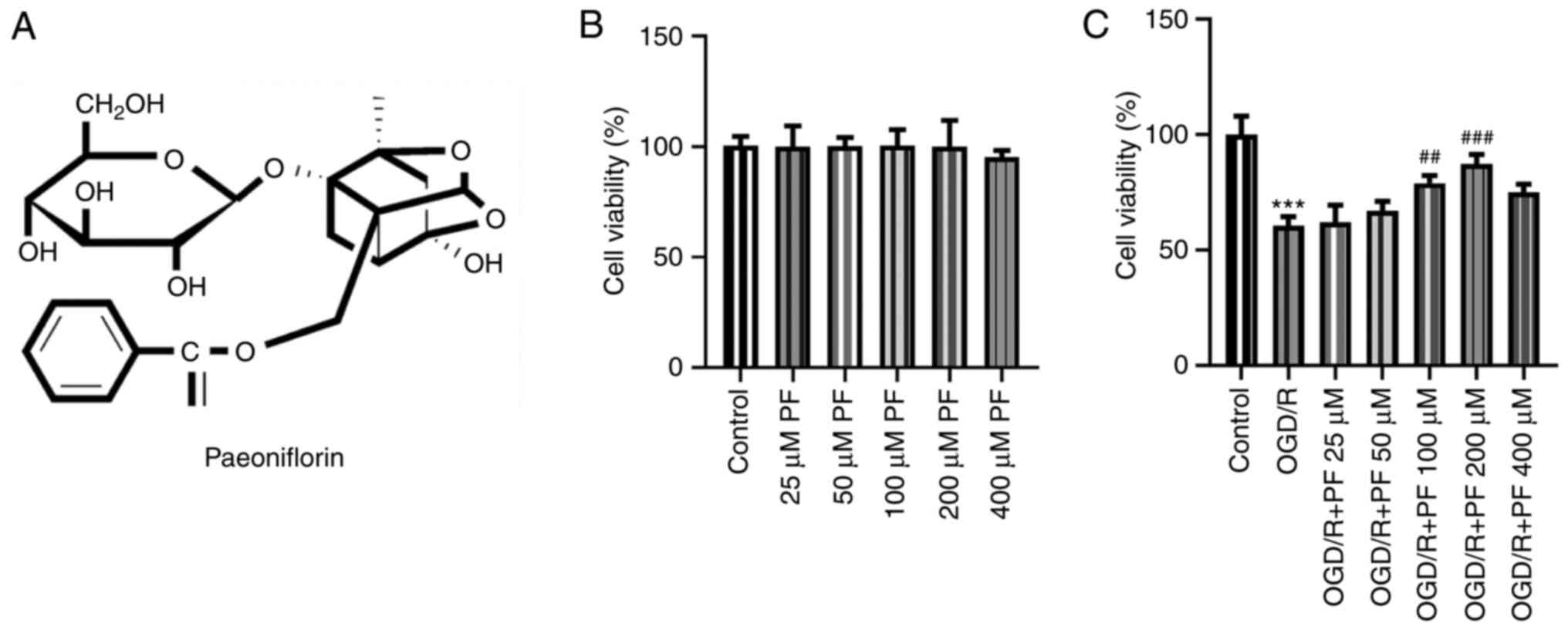
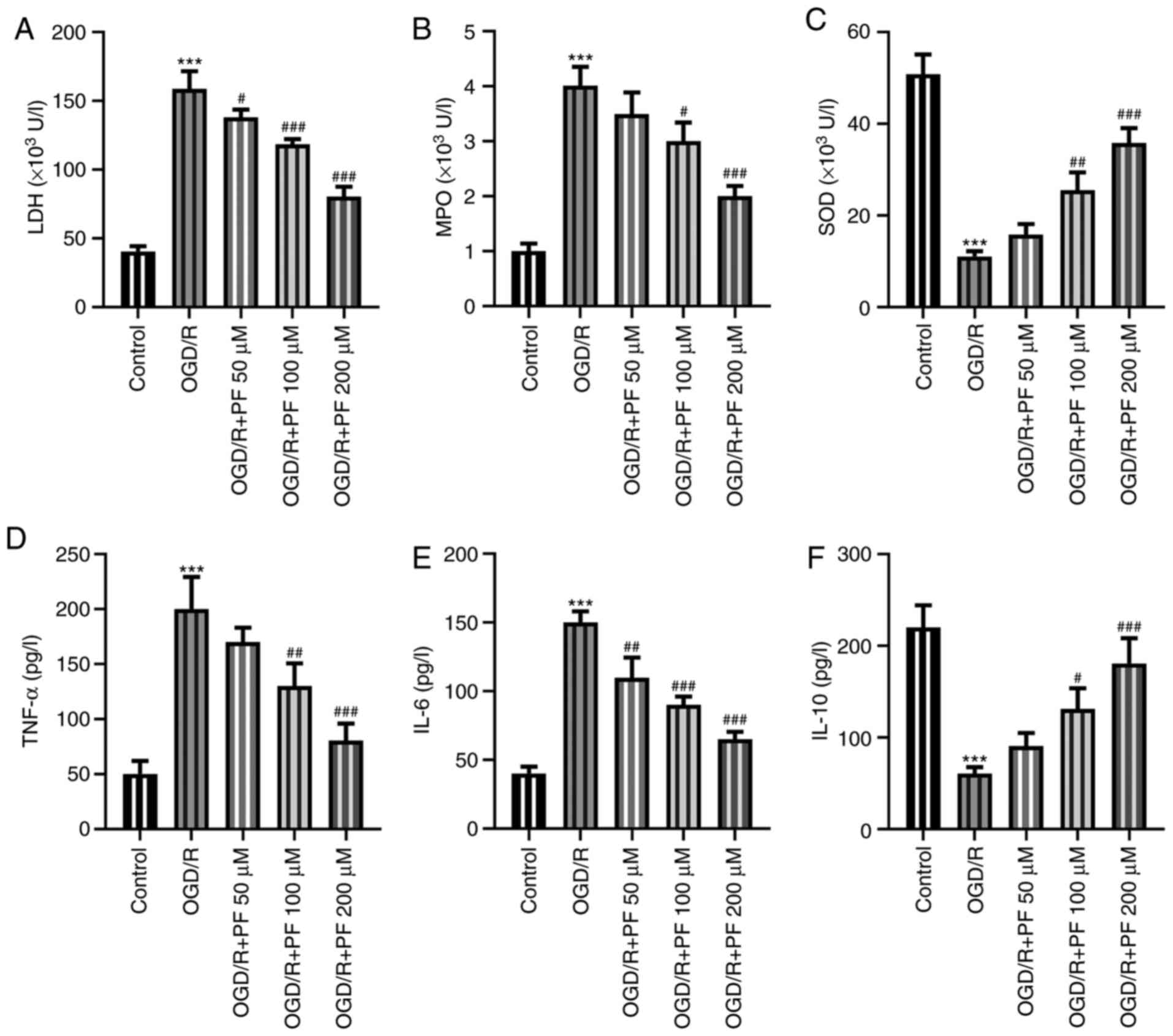
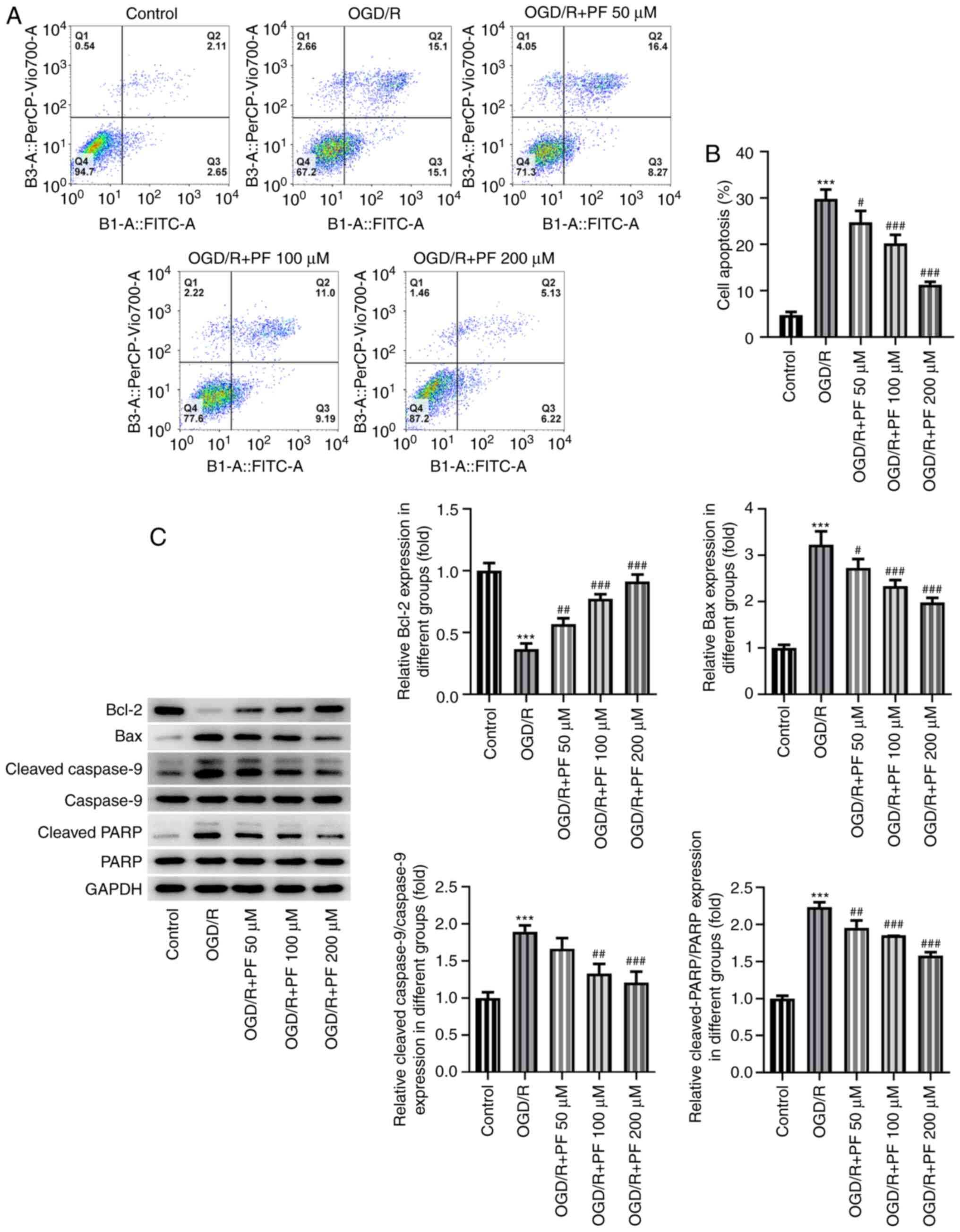
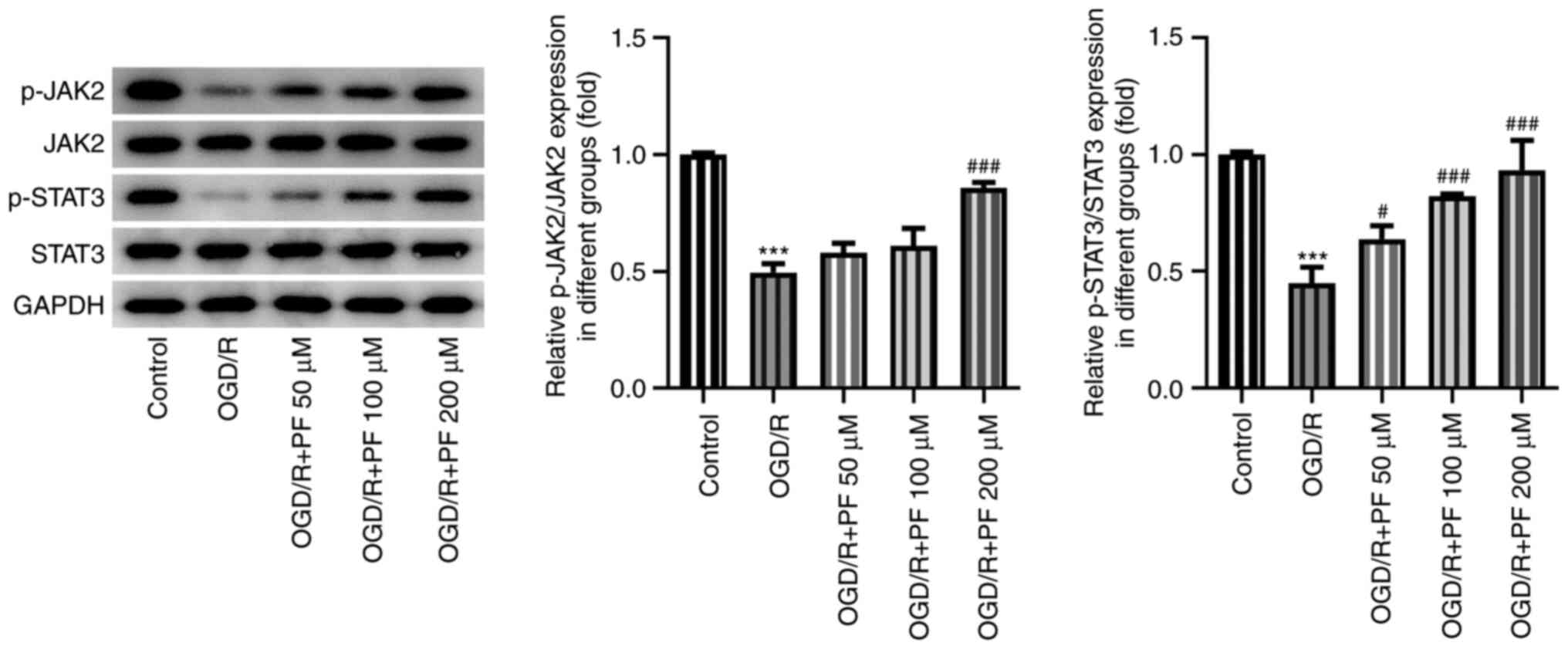
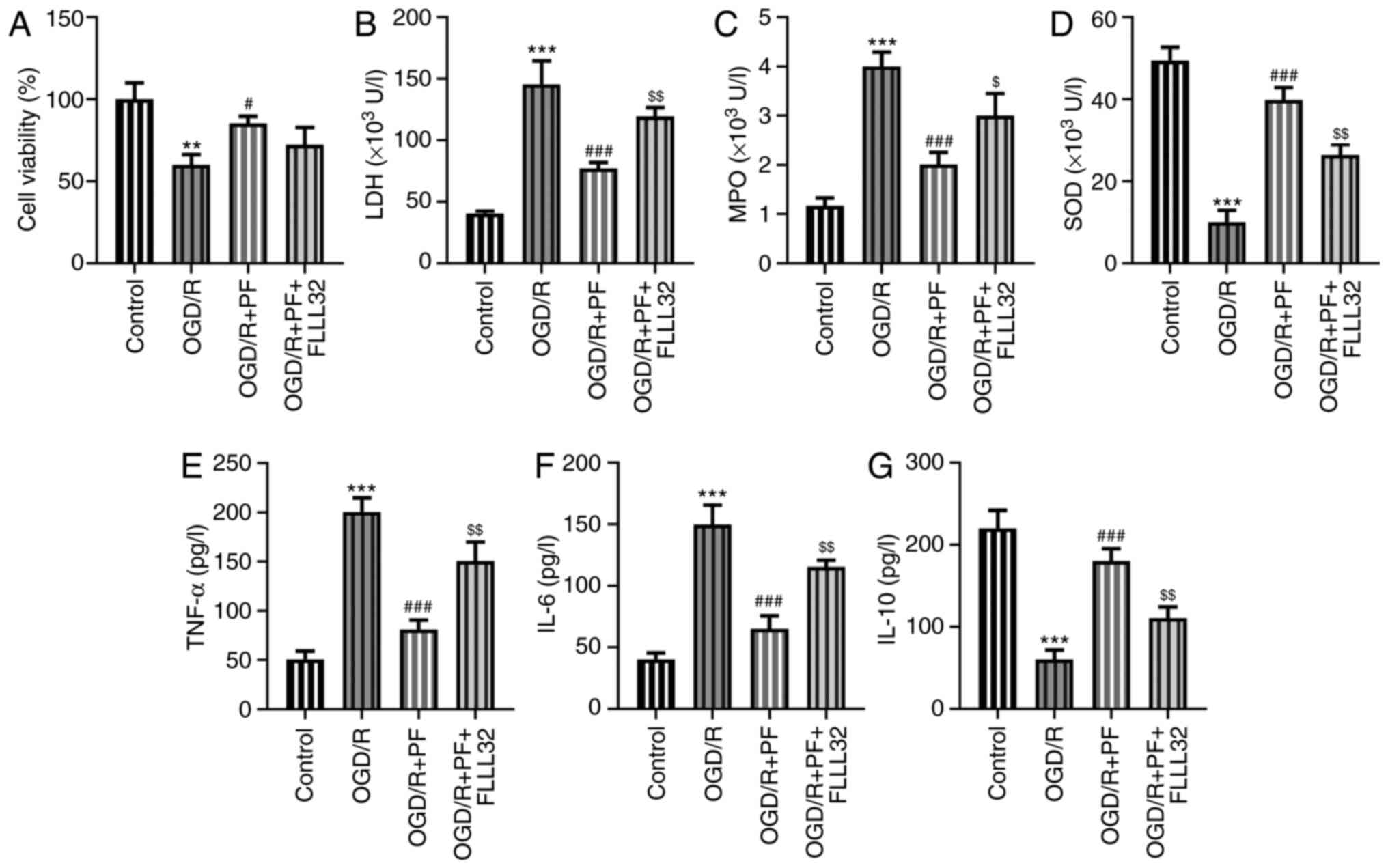
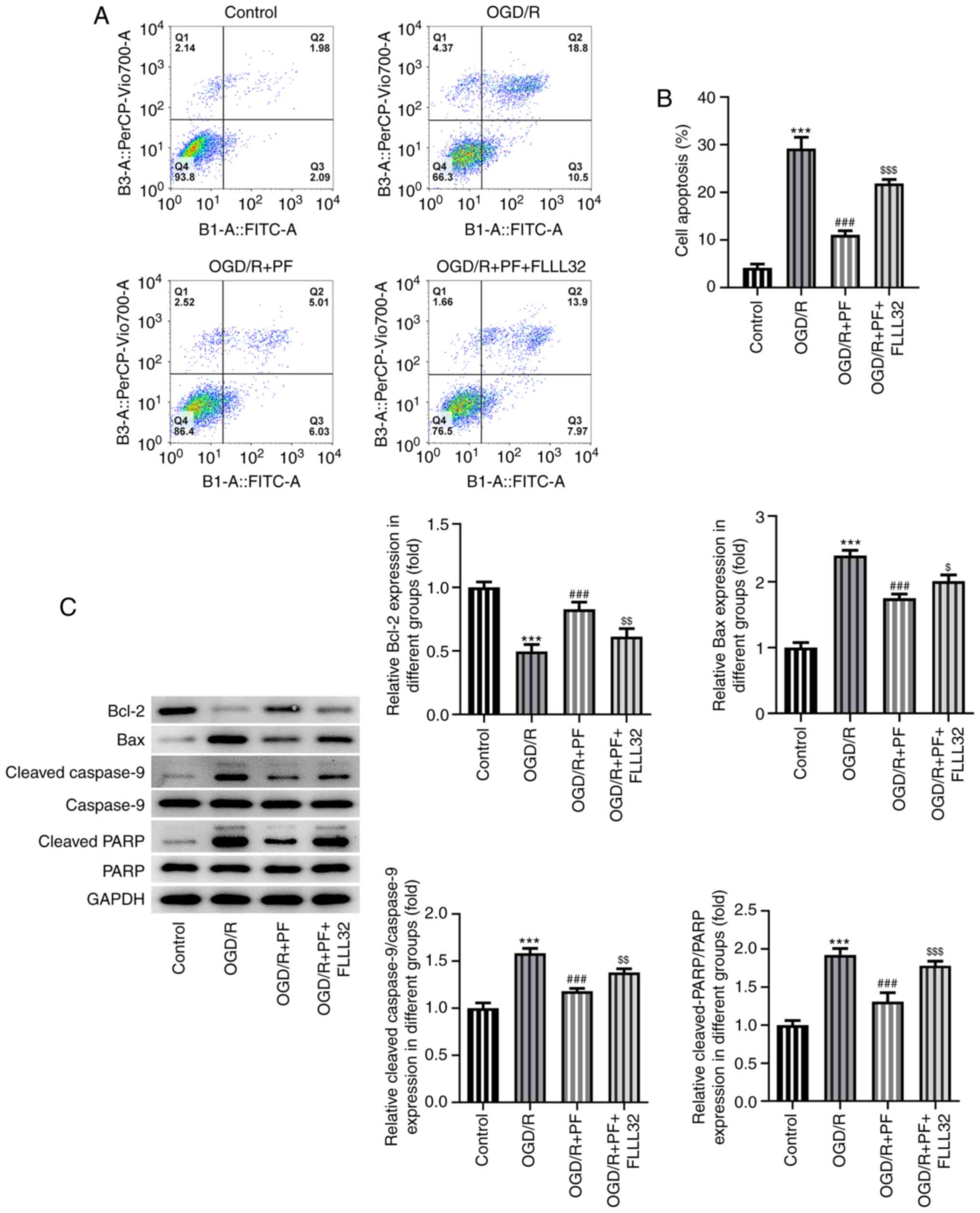
|
1
|
Martini ML, Neifert SN, Lara-Reyna JJ,
Shuman WH, Ladner TR, Hardigan TH, Fifi JT, Mocco J and Yaeger KA:
Trials in thrombectomy for acute ischemic stroke: Describing the
state of clinical research in the field. Clin Neurol Neurosurg.
