1. Introduction
Type 2 diabetes mellitus (T2DM) is characterized by
insulin resistance (IR). Improved risk prediction and understanding
of the pathogenesis underlying IR are crucial for the management of
T2DM. The aetiology of IR is multifactorial. Genetic background and
environmental factors, such as age, lifestyle and diet, are two
classifiable disease risk factors and the interaction between them
contributes to the development of T2DM. Fatty acids (FAs) may have
a key role in the development of IR and T2DM (1-3).
However, the long-term effect of FAs on T2DM has yet to be fully
elucidated (4).
While genomics, transcriptomics and proteomics have
been widely used to improve the current understanding of obesity
and T2DM, lipidomics is a tool that has been used comparatively
less frequently (5). Lipidomics,
which is a subcategory of metabolomics, may enhance the
understanding of the contribution of FAs and lipids towards the
development of health-related complications, particularly IR and
T2DM. Circulating lipids and FAs may reflect an individual's
lifestyle (e.g., diet and exercise) and their gene and protein
activity, all of which may affect the development of IR and T2DM
(6,7). Lipidomic techniques have provided
valuable information on obesity and T2DM-related changes in
adipocyte (8), macrophage (9), skeletal muscle (10), lipoprotein lipid composition
(11) and liver FA metabolism
(12), and have allowed researchers
to better understand the contribution of obesity towards the
development of T2DM. These methods have also enabled researchers to
unravel the underlying mechanisms through which exercise, metformin
and rosiglitazone improve the status of patients with T2DM
(12,13). Lipidomic techniques have revealed
that quantifying specific FAs within lipid fractions [e.g.,
triglycerides (TGs) and phospholipids (PLs)] may provide a more
accurate indication of IR and T2DM (13-15).
It was demonstrated that specific combinations of FAs within PL and
TG exhibited the strongest association with the risk of T2DM,
particularly shorter saturated FAs (SFAs) (15).
2. FAs
FAs may exist as free FAs (FFAs) in the body or they
may combine with other molecules to form lipids, such as
cholesterol esters (CEs), PLs and TGs (16-20).
Low-density lipoprotein (LDL), very LDL and high-density
lipoprotein consist of different relative amounts of CE, PL and TG.
PLs have a hydrophilic phosphate head and two hydrophobic FA tails
linked to a glycerol molecule. TGs contain FFAs that are esterified
to a glycerol molecule. TG is the main form of fat in the diet. CEs
are formed from FA and cholesterol by an ester bond between the
carboxylate group of FAs and the hydroxyl group of cholesterol
(16-20).
In addition to using fat as an energy source, the
human body also synthesizes, desaturates and elongates FAs
endogenously (21). The synthesis
of FAs is low if the dietary intake of fat is moderate or high,
whereas a high dietary intake of carbohydrates stimulates de
novo lipogenesis (22-24).
Several factors affect endogenous FA metabolism and
plasma FA composition, such as age and sex, health status,
epigenetic changes and genes. Desaturation and elongation are steps
of a metabolic pathway in which dietary and endogenous SFAs are
elongated and converted to monounsaturated FAs (MUFAs), while
highly polyunsaturated FAs (PUFAs) are synthesized from dietary n-3
FAs (e.g., α-linolenic acid) and n-6 FAs (e.g., linoleic acid) in
the liver and adipose tissue (25,26).
Desaturases and elongases are enzymes that activate these metabolic
pathways. Desaturases add a double bond to the FA, whereas
elongases lengthen the FA by adding two carbon molecules to the
carbon chain. The metabolism of PUFAs is important, as they are the
essential FAs of the human body (25,26).
A detailed understanding of the molecular structures
and mechanisms implicated in FA synthesis and degradation may
enable nutritional investigators to elucidate how and why specific
dietary patterns and classes of FAs are associated with IR and T2DM
(27). The changes in FA levels in
the circulation caused by IR are summarized in Fig. 1. In a normal condition, when insulin
binds to the insulin receptor it inactivates the enzyme hormone
sensitive lipase (HSL) involved in the hydrolysis of TG to glycerol
and FFA. In an insulin resistant state, there is an increase in the
activity of HSL releasing free fatty acids into circulation to the
liver. In the liver, hepatocytes take up the fatty acids and
channel them into secretory pathways. The enzyme lipoprotein lipase
in the blood vessels hydrolyses monoglycerides and FFA. As this
cycling process continues, FFA also increases (Fig. 1).
