Introduction
Severe acute pancreatitis (SAP) is an acute disease
characterized by a complex pathogenesis that causes rapid failure
of numerous organs (including renal, respiratory and/or
cardiovascular organs) (1). The
global mortality rate of patients with SAP ranges from 13-35%
(2,3)
and can reach 70% in case of pancreatic infection (4-9).
However, surgical treatment of SAP is also associated with high
complication (34-95%) and mortality (11-39%) rates (8,10). The
risk of mortality from multiple organ dysfunction and sepsis
following surgery remains high. Patients with SAP mainly succumb to
infection, mostly secondary pancreatic infection and
infection-related organ failure in the later stages (11). The intestinal tract is the central
organ location of the SAP-induced systemic inflammatory response
syndrome (SIRS). It has been demonstrated that in SAP, the
intestinal mucosa is damaged and intestinal permeability is
increased (12,13), and bacterial translocation (BT)
consists of the entry of intestinal bacteria and endotoxins into
the circulatory and lymphatic systems and distant organs, passing
through the intestinal barrier (14,15). BT
from the gastrointestinal tract is considered as the main cause of
sepsis and systemic infection in patients with SAP (16,17).
Previous studies have reported that in SAP, the intestinal tight
junction (TJ) barrier is destroyed and intestinal mucosal
permeability is increased, thereby facilitating BT (18,19),
indicating that intestinal epithelial TJs serve a crucial role in
the regulation of intestinal mucosal barrier function (20,21).
TJs mainly comprise intercellular transmembrane
proteins, including claudin and occludin, which are linked to
different peripheral membrane proteins located inside the plasma
membrane, mainly to zonula occludens-1 (ZO-1) (22). TJs form a physical barrier between
mucosal epithelial cells. The F-actin cytoskeleton is involved in
numerous important cellular processes, including intercellular
junctions, cell morphology, cell movement and signal transduction,
which are mediated by the interaction between F-actin and the cell
membrane (23). Cell division cycle
42 (Cdc42) is a widely expressed Rho family GTP-binding protein
that regulates actin skeleton assembly and rearrangement, thereby
affecting cell morphology, migration and endocytosis (24).
Carbachol is a cholinergic agonist that promotes
gastrointestinal motility, increases glandular secretion and
protects the intestinal barrier (25-27).
A previous study demonstrated that carbachol increases intestinal
transmembrane electrical resistance and protects the intestinal
barrier in a rat model of inflammatory bowel disease (IBD)
(26). In addition, carbochol can
reduce intestinal barrier permeability and TJ damage induced by
intraperitoneal injection of lipopolysaccharide (LPS) (27). A previous meta-analysis demonstrated
that octreotide, a synthetic somatostatin analogue used to treat
moderate to severe acute pancreatitis, has no significant benefits
on the clinical outcomes of patients (28). Furthermore, a long-term clinical
study reported that the clinical benefits of somatostatin or
octreotide in patients with SAP are not primarily achieved by
inhibiting pancreatic secretion, since a large number of pancreatic
acinar lesions and necrosis in patients with SAP result in
insufficient residual acinar exocrine secretion (29). It is hypothesized that it may be
possible to protect the SAP intestinal barrier by intraperitoneal
injection of carbachol without aggravating pancreatic injury.
Numerous studies reported that carbachol can protect
the intestinal barrier (25-27);
however, its role in the intestinal barrier in patients with SAP
remains unknown. The present study aimed to investigate the effect
of carbachol on the intestinal TJ barrier in a rat model of SAP.
The results suggested that carbachol may prevent intestinal barrier
injury in SAP rats by regulating the Cdc42-F-actin cytoskeleton,
without aggravating pancreatic injury.
Materials and methods
Animal model
A total of 140 7-week old Wistar rats (healthy
males; 230-260 g; Animal Center of Qingdao University) were
acclimated to the laboratory over 3 weeks and were then randomly
assigned into the sham operation (SO) group (n=10), SO + carbachol
group (n=10), SAP group (n=60), and SAP + carbachol group (n=60).
All animals were housed at standard temperatures (25±2˚C) and at a
relative humidity (50-70%) under a 12 h light/dark cycle with free
access to food and water. All experimental protocols were performed
according to the National Institutes of Health Laboratory Animal
Guidelines and were approved by the Institutional Animal Care and
Use Committee of Qingdao University.
The SAP rat model was constructed as previously
described (30). Briefly, the rats
fasted for 12 h, and were then weighed and anesthetized using an
intraperitoneal injection of pentobarbital sodium (3%; 50 mg/kg).
During laparotomy, the biliary pancreatic duct was clipped near the
hepatic portal and duodenal intubation was performed with a
catheter. Sodium taurocholate (30)
(5%; 1 ml/kg) was slowly injected into the biliopancreatic duct to
induce SAP. For the SAP + carbachol group, carbachol (50 µg/kg) was
injected intraperitoneally 12 h following SAP induction. For the
SAP group, rats were intraperitoneally injected with the same
volume of sterile saline 12 h following SAP induction. In the SO
group, the same amount of sterile saline was injected into the
biliopancreatic duct and abdominal cavity of rats. In the SO +
carbachol group, carbachol (50 µg/kg) was injected
intraperitoneally.
All surviving rats were anesthetized 24 h following
SAP induction and treatments. Blood samples were collected from the
inferior vena cava and were divided into two microtubules. One
microtubule was centrifuged at 4˚C and 3,000 x g for 5 min. The
supernatant was collected and kept in an Eppendorf tube at -20˚C
for analysis of serum lipase and amylase. The second microtubule
containing EDTA was stored at -20˚C for bacterial DNA analysis.
Finally, the ileum and pancreatic tissue were isolated and the
ileum was divided into two sections. The first section was stored
at -80˚C for western blot analysis, and the second section, along
with pancreatic tissue, was fixed in 4% paraformaldehyde at room
temperature for 48 h.
Bacterial detection by 16S rRNA
sequencing
BT was assessed in the present study by detecting
the presence of bacteria in rat blood. Bacterial DNA was isolated
from bacterial colonies using a Rapid Bacterial Genomic DNA
Isolation kit (cat. no. B518225; Sangon Biotech Co., Ltd.)
according to the manufacturer's protocol, and isolated DNA was, as
a template, used to amplify the hypervariable regions (V6-V8) of
the 16S rRNA gene. R1521-1539 (5'-AGGAGGTGATCCAACCGCA-3') and
F1169-1187-GC (5'-GC-clamp-AACTGGAGGAAGGTGGGGA-3'; Sangon Biotech
Co., Ltd.) were used as the universal primers. Negative and
positive controls were assessed twice to avoid false-positive
results.
PCR was performed using a touchdown thermocycling
program after adding 100 ng DNA to the reactant. The following
thermocycling conditions were used: Initial denaturation for 5 min
at 95˚C followed by denaturation for 25 cycles for 30 sec at 95˚C,
annealing for 30 sec at 55˚C and final extension for 5 min at 72˚C.
PCR products were visualized on 2% agarose gel. DNA extracted from
E. coli served as a positive control and ddH2O was used as a
negative control. The final products were analyzed using a Roche GS
FLX 454 Sequencer (Roche Diagnostics, Inc.). By using advanced
BLAST (https://blast.ncbi.nlm.nih.gov/Blast.cgi) searches,
the results of 16S rRNA sequence matched with those from GenBank
(http://www.ncbi.nlm.nih.gov/genbank)
and Ribosomal Database Project (http://rdp.cme.msu.edu) from National Center for
Biotechnology Information.
