee of the Children's Hospital of Fudan University (Shanghai,
China). The present study was a retrospective study and the
analysis was approved by the Ethics Committee of the hospital
without the requirement for written informed consent.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Papadimitraki ED and Isenberg DA:
Childhood- and adult-onset lupus: An update of similarities and
differences. Expert Rev Clin Immunol. 5:391–403. 2009.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Takeuchi T, Tsuzaka K, Abe T, Yoshimoto K,
Shiraishi K, Kameda H and Amano K: T cell abnormalities in systemic
lupus erythematosus. Autoimmunity. 38:339–346. 2005.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Rahman A and Isenberg DA: Systemic lupus
erythematosus. N Engl J Med. 358:929–939. 2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Gurevitz SL, Snyder JA, Wessel EK, Frey J
and Williamson BA: Systemic lupus erythematosus: A review of the
disease and treatment options. Consult Pharm. 28:110–121.
2013.PubMed/NCBI View Article : Google Scholar
|
|
5
|
D'Cruz DP, Khamashta MA and Hughes GR:
Systemic lupus erythematosus. Lancet. 369:587–596. 2007.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Huang JL, Yeh KW, Yao TC, Huang YL, Chung
HT, Ou LS, Lee WI and Chen LC: Pediatric lupus in Asia. Lupus.
19:1414–1418. 2010.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tanaka H, Joh K and Imaizumi T: Treatment
of pediatric-onset lupus nephritis: A proposal of optimal therapy.
Clin Exp Nephrol. 21:755–763. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Takei S, Maeno N, Shigemori M, Imanaka H,
Mori H, Nerome Y, Kanekura S, Takezaki T, Hokonohara M, Miyata K
and Fujikawa S: Clinical features of Japanese children and
adolescents with systemic lupus erythematosus: Results of 1980-1994
survey. Acta Paediatr Jpn. 39:250–256. 1997.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bertsias GK, Tektonidou M, Amoura Z,
Aringer M, Bajema I, Berden JH, Boletis J, Cervera R, Dörner T,
Doria A, et al: Joint European league against rheumatism and
European renal association-European dialysis and transplant
association (EULAR/ERA-EDTA) recommendations for the management of
adult and paediatric lupus nephritis. Ann Rheum Dis. 71:1771–1782.
2012.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhou T, Lin S, Yang S and Lin W: Efficacy
and safety of tacrolimus in induction therapy of patients with
lupus nephritis. Drug Des Devel Ther. 13:857–869. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Vadcharavivad S, Praisuwan S,
Techawathanawanna N, Treyaprasert W and Avihingsanon Y: Population
pharmacokinetics of tacrolimus in Thai kidney transplant patients:
Comparison with similar data from other populations. J Clin Pharm
Ther. 41:310–328. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wang D, Chen X, Xu H and Li Z: Population
pharmacokinetics and dosing regimen optimization of tacrolimus in
Chinese pediatric hematopoietic stem cell transplantation patients.
Xenobiotica. 50:178–185. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang DD, Lu JM, Li Q and Li ZP: Population
pharmacokinetics of tacrolimus in paediatric systemic lupus
erythematosus based on real-world study. J Clin Pharm Ther.
43:476–483. 2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Chen X, Wang D, Xu H and Li Z: Initial
dose optimization of tacrolimus for children with systemic lupus
erythematosus based on the CYP3A5 polymorphism and coadministration
with Wuzhi capsule. J Clin Pharm Ther. 45:309–317. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Mizuno T, Fukuda T, Christians U,
Perentesis JP, Fouladi M and Vinks AA: Population pharmacokinetics
of temsirolimus and sirolimus in children with recurrent solid
tumours: A report from the Children's Oncology Group. Br J Clin
Pharmacol. 83:1097–1107. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yang JW, Liao SS, Zhu LQ, Zhao Y, Zhang Y,
Sun XY, Rao W, Qu W, Li WZ and Sun LY: Population pharmacokinetic
analysis of tacrolimus early after Chinese pediatric liver
transplantation. Int J Clin Pharmacol Ther. 53:75–83.
