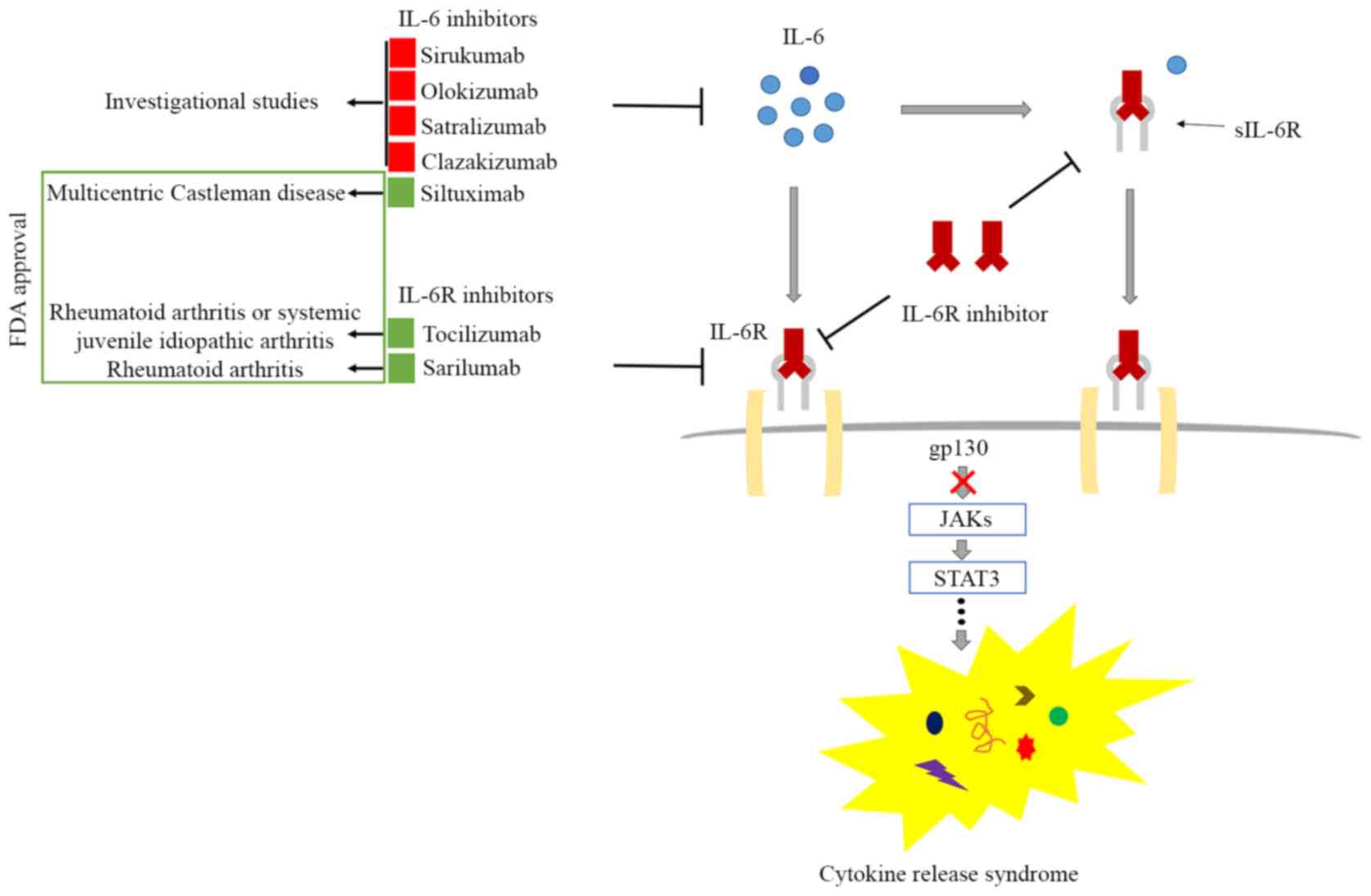
|
1
|
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu
Y, Zhang L, Fan G, Xu J, Gu X, et al: Clinical features of patients
infected with 2019 novel coronavirus in Wuhan, China. Lancet.
