|
1
|
Tagalakis V, Patenaude V, Kahn SR and
Suissa S: Incidence of and mortality from venous thromboembolism in
a Real-world population: The Q-VTE study cohort. Am J Med.
126:832.e13–e21. 2013.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Spencer FA, Emery C, Lessard D, Anderson
F, Emani S, Aragam J, Becker RC and Goldberg RJ: The worcester
venous thromboembolism study: A population-based study of the
clinical epidemiology of venous thromboembolism. J Gen Intern Med.
21:722–727. 2006.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Huang W, Goldberg RJ, Cohen AT, Anderson
FA, Kiefe CI, Gore JM and Spencer FA: Declining Long-term risk of
adverse events after First-time Community-presenting venous
thromboembolism: The Population-based Worcester VTE study (1999 to
2009). Thromb Res. 135:1100–1106. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Hirsh J and Levine MN: Low molecular
weight heparin. Blood. 79:1–17. 1992.PubMed/NCBI
|
|
5
|
Robertson L and Jones LE: Fixed dose
subcutaneous low molecular weight heparins versus adjusted dose
unfractionated heparin for the initial treatment of venous
thromboembolism. Cochrane Database Syst Rev.
2(CD001100)2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kearon C, Akl EA, Ornelas J, Blaivas A,
Jimenez D, Bounameaux H, Huisman M, King CS, Morris TA, Sood N, et
al: Antithrombotic therapy for VTE disease: CHEST Guideline and
expert panel report. Chest. 149:315–352. 2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lindmarker P, Holmström M, Granqvist S,
Johnsson H and Lockner D: Comparison of once-daily subcutaneous
Fragmin with continuous intravenous unfractionated heparin in the
