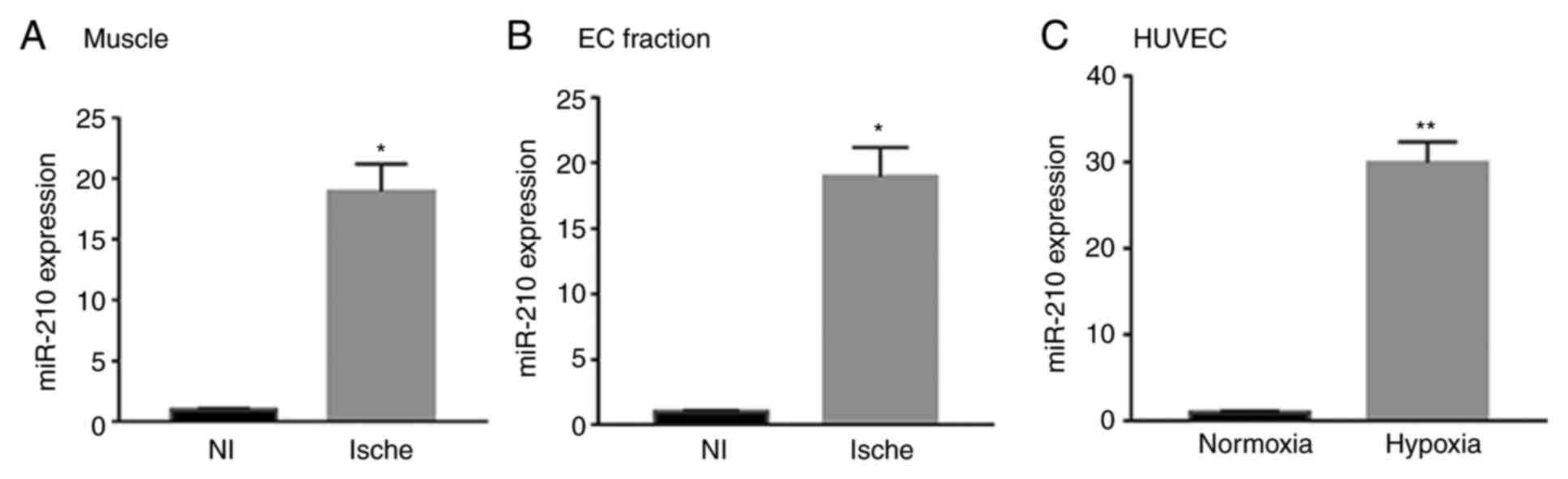
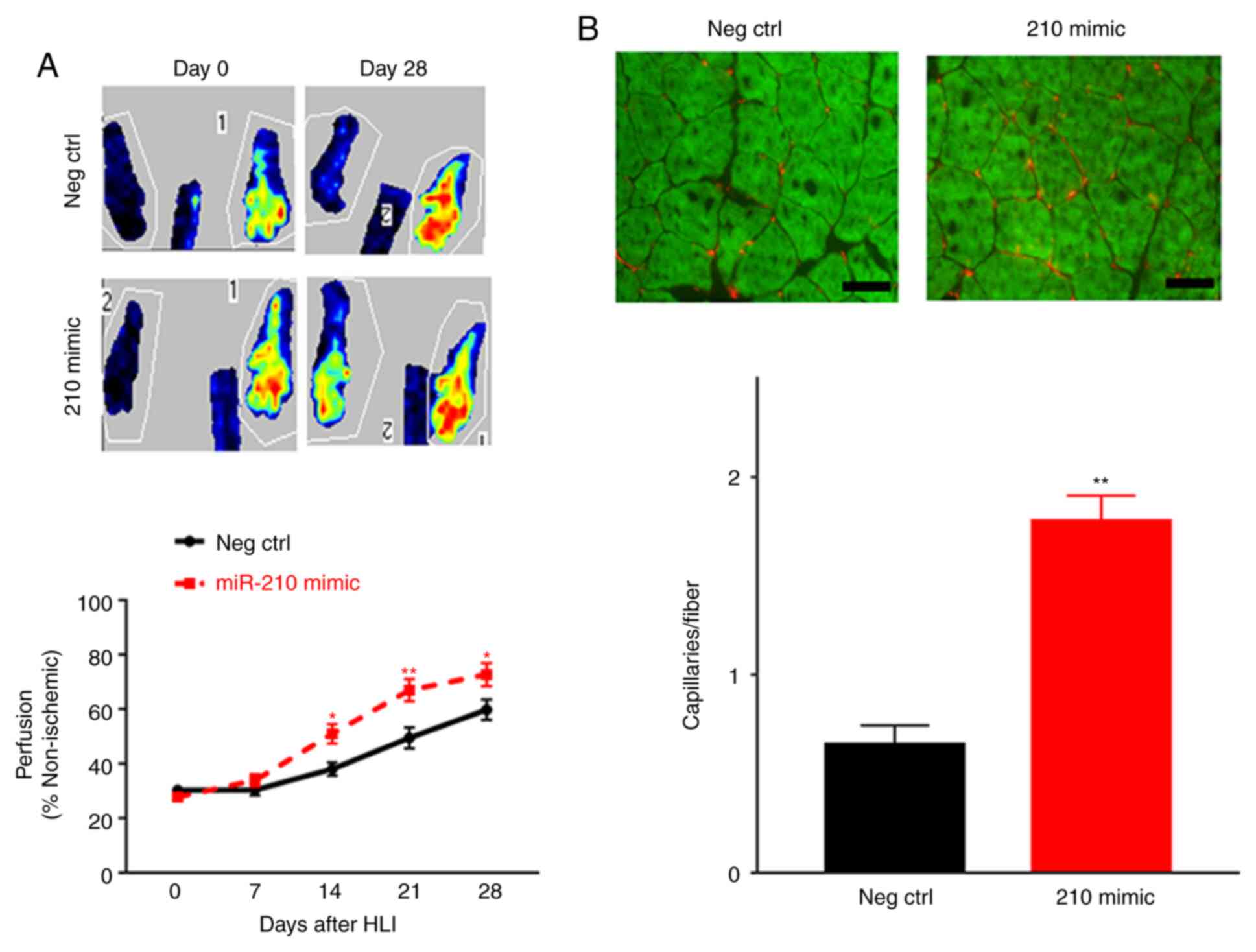
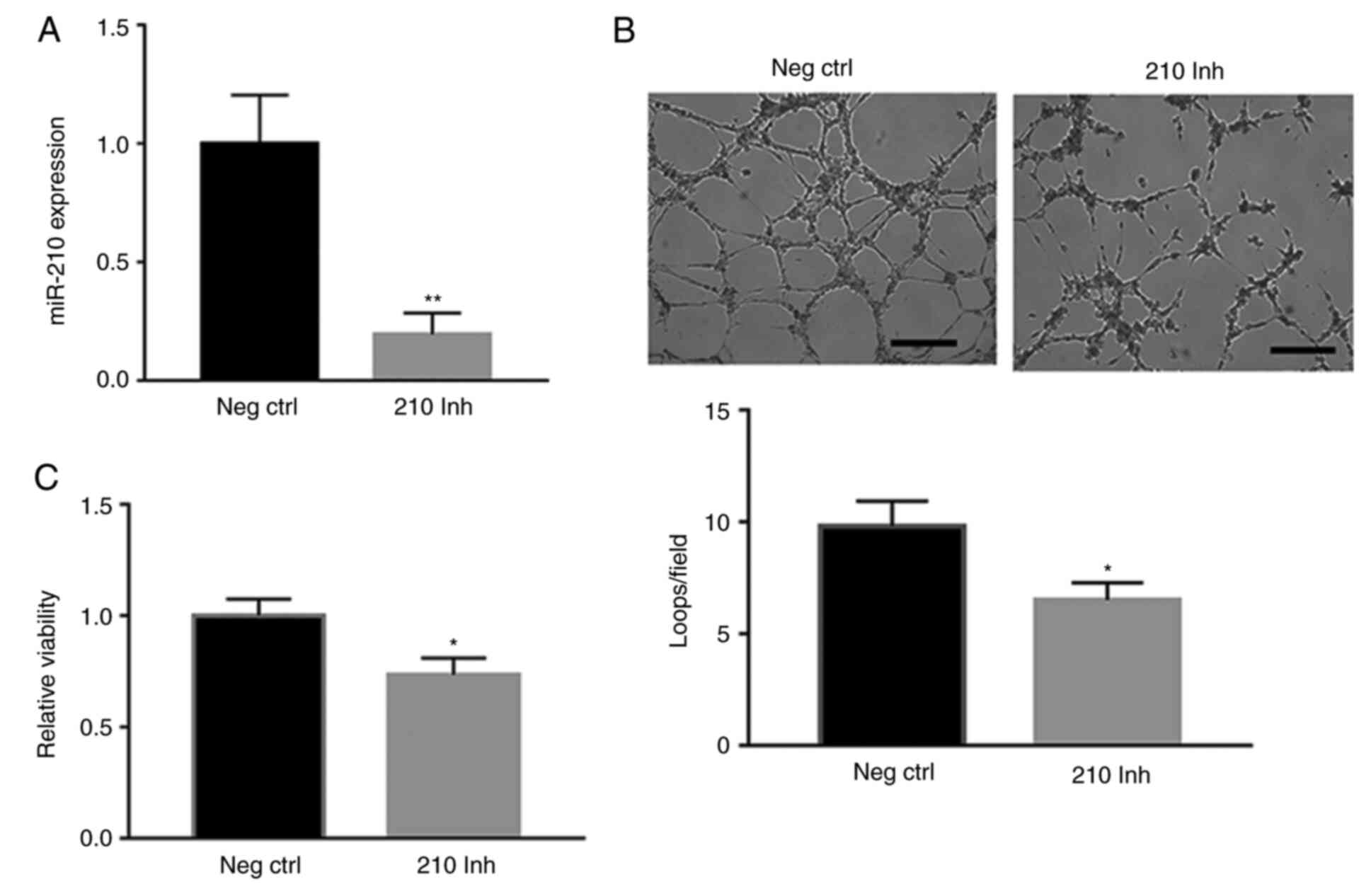
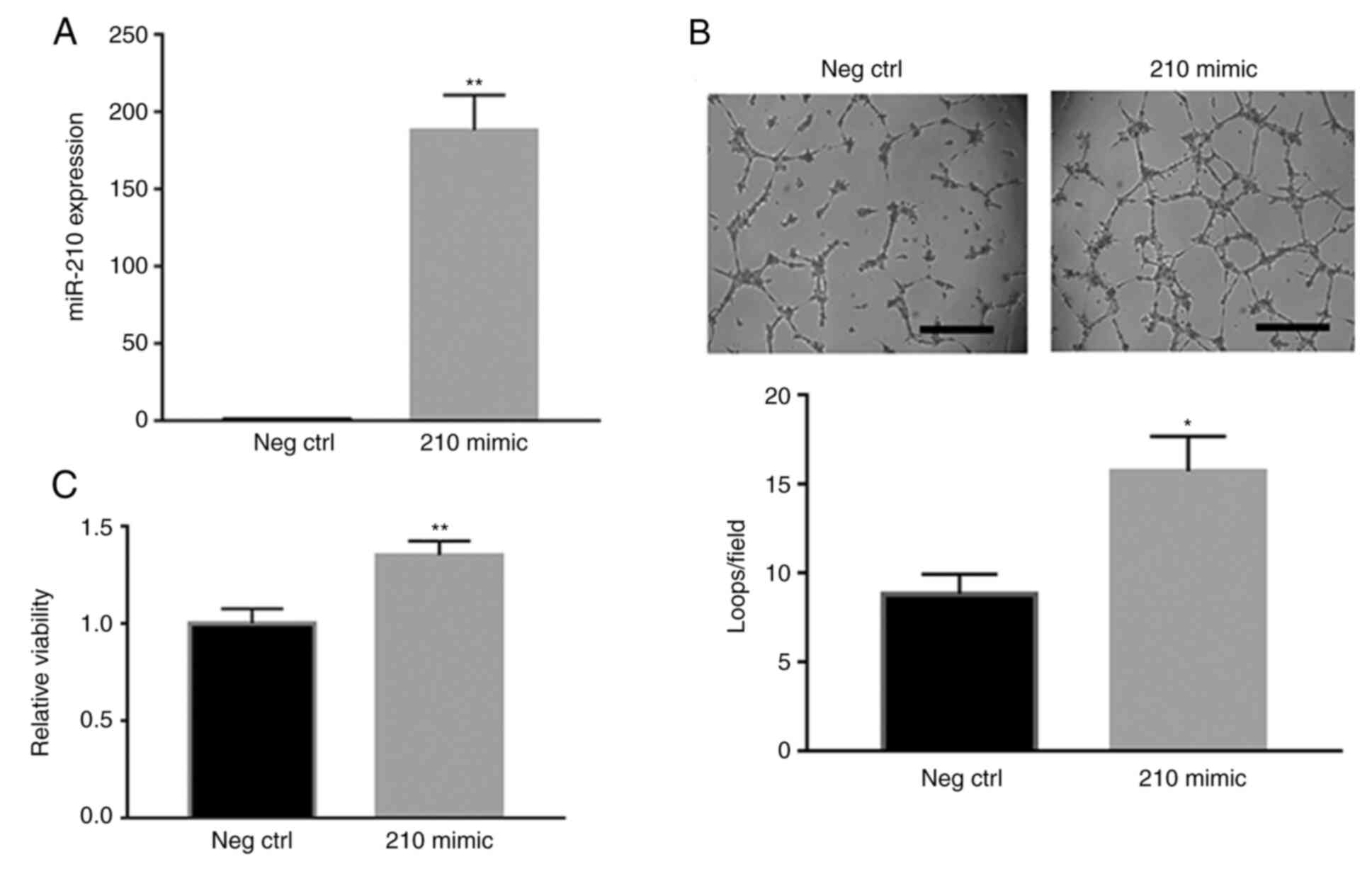
|
1
|
Criqui MH and Aboyans V: Epidemiology of
peripheral artery disease. Circ Res. 116:1509–1526. 2015.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Willey J, Mentias A, Vaughan-Sarrazin M,
McCoy K, Rosenthal G and Girotra S: Epidemiology of lower extremity
peripheral artery disease in veterans. J Vasc Surg. 68:527–535.e5.
2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Fowkes FG, Aboyans V, Fowkes FJ, McDermott