395:497–506. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Wang C, Horby PW, Hayden FG and Gao GF: A
novel coronavirus outbreak of global health concern. Lancet.
395:470–473. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J,
Wang B, Xiang H, Cheng Z, Xiong Y, et al: Clinical characteristics
of 138 hospitalized patients with 2019 novel coronavirus-infected
pneumonia in Wuhan, China. JAMA. 323:1061–1069. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Prompetchara E, Ketloy C and Palaga T:
Immune responses in COVID-19 and potential vaccines: Lessons
learned from SARS and MERS epidemic. Asian Pac J Allergy Immunol.
38:1–9. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Channappanavar R and Perlman S: Pathogenic
human coronavirus infections: Causes and consequences of cytokine
storm and immunopathology. Semin Immunopathol. 39:529–539.
2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wang Y, Wang Y, Chen Y and Qin Q: Unique
epidemiological and clinical features of the emerging 2019 novel
coronavirus pneumonia (COVID-19) implicate special control
measures. J Med Virol. 92:568–576. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xu Z, Shi L, Wang Y, Zhang J, Huang L,
Zhang C, Liu S, Zhao P, Liu H, Zhu L, et al: Pathological findings
of COVID-19 associated with acute respiratory distress syndrome.
Lancet Respir Med. 8:420–422. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Teachey DT, Lacey SF, Shaw PA, Melenhorst
JJ, Maude SL, Frey N, Pequignot E, Gonzalez VE, Chen F, Finklestein
J, et al: Identification of predictive biomarkers for cytokine
release syndrome after chimeric antigen receptor T-cell therapy for
acute lymphoblastic leukemia. Cancer Discov. 6:664–679.
2016.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zhang Y, Li J, Zhan Y, Wu L, Yu X, Zhang
W, Ye L, Xu S, Sun R, Wang Y and Lou J: Analysis of serum cytokines
in patients with severe acute respiratory syndrome. Infect Immun.
72:4410–4415. 2004.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Norelli M, Camisa B, Barbiera G, Falcone
L, Purevdorj A, Genua M, Sanvito F, Ponzoni M, Doglioni C,
Cristofori P, et al: Monocyte-derived IL-1 and IL-6 are
differentially required for cytokine-release syndrome and
neurotoxicity due to CAR T cells. Nat Med. 24:739–748.
2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Schmidt-Arras D and Rose-John S: IL-6
pathway in the liver: From physiopathology to therapy. J Hepatol.
64:1403–1415. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Baran P, Hansen S, Waetzig GH, Akbarzadeh
M, Lamertz L, Huber HJ, Ahmadian MR, Moll JM and Scheller J: The
balance of interleukin (IL)-6, IL-6.soluble IL-6 receptor (sIL-6R),
and IL-6.sIL-6R.sgp130 complexes allows simultaneous classic and
trans-signaling. J Biol Chem. 293:6762–6775. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Rose-John S: IL-6 trans-signaling via the
soluble IL-6 receptor: Importance for the pro-inflammatory
activities of IL-6. Int J Biol Sci. 8:1237–1247. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Banerjee K and Resat H: Constitutive
activation of STAT3 in breast cancer cells: A review. Int J Cancer.
