1. Introduction
Diabetic cardiomyopathy (DCM) is widespread, as is
the necrosis of the myocardium caused by diabetes mellitus (DM),
and leads to cardiac microangiopathy and myocardial metabolic
disorders (1). Early diastolic
dysfunction is usually characterized by decreased myocardial
compliance and blocked diastolic filling (2). Late diastolic dysfunction is mainly
manifested as congestive heart failure (HF) (3). DCM is one of the main complications of
DM, differing from hypertension and other types of cardiovascular
disease, which demonstrates one of the highest incidence rates of
all cardiovascular diseases with an incidence rate of 16.9% in
China (4). In fact, DCM accounts
for >80% of the deaths of diabetic patients (5,6).
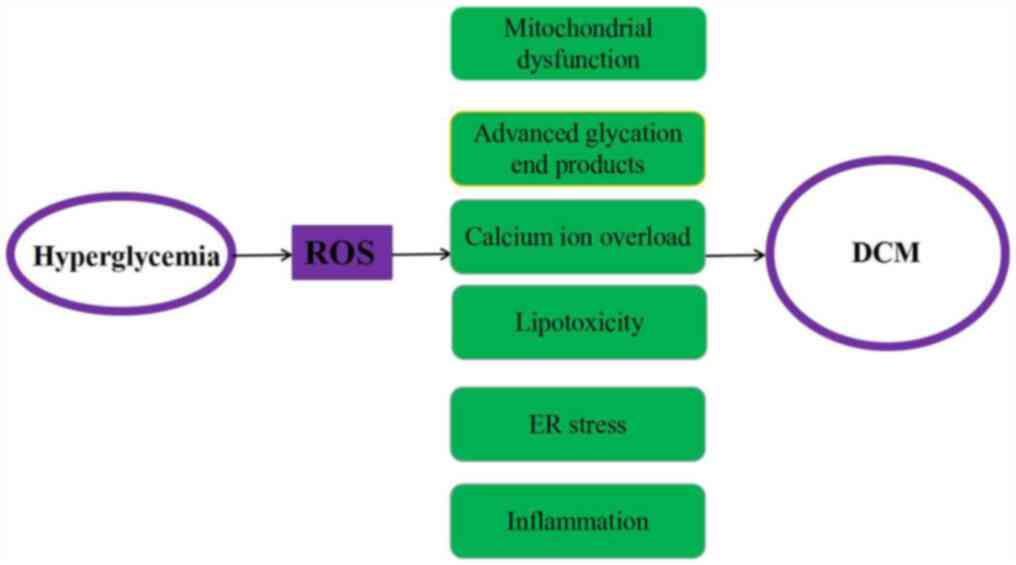
A large number of studies have reported that the
pathogenesis of DCM primarily involves mitochondrial dysfunction,
impaired intracellular calcium regulation, the accumulation of
advanced glycation end products in the heart, abnormal cell
metabolism and endoplasmic reticulum stress (2,7).
Mitochondrial dysfunction has been discovered to lead to the
excessive production of reactive oxygen species (ROS), which in
turn can promote the opening of mitochondrial permeability
transition pores (mPTP), reduce the mitochondrial membrane
potential and impede the respiratory transmission chain (8). Impaired intracellular calcium
regulation has been identified to lead to impaired cardiac
contractility, while the accumulation of advanced glycation end
products in the heart was discovered to lead to a buildup of
extracellular matrix, which in turn leads to diastolic dysfunction
and eventually to functional failure (9). In addition, abnormal cell metabolism
was shown to lead to the accumulation of toxic lipids in the heart,
and endoplasmic reticulum stress is known to mediate cell
apoptosis. It is also well known that metabolic disorders cause
subcellular inflammation of the heart and inflammation is an
important pathogenic feature of DM (Fig. 1) (10).
These pathologies are mainly caused by
hyperglycemia, hyperlipidemia and inflammation, which can stimulate
the production of ROS or reactive nitrogen species (11). ROS are small, highly reactive
molecules, also known as free radicals, which serve important roles
in pathology and physiology. The role of the heart is to provide
sufficient blood flow to organs and tissues to supply them with
oxygen and various nutrients. The heart has high energy
requirements and is prone to oxidative damage caused by
physiological processes (12). The
heart needs more oxygen than other organs and consumes more energy
(6). It has been reported that
oxidative stress served an important role in various types of
cardiac disorder (13). Oxidative
stress is a negative condition caused by the production of free
radicals, and manifests as an imbalance of oxidation and
antioxidant effects in the body (14). The higher the levels of ROS in the
heart, the higher the amount of oxidative stress produced (15). ROS is considered to be responsible
for systolic and endothelial dysfunction, cardiac cell apoptosis
and necrosis (16). Therefore, the
reduction of oxidative stress is an attractive target for the
treatment of cardiac diseases.
Nuclear factor-erythroid-2-related factor 2 (NRF2)
is expressed in a wide range of tissues and organs, including the
heart, brain, liver, kidney and skin (17). A large number of reports have
indicated that NRF2 signaling served important roles in processes,
such as embryonic development, oxidative stress and
ischemia/reperfusion injury (IRI) (14). NRF2 was also reported to regulate
the clearance of free radicals and lipid homeostasis (18). Therefore, NRF2/antioxidant response
element (ARE) signaling has become an attractive target for the
treatment of DCM. The NRF2 signaling pathway is reportedly involved
in DCM via the transcriptional regulation of other signaling
pathways, including PI3K/AKT and JAK/STAT (19). The present review aimed to provide a
comprehensive discussion of the current understanding of the
regulation of NRF2-mediated signaling in the development of
DCM.
2. The NRF2/Kelch-like ECH-associated
protein 1 (Keap1)/ARE signaling pathway
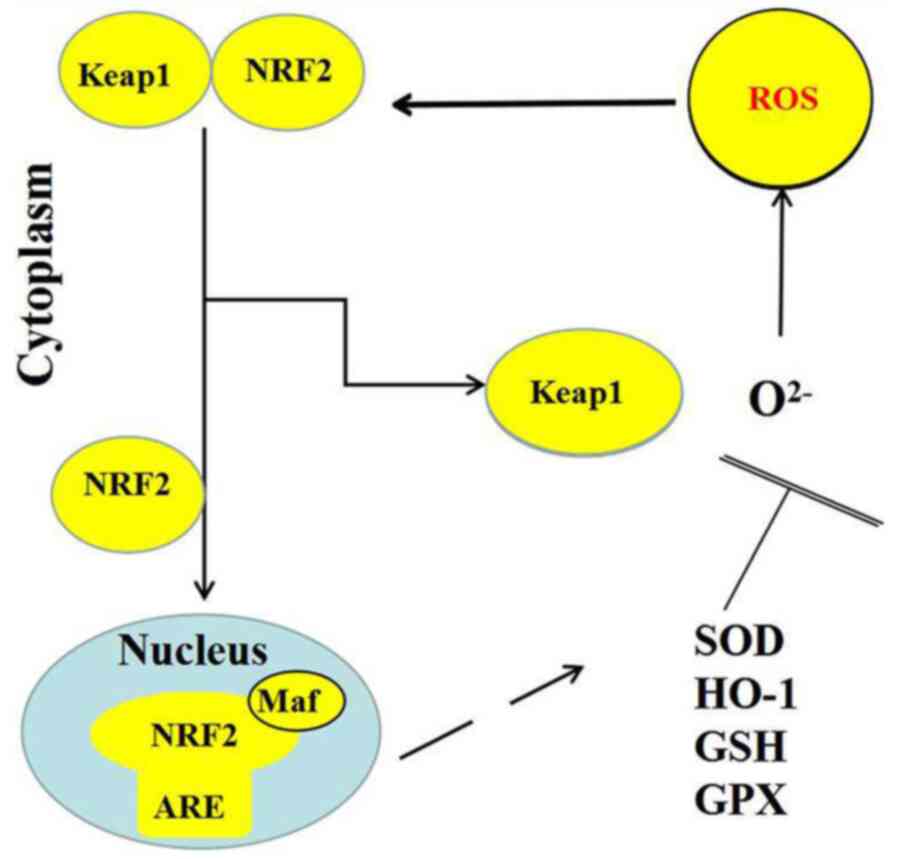
Under the appropriate stimulation by physical and
chemical factors, including inflammation, trauma and fever, NRF2
dissociates from Keap1, which permits the levels of NRF2
degradation to decrease and the simultaneous synthesis of NRF2 to
increase, which subsequently facilitates NRF2 entering the nucleus,
where it specifically recognizes and binds to the core sequence of
AREs (20). As a result of this
series of actions, multiple downstream antioxidants are activated,
where the expression levels of inflammatory proteins and
detoxification enzymes are upregulated (21). The transcription of these genes,
including superoxide dismutase (SOD), GSH, GPX and heme oxygenase
(HO-1), has been identified to suppress ROS production and reduce
oxidative stress (Fig. 2) (22).