2015.PubMed/NCBI View
Article : Google Scholar
|
|
17
|
Lindbom L, Pihlgren P and Jonsson EN:
PsN-Toolkit-a collection of computer intensive statistical methods
for non-linear mixed effect modeling using NONMEM. Comput Methods
Programs Biomed. 79:241–257. 2005.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jusko WJ, Piekoszewski W, Klintmalm GB,
Shaefer MS, Hebert MF, Piergies AA, Lee CC, Schechter P and Mekki
QA: Pharmacokinetics of tacrolimus in liver transplant patients.
Clin Pharmacol Ther. 57:281–290. 1995.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Anderson BJ and Holford NH:
Mechanism-based concepts of size and maturity in pharmacokinetics.
Annu Rev Pharmacol Toxicol. 48:303–332. 2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ho S, Clipstone N, Timmermann L, Northrop
J, Graef I, Fiorentino D, Nourse J and Crabtree GR: The mechanism
of action of cyclosporin A and FK506. Clin Immunol Immunopathol.
80:S40–S45. 1996.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Wang DD, Chen X and Li ZP: Tacrolimus
ameliorates proteinuria in Chinese pediatric lupus nephritis
patients. Int J Clin Exp Med. 12:10931–10937. 2019.
|
|
22
|
Andreu F, Colom H, Grinyo JM, Torras J,
Cruzado JM and Lloberas N: Development of a population PK model of
tacrolimus for adaptive dosage control in stable kidney transplant
patients. Ther Drug Monit. 37:246–255. 2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Benkali K, Rostaing L, Premaud A, Woillard
JB, Saint-Marcoux F, Urien S, Kamar N, Marquet P and Rousseau A:
Population pharmacokinetics and Bayesian estimation of tacrolimus
exposure in renal transplant recipients on a new once-daily
formulation. Clin Pharmacokinet. 49:683–692. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Bergmann TK, Hennig S, Barraclough KA,
Isbel NM and Staatz CE: Population pharmacokinetics of tacrolimus
in adult kidney transplant patients: Impact of CYP3A5 genotype on
starting dose. Ther Drug Monit. 36:62–70. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Han N, Ha S, Yun HY, Kim MG, Min SI, Ha J,
Lee JI, Oh JM and Kim IW: Population
pharmacokinetic-pharmacogenetic model of tacrolimus in the early
period after kidney transplantation. Basic Clin Pharmacol Toxicol.
114:400–406. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Zhao W, Elie V, Roussey G, Brochard K,
Niaudet P, Leroy V, Loirat C, Cochat P, Cloarec S, André JL, et al:
Population pharmacokinetics and pharmacogenetics of tacrolimus in
de novo pediatric kidney transplant recipients. Clin Pharmacol
Ther. 86:609–618. 2009.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zuo XC, Ng CM, Barrett JS, Luo AJ, Zhang
BK, Deng CH, Xi LY, Cheng K, Ming YZ, Yang GP, et al: Effects of
CYP3A4 and CYP3A5 polymorphisms on tacrolimus pharmacokinetics in
Chinese adult renal transplant recipients: A population
pharmacokinetic analysis. Pharmacogenet Genomics. 23:251–261.
2013.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Lu YX, Su QH, Wu KH, Ren YP, Li L, Zhou TY
and Lu W: A population pharmacokinetic study of tacrolimus in
healthy Chinese volunteers and liver transplant patients. Acta
Pharmacol Sin. 36:281–288. 2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Musuamba FT, Guy-Viterbo V, Reding R,
Verbeeck RK and Wallemacq P: Population pharmacokinetic analysis of
tacrolimus early after pediatric liver transplantation. Ther Drug
Monit. 36:54–61. 2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wallin JE, Bergstrand M, Wilczek HE,
Nydert PS, Karlsson MO and Staatz CE: Population pharmacokinetics
of tacrolimus in pediatric liver transplantation: Early
posttransplantation clearance. Ther Drug Monit. 33:663–672.
