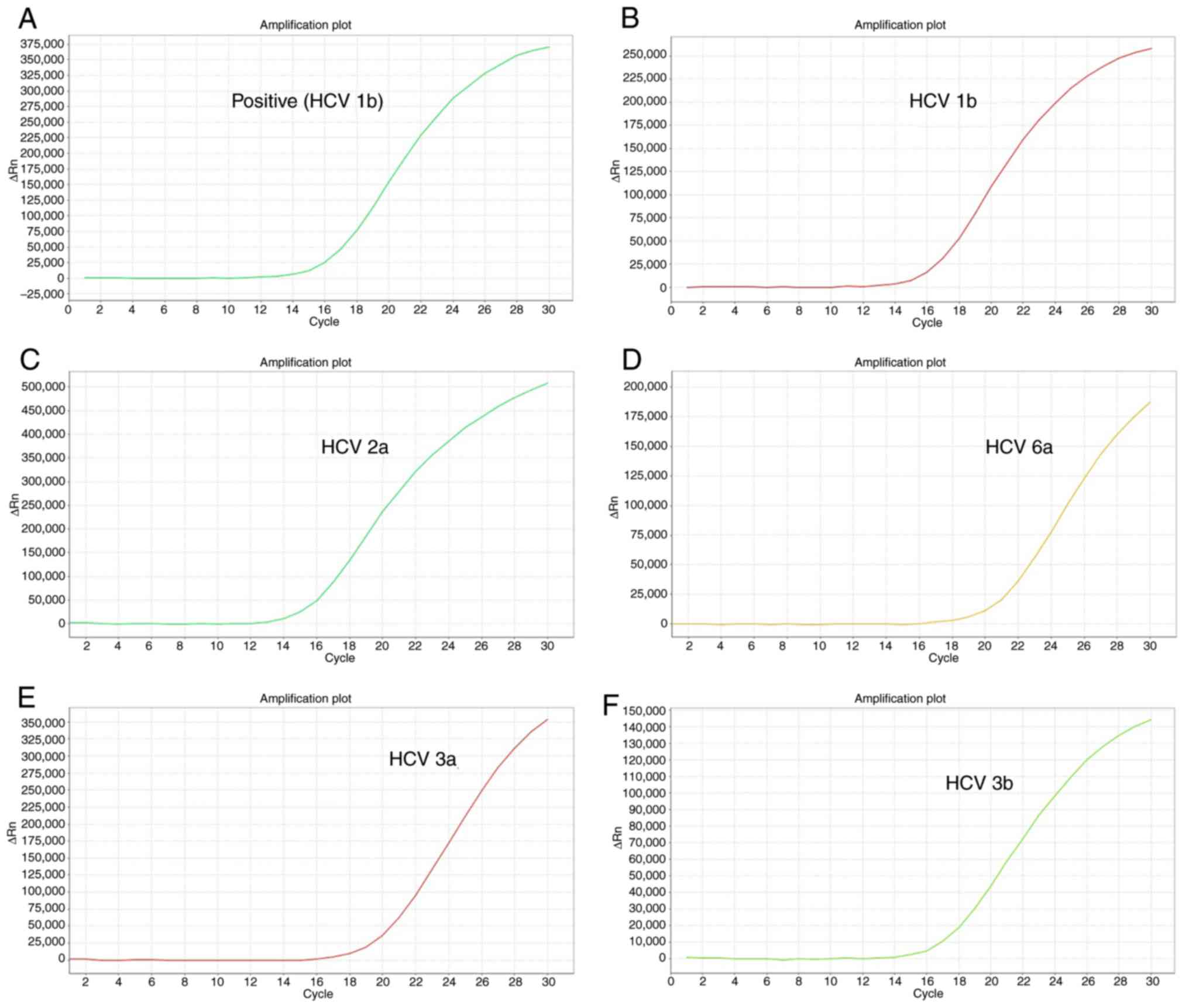
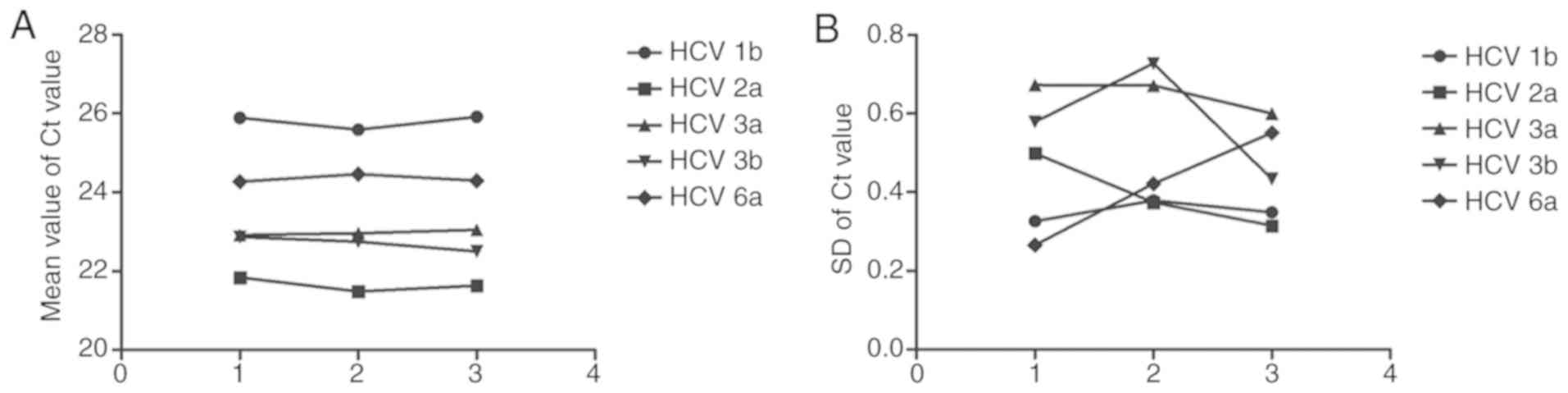
|
1
|
Lavanchy D: Evolving epidemiology of
hepatitis C virus. Clin Microbiol Infect. 17:107–115.
