Introduction
The presence of a right atrial mass (RAM), despite
the advanced diagnostic modalities available, still poses a
diagnostic dilemma. RAM constitutes tumors, thrombus, prominent
crista terminalis, or endocarditis vegetation (1). Cardiac myxomas are benign and are the
most common primary cardiac tumor, 15% of atrial myxomas arise in
the right atrium (RA), and they are usually attached to the
interatrial septum (2,3). Myxomatous disease occurs in all age
groups, and both sexes have an approximate incidence of 0.5-1.0
cases per million inhabitants per year (4,5).
Incidences of CVC-related right atrial thrombosis
(CRAT) are underreported phenomena, but when the thrombus is
present, it carries a mortality risk of 18% in hemodialysis (HD)
patients and greater than 40% risk in non-HD patients (6,7).
Pathogenic mechanisms incriminated for the development of right
atrial thrombosis include mechanical irritation of the myocardial
atrial wall in the presence of CVC, emboli entrapped from
peripheral venous thrombosis migration, cloths propagation from
vena cava, hypercoagulability, and in situ thrombi formation
under low-flow atrial conditions (6,7).
Presentation of CRAT may be asymptomatic or may be associated with
pulmonary embolism, tricuspid valve pathology, leg edema, systemic
embolism, infected thrombi, and right heart failure with
hemodynamic compromise (6). Fatal
complications in CRAT reported are arrhythmias and mechanical
cardiac complications (6). The risk
of right thrombus formation unrelated with CVC is increased in
patients who have antiphospholipid syndrome and who show positive
test results for lupus anticoagulant activity or have medium or
high levels of anticardiolipin antibodies (3). CRAT may be suspected if there is
resistance in blood flow noted during HD.
Transthoracic echocardiography (TTE),
transesophageal echocardiography (TEE), and cardiac magnetic
resonance (MRI) with gadolinium contrast material can be useful
tools for diagnosis and tissue characterization; tumor does not
show gadolinium contrast enhancement. TEE has better sensitivity
and specificity than TTE; if doubt remains after the
echocardiographic examination, cardiac MRI imaging remains a good
alternative that can prevent misdiagnosis and redundant invasive
measures. MRI has high sensitivity and specificity for the
detection of intracardiac mass and can differentiate thrombus,
lipoma, inflammatory masses, and many solid tumors. When cardiac
myxomas are confirmed and surgically resected, they lose their
potential to cause complications, and patients, except for rare
cases of recurrences (8), are
successfully treated. When CRAT was diagnosed, anticoagulation was
the treatment of choice, but when the clot's evolution became
unfavorable, surgery remained the ‘gold standard’ treatment.
In this study, we present the case of a 63-year-old
man with chronic kidney disease, in HD, who underwent TTE and
identification of a tumoral mass of 35x26 mm in the RA.
Case report
Patient
A 64-year-old male with a past medical history of
hypertension, end-stage renal disease due to autosomal dominant
polycystic kidney disease in HD, type 2 diabetes mellitus
insulin-dependent, and paroxysmal atrial fibrillation was referred
to the Department of Cardiovascular Surgery, ‘Prof. Dr. C. C.’
Iliescu Emergency Institute for Cardiovascular Diseases, Bucharest
for intermittent chest pain and shortness of breath worsening over
the last two weeks, especially on exertion. The chest pain was
described as retrosternal and non-radiating. The patient had a
long-term HD catheter placed in the right internal jugular vein and
left brachial-basilic arteriovenous fistula (BBAVF). HD center
staff confirmed maintaining catheter care and heparin usage for
thrombosis prevention. No history of thrombosis or catheter
infection was documented or described by the patient.
On admission, the patient was clinically stable with
no fever, soft abdomen, no peripheral edema, normotensive with
blood pressure 120/60 mmHg. Respiratory rate was 22 breaths/min
with an oxygen saturation of 95%. An electrocardiogram (ECG)
revealed sinus rhythm with a heart rate of 80 bpm. The chest X-ray
was normal. Laboratory tests revealed a mild normochromic,
normocytic anemia (Hb 11.7 g/dl), hyperkalemia (6.2 mmol/l), and
high creatinine levels (12.31 mg/dl). Troponin levels were normal.