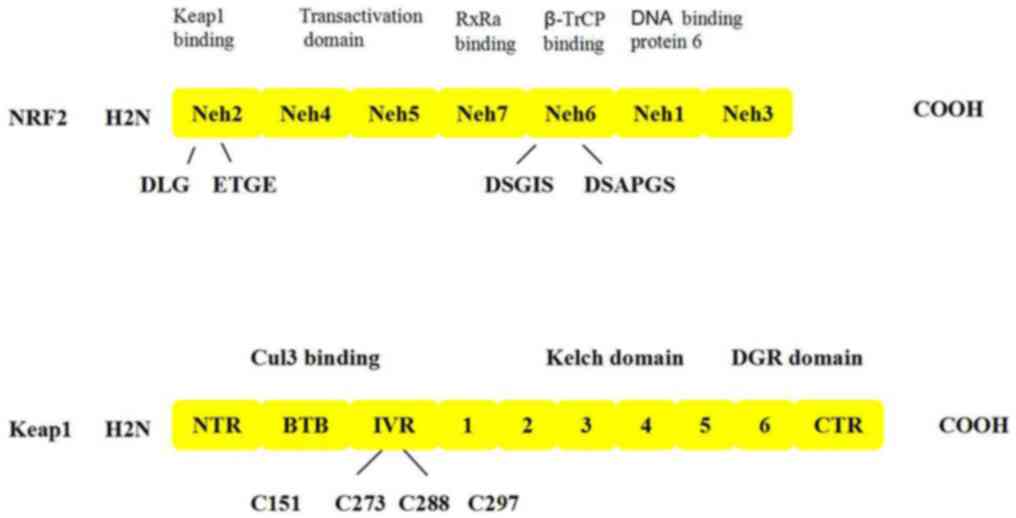
NRF2, which contains 589 amino acids, is a basic
leucine zipper (bZIP) transcription factor belonging to the Cap ‘n’
Collar family that contains seven functional domains,
Neh1-Neh7(23). These structures
serve an important role in regulating the stability of
NRF2(24). The bZIP structure of
the Nehl region can bind to the small Maf protein to form a
heterodimer, which subsequently binds to the DNA molecule through
the ARE (25). Neh2 is the region
in which NRF2 binds to Keap1; the region involves the 69 to 84th
amino acids of NRF2 (a short peptide of 16 amino acids), which
contains an ETGE motif and a DLG motif binding site that binds to
Keap1(26). The Neh3 domain is
located at the carboxy terminus and is highly conserved (27). The agonist chromatin helicase DNA
binding protein 6 binds to the Neh3 domain and upregulates NRF2
target gene expression (Fig. 3)
(28). The Neh4 and Neh5 domains,
which are related to the 596 and 599 amino acids of NRF2, are
involved in the transcriptional activation of the NRF2 target gene
after binding to the coactivator cAMP-response element binding
protein (CREB) (29). The Neh6
region contains a large number of serine residues, which form
redox-insensitive regions. Under oxidative stress conditions, the
degradation of Nrf2 is associated with Neh6, such that Neh6 mediate
B-TrCP-dependent ubiquitination of the NRF2 transcription factor so
that NRF2 can be degraded (30).
Finally, the Neh7 domain can inhibit NRF2 by binding to the
retinoic X receptor α (31).
Keap1 is an important factor regulating the NRF2
response and a central regulator of cellular oxidative stress.
Keap1 contains five domains: i) An N-terminal region domain; ii) a
bric á brac (BTB) dimerization domain; iii) an intervening region
(IVR) domain; iv) a theme containing the six Kelch motifs; and v) a
C-terminal region domain (32).
BTB, which is an evolutionarily conserved motif involved in
protein-protein interactions found in actin-binding proteins and
zinc finger transcription factors, usually forms a dimer with other
BTB regions; this domain is required for Keap1 to dissociate from
NRF2 and prevent phase II gene transcription (33). The IVR domain is rich in cysteine
and contains the most active cysteine residue of Keapl; it is a
functional regulatory region for the entire protein, which is
involved in the reactions of electrophilic compounds and oxidants,
and also participates in the formation of ubiquitination linkages
to stabilize NRF2(31). The DGR
domain contains six bis-glycine repetitive sequences or six Kelch
motifs; the repetitive Kelch motifs form six classical p-helices
with multiple potential protein binding sites for Keapl to bind to
the Neh2 region, and this is where Keapl also binds to cytoplasmic
actin (34).
The ARE is the core protector of cytoprotective
proteins, representing an important antioxidant component of the
human body (35). Under reactive
chemical pressure, ARE cis-acting elements function primarily at
the transcriptional level to regulate the expression levels of
numerous cytoprotective enzymes, including SOD and HO-1(36). The ARE has important structural and
biological characteristics and demonstrates a unique ability to
respond to oxidative stress (37).
It reacts not only to H2O2, but also to
specific chemical compounds, and has the ability to conduct redox
cycles or produce reactive or electrophilic intermediates (38). A number of compounds have a tendency
to react with the sulfhydryl groups, such as diethyl maleate,
dithiothiones and isothiocyanates, which can potently induce ARE
activity (39,40).
NRF2 is a key regulator of antioxidant reactions and
has been demonstrated to regulate both oxidative stress and
inflammation (41,42). There are two types of antioxidant
systems in the body: One is the enzyme antioxidant system, which
includes superoxide, SOD, catalase and glutathione S-transferase;
the other system is a non-enzymatic antioxidant system, involving,
for example, glutathione (43).
NRF2/Keap1 signaling serves an important role in the maintenance of
intracellular redox homeostasis, in addition to serving a vital
role in a variety of cell types and organs in different types of
oxidative damage-related cardiac disease, including myocardial
ischemic disease, HF and cardiac hypertrophy (44). Numerous NRF2 activators are
plant-derived phytochemicals and natural products, such as curcumin
(CUR), sulforaphane (SFN) and resveratrol (RES) compounds (45), although some synthesized NRF2
activators have been identified, including hydrogen sulfate and
4-hydroxynonenal lipoic acid (46).
These chemoprotective compounds protect cells from oxidative stress
by mediating the NRF2 defense response, and activating phase II
detoxification enzymes, transporters and antioxidants (47).
NRF2/ARE signaling has been associated with various
major pathophysiological conditions, including hypoxia, ischemia,
fibrosis and apoptosis (48). It is
also associated with numerous signaling pathways (49). There is increasing evidence to
suggest that NRF2 and PI3K/AKT are important in oxidative stress
injury, where NRF2 is involved in several signaling pathways,
including the NF-κB and other cytokine signaling pathways (50). Therefore, NRF2/ARE signaling is
known to serve an important role in several pathological
conditions.
3. NRF2 signaling and its association with
DCM complications
Ischemic heart disease
Ischemic heart disease is a serious condition, which
among all patients with heart disease in China the death rate can
reach 60% (51). In myocardial IR,
the blood supply to the heart is blocked, following which perfusion
and accompanying reoxygenation resumes (52). IR occurs in acute coronary syndrome,
cardiopulmonary resuscitation, organ transplantation thrombolysis,
coronary bypass and other conditions (53). Calcium and ROS were discovered to be
important molecular species involved in myocardial IRI during
ischemia initiation and reperfusion. Calcium has an important
electrophysiological elements, which enhances myocardial vitality
and participates in heartbeat, whilst ROS plays an important
regulatory role during myocardial ischemia and reperfusion
(39). The mPTP opening is an
important mechanism of myocardial necrosis induced by IRI (22). However, due to the complicated
underlying mechanisms, there are currently no effective methods for
preventing myocardial IRI.
The most critical feature of myocardial IRI
formation is the production of ROS (54). During perfusion in the ischemic
phase, residual oxygen can still reach the tissue and produce ROS
in the myocardium (55). A small
amount of ROS is formed in the early stages of myocardial ischemia,
but levels were found to increase sharply after 20-25 min (56). Myocardial reperfusion is extremely
sensitive to large increases in ROS, which not only damage the
mitochondrial respiratory chain, but also oxidize the mitochondrial
inner membrane cardiolipin and inhibit respiratory chain activity
(57). When ROS levels are
increased, they can also induce lipid peroxidation and
cardiomyocyte oxidative damage, eventually leading to apoptosis
(58).
NRF2 regulates a series of antioxidant enzymes and
cell oxidative stress, and it has been reported that the
overexpression of NRF2 protected against IRI (59). The myocardial infarction (MI) area
was demonstrated to increase, while the degree of cardioprotection
decreased in NRF2 knockout mice following IR (60). Notably, following the activation of
the NRF2 signaling pathway, a large number of compounds can protect
the heart from IR damage (61).
Similarly, reduced NRF2 activity in H9C2 cardiomyoblasts led to a
decrease in cell survival under hypoxic conditions, irrelevant of
whether reoxygenation occurred (62).