2011.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhang XQ, Wang ZW, Fan JW, Li YP, Jiao Z,
Gao JW, Peng ZH and Liu GL: The impact of sulfonylureas on
tacrolimus apparent clearance revealed by a population
pharmacokinetics analysis in Chinese adult liver-transplant
patients. Ther Drug Monit. 34:126–133. 2012.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zhu L, Yang J, Zhang Y, Jing Y, Zhang Y
and Li G: Effects of CYP3A5 genotypes, ABCB1 C3435T and G2677T/A
polymorphism on pharmacokinetics of Tacrolimus in Chinese adult
liver transplant patients. Xenobiotica. 45:840–846. 2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wang DD, Chen X, Fu M, Zheng QS, Xu H and
Li ZP: Model extrapolation to a real-world dataset: Evaluation of
tacrolimus population pharmacokinetics and drug interaction in
pediatric liver transplantation patients. Xenobiotica. 50:371–379.
2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Wang DD, Chen X and Li ZP: Wuzhi capsule
and haemoglobin influence tacrolimus elimination in paediatric
kidney transplantation patients in a population pharmacokinetics
analysis: A retrospective study. J Clin Pharm Ther. 44:611–617.
2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Fay JW, Nash RA, Wingard JR, Przepiorka D,
Collins RH, Anasetti C, Devine SM, Pineiro LA, Storb RF, Aro RM, et
al: FK 506-based immunosuppression for prevention of graft versus
host disease after unrelated donor marrow transplantation.
Transplant Proc. 27(1374)1995.PubMed/NCBI
|
|
36
|
Fay JW, Wingard JR, Antin JH, Collins RH,
Piñeiro LA, Blazar BR, Saral R, Bierer BE, Przepiorka D,
Fitzsimmons WE, et al: FK506 (Tacrolimus) monotherapy for
prevention of graft-versus-host disease after histocompatible
sibling allogenic bone marrow transplantation. Blood. 87:3514–3519.
1996.PubMed/NCBI
|
|
37
|
Nash RA, Etzioni R, Storb R, Furlong T,
Gooley T, Anasetti C, Appelbaum FR, Doney K, Martin P, Slattery J,
et al: Tacrolimus (FK506) alone or in combination with methotrexate
or methylprednisolone for the prevention of acute graft-versus-host
disease after marrow transplantation from HLA-matched siblings: A
single-center study. Blood. 85:3746–3753. 1995.PubMed/NCBI
|
|
38
|
Nash RA, Piñeiro LA, Storb R, Deeg HJ,
Fitzsimmons WE, Furlong T, Hansen JA, Gooley T, Maher RM, Martin P,
et al: FK506 in combination with methotrexate for the prevention of
graft-versus-host disease after marrow transplantation from matched
unrelated donors. Blood. 88:3634–3641. 1996.PubMed/NCBI
|
|
39
|
Przepiorka D, Ippoliti C, Khouri I, Woo M,
Mehra R, Le Bherz D, Giralt S, Gajewski J, Fischer H, Fritsche H,
et al: Tacrolimus and minidose methotrexate for prevention of acute
graft-versus-host disease after matched unrelated donor marrow
transplantation. Blood. 88:4383–4389. 1996.PubMed/NCBI
|
|
40
|
Uberti JP, Silver SM, Adams PT, Jacobson
P, Scalzo A and Ratanatharathorn V: Tacrolimus and methotrexate for
the prophylaxis of acute graft-versus-host disease in allogeneic
bone marrow transplantation in patients with hematologic
malignancies. Bone Marrow Transplant. 19:1233–1238. 1997.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Nash RA, Antin JH, Karanes C, Fay JW,
Avalos BR, Yeager AM, Przepiorka D, Davies S, Petersen FB, Bartels
P, et al: Phase 3 study comparing methotrexate and tacrolimus with
methotrexate and cyclosporine for prophylaxis of acute
graft-versus-host disease after marrow transplantation from
unrelated donors. Blood. 96:2062–2068. 2000.PubMed/NCBI
|
|
42
|
Wang D, Chen X, Xu H and Li Z: Population
pharmacokinetics and dosing regimen optimisation of tacrolimus in
Chinese pediatric hematopoietic stem cell transplantation patients.