138:2570–2578. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Johnson DE, O'Keefe RA and Grandis JR:
Targeting the IL-6/JAK/STAT3 signalling axis in cancer. Nat Rev
Clin Oncol. 15:234–248. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Deisseroth A, Ko CW, Nie L, Zirkelbach JF,
Zhao L, Bullock J, Mehrotra N, Del Valle P, Saber H, Sheth C, et
al: FDA approval: Siltuximab for the treatment of patients with
multicentric castleman disease. Clin Cancer Res. 21:950–954.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yamamura T, Kleiter I, Fujihara K, Palace
J, Greenberg B, Zakrzewska-Pniewska B, Patti F, Tsai CP, Saiz A,
Yamazaki H, et al: Trial of satralizumab in neuromyelitis optica
spectrum disorder. N Engl J Med. 381:2114–2124. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Tanaka T, Narazaki M and Kishimoto T: IL-6
in inflammation, immunity, and disease. Cold Spring Harb Perspect
Biol. 6(a016295)2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
McGonagle D, Sharif K, O'Regan A and
Bridgewood C: The role of cytokines including interleukin-6 in
COVID-19 induced pneumonia and macrophage activation syndrome-like
disease. Autoimmun Rev. 19(102537)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Matthews VB, Allen TL, Risis S, Chan MH,
Henstridge DC, Watson N, Zaffino LA, Babb JR, Boon J, Meikle PJ, et
al: Interleukin-6-deficient mice develop hepatic inflammation and
systemic insulin resistance. Diabetologia. 53:2431–2441.
2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Navarro G, Taroumian S, Barroso N, Duan L
and Furst D: Tocilizumab in rheumatoid arthritis: A meta-analysis
of efficacy and selected clinical conundrums. Semin Arthritis
Rheum. 43:458–469. 2014.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Le RQ, Li L, Yuan W, Shord SS, Nie L,
Habtemariam BA, Przepiorka D, Farrell AT and Pazdur R: FDA approval
summary: Tocilizumab for treatment of chimeric antigen receptor T
cell-induced severe or life-threatening cytokine release syndrome.
Oncologist. 23:943–947. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Jones SA, Richards PJ, Scheller J and
Rose-John S: IL-6 transsignaling: The in vivo consequences. J
Interferon Cytokine Res. 25:241–253. 2005.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kaly L and Rosner I: Tocilizumab-a novel
therapy for non-organ-specific autoimmune diseases. Best Pract Res
Clin Rheumatol. 26:157–165. 2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yokota S, Imagawa T, Mori M, Miyamae T,
Aihara Y, Takei S, Iwata N, Umebayashi H, Murata T, Miyoshi M, et
al: Efficacy and safety of tocilizumab in patients with
systemic-onset juvenile idiopathic arthritis: A randomised,
double-blind, placebo-controlled, withdrawal phase III trial.
Lancet. 371:998–1006. 2008.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Gabay C, Emery P, van Vollenhoven R,
Dikranian A, Alten R, Pavelka K, Klearman M, Musselman D, Agarwal
S, Green J, et al: Tocilizumab monotherapy versus adalimumab
monotherapy for treatment of rheumatoid arthritis (ADACTA): A
randomised, double-blind, controlled phase 4 trial. Lancet.
381:1541–1550. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Yanaihara N, Hirata Y, Yamaguchi N,
Noguchi Y, Saito M, Nagata C, Takakura S, Yamada K and Okamoto A:
Antitumor effects of interleukin-6 (IL-6)/interleukin-6 receptor
(IL-6R) signaling pathway inhibition in clear cell carcinoma of the
ovary. Mol Carcinog. 55:832–841. 2016.PubMed/NCBI View
Article : Google Scholar
|
|
28
|
Goumas FA, Holmer R, Egberts JH,
Gontarewicz A, Heneweer C, Geisen U, Hauser C, Mende MM, Legler K,
Röcken C, et al: Inhibition of IL-6 signaling significantly reduces
primary tumor growth and recurrencies in orthotopic xenograft
models of pancreatic cancer. Int J Cancer. 137:1035–1046.
