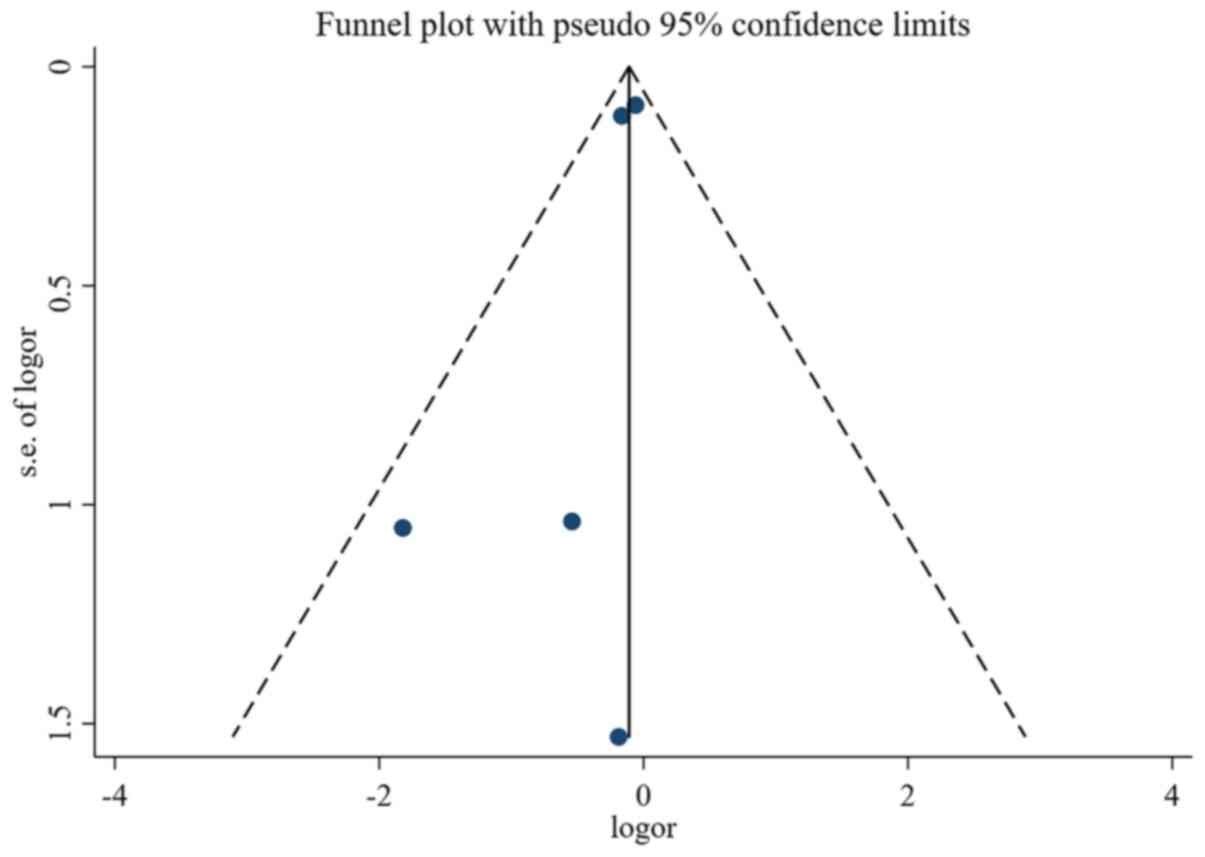
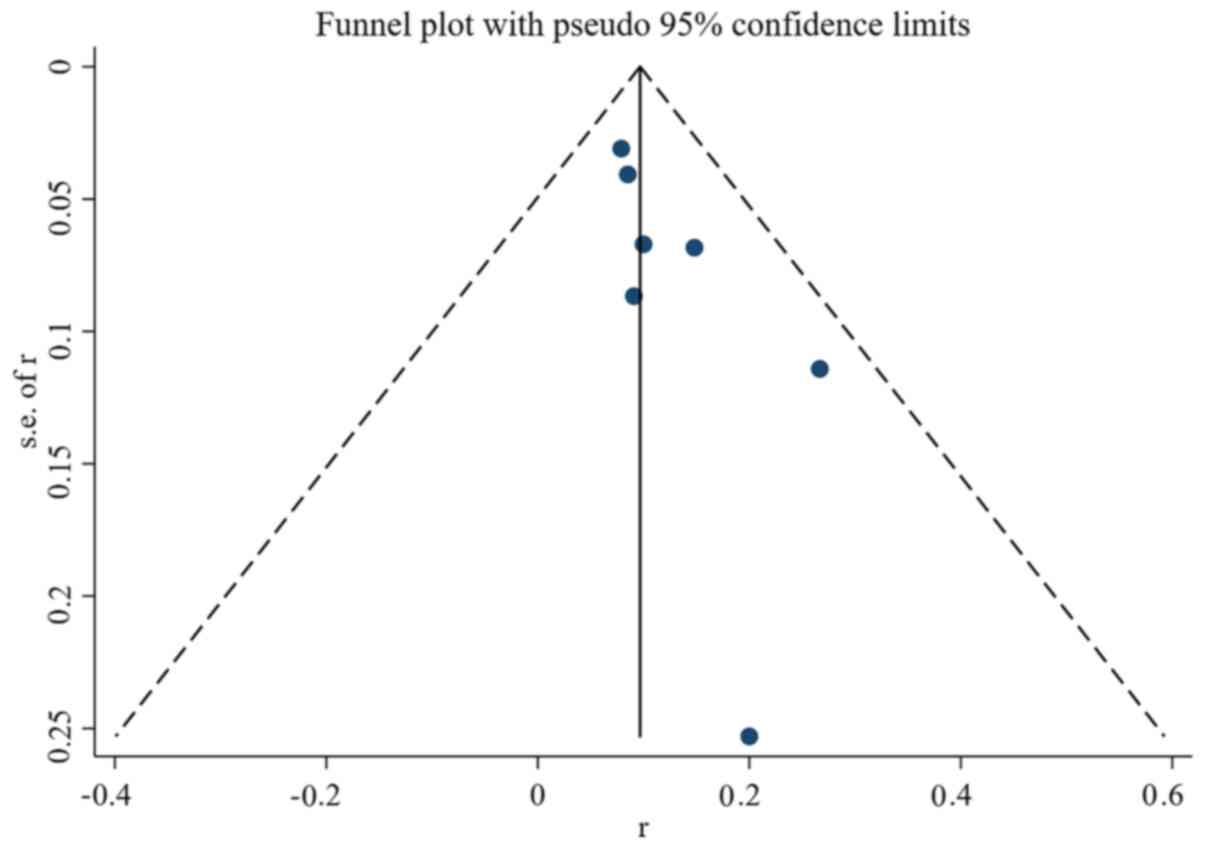
|
1
|
Kawasaki T: Acute febrile mucocutaneous
syndrome with lymphoid involvement with specific desquamation of
the fingers and toes in children. Arerugi. 16:178–222. 1967.(In
Japanese). PubMed/NCBI
|
|
2
|
Miura M, Tamame T, Naganuma T, Chinen S,
Matsuoka M and Ohki H: Steroid pulse therapy for Kawasaki disease
unresponsive to additional immunoglobulin therapy. Paediatr Child
Health. 16:479–484. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Soriano-Ramos M, Martínez-Del Val E,
Negreira Cepeda S, González-Tomé MI, Cedena Romero P,
Fernández-Cooke E, Albert de la Torre L and Blázquez-Gamero D: Risk
of coronary artery involvement in Kawasaki disease. Arch Argent
Pediatr. 114:107–113. 2016.(In English, Spanish). PubMed/NCBI View Article : Google Scholar
|
|
4
|
Koizumi K, Hoshiai M, Katsumata N, Toda T,
Kise H, Hasebe Y, Kono Y, Sunaga Y, Yoshizawa M, Watanabe A, et al:
Infliximab regulates monocytes and regulatory T cells in Kawasaki
disease. Pediatr Int. 60:796–802. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lin Y, Xiao-hui L and Shi L:
Interpretation of the 2017 edition of diagnosis, treatment, and
long-term management of Kawasaki disease: A scientific statement
for health professionals from the American heart association. Chin
J Pract Pediatr, 2017 (In Chinese).
|
|
6
|
Makino N, Nakamura Y, Yashiro M, Sano T,
Ae R, Kosami K, Kojo T, Aoyama Y, Kotani K and Yanagawa H:
Epidemiological observations of Kawasaki disease in Japan,
2013-2014. Pediatr Int. 60:581–587. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Newburger JW, Takahashi M, Gerber MA,
Gewitz MH, Tani LY, Burns JC, Shulman ST, Bolger AF, Ferrieri P,
Baltimore RS, et al: Diagnosis, treatment, and long-term management
of Kawasaki disease: A statement for health professionals from the
committee on rheumatic fever, endocarditis, and Kawasaki disease,
council on cardiovascular disease in the young, American heart
association. Pediatrics. 114:1708–1733. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Lee Y, Schulte DJ, Shimada K, Chen S,
Crother TR, Chiba N, Fishbein MC, Lehman TJ and Arditi M:
Interleukin-1β is crucial for the induction of coronary artery
inflammation in a mouse model of Kawasaki disease. Circulation.