No microorganisms were found in the patient's blood cultures.
Two-dimensional transthoracic echocardiography (TTE) performed
showed normal-sized left ventricle (LV), with an ejection fraction
of 60%, and a mild left and right atrial enlargement (LA=42 mm,
RA=43 mm). The right ventricle was dilated with an end-diastolic
diameter of 41 mm. No valvular disease was found. Mild hypertrophy
of the interventricular septum was present. The most important
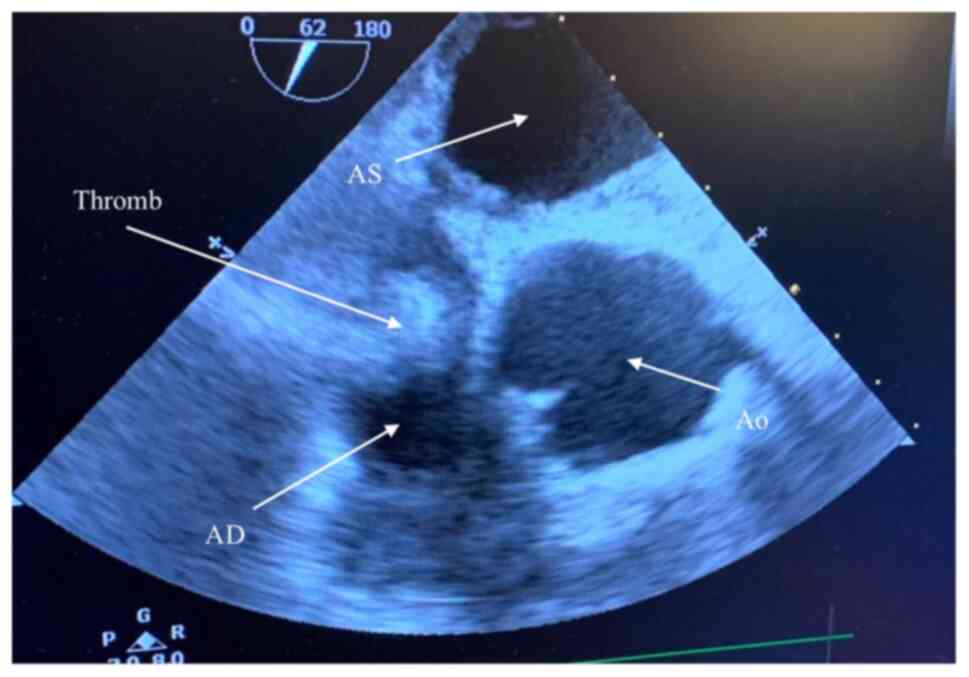
finding revealed by the TTE was a right atrial echogenic mass of
17/18 mm, independent of the catheter tip (Fig. 1). Coronary angiography revealed
normal coronary arteries. No vascularization of the RAM was
identified. In view of these findings, we considered the RAM to be
either a myxoma or a thrombus. Considering the size of the mass and
the high risk of pulmonary embolism, the treatment with
anticoagulant was deemed too risky at the time; thus, surgical
treatment was elected.
Method
A discussion was held with the patient regarding the
risks and benefits involved with the surgical removal of the mass.