treatment of deep vein thrombosis. Thromb Haemost. 72:186–90.
1994.PubMed/NCBI
|
|
8
|
Collignon F, Frydman A, Caplain H, Ozoux
ML, Le Roux Y, Bouthier J and Thébault JJ: Comparison of the
pharmacokinetic profiles of three low molecular mass
heparins-dalteparin, enoxaparin and nadroparin-administered
subcutaneously in healthy volunteers (doses for prevention of
thromboembolism). Thromb Haemost. 73:630–40. 1995.PubMed/NCBI
|
|
9
|
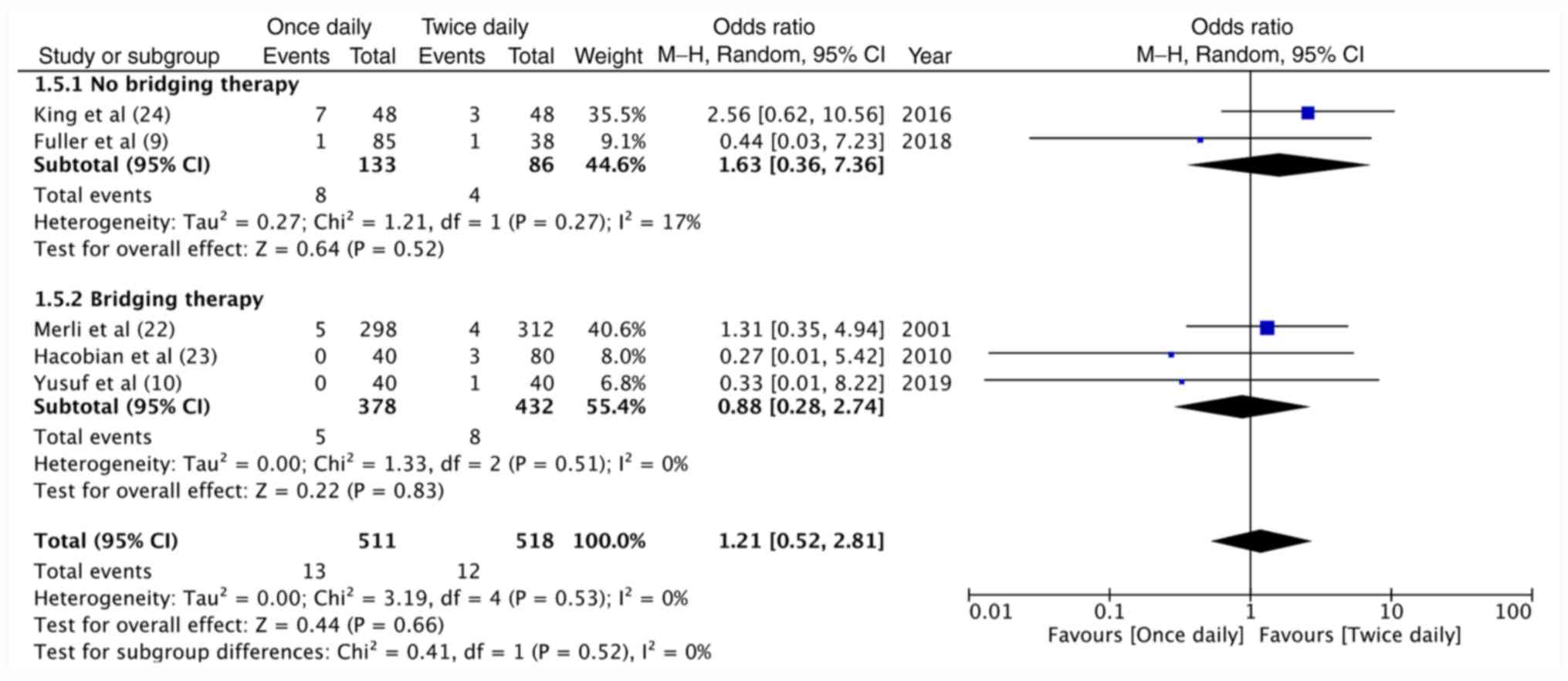
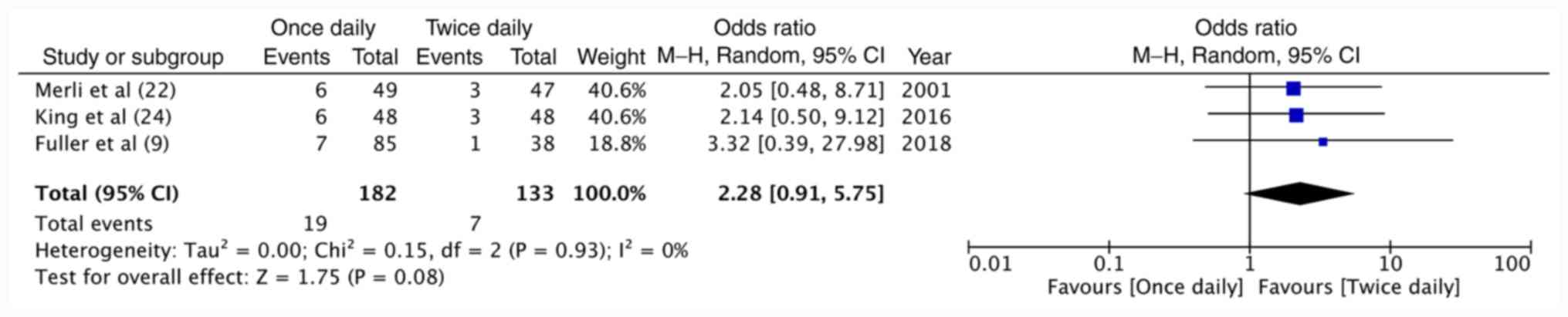
Fuller K, Malecki S, Anselmo L, Borrego
ME, Jakeman B and Burnett A: Once-daily versus Twice-daily
enoxaparin for the treatment of acute venous thromboembolism in
cancer patients. Ann Pharmacother. 52:257–262. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yusuf M, Gouda M, Herz-Allah A, Alkhouly M
and Samir A: Once-daily versus twice-daily enoxaparin for the
initial treatment of acute deep venous thrombosis: A case-control
study. J Med Sci Res. 2:144–147. 2019.
|
|
11
|
Bhutia S and Wong PF: Once versus twice
daily low molecular weight heparin for the initial treatment of
venous thromboembolism. Cochrane Database Syst Rev.
2013(CD003074)2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Moher D, Liberati A, Tetzlaff J, Altman DG
and PRISMA Group: Preferred reporting items for systematic reviews
and Meta-analyses: The PRISMA statement. PLoS Med.
6(e1000097)2009.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Higgins JPT and Green S (eds): Cochrane
Handbook for Systematic Reviews of Interventions. Version 5.1.0
(updated March 2011). The Cochrane Collaboration, 2011.
|
|
14
|
Higgins J, Altman D and Sterne J: Cochrane
statistical methods group and the cochrane bias methods group.