Serum lipase and amylase
Serum lipase and amylase were detected with an
Olympus AU600 automatic biochemical analyzer (Olympus Corporation),
according to the manufacturer's instructions.
Histopathological score
Intestinal and pancreatic samples fixed in
polyformaldehyde were embedded in paraffin. Sections (5-µm thick)
were stained with hematoxylin and eosin at room temperature for 5
min and morphological changes were observed using an optical
microscope (magnification, x200). The degree of pancreatic injury
was assessed as previously described (31). The score ranged between 0 and 16
according to the degree of edema, acinar necrosis, hemorrhage and
fat necrosis, and inflammation. Histological grading of small
intestinal injury was evaluated as previously described (32). The grade ranged between 0 and 5
according to the degree of damage to the mucosal villi,
subepithelial space, lamina propria, dilated capillaries, lifting
epithelial layer and denuded tips of mucosal villi.
Immunofluorescence assay of intestinal
F-actin and TJs
Immunostaining was performed as previously described
(33), with samples that were
previously fixed in PFA. Paraffin-embedded sections (5 µm) were
dried at 37˚C for 15 min and boiled in 2 mM EDTA acid solution for
10 min. Non-specific binding sites were blocked with 1% bovine
serum albumin (Roche Applied Science) and 5% (v/v) normal goat
serum (Gibco; Thermo Fisher Scientific, Inc.) diluted in PBS at
room temperature for 30 min. Sections were incubated overnight at
4˚C with the following primary antibodies: Rabbit anti-claudin-2
(1:500; Zymed; Thermo Fisher Scientific, Inc.; cat. no. 516100),
rabbit anti-ZO-1 (1:500; Abcam; cat. no. ab96587) and rabbit
anti-occludin (1:500; Zymed; Thermo Fisher Scientific, Inc.; cat.
no. 711500). Sections were washed with PBS and were incubated with
phalloidin-iFluor 594 (1:500; Abcam; cat. no. ab176757) and Alexa
Fluor goat anti-rabbit IgG (1:500; 488 wavelength; Abcam; cat. no.
ab150077) for 30 min at room temperature. Nuclei were stained with
DAPI for 5 min. Images were captured using the IX71 fluorescence
inverted microscope (Olympus Corporation). Fluorescence intensity
was analyzed using Image J software (version 1.46; National
Institutes of Health).
Immunohistochemistry (IHC)
IHC staining was performed for Cdc42 as previously
described (34) by using the primary
antibody rabbit anti-Cdc42 (1:500; Abcam; cat. no. ab187643) and
the horseradish peroxidase-conjugated secondary antibody goat
anti-rabbit IgG H&L (1:1,000; Abcam; cat. no. ab150080). Images
were visualized with a fluorescent microscope (magnification, x200;
DM6000B; Leica Microsystems GmbH) (34). IHC staining was scored as previously
described (35) and following the
German ImmunoReactive score system (36) according to the percentage of
positively stained cells and the staining intensity. Staining
intensity was scored as follows: 0, no staining; 1, weak staining;
2, moderate staining; and 3, strong staining. The score was defined
as 0, 1, 2, 3 or 4 for 0, 1-10%, 11-50%, 51-80% and 81-100% of
positively stained cells, respectively. The average of the lower
and the higher staining intensities was calculated when an uneven
distribution between staining intensity or multifocal
immunoreactivity were observed. A final IHC score in the range 0-12
was obtained by multiplying the score by the staining intensity.
IHC scores of 0, 1-4, 5-8 and 9-12 were considered as negative,
weak, moderate and strong, respectively. Data were analyzed using
Image-Pro Plus 6.0 software (Media Cybernetics, Inc.).
Western blotting
Proteins were extracted from the small intestine
using RIPA lysis buffer (Beyotime Institute of Biotechnology) at
4˚C for 30 min. Protein concentration was determined using
bicinchoninic acid protein assay kit. Proteins (50 µg) were
separated by 10% SDS-PAGE and transferred onto a polyvinylidene
fluoride membrane, which was subsequently blocked with 5% skim milk
at room temperature for 1 h. Membranes were incubated at 4˚C
overnight with the following primary antibodies: Mouse anti-F-actin
(1:1,000; Abcam; cat. no. ab205); rabbit anti-claudin-2 (1:1,000;
Zymed; Thermo Fisher Scientific, Inc.; cat. no. 516100); rabbit
anti-Cdc42 (1:1,000; Abcam; cat. no. ab187643); rabbit anti-ZO-1
(1;1,000; Abcam; cat. no. ab96587); rabbit anti-occludin (1;1,000;
Zymed; Thermo Fisher Scientific, Inc.; cat. no. 711500); and
anti-GAPDH (1:1,000; Sigma-Aldrich: Merck KGaA; cat. no. G5262).
Membranes were subsequently incubated with horseradish peroxidase
(HRP)-conjugated Rb IgG (H+L; 1:3,000; OriGene Technologies, Inc.;
cat. no. ZB-2301) or HRP-conjugated anti-mouse IgG (H+L; 1:3,000;
OriGene Technologies, Inc.; cat. no. ZB-2305) at room temperature
for 1 h. Bands were visualized using ECL (OriGene Technologies,
Inc.; cat. no. sc-2048) and the relative expression of bands was
normalized to the endogenous control GAPDH using Image-Pro plus 6.0
software (Media Cybernetics, Inc.).
Statistical analysis
SPSS version 24 (IBM Corp.) was used to analyze the
data. Parametric analysis was performed as the data were normally
distributed. Experiments were repeated independently at least three
times, and the data were expressed as the mean ± standard
deviation. χ2 test was used to analyze the differences
between groups after converting categorical variables into
percentages or frequencies. One-way ANOVA followed by Tukey's post
hoc test was used to analyze differences among three or more
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
Mortality and BT rates
At 24 h following SAP induction, no death was
observed in the SO and SO + carbachol group, whereas 30 rats died
in the SAP group (mortality rate, 50%) and 18 rats died in the SAP
+ carbachol group (mortality rate, 30%). The results from bacterial
species detection from the blood of rats in the SAP and SAP +
carbachol groups are presented in Table
I. No BT was observed in the SO and SO + carbachol group.
However, numerous bacteria were detected in the blood of 18 rats in
the SAP group (BT rate, 60%) and of 14 rats in the SAP + carbachol
group (BT rate, 33.3%). In addition, the mortality (30% vs. 50%;
P<0.05) and BT rates (33.3% vs. 60%; P<0.05) of rats in the
SAP + carbachol group were significantly lower compared with rats
in the SAP group.
 | Table IBacteria species identified from the
blood of rats in the SAP and SAP + carbachol groups. |
Table I
Bacteria species identified from the
blood of rats in the SAP and SAP + carbachol groups.