The patient agreed to the surgical procedure. The patient underwent
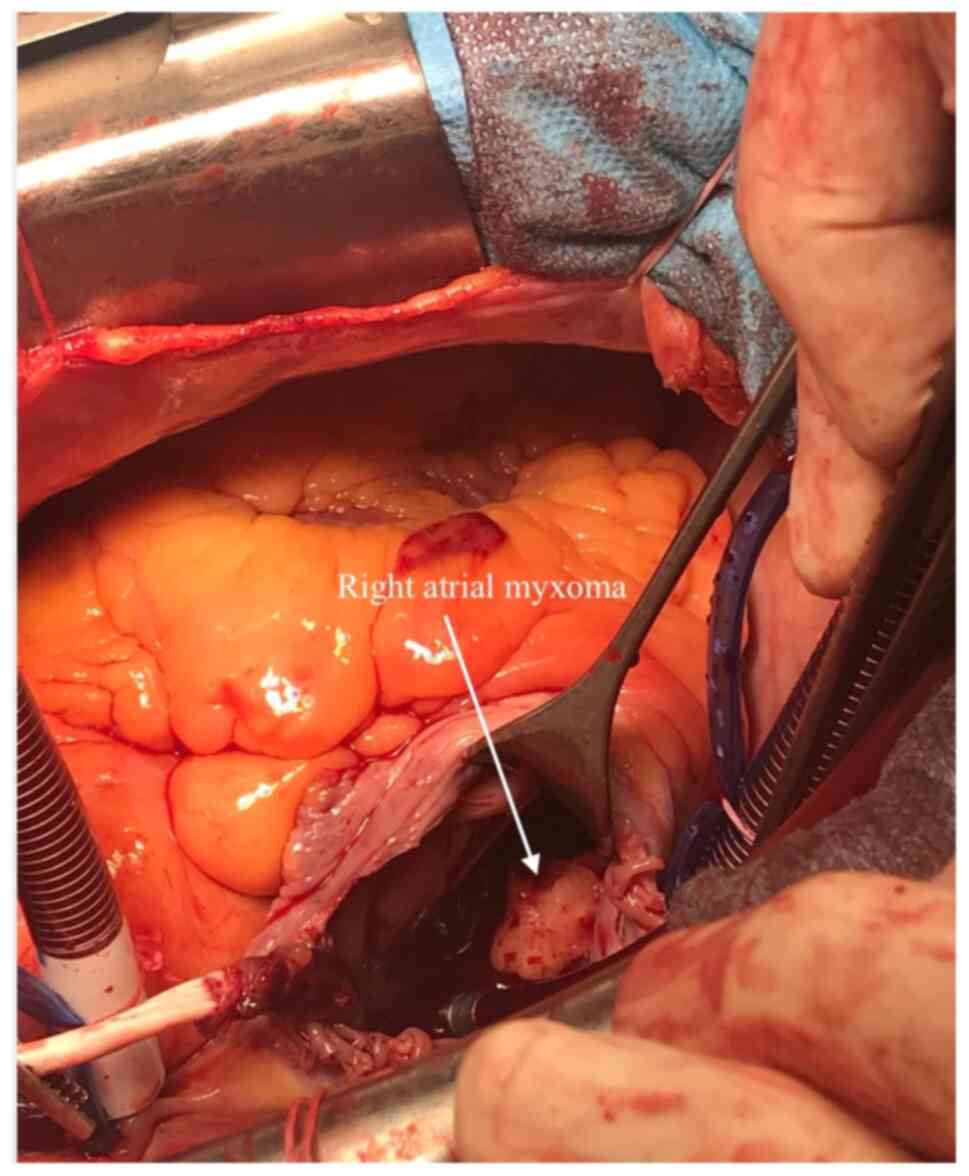
open-heart surgery for the removal of the RAM. After initiation of
cardiopulmonary bypass (CPB), right atriotomy was performed, the
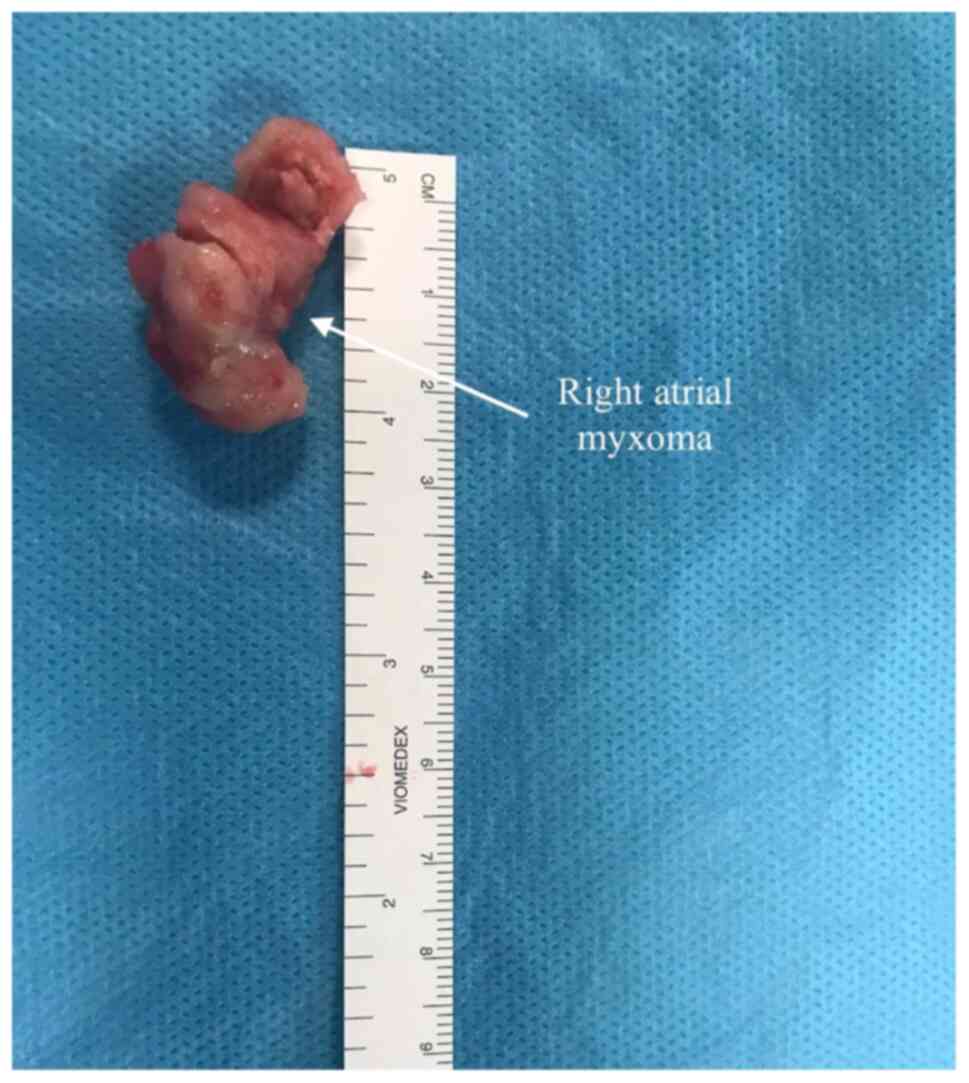
tip of the HD catheter and the mass were removed and sent for
pathology exam (Fig. 2). The mass
was anchored in close proximity to the coronary sinus venosus
orifice (Fig. 3). The next day, the
rest of the right internal jugular vein catheter was removed, and
an HD catheter was inserted in the left femoral vein.
Postoperatively the patient experienced self-remitting atrial
fibrillation and left upper limb edema for which unfractionated
heparin infusion was initiated with good results. Subsequently,
anticoagulant therapy (Acenocoumarol) was initiated.