It has been suggested that NRF2 activity may be
linked to mitochondrial function (63). NRF2 signaling controlled
mitochondrial ROS production by regulating mitochondrial function,
which indicated that the NRF2 signaling pathway may serve a vital
role in cellular redox homeostasis (64). In clinical terms, this may lead to
improved ischemic tolerance and make organs more resistant to
ischemia (65). Following ischemic
injury, organ transplantation and IR are beneficial for restoring
blood flow (66). Although it is
also worthy to note that several drugs can also be used to prevent
IRI (67), as it is important to
remove ROS from cardiomyocytes, the manipulation of the NRF2/Keap1
signaling pathway may represent a novel strategy for the treatment
of IRI (68).
HF
HF is a pathological process in which the cardiac
pump function is reduced due to myocardial contraction and/or
diastolic dysfunction, resulting in a decrease in cardiac output
and an inability of the heart to meet the metabolic needs of the
body tissues (69). HF is
considered the end point of numerous types of cardiovascular
disease. Persistent, abnormal neurohormonal and mechanical stresses
can lead to HF (70); such
pathological stresses include, hypertension, cardiac fibrosis,
cardiac hypertrophy and cardiomyocyte apoptosis (71). These stresses can alter the
microvascular structure and ventricular dilation, ultimately
leading to cardiac dysfunction and HF (12,38).
However, it is common that the heart remains within the normal
range of ejection fraction (72).
It was previously reported that oxidative stress
accelerated the progression of HF (73). In fact, there is some experimental
evidence to suggest that ROS-induced changes in the structure and
function of the heart can lead to HF, which highlights that
oxidative stress and myocardial function are closely related. NRF2
is known to be associated with the inducible expression of various
antioxidant genes and other cytoprotective phase II detoxifying
enzymes, and it was previously reported that NRF2 participated in
the mechanism of HF (74-76).
In addition, several clinical studies have identified that microRNA
(miRNA/miR)-27a expression levels were upregulated in human HF
(63,67,77).
This is interesting as miR-27a was one of the microRNAs predicted
to be a target of NRF2; however, its mechanism of action remains
unknown (69,78). Therefore, it is important to further
investigate the specific role of NRF2 during HF.
Cardiac remodeling
Cardiac remodeling is an adaptive process of
cardiomyocytes to repeated stress in order to maintain homeostasis
(79). Cardiac remodeling
frequently impairs function at the level of molecules, cells,
tissues and organs (80). Numerous
animal and clinical studies have revealed that cardiac hypertrophy,
myocardial apoptosis and interstitial collagen deposition
eventually led to systolic dysfunction (81-83).
During cardiac remodeling, the loss of
cardiomyocytes has been widely ascribed to necrosis, apoptosis or
autophagy (80); however, fibrosis
can also occur through fibroblast proliferation and extracellular
matrix reorganization (84).
Mitochondrial dysfunction and metabolic abnormalities were also
discovered to contribute to the process of cardiac remodeling
(85,86). In fatigued cardiomyocytes, calcium
uptake is impaired, where calcium efflux becomes dysregulated and
involves components such as sarco/endoplasmic reticulum
calcium-ATPase-2a and the lysine receptor (87). Morphological changes also occur in
the heart, with the shape morphing from elliptical to spherical
(82). Cardiac remodeling also
damages the systolic function of the heart (88).
Cardiac remodeling usually leads to left ventricular
mass hypertrophy and a reduction of ejection fraction (89). It has been previously reported that
ROS activated NRF2 signaling to facilitate different cardiac
remodeling processes (90).
Notably, the activation of the NRF2 pathway using allicin treatment
was observed to prevent the development of cardiac remodeling to
cardiac dysfunction (91).
Oxidative stress was also discovered to be closely related to
cardiac remodeling (92). In
addition, antioxidants have been found to prevent cardiac
remodeling and several studies have revealed that oxidative stress
served a regulatory role in myocardial fibrosis (86,88,93).
Therefore, NRF2 may serve as a good potential target for cardiac
remodeling.
MI
MI is a type of myocardial necrosis caused by the
acute and persistent ischemia and hypoxia of the coronary arteries,
which occurs due to an imbalance between the oxygenated blood
supply and demand (92). Every
year, ~10% of patients with symptoms of acute coronary syndrome are
diagnosed with acute MI worldwide (94). Necrosis was considered to be the
main cause of cardiomyocyte death; however, cell apoptosis also
serves an important role in the process of cell death (95). Although rapid reperfusion therapy
was discovered to reduce infarct size and improve left ventricular
function, reperfusion itself resulted in cell necrosis (96). IRI was also revealed to serve an
important role in oxidative stress and myocardial infarction
(97). During oxidative stress,
large amounts of ROS are produced, which was identified to induce
lipid peroxidation and convert oxidizing proteins to an inactive
state, resulting in DNA damage and the exacerbation of IRI
(11). Therefore, excess ROS should
be eliminated as soon as possible. NRF2 is a well-known antioxidant
and has been discovered to be closely related to MI (51). NRF2/Keap1 pathway is an important
antioxidant defense mechanism, which is closely related to DCM
heart remodeling mediated by oxidative stress (98). It has been reported that after
myocardial infarction (MI), the NRF2 protein in the heart is
downregulated, which leads to a decrease in the antioxidant enzymes
targeted by Nrf2(91). At this
stage, the human body will protect the heart tissue from further
damage, so it will trigger a series of reactions to downregulate
NRF2(96). As the heart tissue
continues to undergo irreversible damage, DCM will increase the
transcription and activation of NRF2(14). Therefore, if a gene target that
blocks Nrf2 is found, it may provide a new method for the treatment
of DCM (96,98).
Cardiac hypertrophy
Cardiac hypertrophy is a relatively slow, but
effective, compensatory function, which occurs mainly under
long-term stress overload (99).
Cardiac hypertrophy includes physiological hypertrophy and
pathological hypertrophy (100).
Physiological hypertrophy occurs mainly in healthy individuals,
pregnant women and individuals pursuing long-term high intensity
training and it is a reversible condition; however, pathological
hypertrophy is a compensatory response (101). Different stimuli can provoke
different types of cardiac hypertrophy, including persistent stress
overload (hypertension), excessive volume overload (arteriovenous
shunt) and genetic mutations (cardiomyopathic hypertrophy)
(102).
Cardiomyocytes can enhance myocardial contractility
to a certain extent under the stimulus of various outside factors,
including myocardial ischemia and septic shock (103). When a stimulus persists, the
compensatory mechanisms of the myocardial cells become
decompensated, and eventually HF develops (80). Cardiac hypertrophy has been strongly
linked to HF and cardiac death, indicating that preventing cardiac
hypertrophy may be of utmost importance (53). Previous studies revealed that
oxidative stress activated cardiac hypertrophy and other pathways
including PI3K/AKT, NRF2/ARE signaling (104). Notably, upon the knockdown of NRF2
expression, angiotensin II-induced cardiac hypertrophy was
exaggerated and the degree of oxidative stress in the heart was
exacerbated (105). Other previous
studies have indicated that endogenous antioxidants, such as HO-1,
protected against cardiac hypertrophy (22,68).
For example, in hypertension model rats, an increase in HO-1
effectively reduced left ventricular hypertrophy (106-108).
Myocarditis
Myocarditis can be caused by viral, bacterial or
parasitic infections, or by noninfectious factors, including trauma
and drugs (41). Therefore,
myocarditis is a common infectious or non-infectious myocardial
immunopathological process (109).
Acute viral myocardial inflammatory injury without clinical
symptoms and chronic myocarditis were discovered to lead to
immune-mediated myocardial damage and dysfunction (110). Importantly, myocarditis is a
precursor lesion of DCM and may develop into DCM. It has been
reported that ROS and oxidative stress were involved in the
pathogenesis of myocarditis (78).
ROS and oxidative stress are associated with innate and adaptive
immunity and tissue repair (73).
Persistent myocardial inflammation causes myocardial
remodeling, which eventually develops into DCM. This leads to the
release of cytokines, causing an inflammatory response. Histamine
was discovered to increase the susceptibility of mice to autoimmune
myocarditis (111). In addition,
the activation of cytokines, such as TGFs, activated the
intracellular signaling protein SMAD cascade, increased profibrotic
factors, pathological fibrosis and myocardial remodeling, and
decreased cardiac function, all of which lead to progressive HF
(112). ROS was also identified to
directly alter cardiac contractility; it has also been associated
with cardiac depression in acute inflammation other than chronic
myocarditis (113).
NRF2 is an important regulator of antioxidant
reactions, which regulate oxidative stress (12). Several natural compounds, such as
CUR, have antioxidant properties (53). CUR was discovered to regulate
specific transcription factors, such as NRF2, which subsequently
regulated free radical scavenging and the expression of lipid
homeostasis (102). The induction
of the activity of antioxidative enzymes has been attempted using
gene therapies and nanotechnologies (39). However, there is a lack of clinical
evidence for this effect (71,114).
The clinical efficacy has not been established in human
myocarditis, therefore, further research remains to be undertaken.