| A, SAP rats |
|---|
| Rat number | Bacterial
species |
|---|
| 2 | Escherichia
coli |
| 5 | Citrobacter
freundii |
| 9 | Enterococcus
aerogenes |
| 10 | Streptococcus
pneumonia |
| 12 | Enterococcus
faecium |
| 17 | Escherichia
coli |
| 23 | Enterococcus
aerogenes |
| 28 | Prevotella
copri |
| 31 | Citrobacter
freundii |
| 33 | Escherichia
coli |
| 34 | Enterococcus
faecium |
| 38 | Streptococcus
pneumonia |
| 43 | Prevotella
copri |
| 45 | Escherichia
coli |
| 49 | Enterococcus
aerogenes |
| 52 | Enterococcus
aerogenes |
| 55 | Enterococcus
faecium |
| 57 | Escherichia
coli |
| B, SAP + carbachol
rats |
| 1 | Citrobacter
freundii |
| 3 | Escherichia
coli |
| 8 | Escherichia
coli |
| 12 | Streptococcus
pneumonia |
| 17 | Enterococcus
aerogenes |
| 21 | Enterococcus
faecium |
| 27 | Prevotella
copri |
| 33 | Citrobacter
freundii |
| 38 | Escherichia
coli |
| 42 | Enterococcus
faecium |
| 46 | Prevotella
copri |
| 49 | Escherichia
coli |
| 54 | Escherichia
coli |
| 58 | Enterococcus
aerogenes |
Serum levels of lipase and
amylase
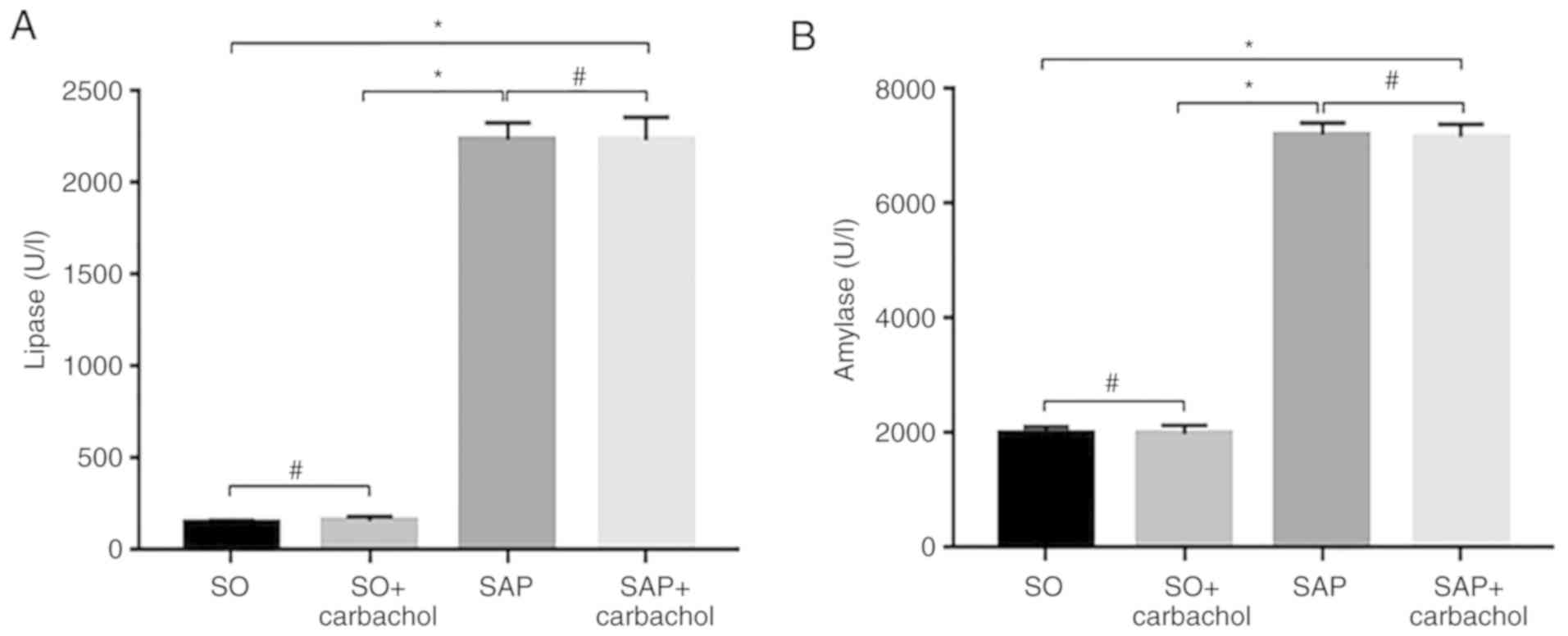
As presented in Fig.
1A, the serum levels of lipase in the SAP and SAP + carbachol
groups were significantly higher compared with the SO group
(P<0.05). In addition, there was no difference between the SAP
and SAP + carbachol groups (P>0.05), and between the SO and SO +
carbachol groups (P>0.05). These results were similar for the
amylase serum level (Fig. 1B).
Intestinal and pancreatic
histopathological scores
The morphology of the intestinal mucosa and pancreas
was evaluated. No injury was identified in the intestinal
epithelium and pancreas of rats in the SO (Fig. 2A, E,
I and J) and SO + carbachol (Fig. 2B and F-J) groups. Pancreatic and intestinal
injury in the SAP and SAP + carbachol groups was significantly
higher compared with SO group (P<0.01, Fig. 2A-D and I; P<0.01, Fig. 2E, G,
H and J). Scores for pancreatic injury were not
different between the SAP + carbachol and SAP groups (P>0.05;
Fig. 2C, D and I);
however, the score for intestinal injury in the SAP group was
significantly higher compared with the SAP + carbachol group
(P<0.01; Fig. 2G, H and J).
Pancreatic tissue from SAP (Fig. 2C)
and SAP + carbachol groups (Fig. 2D)
exhibited hemorrhage and fat necrosis, interstitial edema, a
disordered lobular structure, broad necrosis of acinar cells and
infiltrating inflammatory cells. These results confirmed that the
establishment of the SAP rat model was successful.
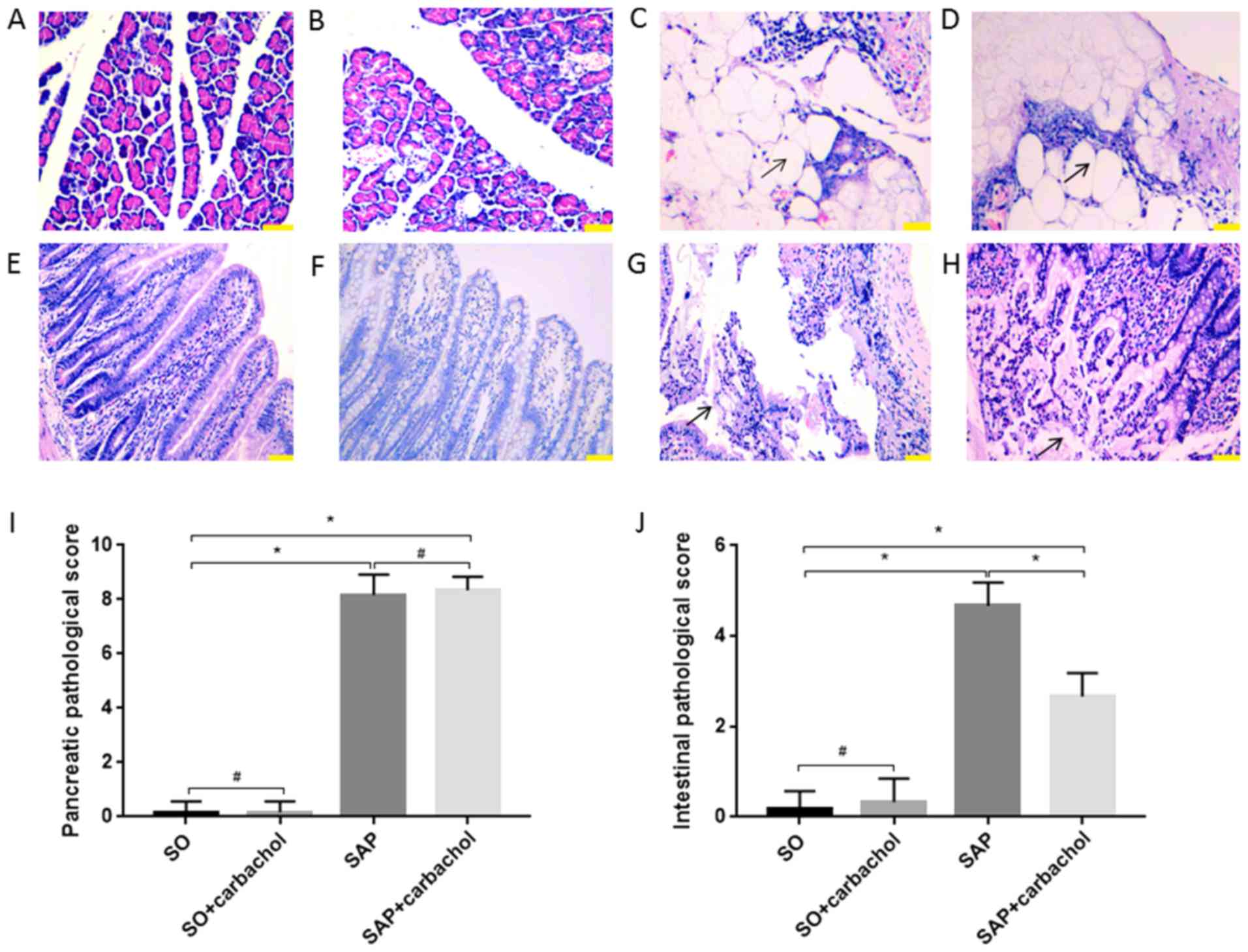 | Figure 2Representative sections of pancreatic
and intestinal tissues stained with hematoxylin and eosin.