2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Grivennikov S, Karin E, Terzic J, Mucida
D, Yu GY, Vallabhapurapu S, Scheller J, Rose-John S, Cheroutre H,
Eckmann L and Karin M: IL-6 and Stat3 are required for survival of
intestinal epithelial cells and development of colitis-associated
cancer. Cancer Cell. 15:103–113. 2009.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Dijkgraaf EM, Santegoets SJ, Reyners AK,
Goedemans R, Wouters MC, Kenter GG, van Erkel AR, van Poelgeest MI,
Nijman HW, van der Hoeven JJ, et al: A phase I trial combining
carboplatin/doxorubicin with tocilizumab, an anti-IL-6R monoclonal
antibody, and interferon-alpha2b in patients with recurrent
epithelial ovarian cancer. Ann Oncol. 26:2141–2149. 2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhou Y, Fu B, Zheng X, Wang D, Zhao C, Qi
Y, Sun R, Tian Z, Xu X and Wei H: Aberrant pathogenic
GM-CSF+T cells and inflammatory CD14+
CD16+ monocytes in severe pulmonary syndrome patients of
a new coronavirus. bioRxiv. Feb 20, 2020 doi: urihttps://doi.org/10.1101/2020.02.12.945576simplehttps://doi.org/10.1101/2020.02.12.945576.
|
|
32
|
Xu X, Han M, Li T, Sun W, Wang D, Fu B,
Zhou Y, Zheng X, Yang Y, Li X, et al: Effective treatment of severe
COVID-19 patients with tocilizumab. Proc Natl Acad Sci USA.
117:10970–10975. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Price CC, Altice FL, Shyr Y, Koff A,
Pischel L, Goshua G, Azar MM, Mcmanus D, Chen SC, Gleeson SE, et
al: Tocilizumab treatment for cytokine release syndrome in
hospitalized COVID-19 patients: Survival and clinical outcomes.
Chest: 15 Jun, 2020 doi: 10.1016/j.chest.2020.06.006 (Epub ahead
for print).
|
|
34
|
Toniati P, Piva S, Cattalini M, Garrafa E,
Regola F, Castelli F, Franceschini F, Airò P, Bazzani C, Beindorf
EA, et al: Tocilizumab for the treatment of severe COVID-19
pneumonia with hyperinflammatory syndrome and acute respiratory
failure: A single center study of 100 patients in Brescia, Italy.
Autoimmun Rev. 19(102568)2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kewan T, Covut F, Al-Jaghbeer MJ, Rose L,
Gopalakrishna KV and Akbik B: Tocilizumab for treatment of patients
with severe COVID-19: A retrospective cohort study.
EClinicalMedicine. 24(100418)2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Eimer J, Vesterbacka J, Svensson AK,
Stojanovic B, Wagrell C, Sonnerborg A and Nowak P: Tocilizumab
shortens time on mechanical ventilation and length of hospital stay
in patients with severe COVID-19: A retrospective cohort study. J
Intern Med: Aug 3, 2020 doi: 10.1111/joim.13162 (Epub ahead for
print).
|
|
37
|
Potere N, Di Nisio M, Rizzo G, La Vella M,
Polilli E, Agostinone A, Spacone A, Di Carlo S, Costantini A,
Abbate A, et al: Low-dose subcutaneous tocilizumab to prevent
disease progression in patients with moderate COVID-19 pneumonia
and hyperinflammation. Int J Infect Dis: Aug 5, 2020 doi:
10.1016/j.ijid.2020.07.078 (Epub ahead for print).
|
|
38
|
Arthritis drug shows ‘significant’ promise
in severe COVID-19 cases: Study (2020, April 28) urihttps://medicalxpress.com/news/2020-04-arthritis-drug-significant-severe-covid-.htmlsimplehttps://medicalxpress.com/news/2020-04-arthritis-drug-significant-severe-covid-.html.
Accessed 30 May 2020.
|
|
39
|
Diagnosis and Treatment Protocol for Novel
Coronavirus Pneumonia (7th Interim edition). China NHCOTPSRO.
urihttp://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989/files/ce3e6945832a438eaae415350a8ce964.pdfsimplehttp://www.nhc.gov.cn/yzygj/s7653p/202003/46c9294a7dfe4cef80dc7f5912eb1989/files/ce3e6945832a438eaae415350a8ce964.pdf.
Accessed 30 May 2020.
|