Although additional studies are required, NRF2 may serve a vital
role in myocarditis.
4. Natural substance therapies for DCM
Oxidative stress, and hence antioxidants, have been
illustrated to serve an important role in the pathogenesis of DCM
(82). NRF2 and its downstream
signaling pathways have been reported to have a key role in
preventing DCM and other cardiovascular events induced by high
glucose (69). In addition, it was
demonstrated that natural or synthetic NRF2 activators had a
therapeutic role in DCM model animals (54). Therefore, NRF2 is considered to be a
target for the treatment of DCM.
CUR. CUR is a naturally occurring polyphenol
isolated from the turmeric plant (115); it has been discovered to contain
antioxidants, anti-inflammatory agents and anti-cancer agents, in
addition to exerting anti-diabetic activity (65). In streptozocin (STZ)-induced
diabetic model mice, CUR intervention was identified to reduce
blood glucose levels (116). Using
malondialdehyde and NAD(P)H quinone dehydrogenase 1 (NQO1) as
indicators, CUR was also reported to inhibit myocardial oxidative
stress by upregulating NRF2 in type 1 DM (T1DM) model rats
(45). The upregulation of the
endogenous antioxidant gene NRF2 also inhibited
hyperglycemia-induced inflammation, macrophage infiltration,
diabetic-induced cardiac dysfunction, endoplasmic reticulum stress,
oxidative stress injury and apoptosis (117).
SFN
As an isothiocyanate, SFN is widely found in
cruciferous vegetables, especially broccoli (118). SFN was discovered to be an
activator of the transcription factor NRF2, and it was observed to
reduce the oxidative damage caused by DCM and improved cardiac
function in T1DM model mice (119). SFN was also identified to
effectively prevent cardiac enlargement and cardiac dysfunction in
type 2 DM model mice by upregulating the expression levels of NRF2
and the subsequent expression levels of the downstream genes, HO-1
and NQO1, and significantly reducing the myocardial inflammatory
response, fibrosis and oxidative damage (120).
RES
RES is a polyphenolic substance widely present in
plants, including grape skins, nuts and knotweed (121). It was shown to have antioxidative
stress and antihypertensive effects in T1DM model rats (122). RES was found to attenuate cardiac
dysfunction, cardiac hypertrophy and myocardial fibrosis (123). By detecting various indicators of
oxidative stress in the heart, it was also illustrated that RES
significantly reduced the degree of oxidative stress damage caused
by DM (124). RES also reduced
oxidative stress and inhibited the cardiac hypertrophy and vascular
fibrosis caused by connective tissue growth factor by upregulating
NRF2 expression levels and the transcriptional activity of its
downstream target genes (125).
DMF
DMF is an oral drug used for the treatment of adult
multiple sclerosis and in Europe for the treatment of psoriasis
(126). In both human and animal
experiments, it was demonstrated that DMF effectively activated
NRF2 and promoted its downstream antioxidative stress activity
(127). In mice with T1DM, the
measurements of the antioxidant enzymes, SOD, HO-1 and other
indicators, suggested that DMF significantly inhibited the levels
of oxidative stress in the DCM myocardium by reducing the source of
free radicals and increasing the expression of enzymes and proteins
involved in oxidative stress (128). Another previous study also
revealed that the protective effect of DMF on DCM was related to
the upregulation of NRF2 and the expression of downstream
antioxidant genes in the cardiomyocytes of T1DM model mice
(129).
Rutin
Rutin is a medicinally active ingredient of a
flavonoid plant that exerts strong pharmacological activity and low
toxicity (130). Rutin reportedly
promoted vasodilation, the antagonization of platelet activating
factor, anti-inflammatory and anti-oxidative processes, and the
protection of the pancreas (131).
It was reported that rutin may also reverse diabetic myocardial
injury (78). In STZ-induced
diabetic model mice, the levels of serum myocardial enzymes and
other indicated were recorded, and hematoxylin and eosin and
Masson's trichrome staining were performed, and rutin was
illustrated to reduce the serum myocardial enzyme content, improve
the pathological morphology of myocardial cells, reduce the degree
of fibrosis and alleviate myocardial injury in mice with DCM
(132).
5. miRNAs as a novel drug therapy for
DCM
miRNA is a type of endogenous, non-coding
single-stranded RNA, which participates in numerous physiological
processes, including angiogenesis, metabolism, cell growth,
survival and death, proliferation and differentiation (133). miRNA can negatively regulate gene
expression by promoting the degradation, and inhibiting the
translation, of target RNA (134).
Notably, the dysregulation of miRNA biological mechanisms has been
shown to promote a variety of diseases, including DCM (135). For example, miR-1 was found to be
abundant in the diabetic myocardium, where it promoted ROS
production and the decline in mitochondrial membrane potential,
whilst upregulating Bax expression levels and inhibiting Bcl-2
expression levels, thereby promoting cardiomyocyte apoptosis
(136). Similarly, Dludla et
al (137) suggested that
inhibiting the expression levels of miR-1 had a protective effect
on the myocardium in patients with diabetes.
As one of the important downstream components of
p53-mediated cell signal transduction pathways, miR-34b was
discovered to serve a proapoptotic role in patients with DM
complicated with HF (138,139). It has been reported that p53 is a
target of miR-30c and miE-181a (124). In addition, in another previous
study, the downregulation of miR-30c and miR-181a in DCM was
closely related to the activation of the p53 pathway in
cardiomyocyte apoptosis (140).
During the course of DM, miRNAs have been discovered
to be involved in numerous aspects of DCM pathogenesis, including
apoptosis, fibrosis, hypertrophy, mitochondrial dysfunction and
epigenetic modifications (141).
Therefore, it is hypothesized that miRNAs may serve an important
role in the pathophysiological processes of DCM. Further research
into miRNAs has the potential to provide new perspectives into the
prevention and treatment of DCM.
6. Conclusions
Oxidative stress is known to serve a significant
role in DCM. Therefore, signaling via the NRF2/Keap1/ARE pathway
may represent a novel drug target for the treatment of DCM. In
addition, research into the relationship between miRNA and DCM has
far-reaching clinical significance. Thus, the role of miRNA in the
pathophysiology of DCM must be further clarified to be applied in
clinical practice.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the National Natural
Science Foundation of China (grant no. 00019509) and the Jiangxi
Provincial Natural Science Foundation (grant no. 700207006).
Availability of data and materials
Not applicable.
Authors' contributions
XW and JL wrote the manuscript, reviewed the draft;
and LH was responsible for analyzing the literature and reviewing
drafts of the manuscript. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Schrier RW, Abdallah JG, Weinberger HH and
Abraham WT: Therapy of heart failure. Kidney Int. 57:1418–1425.
2000.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Rafeian-Kopaei M, Setorki M, Doudi M,
Baradaran A and Nasri H: Atherosclerosis: Process, indicators, risk
factors and new hopes. Int J Prev Med. 5:927–946. 2014.PubMed/NCBI
|
|
3
|
Majerczyk M, Choręza P, Mizia-Stec K,
Bożentowicz-Wikarek M, Brzozowska A, Arabzada H, Owczarek AJ,
Szybalska A, Grodzicki T, Więcek A, et al: Plasma level of
retinol-binding protein 4, N-terminal proBNP and renal function in
older patients hospitalized for heart failure. Cardiorenal Med.
8:237–248. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
McGillicuddy FC, Moll HP, Farouk S,
Damrauer SM, Ferran C and Reilly MP: Translational studies of A20
in atherosclerosis and cardiovascular disease. Adv Exp Med Biol.
809:83–101. 2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lindblom R, Ververis K, Tortorella SM and
Karagiannis TC: The early life origin theory in the development of
cardiovascular disease and type 2 diabetes. Mol Biol Rep.
42:791–797. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Eltzschig HK and Eckle T: Ischemia and
reperfusion-from mechanism to translation. Nat Med. 17:1391–1401.
2011.PubMed/NCBI View
Article : Google Scholar
|
|
7
|
Scolletta S and Biagioli BE: Nergetic
myocardial metabolism and oxidative stress: Let's make them our
friends in the fight against heart failure. Biomed Pharmacother.
64:203–207. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Reczek CR and Chandel NS: ROS-dependent
signal transduction. Curr Opin Cell Biol. 33C:8–13. 2014.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ichihara S: The pathological roles of
environmental and redox stresses in cardiovascular diseases.
Environ Health Prev Med. 18:177–184. 2013.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Brahmanaidu P, Sathibabu U and Ganapathy
S: Diabetic cardiomyopathy: Molecular mechanisms, detrimental
effects of conventional treatment, and beneficial effects of
natural therapy. Heart Fail Rev. 24:279–299. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chakraborty S and Ain R: Nitric-oxide
synthase trafficking inducer is a pleiotropic regulator of
endothelial cell function and signaling. J Biol Chem.