125:1542–1550. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Newburger JW, Takahashi M, Beiser AS,
Burns JC, Bastian J, Chung KJ, Colan SD, Duffy CE, Fulton DR, Glode
MP, et al: A single intravenous infusion of gamma globulin as
compared with four infusions in the treatment of acute Kawasaki
syndrome. N Engl J Med. 324:1633–1639. 1991.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Furukawa T, Kishiro M, Akimoto K, Nagata
S, Shimizu T and Yamashiro Y: Effects of steroid pulse therapy on
immunoglobulin-resistant Kawasaki disease. Arch Dis Child.
93:142–146. 2008.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Matsubara T, Ichiyama T and Furukawa S:
Immunological profile of peripheral blood lymphocytes and
monocytes/macrophages in Kawasaki disease. Clin Exp Immunol.
141:381–387. 2005.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Research Committee of the Japanese Society
of Pediatric Cardiology; Cardiac Surgery Committee for Development
of Guidelines for Medical Treatment of Acute Kawasaki Disease:
Guidelines for medical treatment of acute Kawasaki disease: Report
of the research committee of the Japanese society of pediatric
cardiology and cardiac surgery (2012 revised version). Pediatr Int
56: 135-158, 2014.
|
|
13
|
Ogihara Y, Ogata S, Nomoto K, Ebato T,
Sato K, Kokubo K, Kobayashi H and Ishii M: Transcriptional
regulation by infliximab therapy in Kawasaki disease patients with
immunoglobulin resistance. Pediatr Res. 76:287–293. 2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Saji BT and Kobayashi T: Overview of the
new Japanese guideline2012 for the medical treatment of acute stage
of Kawasaki disease. In: Kawasaki Disease. Saji B, Newburger J,
Burns J, and Takahashi M (eds). Springer, Tokyo, pp103-167,
2017.
|
|
15
|
Xiantao Zeng and Zhu Sun: Tang. H. No. 10
of the Meta Analysis Series: Formulation of Eligibility Criteria.
Chin J Evid Based Cardiovasc Med. 5:e927–e999. 2013.
|
|
16
|
Hirono K, Kemmotsu Y, Wittkowski H, Foell
D, Saito K, Ibuki K, Watanabe K, Watanabe S, Uese K, Kanegane H, et
al: Infliximab reduces the cytokine-mediated inflammation but does
not suppress cellular infiltration of the vessel wall in refractory
Kawasaki disease. Pediatr Res. 65:696–701. 2009.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Jone PN, Anderson MS, Mulvahill MJ, Heizer
H, Glodé MP and Dominguez SR: Infliximab plus intravenous
immunoglobulin (IVIG) versus IVIG alone as initial therapy in
children with kawasaki disease presenting with coronary artery
lesions: Is dual therapy more effective? Pediatr Infect Dis J.
37:976–980. 2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Mori M, Hara T, Kikuchi M, Shimizu H,
Miyamoto T, Iwashima S, Oonishi T, Hashimoto K, Kobayashi N, Waki
K, et al: Infliximab versus intravenous immunoglobulin for
refractory Kawasaki disease: A phase 3, randomized, open-label,
active-controlled, parallel-group, multicenter trial. Sci Rep.
8(1994)2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang P, Tang C, Pan X, Chi H, Sun W and
Qu S: Comparison of the effects of infliximab and IVIG in the
treatment of ineffective IVIG. China Med Front Magazine (electronic
version). 10:66–69. 2018.
|
|
20
|
Son MB, Gauvreau K, Burns JC, Corinaldesi
E, Tremoulet AH, Watson VE, Baker A, Fulton DR, Sundel RP and
Newburger JW: Infliximab for intravenous immunoglobulin resistance
in kawasaki disease: A retrospective study. J Pediatr.
158:644–649.e1. 2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Sonoda K, Mori M, Hokosaki T and Yokota S:
Infliximab plus plasma exchange rescue therapy in Kawasaki disease.
J Pediatr. 164:1128–1132.e1. 2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Tremoulet AH, Jain S, Jaggi P,
Jimenez-Fernandez S, Pancheri JM, Sun X, Kanegaye JT, Kovalchin JP,
Printz BF, Ramilo O and Burns JC: Infliximab for intensification of
primary therapy for Kawasaki disease: A phase 3 randomised,
double-blind, placebo-controlled trial. Lancet. 383:1731–1738.
2014.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Youn Y, Kim J, Hong YM and Sohn S:
Infliximab as the first retreatment in patients with Kawasaki
disease resistant to initial intravenous immunoglobulin. Pediatr
Infect Dis J. 35:457–459. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Hur G, Song MS, Sohn S, Lee HD, Kim GB,
Cho HJ, Yoon KL, Joo CU, Hyun MC and Kim CH: Infliximab treatment
for intravenous immunoglobulin-resistant Kawasaki disease: A
multicenter study in Korea. Korean Circ J. 49:183–191.