Xenobiotica. 50:178–185. 2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Monchaud C, de Winter BC, Knoop C, Estenne
M, Reynaud-Gaubert M, Pison C, Stern M, Kessler R, Guillemain R,
Marquet P and Rousseau A: Population pharmacokinetic modelling and
design of a Bayesian estimator for therapeutic drug monitoring of
tacrolimus in lung transplantation. Clin Pharmacokinet. 51:175–186.
2012.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Han Y, Zhou H, Cai J, Huang J, Zhang J,
Shi SJ, Liu YN and Zhang Y: Prediction of tacrolimus dosage in the
early period after heart transplantation: A population
pharmacokinetic approach. Pharmacogenomics. 20:21–35.
2019.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Chandrasekhara PK, Jayachandran NV, Thomas
J, Agrawal S and Narsimulu G: Successful treatment of pyoderma
gangrenosum associated with juvenile idiopathic arthritis with a
combination of topical tacrolimus and oral prednisolone. Clin
Rheumatol. 28:489–490. 2009.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Shimizu M, Ueno K, Ishikawa S, Tokuhisa Y,
Inoue N and Yachie A: Treatment of refractory polyarticular
juvenile idiopathic arthritis with tacrolimus. Rheumatology
(Oxford). 53:2120–2122. 2014.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Tanaka H, Tsugawa K, Suzuki K, Oki ES,
Nonaka K, Kimura S and Ito E: Treatment of difficult cases of
systemic-onset juvenile idiopathic arthritis with tacrolimus. Eur J
Pediatr. 166:1053–1055. 2007.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Wang D, Chen X and Li Z: Treatment of
patients with systemic-onset juvenile idiopathic arthritis with
tacrolimus. Exp Ther Med. 17:2305–2309. 2019.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Choudhry S, Bagga A, Hari P, Sharma S,
Kalaivani M and Dinda A: Efficacy and safety of tacrolimus versus
cyclosporine in children with steroid-resistant nephrotic syndrome:
A randomized controlled trial. Am J Kidney Dis. 53:760–769.
2009.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Gulati A, Sinha A, Gupta A, Kanitkar M,
Sreenivas V, Sharma J, Mantan M, Agarwal I, Dinda AK, Hari P and
Bagga A: Treatment with tacrolimus and prednisolone is preferable
to intravenous cyclophosphamide as the initial therapy for children
with steroid-resistant nephrotic syndrome. Kidney Int.
82:1130–1135. 2012.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Gulati S, Prasad N, Sharma RK, Kumar A,
Gupta A and Baburaj VP: Tacrolimus: A new therapy for
steroid-resistant nephrotic syndrome in children. Nephrol Dial
Transplant. 23:910–913. 2008.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Loeffler K, Gowrishankar M and Yiu V:
Tacrolimus therapy in pediatric patients with treatment-resistant
nephrotic syndrome. Pediatr Nephrol. 19:281–287. 2004.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Roberti I and Vyas S: Long-term outcome of
children with steroid-resistant nephrotic syndrome treated with
tacrolimus. Pediatr Nephrol. 25:1117–1124. 2010.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Yang EM, Lee ST, Choi HJ, Cho HY, Lee JH,
Kang HG, Park YS, Cheong HI and Ha IS: Tacrolimus for children with
refractory nephrotic syndrome: A one-year prospective, multicenter,
and open-label study of Tacrobell(R), a generic formula. World J
Pediatr. 12:60–65. 2016.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Wang D, Lu J, Li Q and Li Z: Population
pharmacokinetics of tacrolimus in pediatric refractory nephrotic
syndrome and a summary of other pediatric disease models. Exp Ther
Med. 17:4023–4031. 2019.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Duddridge M and Powell RJ: Treatment of
severe and difficult cases of systemic lupus erythematosus with
tacrolimus. A report of three cases. Ann Rheum Dis. 56:690–692.
1997.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Miyasaka N, Kawai S and Hashimoto H:
Efficacy and safety of tacrolimus for lupus nephritis: A
placebo-controlled double-blind multicenter study. Mod Rheumatol.