292:6600–6620. 2017.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Balakumar P, Singh AP and Singh M: Rodent
models of heart failure. J Pharmacol Toxicol Methods. 56:1–10.
2007.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Furfaro AL, Traverso N, Domenicotti C,
Piras S, Moretta L, Marinari UM, Pronzato MA and Nitti M: The
NRF2/HO-1 axis in cancer cell growth andchemoresistance. Oxid Med
Cell Longev. 2016(1958174)2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Suzuki T and Yamamoto M: Molecular basis
of the Keap1-Nrf2 system. Free Radic Biol Med. 88:93–100.
2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Jay PY, Berul CI, Tanaka M, Ishii M,
Kurachi Y and Izumo S: Cardiac conduction and arrhythmia: Insights
from Nkx2.5 mutations in mouse and humans. Novartis Found Symp.
250:227–238. 2003.PubMed/NCBI
|
|
16
|
Namani A, Li Y, Wang XJ and Tang X:
Modulation of Nrf2 signaling pathway by nuclear receptors:
Implications for cancer. Biochim Biophys Acta. 1843:1875–1885.
2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yamamoto M, Kensler TW and Motohashi H:
The KEAP1-NRF2 system: A thiol-based sensor-effector apparatus for
maintaining redox homeostasis. Physiol Rev. 98:1169–1203.
2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Li W and Kong AN: Molecular mechanisms of
Nrf2-mediated antioxidant response. Mol Carcinog. 48:91–104.
2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Nguyen T, Nioi P and Pickett CB: The
Nrf2-antioxidant response element signaling pathway and its
activation by oxidative stress. J Biol Chem. 284:13291–13295.
2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Corradi D, Callegari S, Maestri R, Benussi
S and Alfieri O: Structural remodeling in atrial fibrillation. Nat
Clin Pract Cardiovasc Med. 5:782–796. 2008.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Saracino MR and Lampe JW: Phytochemical
regulation of UDP-glucuronosyltransferases: Implications for cancer
prevention. Nutr Cancer. 59:121–141. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Niture SK, Kaspar JW and Shen J: Nrf2
signaling and cell survival. Toxicol Appl Pharmacol. 244:37–42.
2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Dhamodharan U, Ponjayanthi B, Sireesh D,
Bhakkiyalakshmi E and Ramkumar KM: Association of single-nucleotide
polymorphisms of the KEAP1 gene with the risk of various human
diseases and its functional impact using in silico analysis.
Pharmacol Res. 137:205–218. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Tian W, Rojo de la Vega M, Schmidlin CJ,
Ooi A and Zhang DD: Kelch-like ECH-associated protein 1 (KEAP1)
differentially regulates nuclear factor erythroid-2-related factors
1 and 2 (NRF1 and NRF2). J Biol Chem. 293:2029–2040.
2018.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mohan S and Gupta D: Crosstalk of
toll-like receptors signaling and Nrf2 pathway for regulation of
inflammation. Biomed Pharmacother. 108:1866–1878. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Niture SK, Khatri R and Jaiswal AK:
Regulation of NRF2-an update. Free Radic Biol Med. 66:36–44.
2014.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Katoh Y, Iida K, Kang MI, Kobayashi A,
Mizukami M, Tong KI, McMahon M, Hayes JD, Itoh K and Yamamoto M:
Evolutionary conserved N-terminal domain of Nrf2 is essential for
the Keap1-mediated degradation of the protein by proteasome. Arch
Biochem Biophys. 433:342–350. 2005.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Baird L, Llères D, Swift S and
Dinkova-Kostova AT: Regulatory flexibility in the Nrf2-mediated
stress response is conferred by conformational cycling of the
Keap1-Nrf2 protein complex. Proc Natl Acad Sci USA.
110:15259–15264. 2013.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Hegedűs K, Nagy P, Gáspári Z and Juhász G:
The putative HORMA domain protein Atg101 dimerizes and is required
for starvation-induced and selective autophagy in
Drosophila. Biomed Res Int. 2014(470482)2014.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Baird L and Dinkova-Kostova AT: The
cytoprotective role of the Keap1-Nrf2 pathway. Arch Toxicol.
85:241–272. 2011.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Zhao Q, Liu Z and Huang B: PEDF improves
cardiac function in rats subjected to myocardial
ischemia/reperfusion injury by inhibiting ROS generation via
PEDF-R. Int J Mol Med. 41:3243–3252. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Sporn MB and Liby KT: NRF2 and cancer: The
good, the bad and the importance of context. Nat Rev Cancer.
12:564–571. 2012.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Cameron BD, Sekhar KR, Ofori M and Freeman
ML: The role of Nrf2 in the response to normal tissue radiation
injury. Radiat Res. 190:99–106. 2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Xiang MJ, Namani A, Wu SJ and Wang XL:
Nrf2: Bane or blessing in cancer? J Cancer Res Clin Oncol.
140:1251–1259. 2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Lau YS, Ling WC, Murugan D and Mustafa MR:
Boldine ameliorates vascular oxidative stress and endothelial
dysfunction: Therapeutic implication for hypertension and diabetes.
J Cardiovasc Pharmacol. 65:522–531. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Gillet FX, Bournaud C, Antonino de Souza
Júnior JD and Grossi-de-Sa MF: Plant-parasitic nematodes: Towards
understanding molecular players in stress responses. Ann Bot.
119:775–789. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Lu MC, Ji JA, Jiang ZY and You QD: The
Keap1-Nrf2-ARE pathway as a potential preventive and therapeutic
target: An update. Med Res Rev. 36:924–963. 2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Cheng D, Wu R, Guo Y and Kong AN:
Regulation of Keap1-Nrf2 signaling: The role of epigenetics. Curr
Opin Toxicol. 1:134–138. 2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chiou YS, Huang Q, Ho CT, Wang YJ and Pan
MH: Directly interact with Keap1 and LPS is involved in the
anti-inflammatory mechanisms of (-)-epicatechin-3-gallate in
LPS-induced macrophages and endotoxemia. Free Radic Biol Med.
94:1–16. 2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Itoh K, Ye P, Matsumiya T, Tanji K and
Ozaki T: Emerging functional cross-talk between the Keap1-Nrf2
system and mitochondria. J Clin Biochem Nutr. 56:91–97.
2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zenkov NK, Menshchikova EB and Tkachev VO:
Keap1/Nrf2/ARE redox-sensitive signaling system as a
pharmacological target. Biochemistry (Mosc). 78:19–36.
2013.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Tu J, Zhang X, Zhu Y, Dai Y, Li N, Yang F,
Zhang Q, Brann DW and Wang R: Cell-permeable peptide targeting the
Nrf2-Keap1 interaction: A potential novel therapy for global
cerebral ischemia. Neurosci. 35:14727–14739. 2015.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Wakabayashi N, Slocum SL, Skoko JJ, Shin S
and Kensler TW: When NRF2 talks, who's listening? Antioxid Redox
Signal. 13:1649–1663. 2010.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Thygesen K, Alpert JS, Jaffe AS, Simoons
ML, Chaitman BR and White HD: Task Force for the Universal
Definition of Myocardial Infarction. Third universal definition of
myocardial infarction. Nat Rev Cardiol. 9:620–633. 2012.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Ostadal B, Drahota Z, Houstek J, Milerova
M, Ostadalova I, Hlavackova M and Kolar F: Developmental and sex
difference in cardiac tolerance to ischemia/reperfusion injury: The
role of mitochondria1. Can J Physiol Pharmacol.
97:808–814. 2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Xue M, Momiji H, Rabbani N, Barker G,
Bretschneider T, Shmygol A, Rand DA and Thornalley PJ: Frequency
modulated translocational oscillations of Nrf2 mediate the
antioxidant response element cytoprotective transcriptional
response. Antioxid Redox Signal. 23:613–629. 2015.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Chai D, Zhang L, Xi S, Cheng Y, Jiang H
and Hu R: Nrf2 activation induced by Sirt1 ameliorates acute lung
injury after intestinal ischemia/reperfusion through NOX4-mediated
gene regulation. Cell Physiol Biochem. 46:781–792. 2018.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Schwarz M, Lossow K, Kopp JF, Schwerdtle T
and Kipp AP: Crosstalk of Nrf2 with the trace elements selenium,
iron, zinc, and copper. Nutrients. 11(2112)2019.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Mazzei L, Docherty NG and Manucha W:
Mediators and mechanisms of heat shock protein 70 based
cytoprotection in obstructive nephropathy. Cell Stress Chaperones.
20:893–906. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Mann GE, Bonacasa B, Ishii T and Siow RC:
Targeting the redox sensitive Nrf2-Keap1 defense pathway in
cardiovascular disease: Protection afforded by dietary isoflavones.
