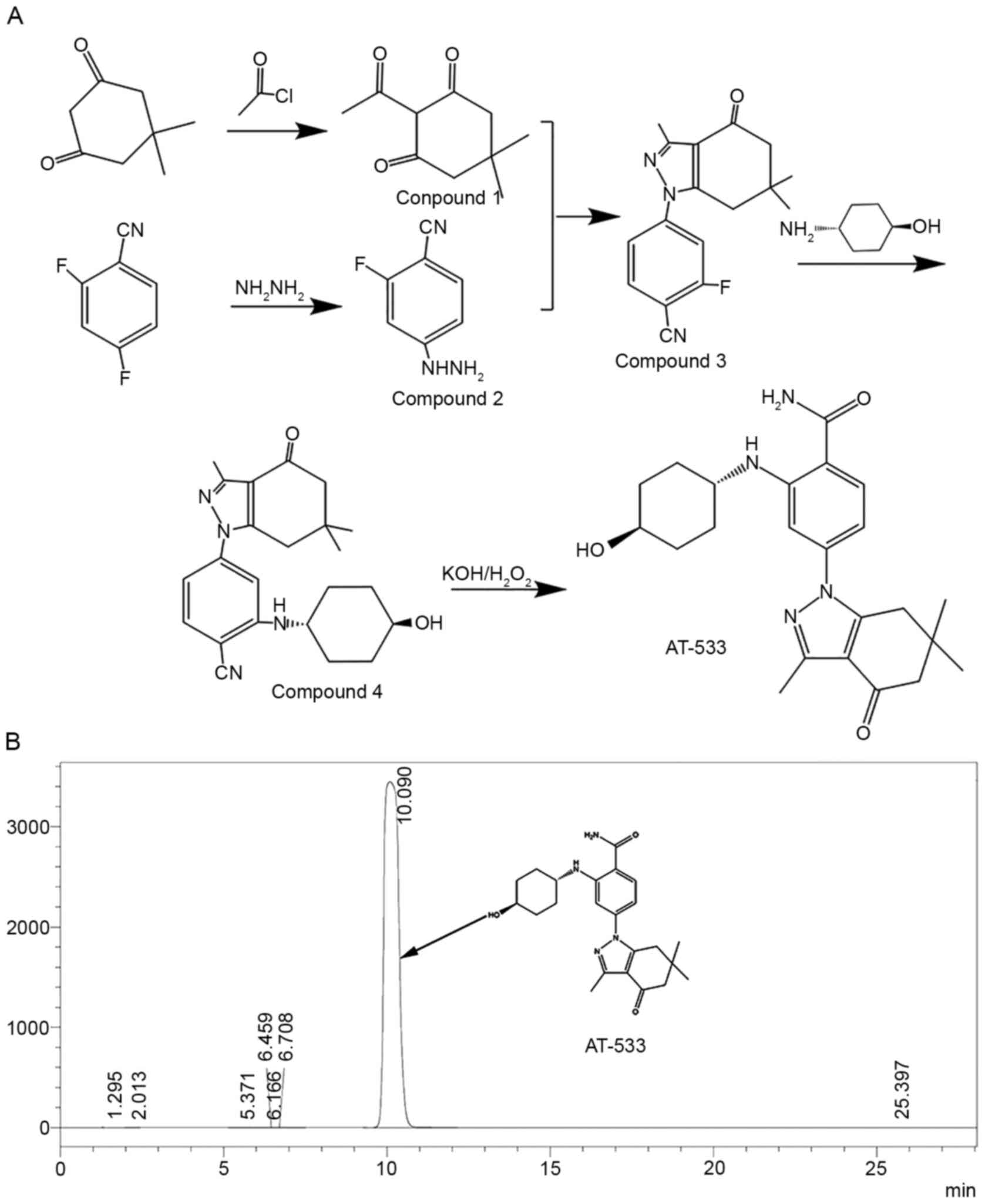
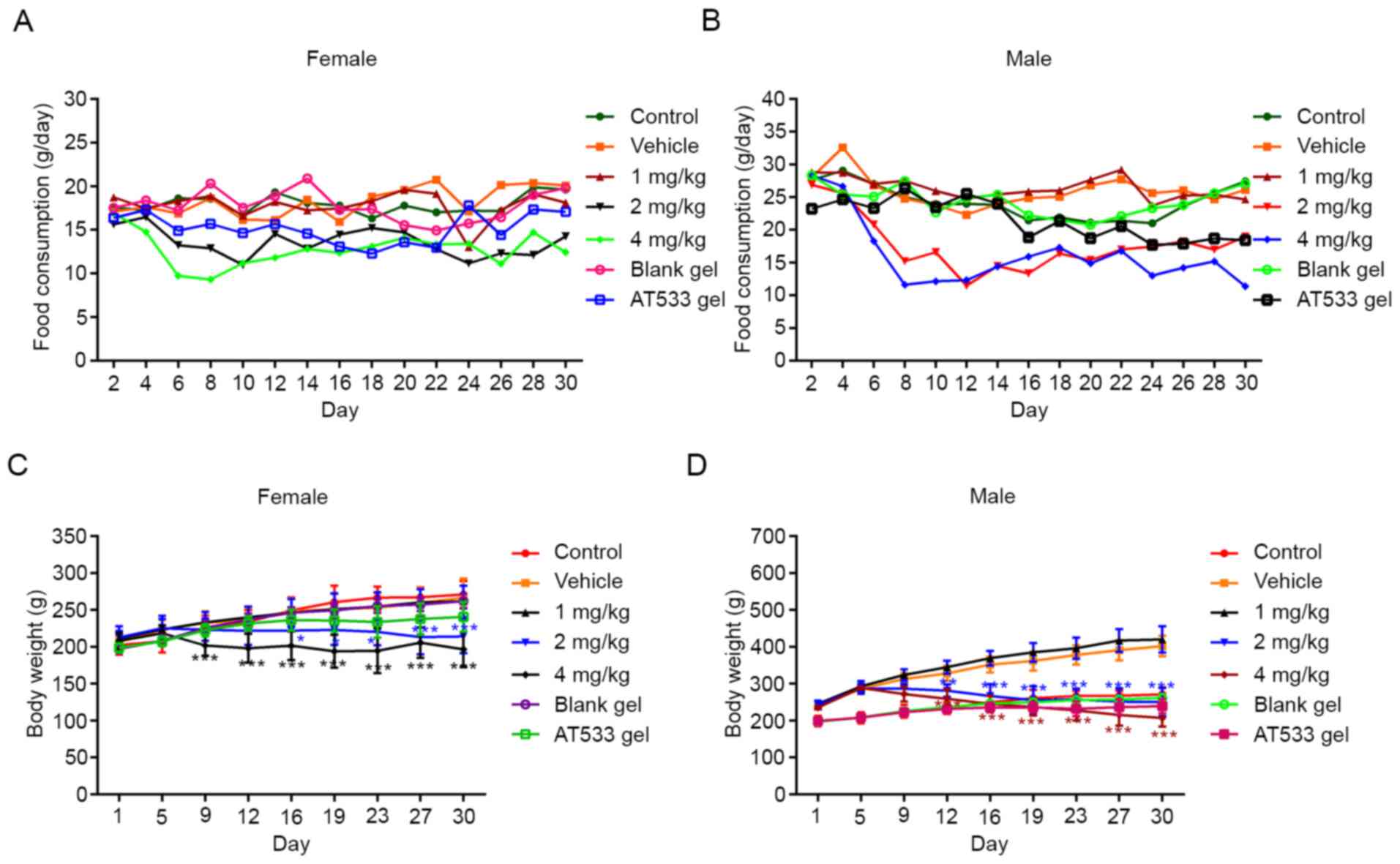
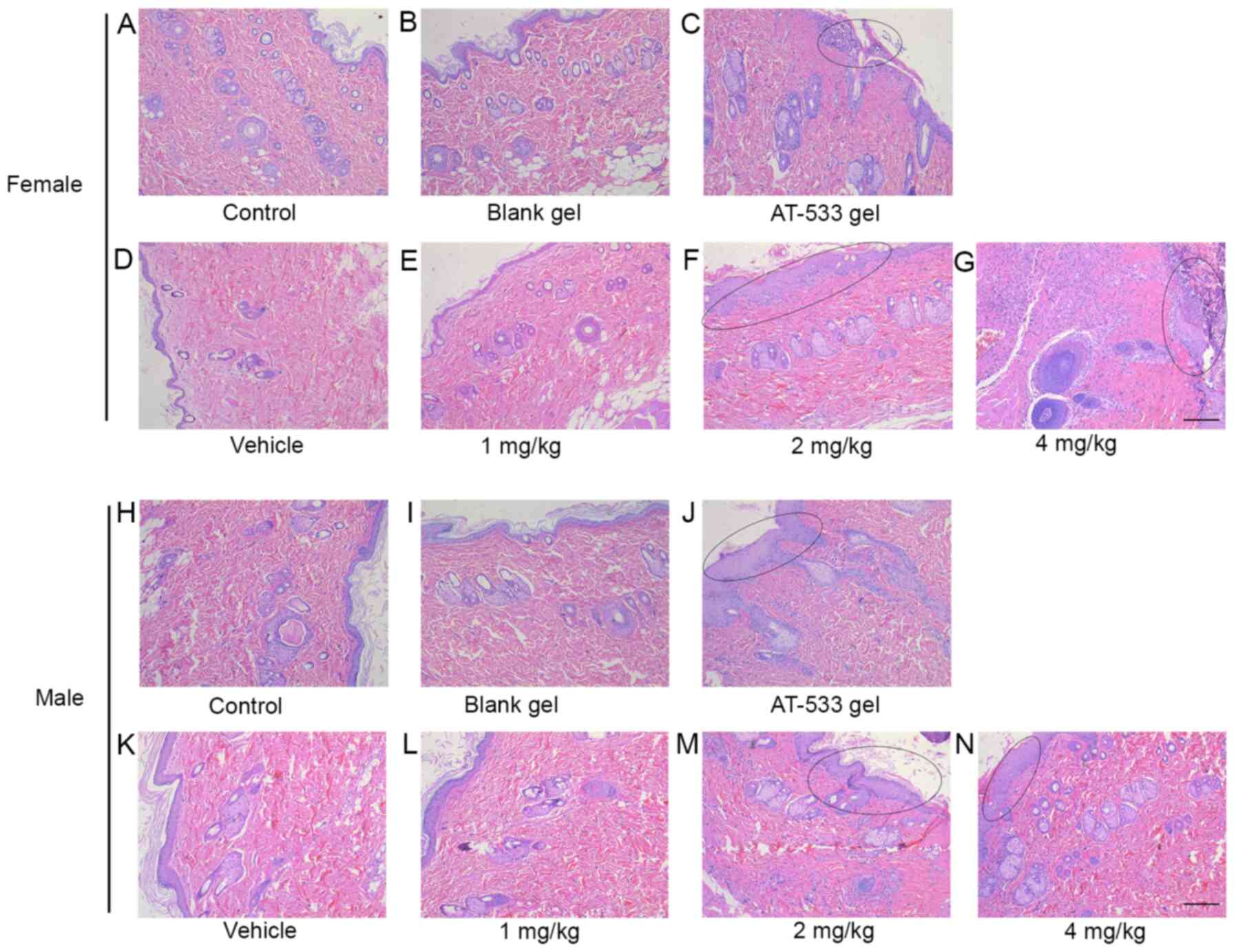
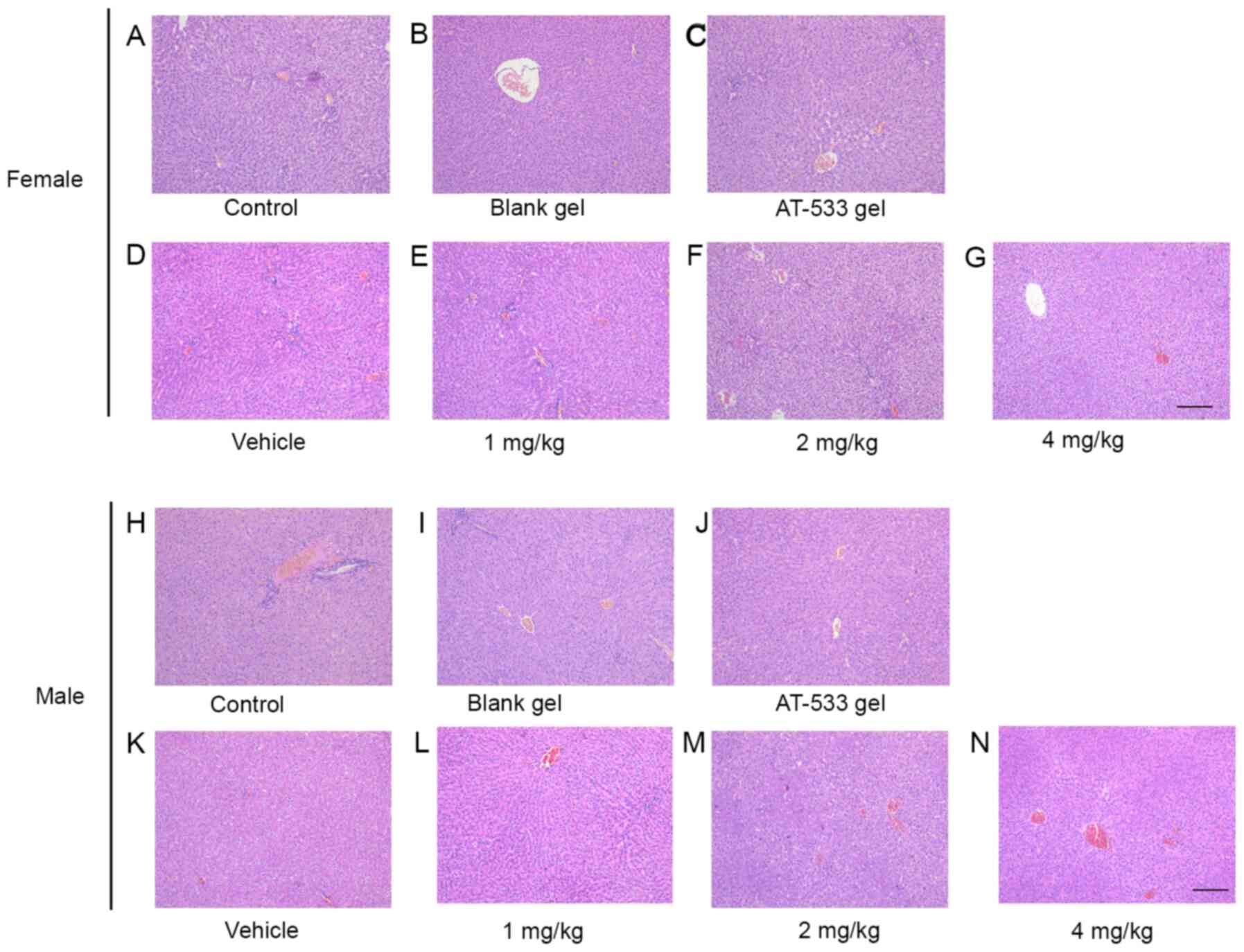
|
1
|
Pearl LH and Prodromou C: Structure and
mechanism of the Hsp90 molecular chaperone machinery. Annu Rev
Biochem. 75:271–294. 2006.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Goetz MP, Toft DO, Ames MM and Erlichman
C: The Hsp90 chaperone complex as a novel target for cancer
therapy. Ann Oncol. 14:1169–1176. 2003.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zininga T, Ramatsui L and Shonhai A: Heat
shock proteins as immunomodulants. Molecules.