Chapter 8: Assessing risk of bias in included studies. In: Cochrane
handbook for systemic reviews of interventions, version 5. The
Cochrane Collaboration, 2011.
|
|
15
|
Kim SY, Park JE, Lee YJ, Seo HJ, Sheen SS,
Hahn S, Jang BH and Son HJ: Testing a tool for assessing the risk
of bias for nonrandomized studies showed moderate reliability and
promising validity. J Clin Epidemiol. 66:408–414. 2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Partsch H, Kechavarz B, Mostbeck A, Köhn H
and Lipp C: Frequency of pulmonary embolism in patients who have
iliofemoral deep vein thrombosis and are treated with once- or
twice-daily low-molecular-weight heparin. J Vasc Surg. 24:774–82.
1996.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Siegbahn A, Y-Hassan S, Boberg J, Bylund
H, Neerstrand HS, Ostergaard P and Hedner U: Subcutaneous treatment
of deep venous thrombosis with low molecular weight heparin. A dose
finding study with LMWH-Novo. Thromb Res. 55:767–78.
1989.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Holmoström M, Berglund MC, Granquist S,
Bratt G, Törnebohm E and Lockner D: Fragmin once or twice daily
subcutaneously in the treatment of deep venous thrombosis of the
leg. Thromb Res. 67:49–55. 1992.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Charbonnier BA, Fiessinger JN, Banga JD,
Wenzel E, d'Azemar P and Sagnard L: Comparison of a once daily with
a twice daily subcutaneous low molecular weight heparin regimen in
the treatment of deep vein thrombosis. FRAXODI group. Thromb
Haemost. 79:897–901. 1998.PubMed/NCBI
|
|
20
|
Narin C, Reyhanoglu H, Tülek B, Onoglu R,
Ege E, Sarigül A, Yeniterzi M and Durmaz I: Comparison of different
dose regimens of enoxaparin in deep vein thrombosis therapy in
pregnancy. Adv Ther. 25:585–594. 2008.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Trujillo-Santos J, Bergmann JF, Bortoluzzi
C, López-Reyes R, Giorgi-Pierfranceschi M, López-Sáez JB, Ferrazzi
P, Bascuñana J, Suriñach JM and Monreal M: Once versus twice daily
enoxaparin for the initial treatment of acute venous
thromboembolism. J Thromb Haemost. 15:429–438. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Merli G, Spiro TE, Olsson CG, Abildgaard
U, Davidson BL, Eldor A, Elias D, Grigg A, Musset D, Rodgers GM, et
al: Subcutaneous enoxaparin once or twice daily compared with
intravenous unfractionated heparin for treatment venous
thromboembolic disease. Ann Intern Med. 134:191–202.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Merli H, Shetty R, Niles CM,
Gerhard-Herman M, Vallurupalli N, Baroletti S, McKean SC, Sonis J,
Parasuraman S, Kosowsky JM and Goldhaber SZ: Once daily enoxaparin
for outpatient treatment of acute venous thromboembolism: A
case-control study. Clin Appl Thromb. 16:21–25. 2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
King AC, Ma MQ, Chisholm G and Toale KM:
Once daily versus twice daily enoxaparin for acute pulmonary
embolism in cancer patients. J Oncol Pharm Pract. 22:265–270.
2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Tazi Mezalek Z, Nejjari C, Essadouni L,
Samkaoui M, Serraj K, Ammouri W, Kanjaa N, Belkhadir Z, Housni B,
Awab M, et al: Evaluation and management of thromboprophylaxis in
Moroccan hospitals at national level: The Avail-MoNa study. J
Thromb Thrombolysis. 46:113–119. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Merli GJ and Groce JB: Pharmacological and
clinical differences between low-molecular-weight heparins:
Implications for prescribing practice and therapeutic interchange.
P T. 35:95–105. 2010.PubMed/NCBI
|
|
27
|
Diaz AH, Rodgers GM and Gilreath JA:
Enoxaparin once daily vs. twice daily dosing for the treatment of
venous thromboembolism in cancer patients: A literature summary. J
Oncol Pharm Pract. 18:264–70. 2012.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Levine M, Gent M, Hirsh J, Leclerc J,
Anderson D, Weitz J, Ginsberg J, Turpie AG, Demers C and Kovacs M:
A comparison of low-molecular-weight heparin administered primarily
at home with unfractionated heparin administered in the hospital
for proximal deep-vein thrombosis. N Engl J Med. 334:677–681.
1996.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Hutten BA, Prins MH, Gent M, Ginsberg J,
Tijssen JG and Büller HR: Incidence of recurrent thromboembolic and
bleeding complications among patients with venous thromboembolism
in relation to both malignancy and achieved international
normalized ratio: A retrospective analysis. J Clin Oncol.
18:3078–3083. 2000.PubMed/NCBI View Article : Google Scholar
|