Curr Opin Pharmacol. 9:139–145. 2009.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Xu B, Zhang J, Strom J, Lee S and Chen QM:
Myocardial ischemic reperfusion induces de novo NRF2 protein
translation. Biochim Biophys Acta. 1842:1638–1647. 2014.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Ashrafian H, Czibik G, Bellahcene M,
Aksentijević D, Smith AC, Mitchell SJ, Dodd MS, Kirwan J, Byme JJ,
Ludwiq C, et al: Fumarate is cardioprotective via activation of the
NRF2 antioxidant pathway. Cell Metab. 15:361–371. 2012.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Farías JG, Carrasco-Pozo C, Carrasco Loza
R, Sepúlveda N, Álvarez P, Quezada M, Quiñones J, Molina V and
Castillo RL: Polyunsaturated fatty acid induces cardioprotection
against ischemia- reperfusion through the inhibition of NF-kappaB
and induction of NRF2. Exp Biol Med. 242:1104–1114. 2017.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Bhogal RH, Weston CJ, Velduis S, G D
Leuvenink H, Reynolds GM, Davies S, Nyguet-Thin L, Alfaifi M,
Shepard EL, Boteon Y, et al: The reactive oxygen species-mitophagy
signaling pathway regulates liver endothelial cell survival during
ischemia/reperfusion injury. Liver Transpl. 24:1437–1452.
2018.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Scherz-Shouval R and Elazar Z: ROS,
mitochondria and the regulation of autophagy. Trends Cell Biol.
17:422–427. 2007.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Dinkova-Kostova AT and Abramov AY: The
emerging role of NRF2 in mitochondrial function. Free Radic Biol
Med. 88:179–188. 2015.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Ludtmann MH, Angelova PR, Zhang Y, Abramov
AY and Dinkova-Kostova AT: NRF2 affects the efficiency of
mitochondrial fatty acid oxidation. Biochem J. 457:415–424.
2014.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Glinka YY and Youdim MB: Inhibition of
mitochondrial complexes I and IV by 6-hydroxydopamine. Eur J
Pharmacol. 292:329–332. 1995.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Baechler BL, Bloemberg D and Quadrilatero
J: Mitophagy regulates mitochondrial network signaling, oxidative
stress, and apoptosis during myoblast differentiation. Autophagy.
15:1606–1619. 2019.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Qiu M, Zhang S, Ke L, Tang H, Zeng X and
Liu J: JS-K enhances chemosensitivity of prostate cancer cells to
Taxol via reactive oxygen species activation. Oncol Lett.
17:757–764. 2019.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Zhang Y, Sano M, Shinmura K, Tamaki K,
Katsumata Y, Matsuhashi T, Morizane S, Ito H, Hishiki T, Endo J, et
al: 4-hydroxy-2-nonenal protects against cardiac
ischemiareperfusion injury via the NRF2-dependent pathway. J Mol
Cell Cardiol. 49:576–586. 2010.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Anedda A, López-Bernardo E, Acosta-Iborra
B, Saadeh Suleiman M, Landázuri MO and Cadenas S: The transcription
factor NRF2 promotes survival by enhancing the expression of
uncoupling protein 3 under conditions of oxidative stress. Free
Radic Biol Med. 61C:395–407. 2013.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Curfman G: Stem cell therapy for heart
failure: An unfulfilled promise? JAMA. 321:1186–1187.
2019.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Chen YL, Fan J, Cao L, Han TL, Zeng M, Xu
Y, Ling Z and Yin Y: Unique mechanistic insights into the
beneficial effects of angiotensin-(1-7) on the prevention of
cardiac fibrosis: A metabolomic analysis of primary cardiac
fibroblasts. Exp Cell Res. 378:158–170. 2019.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Ambrosi N, Guerrieri D, Caro F, Sanchez F,
Haeublein G, Casadei D, Incardona C and Chuluyan E: Alpha lipoic
acid: A therapeutic strategy that tend to limit the action of free
radicals in transplantation. Int J Mol Sci. 19(102)2018.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Kageyama S, Saito T, Obata M, Koide RH,
Ichimura Y and Komatsu M: Negative regulation of the Keap1-Nrf2
pathway by a p62/Sqstm1 splicing variant. Mol Cell Biol.
38:e00642–17. 2018.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Erpicum P, Rowart P, Defraigne JO,
Krzesinski JM and Jouret F: What we need to know about
lipid-associated injury in case of renal ischemia-reperfusion. Am J
Physiol Renal Physiol. 315:F1714–F1719. 2018.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Lee LY, Harberg C, Matkowskyj KA, Cook S,
Roenneburg D, Werner S, Johnson J and Foley DP: Overactivation of
the nuclear factor (erythroid-derived 2)-like 2-antioxidant
response element pathway in hepatocytes decreases hepatic
ischemia/reperfusion injury in mice. Liver Transpl. 22:91–102.
2015.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Kaplinsky E: DAPA-HF trial: Dapagliflozin
evolves from a glucose-lowering agent to a therapy for heart
failure. Drugs Context. 9(2019-11-3)2020.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Virani SA, Sharma V, McCann M, Koehler J,
Tsang B and Zieroth S: Prospective evaluation of integrated device
diagnostics for heart failure management: Results of the TRIAGE-HF
study. ESC Heart Fail. 5:809–817. 2018.PubMed/NCBI View Article : Google Scholar
|
|
71
|
McMurray JJ, Adamopoulos S, Anker SD,
Auricchio A, Böhm M, Dickstein K, Falk V, Filippatos G, Fonseca C,
Gomez-Sanchez MA, et al: ESC guidelines for the diagnosis and
treatment of acute and chronic heart failure 2012: The task force
for the diagnosis and treatment of acute and chronic heart failure
2012 of the European Society of Cardiology. Developed in
collaboration with the Heart Failure Association (HFA) of the ESC.
Eur Heart J. 33:1787–1847. 2012.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Zhu X, Oseghale AR, Nicole LH, Li B and
Pace BS: Mechanisms of NRF2 activation to mediate fetal hemoglobin
induction and protection against oxidative stress in sickle cell
disease. Exp Biol Med (Maywood). 244:171–182. 2019.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Rajasekaran NS, Varadharaj S, Khanderao
GD, Davidson CJ, Kannan S, Firpo MA, Zweier JL and Benjamin IJ:
Sustained activation of nuclear erythroid 2-related factor
2/antioxidant response element signaling promotes reductive stress
in the human mutant protein aggregation cardiomyopathy in mice.
Antioxid Redox Signal. 14:957–971. 2011.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Ichikawa T, Li J, Meyer CJ, Janicki JS,
Hannink M and Cui T: Dihydro-CDDO-trifluoroethyl amide (dh404), a
novel Nrf2 activator, suppresses oxidative stress in
cardiomyocytes. PLoS One. 4(e8391)2009.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Li J, Zhang C, Xing Y, Janicki JS,
Yamamoto M, Wang XL, Tang DQ and Cui T: Up-regulation of p27(kip1)
contributes to Nrf2-mediated protection against angiotensin
II-induced cardiac hypertrophy. Cardiovasc Res. 90:315–324.
2011.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Burchfield JS, Xie M and Hill JA:
Pathological ventricular remodeling: Mechanisms: Part 1 of 2.
Circulation. 128:388–400. 2013.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Takimoto E and Kass DA: Role of oxidative
stress in cardiac hypertrophy and remodeling. Hypertension.
49:241–248. 2007.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Guan Y, Zhou L, Zhang Y, Tian H, Li A and
Han X: Effects of PP2A/Nrf2 on experimental diabetes
mellitus-related cardiomyopathy by regulation of autophagy and
apoptosis through ROS dependent pathway. Cell Signal.
62(109339)2019.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Murdoch CE, Zhang M, Cave AC and Shah AM:
NADPH oxidase-dependent redox signalling in cardiac hypertrophy,
remodelling and failure. Cardiovasc Res. 71:208–215.
2006.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Shuai W, Kong B, Fu H, Shen C, Jiang X and
Huang H: MD1 Deficiency promotes inflammatory atrial remodelling
induced by high-fat diets. Can J Cardiol. 35:208–216.
2019.PubMed/NCBI View Article : Google Scholar
|
|
81
|
Yu C, Lin H, Yang H, Kong SL, Zhang Q and
Lee SW: Progression of systolic abnormalities in patients with
‘isolated’ diastolic heart failure and diastolic dysfunction.
Circulation. 105:1195–1201. 2002.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Cai L, Li W, Wang G, Guo L, Jiang Y and
Kang YJ: Hyperglycemia-induced apoptosis in mouse myocardium:
Mitochondrial cytochrome C-mediated caspase-3 activation pathway.
Diabetes. 51:1938–1948. 2002.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Fassett J, Xu X, Kwak D, Zhu G, Fassett
EK, Zhang P, Wang H, Maver B, Bache RJ and Chen Y: Adenosine kinase
attenuates cardiomyocyte microtubule stabilization and protects
against pressure overload-induced hypertrophy and LV dysfunction. J
Mol Cell Cardiol. 130:49–58. 2019.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Hafstad AD, Nabeebaccus AA and Shah AM:
Novel aspects of ROS signalling in heart failure. Basic Res
Cardiol. 108(359)2013.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Gupta S, Das B and Sen S: Cardiac
hypertrophy: Mechanisms and therapeutic opportunities. Antioxid
Redox Signal. 9:623–652. 2007.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Sabri A, Hughie HH and Lucchesi PA:
Regulation of hypertrophic and apoptotic signaling pathways by
reactive oxygen species in cardiac myocytes. Antioxid Redox Signal.