Pancreatic tissue from the (A) SO and (B) SO + carbachol groups; no
notable pathological changes were seen. Pancreatic tissue from the
(C) SAP and (D) SAP + carbachol groups; hemorrhage and fat
necrosis, interstitial edema, a fuzzy lobular structure, broad
necrosis of acinar cells and infiltrating inflammatory cells were
observed. Black arrows indicate adipocyte necrosis. Intestinal
tissue of the (E) SO and (F) SO + carbachol groups; no significant
pathological injury was observed. Intestinal tissue of the (G) SAP
group; numerous inflammatory cells were observed to be infiltrating
the thinning intestinal wall, as well as denuded villi with the
lamina propria and dilated capillaries, and digestion and
disintegration of the lamina propria. Black arrows indicate
digestion and disintegration of the lamina propria. Intestinal
tissue of the (H) SAP + carbachol group; mucosal glands reduced,
tips denuded, local bleeding and necrosis, inflammatory cells
infiltrated in the thinning intestinal wall. Black arrows indicate
denuded tips of mucosal villi. Comparison of the pathological
scores of (I) pancreatic and (J) intestinal tissues. Magnification,
x200. Scale bar, 100 µm. Results are expressed as the mean ±
standard deviation (n=6 randomly selected mice in each group).
*P<0.01 and #P>0.05. SAP, severe acute
pancreatitis; SO, sham operation. |
Expression of TJ proteins, F-actin and
Cdc42 in intestine
The expression of Cdc42, F-actin and TJ proteins in
the small intestine of rats in all groups was investigated by
western blotting. Whereas claudin-2 expression was significantly
increased (P<0.05; Fig. 3A and
F), ZO-1 and occludin expression
were significantly decreased in the SAP and SAP + carbachol groups
(P<0.05; Fig. 3A, C and E)
compared with the SO group. Furthermore, claudin-2 expression was
significantly decreased (P<0.05; Fig.
3A and F), but ZO-1 and occludin
expressions were significantly increased in the SO + carbachol
group compared with the SAP and SAP + carbachol groups (P<0.05;
Fig. 3A, C and E).
Expression of ZO-1 and occludin was significantly increased in the
SAP + carbachol group compared with the SAP group (P<0.05;
Fig. 3A, C and E),
whereas claudin-2 expression was significantly decreased in the SAP
+ carbachol group compared with the SAP group (P<0.05; Fig. 3A and F). In addition, Cdc42 and F-actin
expression in the SAP and SAP + carbachol groups was significantly
increased compared with the SO group (P<0.05; Fig. 3A, B
and D); however, expression of these
proteins was significantly decreased in the SO + carbachol group
compared with the SO group (P<0.05; Fig. 3A, B
and D). Expression of Cdc42 and
F-actin was significantly decreased in the SAP + carbachol group
compared with the SAP group (P<0.05; Fig. 3A, B
and D).
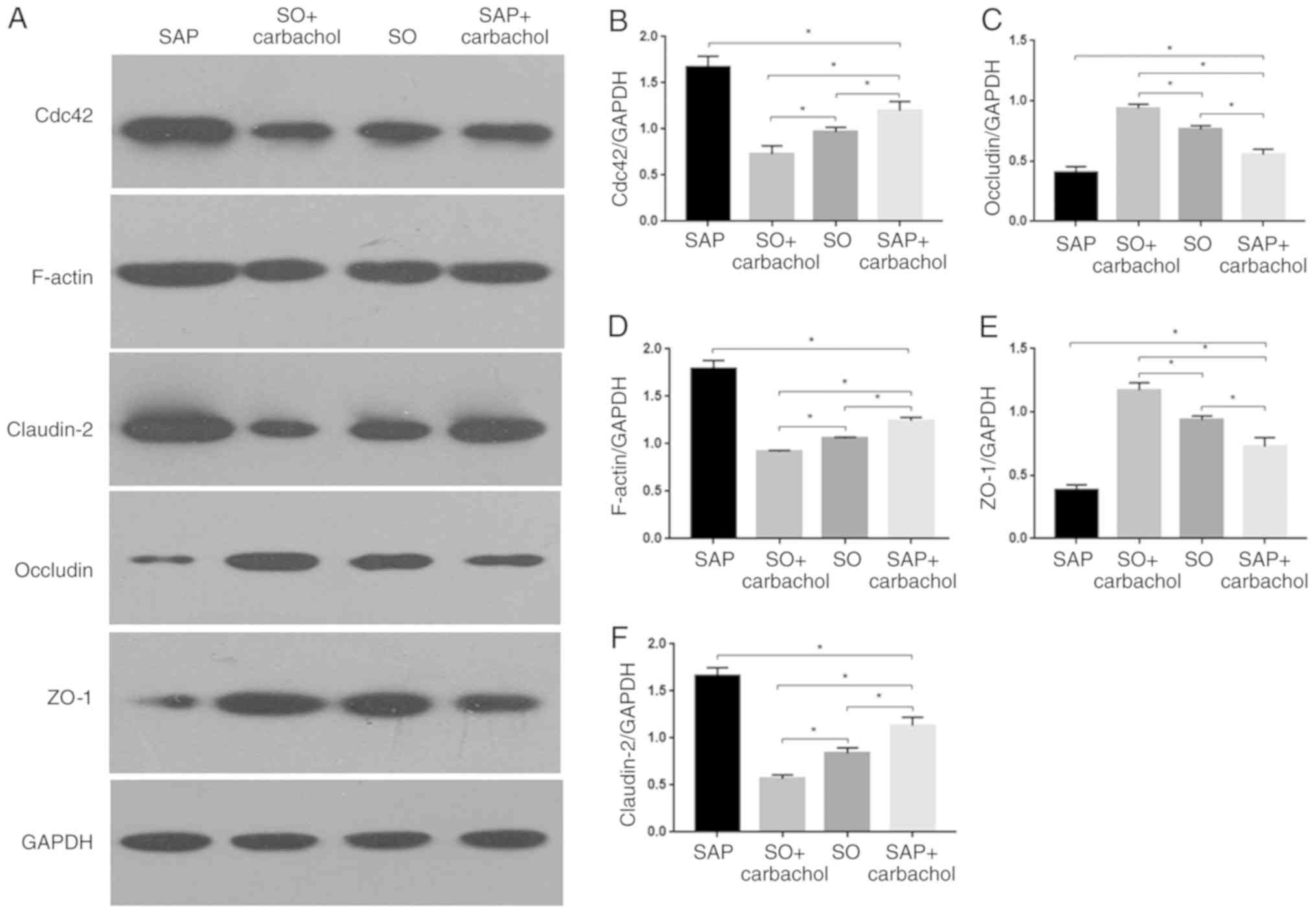 | Figure 3Western blotting analysis.