Postoperative TTE showed a free RA. In the six days
after surgery, the patient's lab tests revealed leukocytosis
(15x103/mmc) and a CPR (C-reactive protein) of 199.2
mg/l. Antibiotics were started (Ceftazidime and Levofloxacin) with
the remission of the inflammatory syndrome and a clinically stable
patient. After consultation with the nephrology department, it was
decided that after every HD session 1 g of Ceftazidime and 250 mg
of Levofloxacin would be administrated. The patient was discharged
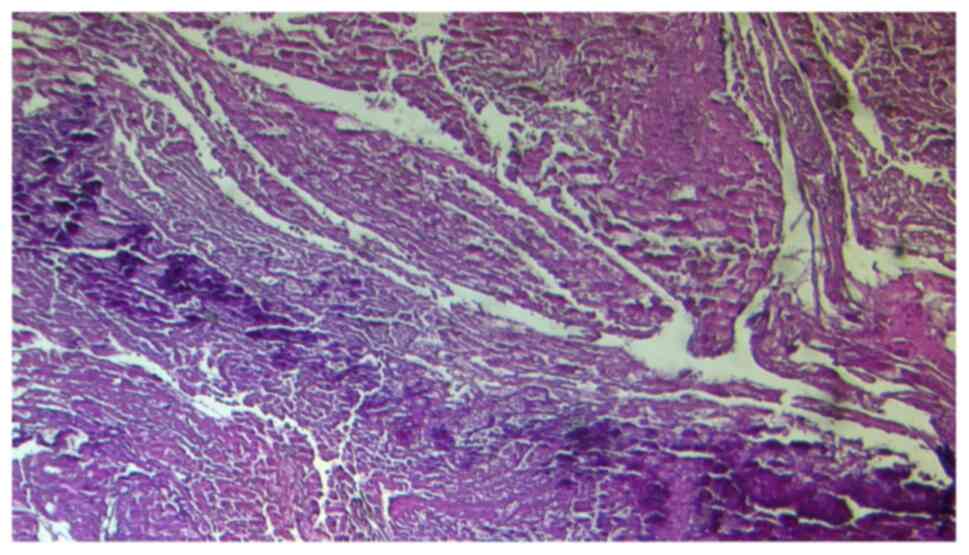
on the 14th postoperative day. After three weeks, the pathology
exam confirmed that the RAM was a recent clot with variable amounts
of fibrin (Fig. 4). Three months
later, a new long-term HD catheter was placed in the left internal
jugular vein, followed by removal of the left femoral catheter.