19:606–615. 2009.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Watanabe H, Yamanaka R, Sada KE, Zeggar S,
Katsuyama E, Katsuyama T, Narazaki MT, Tatebe NT, Sugiyama K,
Watanabe KS, et al: The efficacy of add-on tacrolimus for minor
flare in patients with systemic lupus erythematosus: A
retrospective study. Lupus. 25:54–60. 2016.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Yoon KH: Efficacy and cytokine modulating
effects of tacrolimus in systemic lupus erythematosus: A review. J
Biomed Biotechnol. 2010(686480)2010.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Szeto CC, Kwan BC, Lai FM, Tam LS, Li EK,
Chow KM, Gang W and Li PK: Tacrolimus for the treatment of systemic
lupus erythematosus with pure class V nephritis. Rheumatology
(Oxford). 47:1678–1681. 2008.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Kusunoki Y, Tanaka N, Kaneko K, Yamamoto
T, Endo H and Kawai S: Tacrolimus therapy for systemic lupus
erythematosus without renal involvement: A preliminary
retrospective study. Mod Rheumatol. 19:616–621. 2009.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Alsuwaida A: Successful management of
systemic lupus erythematosus nephritis flare-up during pregnancy
with tacrolimus. Mod Rheumatol. 21:73–75. 2011.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Li H, Zhang X and Chen J: Successful
treatment of steroid-refractory systemic lupus
erythematosus-associated protein-losing enteropathy using
combination therapy with tacrolimus and steroid. Lupus.
20:1109–1111. 2011.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Kaieda S, Kobayashi T, Moroki M, Honda S,
Yuge K, Kawano H, Mitsuyama K, Sata M, Ida H, Hoshino T and Fukuda
T: Successful treatment of rectal ulcers in a patient with systemic
lupus erythematosus using corticosteroids and tacrolimus. Mod
Rheumatol. 24:357–360. 2014.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Politt D, Heintz B, Floege J and Mertens
PR: Tacrolimus-(FK 506) based immunosuppression in severe systemic
lupus erythematosus. Clin Nephrol. 62:49–53. 2004.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Zhang Z, Yang C, Zhang L, Yi Q and Hao Z:
Efficacy and safety of tacrolimus in myasthenia gravis: A
systematic review and meta-analysis. Ann Indian Acad Neurol.
20:341–347. 2017.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Wang L, Zhang S, Xi J, Li W, Zhou L, Lu J,
Lu J, Zhang T and Zhao C: Efficacy and safety of tacrolimus for
myasthenia gravis: A systematic review and meta-analysis. J Neurol.
264:2191–2200. 2017.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Komaki Y, Komaki F, Ido A and Sakuraba A:
Efficacy and safety of tacrolimus therapy for active ulcerative
colitis; A systematic review and meta-analysis. J Crohns Colitis.
10:484–494. 2016.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Matsuoka K, Saito E, Fujii T, Takenaka K,
Kimura M, Nagahori M, Ohtsuka K and Watanabe M: Tacrolimus for the
treatment of ulcerative colitis. Intest Res. 13:219–226.
2015.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Hanouneh M, Ritchie MM, Ascha M, Ascha MS,
Chedid A, Sanguankeo A, Zein NN and Hanouneh IA: A review of the
utility of tacrolimus in the management of adults with autoimmune
hepatitis. Scand J Gastroenterol. 54:76–80. 2019.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Scott LJ, McKeage K, Keam SJ and Plosker
GL: Tacrolimus: A further update of its use in the management of
organ transplantation. Drugs. 63:1247–1297. 2003.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Naesens M, Kuypers DR and Sarwal M:
Calcineurin inhibitor nephrotoxicity. Clin J Am Soc Nephrol.