200(106360)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Chen H, Yoshioka H, Kim GS, Jung JE, Okami
N, Sakata H, Maier CM, Narasimhan P, Goeders CE and Chan PH:
Oxidative stress in ischemic brain damage: Mechanisms of cell death
and potential molecular targets for neuroprotection. Antioxid Redox
Signal. 14:1505–1517. 2011.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Bavarsad K, Barreto GE, Hadjzadeh MA and
Sahebkar A: Protective effects of curcumin against
ischemia-reperfusion injury in the nervous system. Mol Neurobiol.
56:1391–1404. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Chen A, Wang H, Zhang Y, Wang X, Yu L, Xu
W, Xu W and Lin Y: Paeoniflorin exerts neuroprotective effects
against glutamate induced PC12 cellular cytotoxicity by inhibiting
apoptosis. Int J Mol Med. 40:825–833. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Zhai A, Zhang Z and Kong X: Paeoniflorin
alleviates H2O2-induced oxidative injury
through down-regulation of microRNA-135a in HT-22 cells. Neurochem
Res. 44:2821–2831. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wen Z, Hou W, Wu W, Zhao Y, Dong X, Bai X,
Peng L and Song L: 6'-O-Galloylpaeoniflorin attenuates cerebral
ischemia reperfusion-induced neuroinflammation and oxidative stress
via PI3K/Akt/Nrf2 activation. Oxid Med Cell Longev.
2018(8678267)2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Liu H, Yu C, Xu T, Zhang X and Dong M:
Synergistic protective effect of paeoniflorin and β-ecdysterone
against rotenone-induced neurotoxicity in PC12 cells. Apoptosis.
21:1354–1365. 2016.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Shio MT, Christian JG, Jung JY, Chang KP
and Olivier M: PKC/ROS-mediated NLRP3 inflammasome activation is
attenuated by leishmania zinc-metalloprotease during Infection.
PLoS Negl Trop Dis. 9(e0003868)2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Li J, Ji X, Zhang J, Shi G, Zhu X and Wang
K: Paeoniflorin attenuates Aβ25-35-induced neurotoxicity in PC12
cells by preventing mitochondrial dysfunction. Folia Neuropathol.
52:285–290. 2014.PubMed/NCBI
|
|
10
|
Wang D, Wong HK, Feng YB and Zhang ZJ:
Paeoniflorin, a natural neuroprotective agent, modulates multiple
anti-apoptotic and pro-apoptotic pathways in differentiated PC12
cells. Cell Mol Neurobiol. 33:521–529. 2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hou Y, Wang K, Wan W, Cheng Y, Pu X and Ye
X: Resveratrol provides neuroprotection by regulating the
JAK2/STAT3/PI3K/AKT/mTOR pathway after stroke in rats. Genes Dis.