Representative western blotting images of (A) claudin-2, occludin,
ZO-1, F-actin and Cdc42 in the intestinal epithelium. Relative
expression of (B) Cdc42, (C) occludin, (D) F-actin, (E) ZO-1 and
(F) claudin-2 determined by optical densitometry. Results are
expressed as the mean ± standard deviation (n=6 randomly selected
mice in each group). *P<0.05. SAP, severe acute
pancreatitis; SO, sham operation; ZO-1, zonula occludens-1; Cdc42,
cell division cycle 42. |
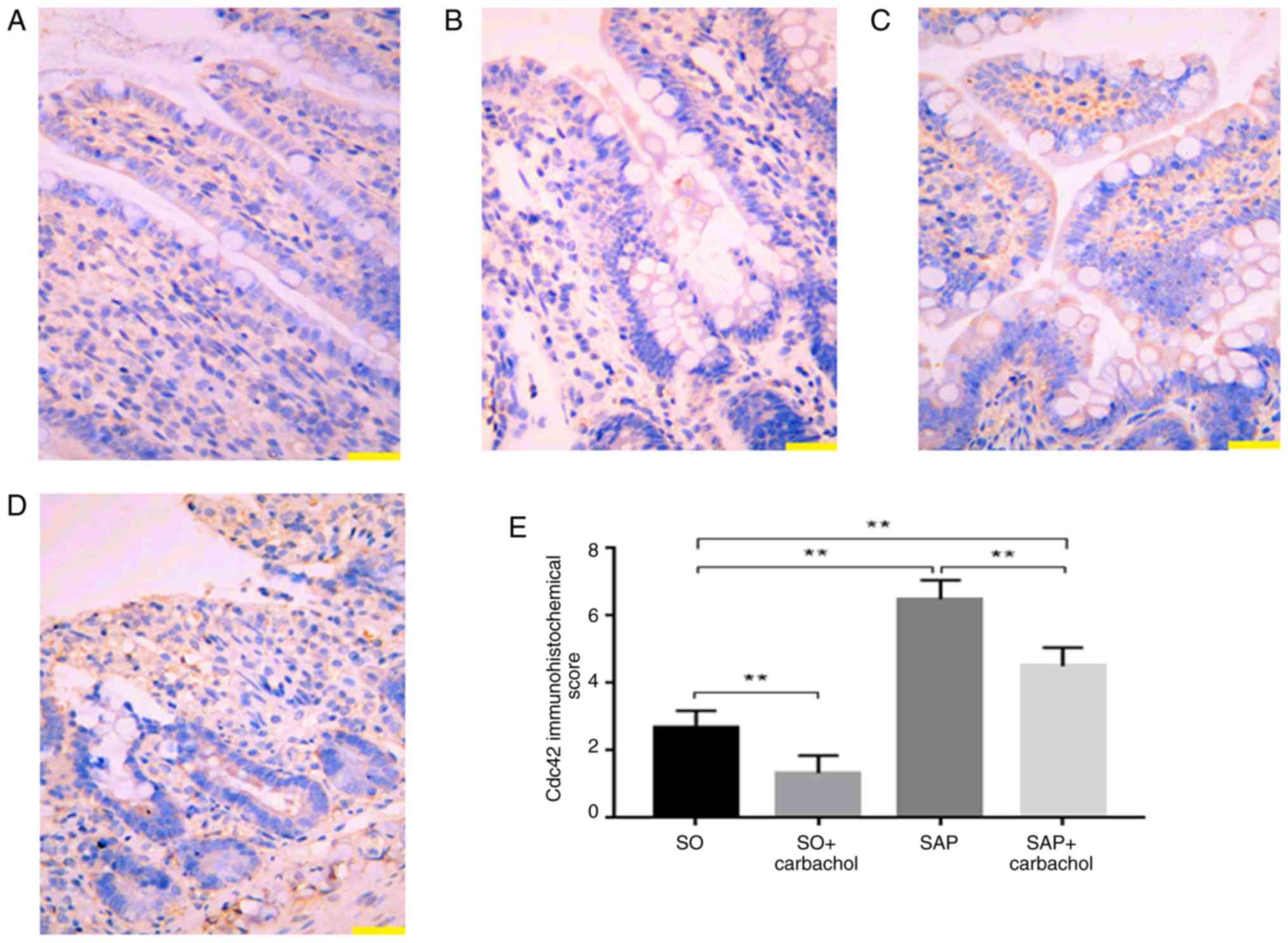
Detection of Cdc42 by IHC
The detection of Cdc42 in rat intestinal epithelium
was performed by IHC. As presented in Fig. 4, Cdc42 expression was significantly
increased in the SAP and SAP + carbachol groups compared with the
SO group (P<0.01; Fig. 4A and
C-E). Furthermore, Cdc42 expression,
in the SO + carbachol group was the lowest (P<0.01; Fig. 4B and E), and expression of Cdc42 was
significantly decreased in the SAP + carbachol group compared with
the SAP group, (P<0.01; Fig.
4C-E). These results were consistent with the results from the
western blotting.
Detection of F-actin and TJ proteins
by fluorescence microscopy
The expression of TJ proteins and F-actin in
intestinal tissue, as well as nuclear staining, was detected by
immunofluorescence triple staining. Claudin-2 (green), occluding
(green) and ZO-1 (green) (Fig. 5A-C,
respectively) were observed at the junction among intestinal
epithelial cells and F-actin (red) was observed in the cytoplasm of
intestinal epithelial cells. As presented in Fig. 5, occludin and ZO-1 staining intensity
in the SAP + carbachol group was significantly higher compared with
that in the SAP group (P<0.05; Fig.
5B, C, E and F).
However, occludin and ZO-1 staining intensity was significantly
higher in the SO + carbachol group compared with the SO group
(P<0.05; Fig. 5B, C, E and
F). Conversely, claudin-2 and
F-actin staining intensity was significantly higher in the SAP
group compared with the SAP + carbachol group (P<0.05; Fig. 5A, D
and G), and significantly lower in
the SO + carbachol group compared with the SAP + carbachol group
(P<0.05; Fig. 5A, D and G).
Combined with pathological results, the degree of intestinal damage
was determined to be associated with the expression of TJ protein.
These results were consistent with the results from the western
blotting.