23(2846)2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Whitesell L and Lindquist SL: HSP90 and
the chaperoning of cancer. Nat Rev Cancer. 5:761–772.
2005.PubMed/NCBI View
Article : Google Scholar
|
|
5
|
Hoter A, El-Sabban ME and Naim HY: The
HSP90 family: Structure, regulation, function, and implications in
health and disease. Int J Mol Sci. 19(2560)2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Workman P: Pharmacogenomics in cancer drug
discovery and development: Inhibitors of the Hsp90 molecular
chaperone. Cancer Detect Prev. 26:405–410. 2002.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Schulte TW, Akinaga S, Soga S, Sullivan W,
Stensgard B, Toft D and Neckers LM: Antibiotic radicicol binds to
the N-terminal domain of Hsp90 and shares important biologic
activities with geldanamycin. Cell Stress Chaperones. 3:100–108.
1998.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Vilenchik M, Solit D, Basso A, Huezo H,
Lucas B, He H, Rosen N, Spampinato C, Modrich P and Chiosis G:
Targeting wide-range oncogenic transformation via PU24FCl, a
specific inhibitor of tumor Hsp90. Chem Biol. 11:787–797.
2004.PubMed/NCBI View Article : Google Scholar
|
|
9
|
He H, Zatorska D, Kim J, Aguirre J,
Llauger L, She Y, Wu N, Immormino RM, Gewirth DT and Chiosis G:
Identification of potent water soluble purine-scaffold inhibitors
of the heat shock protein 90. J Med Chem. 49:381–390.
2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Llauger L, He H, Kim J, Aguirre J, Rosen
N, Peters U, Davies P and Chiosis G: Evaluation of 8-arylsulfanyl,
8-arylsulfoxyl, and 8-arylsulfonyl adenine derivatives as
inhibitors of the heat shock protein 90. J Med Chem. 48:2892–2905.
2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Kasibhatla SR, Hong K, Biamonte MA, Busch
DJ, Karjian PL, Sensintaffar JL, Kamal A, Lough RE, Brekken J,
Lundgren K, et al: Rationally designed high-affinity
2-amino-6-halopurine heat shock protein 90 inhibitors that exhibit
potent antitumor activity. J Med Chem. 50:2767–2778.
2007.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Pearl LH: Review: The HSP90 molecular
chaperone-an enigmatic ATPase. Biopolymers. 105:594–607.
2016.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Prodromou C and Pearl LH: Structure and
functional relationships of Hsp90. Curr Cancer Drug Targets.
3:301–323. 2003.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Huang KH, Veal JM, Fadden RP, Rice JW,
Eaves J, Strachan JP, Barabasz AF, Foley BE, Barta TE, Ma W, et al:
Discovery of novel 2-aminobenzamide inhibitors of heat shock
protein 90 as potent, selective and orally active antitumor agents.
J Med Chem. 52:4288–4305. 2009.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Barta TE, Veal JM, Rice JW, Partridge JM,
Fadden RP, Ma W, Jenks M, Geng L, Hanson GJ, Huang KH, et al:
Discovery of benzamide tetrahydro-4H-carbazol-4-ones as novel small
molecule inhibitors of Hsp90. Bioorg Med Chem Lett. 18:3517–3521.
2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang S, Wang X, Du Z, Liu Y, Huang D,
Zheng K, Liu K, Zhang Y, Zhong X and Wang Y: SNX-25a, a novel Hsp90
inhibitor, inhibited human cancer growth more potently than 17-AAG.
Biochem Biophys Res Commun. 450:73–80. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Li F, Jin F and Wang Y, Zheng D, Liu J,
Zhang Z, Wang R, Dong D, Zheng K and Wang Y: Hsp90 inhibitor AT-533
blocks HSV-1 nuclear egress and assembly. J Biochem. 164:397–406.
2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wang Y, Wang R, Li F and Wang Y, Zhang Z,
Wang Q, Ren Z, Jin F, Kitazato K and Wang Y: Heat-shock protein 90α
is involved in maintaining the stability of VP16 and VP16-mediated
transactivation of α genes from herpes simplex virus-1. Mol Med.