5:245–255. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
de Couto G, Liu W, Tseliou E, Sun B,
Makkar N, Kanazawa H, Arditi M and Marbán E: Macrophages mediate
cardioprotective cellular postconditioning in acute myocardial
infarction. J Clin Invest. 125:3147–3162. 2015.PubMed/NCBI View
Article : Google Scholar
|
|
13
|
Li X, Wang Y, Wang K and Wu Y: Renal
protective effect of Paeoniflorin by inhibition of JAK2/STAT3
signaling pathway in diabetic mice. Biosci Trends. 12:168–176.
2018.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yang B, Zang LE, Cui JW, Zhang MY, Ma X
and Wei LL: Melatonin plays a protective role by regulating
miR-26a-5p-NRSF and JAK2-STAT3 pathway to improve autophagy,
inflammation and oxidative stress of cerebral ischemia-reperfusion
injury. Drug Des Devel Ther. 14:3177–3188. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yuan J, Zeng L, Sun Y, Wang N, Sun Q,
Cheng Z and Wang Y: SH2B1 protects against OGD/R induced apoptosis
in PC12 cells via activation of the JAK2/STAT3 signaling pathway.
Mol Med Rep. 18:2613–2620. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zhang LL, Wei W, Wang NP, Wang QT, Chen
JY, Chen Y, Wu H and Hu XY: Paeoniflorin suppresses inflammatory
mediator production and regulates G protein-coupled signaling in
fibroblast-like synoviocytes of collagen induced arthritic rats.
Inflamm Res. 57:388–395. 2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ling L, Tong J and Zeng L: Paeoniflorin
improves acute lung injury in sepsis by activating Nrf2/Keap1
signaling pathway. Sichuan Da Xue Xue Bao Yi Xue Ban. 51:664–669.
2020.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
18
|
Zhou H, Bian D, Jiao X, Wei Z, Zhang H,
Xia Y, He Y and Dai Y: Paeoniflorin protects against
lipopolysaccharide-induced acute lung injury in mice by alleviating
inflammatory cell infiltration and microvascular permeability.
Inflamm Res. 60:981–990. 2011.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Jiang WL, Chen XG, Zhu HB, Gao YB, Tian JW
and Fu FH: Paeoniflorin inhibits systemic inflammation and improves
survival in experimental sepsis. Basic Clin Pharmacol Toxicol.
105:64–71. 2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Wei S, Ma X, Niu M, Wang R, Yang T, Wang
D, Wen J, Li H and Zhao Y: Mechanism of paeoniflorin in the
treatment of bile duct ligation-induced cholestatic liver injury
using integrated metabolomics and network pharmacology. Front
Pharmacol. 11(586806)2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Poprac P, Jomova K, Simunkova M, Kollar V,
Rhodes CJ and Valko M: Targeting free radicals in oxidative
stress-related human diseases. Trends Pharmacol Sci. 38:592–607.
2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chen S, Chen H, Du Q and Shen J: Targeting
myeloperoxidase (MPO) mediated oxidative stress and inflammation
for reducing brain ischemia injury: Potential application of
natural compounds. Front Physiol. 11(433)2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yu G, Liang Y, Huang Z, Jones DW,
Pritchard KA Jr and Zhang H: Erratum to: Inhibition of
myeloperoxidase oxidant production by N-acetyl lysyltyrosylcysteine
amide reduces brain damage in a murine model of stroke. J
Neuroinflammation. 13(166)2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Briyal S, Gulati K and Gulati A: Repeated
administration of exendin-4 reduces focal cerebral ischemia-induced
infarction in rats. Brain Res. 1427:23–34. 2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhao L, Xu J, Wang Q, Qian Z, Feng W, Yin
X and Fang Y: Protective effect of rhGLP-1 (7-36) on brain
ischemia/reperfusion damage in diabetic rats. Brain Res.