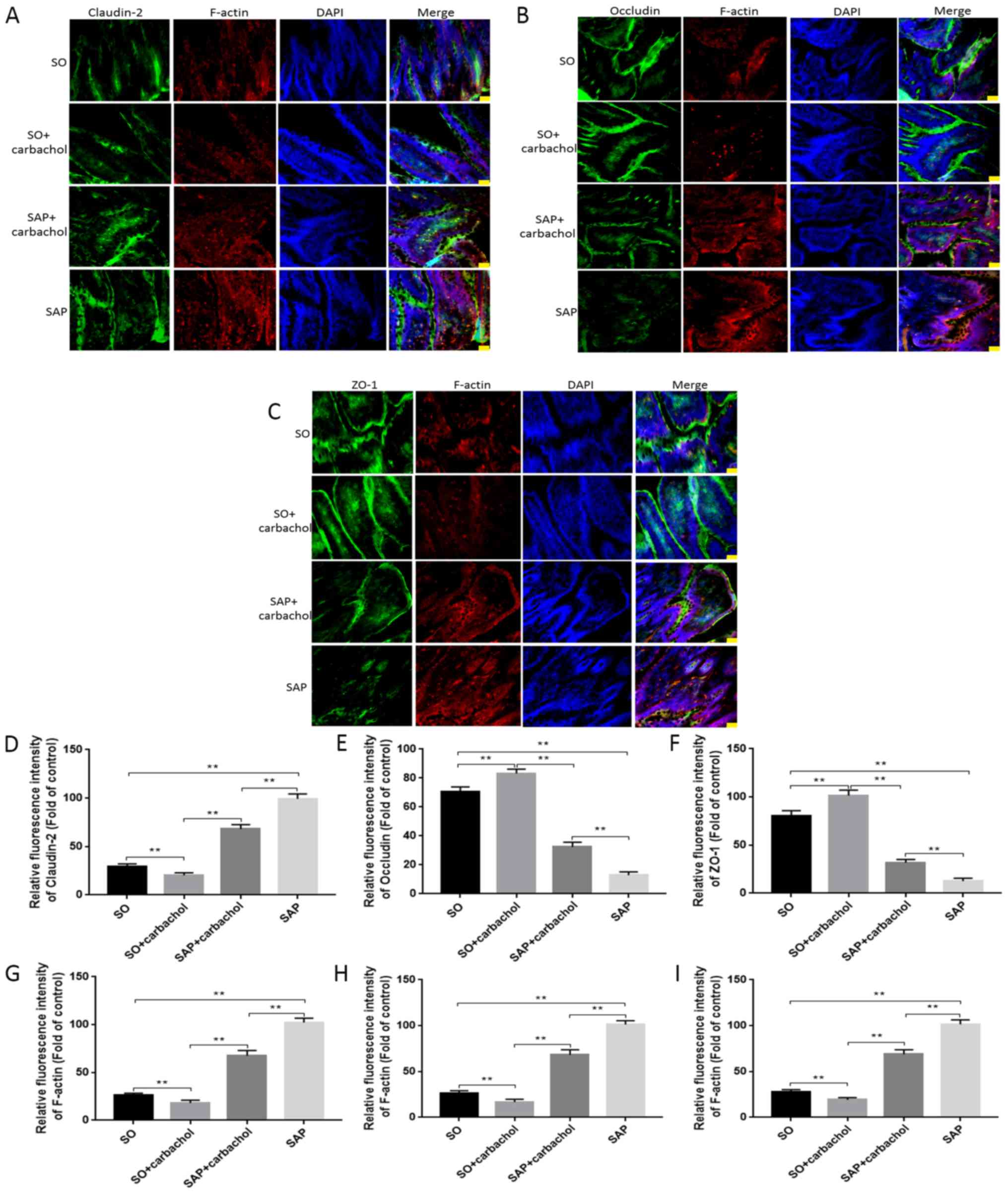 | Figure 5Immunofluorescent detection.
Intestinal sections from rats in the SAP, SAP + carbachol, SO +
carbachol and SO groups were triple stained with F-actin (red),
DAPI (blue), and ZO-1 (green), occluding (green) or claudin-2
(green). (A) Claudin-2, F-actin and DAPI staining. (B) Occludin,
F-actin and DAPI staining. (C) ZO-1, F-actin and DAPI staining. The
fluorescence intensity of (D) claudin-2, (E) occludin and (F) ZO-1,
and the respective F-actin fluorescence intensity from the staining
experiments for (G) claudin-2, (H) occludin and (I) ZO-1, were
analyzed. The results are expressed as the mean ± standard
deviation (n=6 randomly selected mice in each group).
Magnification, x200. Scale bar, 100 µm. **P<0.01.
SAP, severe acute pancreatitis; SO, sham operation; ZO-1, zonula
occludens-1. |
Discussion
SAP is a critical condition characterized by the
rapid failure of numerous organs and a high mortality rate
(1). Infection of the pancreas and
peripancreatic necrosis is the main cause of mortality in patients
with late SAP (37).
Previous studies reported a significant correlation
between the increased intestinal mucosal permeability and a poorer
prognosis of SAP in patients (38,39). In
cases of sepsis and multiple organ failure, there is a poor
prognosis in surgical treatment of SAP (8,10). BT
within the intestine is the main cause of systemic infection in
patients with SAP. It was demonstrated that the main factor
promoting BT is the destruction of the intestinal barrier (40-42).
It is therefore crucial to protect the function of the intestinal
barrier in patients with SAP in order to prevent and treat BT and
subsequent systemic infection. The present study investigated the
protective effect of carbachol on intestinal epithelial barrier
function in rats with SAP and the underlying role of
Cdc42/F-actin.
Carbachol is a cholinergic agonist that has been
reported to serve a protective role in intestinal barrier
dysfunction induced by LPS, TNF-α or following IBD (26,27).
Octreotide is a synthetic somatostatin analogue, which inhibits
glandular secretion. Previous studies reported that octreotide
cannot significantly improve the incidence rate of complications
and mortality associated with moderate to severe acute pancreatitis
(43-45).
Previous studies have revealed extensive necrosis of pancreatic
acini or atrophy of pancreatic acini with a reduction of enzyme
content in SAP (46,47). Li et al (29) therefore hypothesized that a large
number of pancreatic acinar lesions and necrosis in patients with
SAP lead to insufficient secretion from residual acinar tissue. At
present, the protective effect of carbachol on intestinal barrier
is clear. There was no significant difference in the severity of
pancreatic injury between the SAP and SAP + carbachol treated
groups. Furthermore, the severity of intestinal injury in the SAP +
carbachol group was lower compared with that in the SAP group.
Similarly, the BT and mortality rates in the SAP + carbachol group
were lower compared with the SAP group. Serum levels of lipase and
amylase in the SAP and SAP + carbachol groups were significantly
higher compared with the SO group. In addition, no differences were
identified in the serum levels of lipase and amylase between the
SAP and SAP + carbachol groups, and between the SO and SO +
carbachol groups. Further investigation using a larger sample size
is required to validate these results. These findings suggested
that carbachol may protect the function of the intestinal barrier
in rats with SAP without aggravating pancreatic injury.
TJ proteins are transmembrane proteins localized
between intestinal epithelial cells. These proteins are anchored by
the peripheral membrane protein ZO-1 to cytoplasmic F-actin-based
cytoskeleton proteins. This structure forms a physical barrier
between intestinal epithelial cells, which is essential to maintain
intestinal function (48,49). Cdc42 is a GTP-binding protein from
the Rho family that regulates actin skeleton assembly and
rearrangement (50,51). Proteins from this family have been
demonstrated to regulate cell morphology, migration and endocytosis
(52,53). The results from the present study
confirmed the effect of Cdc42 on TJ proteins and F-actin. In the
SAP + carbachol group, intestinal tissue injury was decreased, and
the expression of ZO-1 and occludin was higher, whereas the
expression of claudin-2, Cdc42 and F-actin was lower compared with
the SAP group. BT rate was also lower in the SAP + carbachol group
compared with the SAP group. These results indicated that ZO-1 and
occludin may serve a crucial role in maintaining the intestinal
barrier. However, this study demonstrated that claudin-2 was
associated with intestinal barrier dysfunction, which was
consistent with previous studies (19,30,54). The
results of the current study confirmed that carbachol reduces the
expression of Claudin-2, Cdc42 and F-actin, and increases the
expression of ZO-1 and occludin in the small intestine of SAP rats.