5:731–740. 2003.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Zhang X, Xiao Z, Yao J, Zhao G, Fa X and
Niu J: Participation of protein kinase C in the activation of Nrf2
signaling by ischemic preconditioning in the isolated rabbit heart.
Mol Cell Biochem. 372:169–179. 2013.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Zhao WY, Zhao TQ, Chen YJ, Ahokas RA and
Sun Y: Oxidative stress mediates cardiac fbrosis by enhancing
transforming growth factor-beta1 in hypertensive rats. Mol Cell
Biochem. 317:43–50. 2008.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Yang T, Sun Y, Mao L, Zhang M, Li Q, Zhang
L, Shi Y, Leak RK, Chen J and Zhang F: Brain ischemic
preconditioning protects against ischemic injury and preserves the
blood-brain barrier via oxidative signaling and Nrf2 activation.
Redox Biol. 17:323–337. 2018.PubMed/NCBI View Article : Google Scholar
|
|
90
|
James PA, Oparil S, Carter BL, Cushman WC,
Dennison-Himmelfarb C, Handler J, Lackland DT, LeFevre ML,
MacKenzie TD, Oqedeqbe O, et al: Evidence-based guideline for the
management of high blood pressure in adults: Report from the panel
members appointed to the Eighth Joint National Committee. JAMA.
311:507–520. 2014.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Hu CM, Chen YH, Chiang MT and Chau LY:
Heme oxygenase-1 inhibits angiotensin II-induced cardiac
hypertrophy in vitro and in vivo. Circulation. 110:309–316.
2004.PubMed/NCBI View Article : Google Scholar
|
|
92
|
Mancusi C, Canciello G, Izzo R, Damiano S,
Grimaldi MG, Luca N, Simone G, Trimarco B and Losi MA: Left atrial
dilatation: A target organ damage in young to middle-age
hypertensive patients. The Campania Salute Network. Int J Cardiol.
265:229–233. 2018.PubMed/NCBI View Article : Google Scholar
|
|
93
|
Wang ZH, Liu JL, Wu L, Yu Z and Yang HT:
Concentration-dependent wrestling between detrimental and
protective effects of H2O2 during myocardial
ischemia/reperfusion. Cell Death Dis. 5(e1297)2014.PubMed/NCBI View Article : Google Scholar
|
|
94
|
Yao SY, Liu J, Li Y, Wang M, Wang C and
Xue H: Association between plasma microRNA-29a and left ventricular
hypertrophy in patients with hypertension. Zhonghua Xin Xue Guan
Bing Za Zhi. 47:215–220. 2019.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
95
|
Mitra A, Basak T, Datta K, Naskar S,
Sengupta S and Sarkar S: Role of a-crystallin B as a regulatory
switch in modulating cardiomyocyte apoptosis by mitochondria or
endoplasmic reticulum during cardiac hypertrophy and myocardial
infarction. Cell Death Dis. 4(e582)2013.PubMed/NCBI View Article : Google Scholar
|
|
96
|
Delmar M and Makita N: Cardiac connexins,
mutations and arrhythmias. Curr Opin Cardiol. 27:236–241.
2012.PubMed/NCBI View Article : Google Scholar
|
|
97
|
Stout JM, Gousset MU, Drummond HA, Gray W
III, Pruett BE and Stec DE: Sex-specific effects of heme
oxygenase-2 deficiency on renovascular hypertension. J Am Soc
Hypertens. 7:328–335. 2013.PubMed/NCBI View Article : Google Scholar
|
|
98
|
Tian C, Gao L, Zimmerman MC and Zucker IH:
Myocardial infarction-induced microRNA-enriched exosomes contribute
to cardiac Nrf2 dysregulation in chronic heart failure. Am J
Physiol Heart Circ Physiol. 314:H928–H939. 2018.PubMed/NCBI View Article : Google Scholar
|
|
99
|
Tham YK, Bernardo BC, Ooi JY, Weeks KL and
McMullen JR: Pathophysiology of cardiac hypertrophy and heart
failure: Signaling pathways and novel therapeutic targets. Arch
Toxicol. 89:1401–1438. 2015.PubMed/NCBI View Article : Google Scholar
|
|
100
|
Yang KC and Dudley JSC: Oxidative stress
and atrial fibrillation: Finding a missing piece to the puzzle.
Circulation. 128:1724–1726. 2013.PubMed/NCBI View Article : Google Scholar
|
|
101
|
Barry SP, Davidson SM and Townsend PA:
Molecular regulation of cardiac hypertrophy. Int J Biochem Cell
Biol. 40:2023–2039. 2008.PubMed/NCBI View Article : Google Scholar
|
|
102
|
Maulik SK and Kumar S: Oxidative stress
and cardiac hypertrophy: A review. Toxicol Mech Methods.
22:359–366. 2012.PubMed/NCBI View Article : Google Scholar
|
|
103
|
Baruteau AE, Probst V and Abriel H:
Inherited progressive cardiac conduction disorders. Curr Opin
Cardiol. 30:33–39. 2015.PubMed/NCBI View Article : Google Scholar
|
|
104
|
Chang YJ, Hsiao HJ, Hsia SH, Lin JJ, Hwang
MS, Chung HT, Chen CL, Huang YC and Tsai MH: Analysis of clinical
parameters and echocardiography as predictors of fatal pediatric
myocarditis. PLoS One. 14(e0214087)2019.PubMed/NCBI View Article : Google Scholar
|
|
105
|
Zhao YS, An JR, Yang S, Guan P, Yu FY, Li
W, Li JR, Guo Y, Sun ZM and Ji ES: Hydrogen and oxygen mixture to
improve cardiac dysfunction and myocardial pathological changes
induced by intermittent hypoxia in rats. Oxid Med Cell Longev.
2019(7415212)2019.PubMed/NCBI View Article : Google Scholar
|
|
106
|
Nakamura M and Sadoshima J: Mechanisms of
physiological and pathological cardiac hypertrophy. Nat Rev
Cardiol. 15:387–407. 2018.PubMed/NCBI View Article : Google Scholar
|
|
107
|
Li J, Ichikawa T, Villacorta L, Janicki
JS, Brower GL, Yamamoto M and Cui T: Nrf2 protects against
maladaptive cardiac responses to hemodynamic stress. Arterioscler
Thromb Vasc Biol. 29:1843–1850. 2009.PubMed/NCBI View Article : Google Scholar
|
|
108
|
Nakamura M and Sadoshima J: Cardiomyopathy
in obesity, insulin resistance or diabetes. J Physiol.
598:2977–2993. 2020.PubMed/NCBI View Article : Google Scholar
|
|
109
|
Felker GM, Thompson RE, Hare JM, Hruban
RH, Clemetson DE, Howard DL, Baughman K and Kasper EK: Underlying
causes and long-term survival in patients with initially
unexplained cardiomyopathy. N Engl J Med. 342:1077–1184.
2000.PubMed/NCBI View Article : Google Scholar
|
|
110
|
Quinaglia T, Oliveira DC, Matos-Souza JR
and Sposito AC: Diabetic cardiomyopathy: Factual or factoid? Rev
Assoc Med Bras (1992). 65:61–69. 2019.PubMed/NCBI View Article : Google Scholar
|
|
111
|
Cooper LT Jr: Myocarditis. N Engl J Med.
360:1526–1538. 2009.PubMed/NCBI View Article : Google Scholar
|
|
112
|
Althunibat OY, Al Hroob AM, Abukhalil MH,
Germoush MO, Bin-Jumah M and Mahmoud AM: Fisetin ameliorates
oxidative stress, inflammation and apoptosis in diabetic
cardiomyopathy. Life Sci. 221:83–92. 2019.PubMed/NCBI View Article : Google Scholar
|
|
113
|
Zhao X, Cai A, Peng Z, Liang W, Xi H, Li
P, Chen G, Yu J and Chen L: JS-K induces reactive oxygen
species-dependent anti-cancer effects by targeting mitochondria
respiratory chain complexes in gastric cancer. J Cell Mol Med.
23:2489–2504. 2019.PubMed/NCBI View Article : Google Scholar
|
|
114
|
Ansley DM and Wang B: Oxidative stress and
myocardial injury in the diabetic heart. J Pathol. 229:232–241.