1602:153–159. 2015.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Fang Y, Liu X, Zhao L, Wei Z, Jiang D,
Shao H, Zang Y, Xu J, Wang Q, Liu Y, et al: RhGLP-1 (7-36) protects
diabetic rats against cerebral ischemia-reperfusion injury via
up-regulating expression of Nrf2/HO-1 and increasing the activities
of SOD. Korean J Physiol Pharmacol. 21:475–485. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Dai X, Wang LW, Jia XY, Chang Y, Wu HX,
Wang C and Wei W: Paeoniflorin regulates the function of human
peripheral blood mononuclear cells stimulated by rhIL-1β by
up-regulating Treg expression. Immunopharmacol Immunotoxicol.
37:252–257. 2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Jin L, Zhang LM, Xie KQ, Ye Y and Feng L:
Paeoniflorin suppresses the expression of intercellular adhesion
molecule-1 (ICAM-1) in endotoxin-treated human monocytic cells. Br
J Pharmacol. 164:694–703. 2011.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zhai T, Sun Y, Li H, Zhang J, Huo R, Li H,
Shen B and Li N: Unique immunomodulatory effect of paeoniflorin on
type I and II macrophages activities. J Pharmacol Sci. 130:143–150.
2016.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Shi D, Wang Q, Zheng H, Li D, Shen Y, Fu
H, Li T, Mei H, Lu G, Qiu Y, et al: Paeoniflorin suppresses
IL-6/Stat3 pathway via upregulation of Socs3 in dendritic cells in
response to 1-chloro-2,4-dinitrobenze. Int Immunopharmacol.
38:45–53. 2016.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Adams JM and Cory S: The Bcl-2 protein
family: Arbiters of cell survival. Science. 281:1322–1326.
1998.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Shyu WC, Lin SZ, Chiang MF, Chen DC, Su
CY, Wang HJ, Liu RS, Tsai CH and Li H: Secretoneurin promotes
neuroprotection and neuronal plasticity via the Jak2/Stat3 pathway
in murine models of stroke. J Clin Invest. 118:133–148.
2008.PubMed/NCBI View
Article : Google Scholar
|
|
33
|
Hossain E, Li Y and Anand-Srivastava MB:
Role of JAK2/STAT3 pathway in angiotensin II-induced enhanced
expression of Gialpha proteins and hyperproliferation of aortic
vascular smooth muscle cells. Can J Physiol Pharmacol. 99:237–246.
2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
El-Ghafar OAMA, Helal GK and Abo-Youssef
AM: Apixaban exhibits anti-arthritic effects by inhibiting
activated factor X-mediated JAK2/STAT3 and MAPK phosphorylation
pathways. Inflammopharmacology. 28:1253–1267. 2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Pan S, Chen Y, Zhang X and Xie Y: The
JAK2/STAT3 pathway is involved in dexmedetomidine-induced
myocardial protection in rats undergoing cardiopulmonary bypass.
Ann Transl Med. 8(483)2020.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Wu QY, Ma MM, Fu L, Zhu YY, Liu Y, Cao J,
Zhou P, Li ZY, Zeng LY, Li F, et al: Roles of germline JAK2
activation mutation JAK2 V625F in the pathology of
myeloproliferative neoplasms. Int J Biol Macromol. 116:1064–1073.
2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Leong PL, Andrews GA, Johnson DE, Dyer KF,
Xi S, Mai JC, Robbins PD, Gadiparthi S, Burke NA, Watkins SF, et
al: Targeted inhibition of Stat3 with a decoy oligonucleotide
abrogates head and neck cancer cell growth. Proc Natl Acad Sci USA.
100:4138–4143. 2003.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Li L, Li H and Li M: Curcumin protects
against cerebral ischemia-reperfusion injury by activating
JAK2/STAT3 signaling pathway in rats. Int J Clin Exp Med.
8:14985–14991. 2015.PubMed/NCBI
|
|
39
|
Li L, Sun L, Qiu Y, Zhu W, Hu K and Mao J:
Protective effect of stachydrine against cerebral
ischemia-reperfusion injury by reducing inflammation and apoptosis
through P65 and JAK2/STAT3 signaling pathway. Front Pharmacol.
11(64)2020.PubMed/NCBI View Article : Google Scholar
|