2019.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Dionne A, Burns JC, Dahdah N, Tremoulet
AH, Gauvreau K, de Ferranti SD, Baker AL, Son MB, Gould P, Fournier
A, et al: Treatment intensification in patients with Kawasaki
disease and coronary aneurysm at diagnosis. Pediatrics. 143(pii:
e20183341)2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Mori M, Imagawa T, Hara R, Kikuchi M, Hara
T, Nozawa T, Miyamae T and Yokota S: Efficacy and limitation of
infliximab treatment for children with Kawasaki disease intractable
to intravenous immunoglobulin therapy: Report of an open-label case
series. J Rheumatol. 39:864–867. 2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Han CL and Zhao SL: Intravenous
immunoglobulin gamma (IVIG) versus IVIG plus infliximab in young
children with Kawasaki disease. Med Sci Monit. 24:7264–7270.
2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Masuda H, Kobayashi T, Hachiya A,
Nakashima Y, Shimizu H, Nozawa T, Ogihara Y, Ito S, Takatsuki S,
Katsumata N, et al: Infliximab for the treatment of refractory
Kawasaki disease: A nationwide survey in Japan. J Pediatr.
195:115–120.e3. 2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Song MS, Lee SB, Sohn S, Oh JH, Yoon KL,
Han JW and Kim CH: Infliximab treatment for refractory kawasaki
disease in korean children. Korean Circ J. 40:334–338.
2010.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Burns JC, Song Y, Bujold M, Shimizu C,
Kanegaye JT, Tremoulet AH and Franco A: Immune-monitoring in
Kawasaki disease patients treated with infliximab and intravenous
immunoglobulin. Clin Exp Immunol. 174:337–344. 2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Galeotti C, Kaveri SV, Cimaz R, Koné-Paut
I and Bayry J: Predisposing factors, pathogenesis and therapeutic
intervention of Kawasaki disease. Drug Discov Today. 21:1850–1857.
2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Moffett BS, Syblik D, Denfield S, Altman C
and Tejtel-Sexson K: Epidemiology of immunoglobulin resistant
Kawasaki disease: Results from a large, national database. Pediatr
Cardiol. 36:374–378. 2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Furusho K, Kamiya T, Nakano H, Kiyosawa N,
Shinomiya K, Hayashidera T, Tamura T, Hirose O, Manabe Y, Yokoyama
T, et al: High-dose intravenous gammaglobulin for Kawasaki disease.
Lancet. 2:1055–1058. 1984.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Rasouli M, Heidari B and Kalani M:
Downregulation of Th17 cells and the related cytokines with
treatment in Kawasaki disease. Immunol Lett. 162:269–275.
2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Guo MH, Tseng WN, Ko CH, Pan HM, Hsieh KS
and Kuo HC: Th17- and Treg-related cytokine and mRNA expression are
associated with acute and resolving Kawasaki disease. Allergy.
70:310–318. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bai X, Zhang R and Zhong J: Prediction of
unresponsiveness to intravenous immunoglobulin retreatmentin
patients with Kawasaki disease. Chin J Evid Based Pediatr.
11:16–20. 2016.(In Cbinese).
|
|
37
|
Burns JC, Capparelli EV, Brown JA,
Newburger JW and Glode MP: Intravenous gamma-globulin treatment and
retreatment in Kawasaki disease. US/Canadian Kawasaki syndrome
study group. Pediatr Infect Dis J. 17:1144–1148. 1998.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Newburger JW, Takahashi M, Gerber MA,
Gewitz MH, Tani LY, Burns JC, Shulman ST, Bolger AF, Ferrieri P,
Baltimore RS, et al: Diagnosis, treatment, and long-term management
of Kawasaki disease: A statement for health professionals from the
committee on rheumatic fever, endocarditis and Kawasaki disease,
council on cardiovascular disease in the young, American heart
association. Circulation. 110:2747–2771. 2004.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Matsubara T, Furukawa S and Yabuta K:
Serum levels of tumor necrosis factor, interleukin 2 receptor, and
interferon-gamma in Kawasaki disease involved coronary-artery
lesions. Clin Immunol Immunopathol. 56:29–36. 1990.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Ahn SY, Jang GC, Shin JS, Shin KM and Kim
DS: Tumor necrosis factor-alpha levels and promoter polymorphism in
patients with Kawasaki disease in Korea. Yonsei Med J.
44:1021–1026. 2003.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Hui-Yuen JS, Duong TT and Yeung RS:
TNF-alpha is necessary for induction of coronary artery
inflammation and aneurysm formation in an animal model of Kawasaki
disease. J Immunol. 176:6294–6301. 2006.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yamaji N, da Silva Lopes K, Shoda T,
Ishitsuka K, Kobayashi T, Ota E and Mori R: TNF-α blockers for the
treatment of Kawasaki disease in children. Cochrane Database Syst
Rev. 8(CD012448)2019.PubMed/NCBI View Article : Google Scholar
|