Control TEE was performed at 3 and 6 months, which revealed a free
RA. The patient was symptom-free and was kept on anticoagulation
treatment with Acenocoumarol.
Discussion
The differential diagnosis of RAMs remains a
challenge, despite having new imaging techniques. Cardiac mass
detection is based on the use of echocardiography, computed
tomography and cardiac MRI, with echocardiography being the primary
choice (1,9). Right atrial myxomas can be hard to
differentiate from right atrial thrombus (10), as was evident in this case report.
The risk of intracardiac thrombosis is increased for patients who
have a history of atrial fibrillation or indwelling catheters in
the RA (11,12). Echocardiographic features, used to
differentiate atrial myxoma from thrombus, are represented by an
irregular shape of the thrombus, immobile mass with a broad base
attached to the posterior atrial wall in case of in situ
thrombi and a spherical, ovoid mobile mass in case of secondary
thrombi. Large mobile thrombi may appear to have no attachment to
the atrial wall and they may be mistaken for myxoma (13,14).
CRAT is a rare complication, but it is associated
with a high mortality rate. Furthermore, there are no clear
guidelines for the treatment of CRAT (6). Thus, it is a decision made on
imagistic findings correlated with clinical findings. Right atrial
thrombi can develop from venous emboli, they may develop under
low-flow conditions, in situ (3) or from around foreign bodies in the
right heart (6). In this case, the
patient had no history of thrombosis, he had documented heparin
usage at HD and no primary cardiac disease was identified on the
echocardiography which could lead to cardiac thrombus formation.
These findings made the diagnosis even more challenging.
Subsequently, it was of the utmost importance to correlate the
imagistic and clinical findings, to establish a therapeutic
attitude. The size of the mass and the risk of pulmonary embolism
are important markers that favor the surgical procedure. In case of
a thrombus, Hussain et al postulated that clot dimensions
have a negative influence on the medical treatment (6). It remains unclear which is the best
therapeutic approach, in particular for patients on HD for whom
anticoagulation can be challenging. According to Elliott et
al warfarin in these patients is linked with a high bleeding
risk. A systematic review with 177 patients reported that all
individuals that did not receive any treatment, died (15). An important aspect is related to the
removal of the atrial catheter, in particular, the timing of
extraction. In this patient it was deemed appropriate to remove it
in a two-stage procedure and by doing so, minimizing the risks of
thrombus displacement. Differentiating RAM still poses
difficulties, especially in patients with CVC, and no history of
thrombosis, in whom echocardiographic and clinical findings do not
support a certain diagnosis. CRAT should always be considered in
patients with a RAM and long-term CVC. According to the
abovementioned, we consider that surgical removal of big atrial
masses, is the preferred treatment.