2013.PubMed/NCBI View Article : Google Scholar
|
|
115
|
Hernández M, Wicz S and Corral RS:
Cardioprotective actions of curcumin on the pathogenic
NFAT/COX-2/prostaglandin E2 pathway induced during Trypanosoma
cruzi infection. Phytomedicine. 23:1392–1400. 2016.PubMed/NCBI View Article : Google Scholar
|
|
116
|
Ndisang JF, Lane N, Syed N and Jadhav A:
Up-regulating the heme oxygenase system with hemin improves insulin
sensitivity and glucose metabolism in adult spontaneously
hypertensive rats. Endocrinology. 151:549–560. 2010.PubMed/NCBI View Article : Google Scholar
|
|
117
|
Jiménez-Osorio AS, García-Niño WR,
González-Reyes S, Álvarez-Mejía AE, Guerra-León S, Salazar-Segovia
J, Falcón I, Montes de Oca-Solano H, Madero M and Pedraza-Chaverri
J: The effect of dietary supplementation with curcumin on redox
status and Nrf2 activation in patients with nondiabetic or diabetic
proteinuric chronic kidney disease: A pilot study. J Ren Nutr.
26:237–244. 2016.PubMed/NCBI View Article : Google Scholar
|
|
118
|
Soundararajan P and Kim JS:
Anti-carcinogenic glucosinolates in cruciferous vegetables and
their antagonistic effects on prevention of cancers. Molecules.
23(2983)2018.PubMed/NCBI View Article : Google Scholar
|
|
119
|
Wang J, Wang S, Wang W, Chen J, Zhang Z,
Zheng Q, Liu Q and Cai L: Protection against diabetic
cardiomyopathy is achieved using a combination of sulforaphane and
zinc in type 1 diabetic OVE26 mice. J Cell Mol Med. 23:6319–6330.
2019.PubMed/NCBI View Article : Google Scholar
|
|
120
|
Zhang Z, Wang S, Zhou S, Yan X, Wang Y,
Chen J, Mellen N, Kong M, Gu J, Tan Y, et al: Sulforaphane prevents
the development of cardiomyopathy in type 2 diabetic mice probably
by reversing oxidative stress-induced inhibition of LKB1/AMPK
pathway. J Mol Cell Cardiol. 77:42–52. 2014.PubMed/NCBI View Article : Google Scholar
|
|
121
|
Bai Y, Wang X, Zhao S, Ma C, Cui J and
Zheng Y: Sulforaphane protects against cardiovascular disease via
Nrf2 activation. Oxid Med Cell Longev. 2015(407580)2015.PubMed/NCBI View Article : Google Scholar
|
|
122
|
Delucchi F, Berni R, Frati C, Cavalli S,
Graiani G, Sala R, Chaponnier C, Gabbiani G, Calani L, Rio DD, et
al: Resveratrol treatment reduces cardiac progenitor cell
dysfunction and prevents morpho-functional ventricular remodeling
in type-1 diabetic rats. PLoS One. 7(e39836)2012.PubMed/NCBI View Article : Google Scholar
|
|
123
|
Sun X, Shan A, Wei Z and Xu B: Intravenous
mesenchymal stem cell-derived exosomes ameliorate myocardial
inflammation in the dilated cardiomyopathy. Biochem Biophys Res
Commun. 503:2611–2618. 2018.PubMed/NCBI View Article : Google Scholar
|
|
124
|
Ge ZD, Lian Q, Mao X and Xia Z: Current
status and challenges of NRF2 as a potential therapeutic target for
diabetic cardiomyopathy. Int Heart J. 60:512–520. 2019.PubMed/NCBI View Article : Google Scholar
|
|
125
|
El-Agamy DS, El-Harbi KM, Khoshhal S,
Ahmed N, Elkablawy MA, Shaaban AA and Abo-Haded HM: Pristimerin
protects against doxorubicin-induced cardiotoxicity and fibrosis
through modulation of Nrf2 and MAPK/NF-κB signaling pathways.
Cancer Manag Res. 11:47–61. 2018.PubMed/NCBI View Article : Google Scholar
|
|
126
|
Zhou S, Jin J, Bai T, Sachleben LR Jr, Cai
L and Zheng Y: Potential drugs which activate nuclear factor
E2-related factor 2 signaling to prevent diabetic cardiovascular
complications: A focus on fumaric acid esters. Life Sci. 134:56–62.
2015.PubMed/NCBI View Article : Google Scholar
|
|
127
|
Saidu NE, Noé G, Cerles O, Cabel L,
Kavian-Tessler N, Chouzenoux S, Bahuaud M, Chéreau C, Nicco C,
Leroy K, et al: Dimethyl fumarate controls the NRF2/DJ-1 axis in
cancer cells: Therapeutic applications. Mol Cancer Ther.
16:529–539. 2017.PubMed/NCBI View Article : Google Scholar
|
|
128
|
Brennan MS, Patel H, Allaire N, Thai A,
Cullen P, Rvan S, Lukashev M, Bista P, Huang R, Rhodes KJ and
Scannevin RH: Pharmacodynamics of dimethyl fumarate are tissue
specific and involve Nrf2-dependent and -independent mechanisms.
Antioxid Redox Signal. 24:1058–1071. 2016.PubMed/NCBI View Article : Google Scholar
|
|
129
|
Bomprezzi R: Dimethyl fumarate in the
treatment of relapsing-remitting multiple sclerosis: An overview.
Ther Adv Neurol Disord. 8:20–30. 2015.PubMed/NCBI View Article : Google Scholar
|
|
130
|
Ganeshpurkar A and Saluja AK: The
pharmacological potential of rutin. Saudi Pharm J. 25:149–164.
2017.PubMed/NCBI View Article : Google Scholar
|
|
131
|
Moore PK, Griffiths RJ and Lofts FJ: The
effect of some flavone drugs on the conversion of prostacyclin to
6-oxoprostaglandin E1. Biochem Pharmacol. 32:2813–2817.
1983.PubMed/NCBI View Article : Google Scholar
|
|
132
|
Gao HC, Zhu K, Gao HM, Miao CS, Zhang LN,
Liu W and Xin H: Role of tissue transglutaminase in the
pathogenesis of diabetic cardiomyopathy and the intervention effect
of rutin. Exp Ther Med. 9:1103–1108. 2015.PubMed/NCBI View Article : Google Scholar
|
|
133
|
Sayed AS, Xia K, Salma U, Yang T and Peng
J: Diagnosis, prognosis and therapeutic role of circulating miRNAs
in cardiovascular diseases. Heart Lung Circ. 23:503–510.
2014.PubMed/NCBI View Article : Google Scholar
|
|
134
|
Chen S, Puthanveetil P, Feng B, Matkovich
SJ, Dorn GW II and Chakrabarti S: Cardiac miR-133a overexpression
prevents early cardiac fibrosis in diabetes. J Cell Mol Med.
18:415–421. 2014.PubMed/NCBI View Article : Google Scholar
|
|
135
|
Chen K, Ma Y, Wu S, Zhuang Y, Liu X, Lv L
and Zhang G: Construction and analysis of a lncRNA-miRNA-mRNA
network based on competitive endogenous RNA reveals functional
lncRNAs in diabetic cardiomyopathy. Mol Med Rep. 20:1393–1403.
2019.PubMed/NCBI View Article : Google Scholar
|
|
136
|
Lin M and Mao ZJ: lncRNA-mRNA competing
endogenous RNA network in IR-hepG2 cells ameliorated by APBBR
decreasing ROS levels: A systematic analysis. PeerJ.
8(e8604)2020.PubMed/NCBI View Article : Google Scholar
|
|
137
|
Dludla PV, Nkambule BB, Dias SC and
Johnson R: Cardioprotective potential of N-acetyl cysteine against
hyperglycaemia-induced oxidative damage: A protocol for a
systematic review. Syst Rev. 6(96)2017.PubMed/NCBI View Article : Google Scholar
|
|
138
|
Mansueto G, Benincasa G, Della Mura N,
Nicoletti GF and Napoli C: Epigenetic-sensitive liquid biomarkers
and personalised therapy in advanced heart failure: A focus on
cell-free DNA and microRNAs. J Clin Pathol. 73:535–543.
2020.PubMed/NCBI View Article : Google Scholar
|
|
139
|
Zhang X, Dong S, Jia Q, Zhang A, Li Y, Zhu
Y, Lv S and Zhang J: The microRNA in ventricular remodeling: The
miR-30 family. Biosci Rep. 39(BSR20190788)2019.PubMed/NCBI View Article : Google Scholar
|
|
140
|
Raut SK, Singh GB, Rastogi B, Saikia UN,
Mittal A, Dogra N, Singh S, Prasad R and Khullar M: miR-30c and
miR-181a synergistically modulate p53-p21 pathway in diabetes
induced cardiac hypertrophy. Mol Cell Biochem. 417:191–203.
2016.PubMed/NCBI View Article : Google Scholar
|
|
141
|
Li M, Chen X, Chen L, Chen K, Zhou J and
Song J: MiR-1-3p that correlates with left ventricular function of
HCM can serve as a potential target and differentiate HCM from DCM.
J Transl Med. 16(161)2018.PubMed/NCBI View Article : Google Scholar
|