MM, Sampson UK and Criqui MH: Peripheral artery disease:
Epidemiology and global perspectives. Nat Rev Cardiol. 14:156–170.
2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Thiruvoipati T, Kielhorn CE and Armstrong
EJ: Peripheral artery disease in patients with diabetes:
Epidemiology, mechanisms, and outcomes. World J Diabetes.
6:961–969. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Espinola-Klein C and Savvidis S:
Peripheral arterial disease. Epidemiology, symptoms and diagnosis.
Internist (Berl). 50:919–926. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Joh JH, Joo SH and Park HC: Simultaneous
hybrid revascularization for symptomatic lower extremity arterial
occlusive disease. Exp Ther Med. 7:804–810. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Tanaka M, Taketomi K and Yonemitsu Y:
Therapeutic angiogenesis: Recent and future prospects of gene
therapy in peripheral artery disease. Curr Gene Ther. 14:300–308.
2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Grochot-Przeczek A, Dulak J and Jozkowicz
A: Therapeutic angiogenesis for revascularization in peripheral
artery disease. Gene. 525:220–228. 2013.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Aviles RJ, Annex BH and Lederman RJ:
Testing clinical therapeutic angiogenesis using basic fibroblast
growth factor (FGF-2). Brit J Pharmacol. 140:637–646.
2003.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Owens CD and Conte MS: Medical management
of peripheral arterial disease bridging the ‘Gap’? Circulation.
126:1319–1321. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Annex BH: Therapeutic angiogenesis for
critical limb ischaemia. Nat Rev Cardiol. 10:387–396.