2011.PubMed/NCBI View Article : Google Scholar
|
|
2
|
World Health Organization: Guidelines for
the Screening Care and Treatment of Persons with Chronic Hepatitis
C Infection: Updated Version, Geneva, 2016. https://www.who.int/hepatitis/publications/hep-c-guidelines-2016_170.png.
|
|
3
|
Razavi H, Waked I, Sarrazin C, Myers RP,
Idilman R, Calinas F, Vogel W, Mendes Correa MC, Hézode C, Lázaro
P, et al: The present and future disease burden of hepatitis C
virus (HCV) infection with today's treatment paradigm. J Viral
Hepat. 21 (Suppl 1)(S34-S59)2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Petruzziello A, Marigliano S, Loquercio G,
Cozzolino A and Cacciapuoti C: Global epidemiology of hepatitis C
virus infection: An up-date of the distribution and circulation of
hepatitis C virus genotypes. World J Gastroenterol. 22:7824–7840.
2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Polaris Observatory HCV Collaborators;
Blach S, Zeuzem S, Manns M, Altraif I, Duberg AS, Muljono DH, Waked
I, Alavian SM, Lee MH, et al: Global prevalence and genotype
distribution of hepatitis C virus infection in 2015: A modelling
study. Lancet Gastroenterol Hepatol. 2:161–176. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Tong YQ, Liu B, Liu H, Zheng HY, Gu J, Liu
H, Song EJ, Song C and Li Y: Accurate genotyping of hepatitis C
virus through nucleotide sequencing and identification of new HCV
subtypes in China population. Clin Microbiol Infect.
21(874.e9-e847.e21)2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Grebely J and Dore GJ: What is killing
people with hepatitis C virus infection? Semin Liver Dis.
31:331–339. 2011.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Schinazi R, Halfon P, Marcellin P and
Asselah T: HCV direct-acting antiviral agents: The best
interferon-free combinations. Liver Int. 34 (Suppl
1)(S69-S78)2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
De Clercq E: Current race in the
development of DAAs (direct-acting antivirals) against HCV. Biochem
Pharmacol. 89:441–452. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
European Association for the Study of the
Liver. EASL recommendations on treatment of hepatitis C 2018. J
Hepatol. 69:461–511. 2018.
|
|
11
|
Manns MP, Buti M, Gane E, Pawlotsky JM,
Razavi H, Terrault N and Younossi Z: Hepatitis C virus infection.
Nat Rev Dis Primers. 3(17006)2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
European Association for the Study of the
Liver. EASL recommendations on treatment of Hepatitis C 2016. J
Hepatol. 66:153–194. 2017.
|
|
13
|
Yang R and Wei L: Profile of the VERSANT
HCV genotype 2.0 assay. Expert Rev Mol Diagn. 18:995–1004.
2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Verbeeck J, Stanley MJ, Shieh J, Celis L,
Huyck E, Wollants E, Morimoto J, Farrior A, Sablon E,
Jankowski-Hennig M, et al: Evaluation of versant Hepatitis C virus
genotype assay (LiPA) 2.0. J Clin Microbiol. 46:1901–1906.
2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gryadunov D, Nicot F, Dubois M,
Mikhailovich V, Zasedatelev A and Izopet J: Hepatitis C virus
genotyping using an oligonucleotide microarray based on the NS5B
sequence. J Clin Microbiol. 48:3910–3917. 2010.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Rodriguez-Frias F, Nieto-Aponte L, Gregori
J, Garcia-Cehic D, Casillas R, Tabernero D, Homs M, Blasi M, Vila
M, Chen Q, et al: High HCV subtype heterogeneity in a chronically
infected general population revealed by high-resolution hepatitis C
virus subtyping. Clin Microbiol Infect.
23(775.e1-775.e6)2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Soria ME, Gregori J, Chen Q, García-Cehic
D, Llorens M, de Ávila AI, Beach NM, Domingo E, Rodríguez-Frías F,
Buti M, et al: Pipeline for specific subtype amplification and drug
resistance detection in hepatitis C virus. BMC Infect Dis.
18(446)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Roy F, Mendoza L, Hiebert J, McNall RJ,
Bankamp B, Connolly S, Lüdde A, Friedrich N, Mankertz A, Rota PA
and Severini A: Rapid identification of measles virus vaccine
genotype by real-time PCR. J Clin Microbiol. 55:735–743.
2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Warkad SD, Nimse SB, Song KS and Kim T:
HCV detection, discrimination, and genotyping technologies. Sensors
(Basel). 18(pii: E3423)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Nyan DC and Swinson KL: A method for rapid
detection and genotype identification of hepatitis C virus 1-6 by
one-step reverse transcription loop-mediated isothermal
amplification. Int J Infect Dis. 43:30–36. 2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang Y, Chen LM and He M: Hepatitis C
Virus in mainland China with an emphasis on genotype and subtype
distribution. Virol J. 14(41)2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chen Y, Yu C, Yin X, Guo X, Wu S and Hou
J: Hepatitis C virus genotypes and subtypes circulating in Mainland
China. Emerg Microbes Infect. 6(e95)2017.PubMed/NCBI View Article : Google Scholar
|