The results indicated that carbachol may protect the intestinal TJ
barrier of rats with SAP by regulating the expression of Cdc42 and
F-actin.
The present study had some limitations in terms of
the evaluation of SAP-related intestinal injury and BT. A further
study could collect ascites and detect GFP-Escherichia coli
in an SAP rat model and discern whether ascites and bacteria
translocation occurred in the abdominal cavity of rats. In this
way, it would be possible to not only understand BT indirectly by
16S rRNA technology, but also to observe BT visually by microscopy.
The results from the present study suggested that carbachol may
serve a role in the protection of the intestinal barrier in SAP via
Cdc42/F-actin; however the underlying mechanisms remain
unknown.
In conclusion, to the best of our knowledge, the
present study demonstrated for the first time that carbachol may
protect the intestinal barrier in a rat model of SAP without
aggravating pancreatic injury. Furthermore, the present study
demonstrated the potential regulatory role of Cdc42/F-actin in the
protection of the intestinal barrier function in SAP. Further
investigation on the protective role of carbachol against
intestinal barrier injury in SAP is essential to develop novel
therapeutic strategies for patients with SAP.
Acknowledgements
Not applicable.
Funding
This work was supported by the National Natural
Science Foundation of China (grant no. 81470890).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Author's contributions
HW and DZ conceived and designed the current study.
HW, YJ, HL and CL acquired the data. HW, YJ and JW analyzed and
interpreted the data. HL, JW and CL wrote the manuscript. HW and DZ
approved the final version of manuscript, all authors. All authors
are accountable for all aspects of the study.
Ethics approval and consent to
participate
All experimental protocols were performed according
to the National Institutes of Health Laboratory Animal Guidelines
and were approved by the Institutional Animal Care and Use
Committee of Qingdao University.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Leppäniemi A, Tolonen M, Tarasconi A,
Segovia-Lohse H, Gamberini E, Kirkpatrick AW, Ball CG, Parry N,
Sartelli M, Wolbrink D, et al: 2019 WSES guidelines for the
management of severe acute pancreatitis. World J Emerg Surg.
14(27)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Banks PA and Freeman ML: Practice
parameters committee of the American college of gastroenterology.
Practice guidelines in acute pancreatitis. Am J Gastroenterol.
101:2379–2400. 2006.PubMed/NCBI View Article : Google Scholar
|
|
3
|
van Dijk SM, Hallensleben NDL, van
Santvoort HC, Fockens P, van Goor H, Bruno MJ and Besselink MG:
Dutch Pancreatitis Study Group. Acute pancreatitis: Recent advances
through randomised trials. Gut. 66:2024–2032. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Uhl W, Warshaw A, Imrie C, Bassi C, McKay
CJ, Lankisch PG, Carter R, Magno ED, Banks PA, Whitcomb DC, et al:
IAP guidelines for the surgical management of acute pancreatitis.
Pancreatology. 2:565–573. 2002.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Berger HG, Rau B, Mayer J and Pralle U:
Natural course of acute pancreatitis. World J Surg. 21:130–135.
1997.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bradley EL III: A fifteen year experience
with open drainage for infected pancreatic necrosis. Surg Gynecol
Obstet. 177:215–222. 1993.PubMed/NCBI
|
|
7
|
Mier J, León EL, Castillo A, Robledo F and
Blanco R: Early versus late necrosectomy in severe necrotizing
pancreatitis. Am J Surg. 173:71–75. 1997.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Nieuwenhuijs VB, Besselink MG, van Minnen
LP and Gooszen HG: Surgical management of acute necrotizing
pancreatitis: A 13-year experience and a systematic review. Scand J
Gastroenterol. Suppl:111–116. 2003.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Werner J, Feuerbach S, Uhl W and Büchler
MW: Management of acute pancreatitis: From surgery to
interventional intensive care. Gut. 54:426–436. 2005.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Rau B, Bothe A and Beger HG: Surgical
treatment of necrotizing pancreatitis by necrosectomy and closed
lavage: Changing patient characteristics and outcome in a 19-year,
single-center series. Surgery. 138:28–39. 2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Banks PA, Bollen TL, Dervenis C, Gooszen
HG, Johnson CD, Sarr MG, Tsiotos GG and Vege SS: Acute Pancreatitis
Classification Working Group. Classification of acute
pancreatitis-2012: Revision of the Atlanta classification and
definitions by international consensus. Gut. 62:102–111.
2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Fishman JE, Levy G, Alli V, Zheng X, Mole
DJ and Deitch EA: The intestinal mucus layer is a critical
component of the gut barrier that is damaged during acute
pancreatitis. Shock. 42:264–270. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Barbeiro DF, Koike MK, Coelho AM, Pinheiro
da Silva F and Machado MC: Intestinal barrier dysfunction and
increased COX-2 gene expression in the gut of elderly rats with
acute pancreatitis. Pancreatology. 16:52–56. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Simsek I, Mas MR, Yasar M, Ozyurt M,
Saglamkaya U, Deveci S, Comert B, Basustaoglu A, Kocabalkan F and
Refik M: Inhibition of inducible nitric oxide synthase reduces
bacterial translocation in a rat model of acute pancreatitis.
Pancreas. 23:296–301. 2001.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Schwarz M, Poch B, Isenmann R, Kriese D,
Rozdzinski E, Beger HG and Gansauge F: Effect of early and late
antibiotic treatment in experimental acute pancreatitis in rats.
Langenbecks Arch Surg. 392:365–370. 2007.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Schmid SW, Uhl W, Friess H, Malfertheiner
P and Büchler MW: The role of infection in acute pancreatitis. Gut.
45:311–316. 1999.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Medich DS, Lee TK, Melhem MF, Rowe MI,
Schraut WH and Lee KK: Pathogenesis of pancreatic sepsis. Am J
Surg. 165:46–52. 1993.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wang J, Li C, Jiang YJ, Zheng HM, Li DH,
Liang YB, Deng WS and Zhang DL: Effect of ceramide-1-phosphate
transfer protein on intestinal bacterial translocation in severe
acute pancreatitis. Clin Res Hepatol Gas. 41:86–92. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Deng WS, Zhang J, Ju H, Zheng HM, Wang J,
Wang S and Zhang DL: Arpin contributes to bacterial translocation
and development of severe acute pancreatitis. World J
Gastroenterol. 21:4293–4301. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Buckley A and Turner JR: Cell biology of
tight junction barrier regulation and mucosal disease. Cold Spring
Harb Perspect Biol. 10(a029314)2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Dokladny K, Zuhl MN and Moseley PL:
Intestinal epithelial barrier function and tight junction proteins
with heat and exercise. J Appl Physiol (1985). 120:692–701.
2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Anderson JM and Van Itallie CM: Physiology
and function of the tight junction. Cold Spring Harb Perspect Biol.
1(a002584)2009.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Doherty GJ and McMahon HT: ‘Mediation,
modulation, and consequences of membrane-cytoskeleton
interactions’. Annu Rev Biophys. 37:65–95. 2008.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Qadir MI, Parveen A and Ali M: Cdc42: Role
in cancer management. Chem Biol Drug Des. 86:432–439.