2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Stather PW, Sylvius N, Wild JB, Choke E,
Sayers RD and Bown MJ: Differential MicroRNA expression profiles in
peripheral arterial disease. Circ Cardiovasc Genet. 6:490–497.
2013.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Guo S, Bai R, Liu W, Zhao A, Zhao Z, Wang
Y, Wang Y, Zhao W and Wang W: MicroRNA-210 is upregulated by
hypoxia-inducible factor-1α in the stromal cells of giant cell
tumors of bone. Mol Med Rep. 12:6185–6192. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ikeda S, Kitadate A, Abe F, Saitoh H,
Michishita Y, Hatano Y, Kawabata Y, Kitabayashi A, Teshima K, Kume
M, et al: Hypoxia-inducible microRNA-210 regulates the DIMT1-IRF4
oncogenic axis in multiple myeloma. Cancer Sci. 108:641–652.
2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pulkkinen K, Malm T, Turunen M, Koistinaho
J and Ylä-Herttuala S: Hypoxia induces microRNA miR-210 in vitro
and in vivo ephrin-A3 and neuronal pentraxin 1 are potentially
regulated by miR-210. FEBS Lett. 582:2397–2401. 2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Signorelli SS, Volsi GL, Pitruzzella A,
Fiore V, Mangiafico M, Vanella L, Parenti R, Rizzo M and Volti GL:
Circulating miR-130a, miR-27b, and miR-210 in patients with
peripheral artery disease and their potential relationship with
oxidative stress. Angiology. 67:945–950. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zhang J, Wang Q, Rao G, Qiu J and He R:
Curcumin improves perfusion recovery in experimental peripheral
arterial disease by upregulating microRNA-93 expression. Exp Ther
Med. 17:798–802. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Albadawi H, Oklu R, Cormier NR, O'Keefe
RM, Heaton JT, Kobler JB, Austen WG and Watkins MT: Hind limb
ischemia-reperfusion injury in diet-induced obese mice. J Surg Res.
190:683–691. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Chen L, Liu C, Sun D, Wang T, Zhao L, Chen
W, Yuan M, Wang J and Lu W: MicroRNA-133a impairs perfusion
recovery after hindlimb ischemia in diabetic mice. Biosci Rep.
38(BSR20180346)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Wang T, Cunningham A, Dokun AO, Hazarika
S, Houston K, Chen L, Lye RJ, Spolski R, Leonard WJ and Annex BH:
Loss of interleukin-21 receptor activation in hypoxic endothelial
cells impairs perfusion recovery after hindlimb ischemia.
Arterioscler Thromb Vasc Bio. 35:1218–1225. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhou Y, Yang J, Zhang Q, Xu Q, Lu L, Wang
J and Xia W: P4HB knockdown induces human HT29 colon cancer cell
apoptosis through the generation of reactive oxygen species and
inactivation of STAT3 signaling. Mol Med Rep. 19:231–237.
2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Pirinccioglu AG, Gökalp D, Pirinccioglu M,
Kizil G and Kizil M: Malondialdehyde (MDA) and protein carbonyl
(PCO) levels as biomarkers of oxidative stress in subjects with
familial hypercholesterolemia. Clin Biochem. 43:1220–1224.
2010.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Steven S, Daiber A, Dopheide JF, Munzel T
and Espinola-Klein C: Peripheral artery disease, redox signaling,
oxidative stress-Basic and clinical aspects. Redox Biol.
12:787–797. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zaccagnini G, Maimone B, Di Stefano V,
Fasanaro P, Greco S, Perfetti A, Capogrossi MC, Gaetano C and
Martelli F: Hypoxia-induced miR-210 modulates tissue response to
acute peripheral ischemia. Antioxid Redox Signal. 21:1177–1188.
2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Hosoda T, Zheng H, Cabral-da-Silva M,
Sanada F, Ide-Iwata N, Ogórek B, Ferreira-Martins J, Arranto C,
D'Amario D, del Monte F, et al: Human cardiac stem cell
differentiation is regulated by a mircrine mechanism. Circulation.
123:1287–1296. 2011.PubMed/NCBI View Article : Google Scholar
|