The diagnosis of a RAM can pose a dilemma despite
all the investigations available. In this case, preoperative
investigations could not distinguish between myxoma and thrombus,
and the exact diagnosis was made postoperatively. Right atrial
thrombus should be taken into consideration in the differential
diagnosis of RAM, particularly in the presence of a CVC and
thrombosis risk factors and in the absence of surgical
contraindications should undergo surgical resection.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
OS, RD, RCG, VAI performed surgical procedures. AT,
AC, LR, NB, and ABS reviewed literature data. AT, LR, CD, and CS
conducted the preoperative investigation of the patient. IB, NB,
and OS prepared the draft of the article. VAI was the advisor for
the surgical procedures. OS, NB, and VAI reviewed the final version
of the article. All authors have read and approved the final
version of the article.
Patient consent for publication
Obtained and signed by the patient on
03.02.2020.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Wang J, Wang G, Bi X, Zhang R and Liu C:
An unusual presentation of prominent crista terminalis mimicking a
right atrial mass: A case report. BMC Cardiovasc Disord.
18(210)2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
McAllister HA Jr, Hall RJ and Cooley DA:
Tumors of the heart and pericardium. Curr Probl Cardiol. 24:57–116.
1999.PubMed/NCBI
|
|
3
|
Cianciulli TF, Saccheri MC, Redruello HJ,
Cosarinsky LA, Celano L, Trila CS, Parisi CE and Prezioso HA: Right
atrial thrombus mimicking myxoma with pulmonary embolism in a
patient with systemic lupus erythematosus and secondary
antiphospholipid syndrome. Tex Heart Inst J. 35:454–457.
2008.PubMed/NCBI
|
|
4
|
Iliescu VA, Dorobantu LF, Stiru O,
Iosifescu AG, Coman I, Marin S and Filipescu D: Second recurrence
of cardiac myxoma 7 years after the initial operation. Chirurgia
(Bucur). 103:239–241. 2008.PubMed/NCBI(In Romanian).
|
|
5
|
Gosev I, Paic F, Duric Z, Gosev M, Ivcevic
S, Jakus FB and Biocina B: Cardiac myxoma the great imitators:
Comprehensive histopathological and molecular approach. Int J
Cardiol. 164:7–20. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Hussain N, Shattuck PE, Senussi MH,
Velasquez Kho E, Mohammedabdul M, Sanghavi DK, Mustafa U,
Balavenkataraman A and Obradovic DM: Large right atrial thrombus
associated with central venous catheter requiring open heart
surgery. Case Rep Med. 2012(501303)2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
van Laecke S, Dhondt A, de Sutter J and
Vanholder R: Right atrial thrombus in an asymptomatic hemodialysis
patient with malfunctioning catheter and patent foramen ovale.
Hemodial Int. 9:236–240. 2005.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Dorobantu LF, Stiru O, Prodea A, Cioranu
R, Georgescu A, Filipescu D and Iliescu VA: Unique case of primary
malignant fibrous histiocytoma of the right ventricle with
moderator band involvement. Heart Surg Forum. 14:E245–E248.
2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Catez E, Tatnga Ngatcha V and Catez S: An
unusual right atrial mass: Case report and review of the
literature. Acta Cardiol. 65:477–480. 2010.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Matsukuma S, Yamaguchi H and Hamawaki M:
Floating and entangled long right atrial thrombus mimicking myxoma.
J Echocardiogr. 8:146–147. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kingdon EJ, Holt SG, Davar J, Pennell D,
Baillod RA, Burns A, Sweny P and Davenport A: Atrial thrombus and
central venous dialysis catheters. Am J Kidney Dis. 38:631–639.
2001.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zhao C, Sathya B, Nadal Rios R, Arai AE,
Brofferio A, Thein SL and Ruhl AP: Catheter-related right atrial
thrombus in sickle cell disease. Clin Case Rep. 5:1898–1900.
2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Nemani L and Killi S: Right atrial
thrombus mimicking myxoma. Indian J Cardiovasc Dis Women-WINCARS.
2:86–90. 2017.
|
|
14
|
Yamane Y, Morimoto H, Okubo S, Koshiyama H
and Mukai S: Right atrial calcified ball thrombus mimicking a
myxoma. Heart Lung Circ. 25:e21–e23. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Elliott MJ, Zimmerman D and Holden RM:
Warfarin anticoagulation in hemodialysis patients: A systematic
review of bleeding rates. Am J Kidney Dis. 50:433–440.
2007.PubMed/NCBI View Article : Google Scholar
|