24(65)2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Xiang YF, Qian CW, Xing GW, Hao J, Xia M
and Wang YF: Anti-herpes simplex virus efficacies of
2-aminobenzamide derivatives as novel HSP90 inhibitors. Bioorg Med
Chem Lett. 22:4703–4706. 2012.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Li F, Song X, Su G and Wang Y, Wang Z,
Qing S, Jia J and Wang Y, Huang L, Zheng K and Wang Y: AT-533, a
Hsp90 inhibitor, attenuates HSV-1-induced inflammation. Biochem
Pharmacol. 166:82–92. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang X, Cheng Y, Zhou Q, Huang H, Dong Y,
Yang Y, Zhao M and He Q: The effect of Chinese traditional medicine
huaiqihuang (HQH) on the protection of nephropathy. Oxid Med Cell
Longev. 2020(2153912)2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
SFDA: Technical guidelines of chronic
toxicity for chemical drugs. SFDA Guidelines No. [H]
GPT2-12005.
|
|
23
|
Chinese Pharmacopoeia Commission: Chinese
pharmacopoeia. China Medical Science and Technology Press, Beijing,
pp291-353, 2015.
|
|
24
|
Li CL, Shu XJ, Chen XQ, Liu YW, Yi HL and
Ma BM: Determination and comparison of blood physiological and
biochemical indicators in SPF SD rats of different ages and
genders. J Jianghan Univ (Nat Sci Ed). 44:58–63. 2016.
|
|
25
|
Wang R, Wen XT, Wang H, Tang JL, Guan B
and Zeng YL: Establishment of blood routine reference interval in
SD rats. Med Equip. 28:11–15. 2015.
|
|
26
|
Wang LM, Wang YH, Han XF, Yang RH and Cao
X: Determination and analysis of blood biochemical indicators in SD
rats of different ages and genders. Heilongjiang Anim Sci Vet Med.
214-216:268–269. 2018.
|
|
27
|
He XY, Wang H, Tang JL, Guan B, Zeng YL
and Shi JF: Preliminary establishment of normal interval of blood
biochemical indicators in SD rats. Med Equip. 28:13–15. 2015.
|
|
28
|
Dong YS, Ying JY, Chen C, Zhai WS, Yuan
BL, Gao XX, Ding RG and Wang HM: Establishment and application of
the normal reference values of organ masses and organ/body
coefficients in SD rats. Mil Med Sci. 36:351–353. 2012.
|
|
29
|
Liu YE, Yao BY, Yang DH and Wang J:
Analysis on organ weights and their changes trend in Sprague Dawley
rats with different ages. Chin J Comp Med. 22:22–27. 2012.
|
|
30
|
SFDA: Technical guidelines of acute
toxicity testing for chemical drugs. SFDA Guidelines No. [H]
GPT1-12005.
|
|
31
|
Bigoniya P, Sahu T and Tiwari V:
Hematological and biochemical effects of sub-chronic artesunate
exposure in rats. Toxicol Rep. 2:280–288. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Tan YJ, Ren YS, Gao L, Li LF, Cui LJ, Li
B, Li X, Yang J, Wang MZ, Lv YY, et al: 28-Day oral chronic
toxicity study of arctigenin in rats. Front Pharmacol.
9(1077)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Yao Q, Sun R, Bao S, Chen R and Kou L:
Bilirubin protects transplanted islets by targeting ferroptosis.
Front Pharmacol. 11(907)2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Maher PA: Using plants as a source of
potential therapeutics for the treatment of Alzheimer's disease.
Yale J Biol Med. 93:365–373. 2020.PubMed/NCBI
|
|
35
|
Charoonratana T, Puntarat J,
Vinyoocharoenkul S, Sudsai T and Bunluepuech K: Innocuousness of a
polyherbal formulation: A case study using a traditional Thai
antihypertensive herbal recipe in rodents. Food Chem Toxicol.
112:458–465. 2018.PubMed/NCBI View Article : Google Scholar
|