4:481–508. 2009.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Entani C, Izumino K, Iida H, Fujita M,
Asaka M, Takata M and Sasayama S: Effect of a novel
immunosuppressant, FK506, on spontaneous lupus nephritis in
MRL/MpJ-lpr/lpr mice. Nephron. 64:471–475. 1993.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Jusko WJ, Thomson AW, Fung J, McMaster P,
Wong SH, Zylber-Katz E, Christians U, Winkler M, Fitzsimmons WE,
Lieberman R, et al: Consensus document: Therapeutic monitoring of
tacrolimus (FK-506). Ther Drug Monit. 17:606–614. 1995.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Venkataramanan R, Swaminathan A, Prasad T,
Jain A, Zuckerman S, Warty V, McMichael J, Lever J, Burckart G and
Starzl T: Clinical pharmacokinetics of tacrolimus. Clin
Pharmacokinet. 29:404–430. 1995.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Zheng QS and Li LJ: Pharmacometrics: A
quantitative tool of pharmacological research. Acta Pharmacol Sin.
33:1337–1338. 2012.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Chen CY, Zhou Y, Cui YM, Yang T, Zhao X
and Wu Y: Population pharmacokinetics and dose simulation of
oxcarbazepine in Chinese paediatric patients with epilepsy. J Clin
Pharm Ther. 44:300–311. 2019.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Chen Y, Wu D, Dong M, Zhu Y, Lu J, Li X,
Chen C and Li Z: Population pharmacokinetics of vancomycin and
AUC-guided dosing in Chinese neonates and young infants. Eur J Clin
Pharmacol. 74:921–930. 2018.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Zheng Y, Liu SP, Xu BP, Shi ZR, Wang K,
Yang JB, Huang X, Tang BH, Chen XK, Shi HY, et al: Population
pharmacokinetics and dosing optimization of azithromycin in
children with community-acquired pneumonia. Antimicrob Agents
Chemother. 62(e00686-18)2018.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Wang DD, Ye QF, Chen X, Xu H and Li ZP:
Population pharmacokinetics and initial dosing regimen optimization
of cyclosporin in pediatric hemophagocytic lymphohistiocytosis
patients. Xenobiotica. 50:435–441. 2020.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Lu T, Zhu X, Xu S, Zhao M, Huang X, Wang Z
and Zhao L: Dosage optimization based on population pharmacokinetic
analysis of tacrolimus in chinese patients with nephrotic syndrome.
Pharm Res. 36(45)2019.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Wang X, Han Y, Chen C, Ma L, Xiao H, Zhou
Y, Cui Y, Wang F, Su B, Yao Y and Ding J: Population
pharmacokinetics and dosage optimization of tacrolimus in pediatric
patients with nephrotic syndrome. Int J Clin Pharmacol Ther.
57:125–134. 2019.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Wei H, Tao X, Di P, Yang Y, Li J, Qian X,
Feng J and Chen W: Effects of traditional chinese medicine Wuzhi
capsule on pharmacokinetics of tacrolimus in rats. Drug Metab
Dispos. 41:1398–1403. 2013.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Xin HW, Li Q, Wu XC, He Y, Yu AR, Xiong L
and Xiong Y: Effects of Schisandra sphenanthera extract on the
blood concentration of tacrolimus in renal transplant recipients.
Eur J Clin Pharmacol. 67:1309–1311. 2011.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Xin HW, Wu XC, Li Q, Yu AR, Zhu M, Shen Y,
Su D and Xiong L: Effects of Schisandra sphenanthera extract on the
pharmacokinetics of tacrolimus in healthy volunteers. Br J Clin
Pharmacol. 64:469–475. 2007.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Qin XL, Bi HC, Wang CX, Li JL, Wang XD,
Liu LS, Chen X and Huang M: Study of the effect of Wuzhi tablet
(Schisandra sphenanthera extract) on tacrolimus tissue distribution
in rat by liquid chromatography tandem mass spectrometry method.
Biomed Chromatogr. 24:399–405. 2010.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Qin XL, Bi HC, Wang XD, Li JL, Wang Y, Xue
XP, Chen X, Wang CX, Xu le J, Wang YT and Huang M: Mechanistic
understanding of the different effects of Wuzhi Tablet (Schisandra
sphenanthera extract) on the absorption and first-pass intestinal
and hepatic metabolism of Tacrolimus (FK506). Int J Pharm.
389:114–121. 2010.PubMed/NCBI View Article : Google Scholar
|