2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Cao WH, Chai JK, Hu S, Yang H, Sun T, Zou
X and Sheng Z: Influence of carbachol on intestinal dysfunction
after traumatic or burn injury. Zhonghua Shao Shang Za Zhi.
22:168–171. 2006.PubMed/NCBI(In Chinese).
|
|
26
|
Khan MR, Uwada J, Yazawa T, Islam MT, Krug
SM, Fromm M, Karaki S, Suzuki Y, Kuwahara A, Yoshiki H, et al:
Activation of muscarinic cholinoceptor ameliorates tumor necrosis
factor-a-induced barrier dysfunction in intestinal epithelial
cells. FEBS Lett. 589:3640–3647. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zhang Y and Li J: Carbachol ameliorates
lipopolysaccharide-induced intestinal epithelial tight junction
damage by down-regulating NF-κβ and myosin light-chain kinase
pathways. Biochem Biophys Res Commun. 428:321–326. 2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Xu W, Zhou YF and Xia SH: Octreotide for
primary moderate to severe acute pancreatitis: A meta-analysis.
Hepatogastroenterology. 60:1504–1508. 2013.PubMed/NCBI
|
|
29
|
Li J, Wang R and Tang C: Somatostatin and
octreotide on the treatment of acute pancreatitis-basic and
clinical studies for three decades. Curr Pharm Des. 17:1594–1601.
2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Huang L, Jiang Y, Sun Z, Gao Z, Wang J and
Zhang D: Autophagy strengthens intestinal mucosal barrier by
attenuating oxidative stress in severe acute pancreatitis. Dig Dis
Sci. 63:910–919. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Schmidt J, Rattner DW, Lewandrowski K,
Compton CC, Mandavilli U, Knoefel WT and Warshaw AL: A better model
of acute pancreatitis for evaluating therapy. Ann Surg. 215:44–56.
1992.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Chiu CJ, McArdle AH, Brown R, Scott HJ and
Gurd FN: Intestinal mucosal lesion in low-flow states. I. A
morphological, hemodynamic, and metabolic reappraisal. Arch Surg.
101:478–483. 1970.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Amasheh S, Dullat S, Fromm M, Schulzke JD,
Buhr HJ and Kroesen AJ: Infamed pouch mucosa possesses altered
tight junctions indicating recurrence of infammatory bowel disease.
Int J Colorectal Dis. 24:1149–1156. 2009.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Shi C, Ren L, Sun C, Yu L, Bian X, Zhou X,
Wen Y, Hua D, Zhao S, Luo W, et al: miR-29a/b/c function as
invasion suppressors for gliomas by targeting CDC42 and predict the
prognosis of patients. Br J Cancer. 117:1036–1047. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Soslow RA, Dannenberg AJ, Rush D, Woerner
BM, Khan KN, Masferrer J and Koki AJ: Cox-2 is expressed in human
pulmonary, colonic, and mammary tumors. Cancer. 89:2637–2645.
2000.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Remmele W and Schicketanz KH:
Immunohistochemical determination of estrogen and progesterone
receptor content in human breast cancer Computer-assisted image
analysis (QIC score) vs. subjective grading (IRS). Pathol Res
Pract. 189:862–866. 1993.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Werge M, Novovic S, Schmidt PN and Gluud
LL: Infection increases mortality in necrotizing pancreatitis: A
systematic review and meta-analysis. Pancreatology. 16:698–707.
2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Nagpal K, Minocha VR, Agrawal V and Kapur
S: Evaluation of intestinal mucosal permeability function in
patients with acute pancreatitis. Am J Surg. 192:24–28.
2006.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ammori BJ, Leeder PC, King RF, Barclay GR,
Martin IG, Larvin M and McMahon MJ: Early increase in intestinal
permeability in patients with severe acute pancreatitis:
Correlation with endotoxemia, organ failure, and mortality. J
Gastrointest Surg. 3:252–262. 1999.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Schmitz H, Barmeyer C, Fromm M, Runkel N,
Foss HD, Bentzel CJ, Riecken EO and Schulzke JD: Altered tight
junction structure contributes to the impaired epithelial barrier
function in ulcerative colitis. Gastroenterology. 116:301–309.
1999.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Ma TY: Intestinal epithelial barrier
dysfunction in Crohn's disease. Proc Soc Exp Biol Med. 214:318–327.
1997.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Jiang Y, Lin J, Zhang D, Yu Z, Li Q, Jiang
J and Li J: Bacterial translocation contributes to cachexia and its
possible pathway in patients with colon cancer. J Clin
Gastroenterol. 48:131–137. 2014.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Paran H, Mayo A, Paran D, Neufeld D,
Shwartz I, Zissin R, Singer P, Kaplan O, Skornik Y and Freund U:
Octreotide treatment in patients with severe acute pancreatitis.
Digest Dis Sci. 45:2247–2251. 2000.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Uhl W, Buchler MW, Malfertheiner P, Beger
HG, Adler G and Gaus W: A randomised, double blind, multicentre
trial of octreotide in moderate to severe acute pancreatitis. Gut.
45:97–104. 1999.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Mckay C, Baxter J and Imrie C: A
randomized, controlled trial of octreotide in the management of
patients with acute pancreatitis. Int J Pancreatol. 21:13–19.
1997.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Bhatia M: Apoptosis versus necrosis in
acute pancreatitis. Am J Physiol Gastrointest Liver Physiol.
286:G189–G196. 2004.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Chen Y, Chong MM, Darwiche R, Thomas HE
and Kay TW: Severe pancreatitis with exocrine destruction and
increased islet neogenesis in mice with suppressor of cytokine
signaling-1 deficiency. Am J Pathol. 165:913–921. 2004.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Feng Y, Wang Y, Wang P, Huang Y and Wang
F: Short-chain fatty acids manifest stimulative and protective
effects on intestinal barrier function through the inhibition of
NLRP3 inflammasome and autophagy. Cell Physiol Biochem. 49:190–205.
2018.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Zhao J, Zhao R, Cheng L, Yang J and Zhu L:
Peroxisome proliferator-activated receptor gamma activation
promotes intestinal barrier function by improving mucus and tight
junctions in a mouse colitis model. Dig Liver Dis. 50:1195–1204.
2018.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Ma L, Rohatgi R and Kirschner MW: The
Arp2/3 complex mediates actin polymerization induced by the small
GTP-binding protein Cdc42. Proc Natl Acad Sci. 95:15362–15367.
1998.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Rohatgi R, Ma L, Miki H, Lopez M,
Kirchhausen T, Takenawa T and Kirschner MW: The interaction between
NWASP and the Arp2/3 complex links Cdc42-dependent signals to actin
assembly. Cell. 97:221–231. 1999.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Hall A: Small GTP-binding proteins and the
regulation of the actin cytoskeleton. Annu Rev Cell Biol. 10:31–54.
1994.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Nobes CD and Hall A: Rho, Rac, and cdc42
GTPases regulate the assembly of multimolecular focal complexes
associated with actin stress fibers, lamellipodia, and filopodia.
Cell. 81:53–62. 1995.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Ren X, Zhu Y, Gamallat Y, Ma S, Chiwala G,
Meyiah A and Xin Y: E. coli O124 K72 alters the intestinal barrier
and the tight junctions proteins of guinea pig intestine. Biomed
Pharmacother. 94:468–473. 2017.PubMed/NCBI View Article : Google Scholar
|