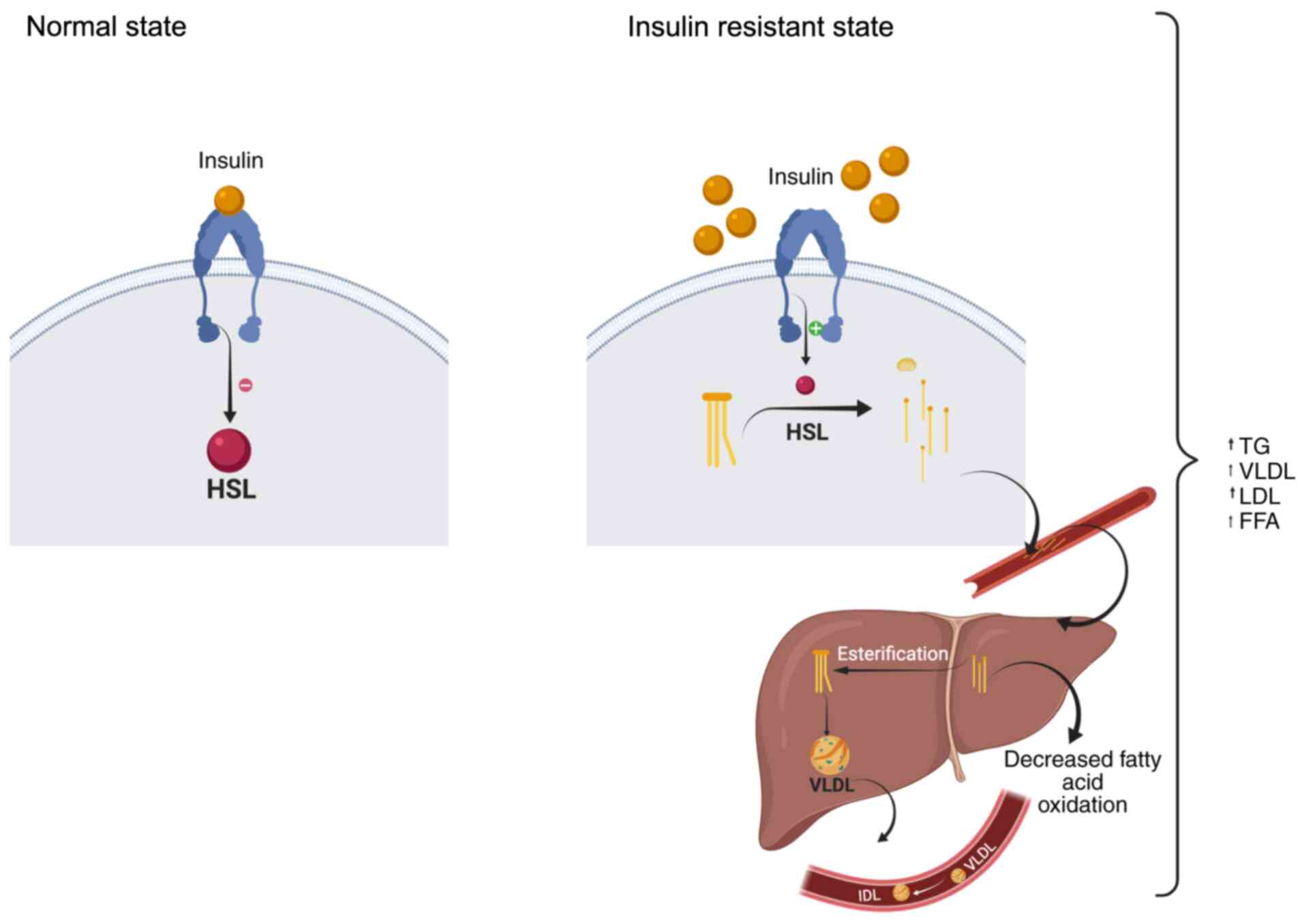 | Figure 1IR and changes in FA composition. In
adipose tissue, in the normal state, insulin binds to the insulin
receptor, inhibiting the enzyme HSL in the adipocytes of the
adipose tissue. This HSL hydrolyses lipids such as TGs. In the
state of IR, insulin is unable to bind to insulin receptors,
thereby suppressing intracellular signals. In turn, it activates
the HSL enzyme, which hydrolyse TGs to glycerol and FFAs, which are
released into the circulation towards the liver. Liver hepatocytes
take up FFAs to channel them to their secretory pathways. In the
case of hyperinsulinemia/IR, esterification is increased. In the
blood vessels, the enzyme LPL hydrolyses monoglycerides and FFAs. A
certain proportion of these are delivered to liver LPL
(hydrolysis). This process continues and more LDL and FFAs are
formed (schematic created with BioRender.com). FFA, free fatty acid; HSL,
hormone-sensitive lipase; LPL, lipoprotein lipase; LDL, low-density
lipoprotein; VLDL, very low-density lipoprotein; TG, triglyceride;
IR, insulin resistance. |
Fig. 2 depicts how
FFAs affect the insulin signalling pathway by increasing
diacylglycerol (DAG), reactive oxygen species and protein kinase C
(PKC), which in turn increases insulin receptor substrate-1 (IRS-1)
serine phosphorylation and decreases IRS-1 tyrosine
phosphorylation, thereby inhibiting the activity of PI3K. This
finally leads to disturbance of the fragile balance between β-cell
function and peripheral insulin resistance, which eventually
results in the clinical manifestation of T2DM.
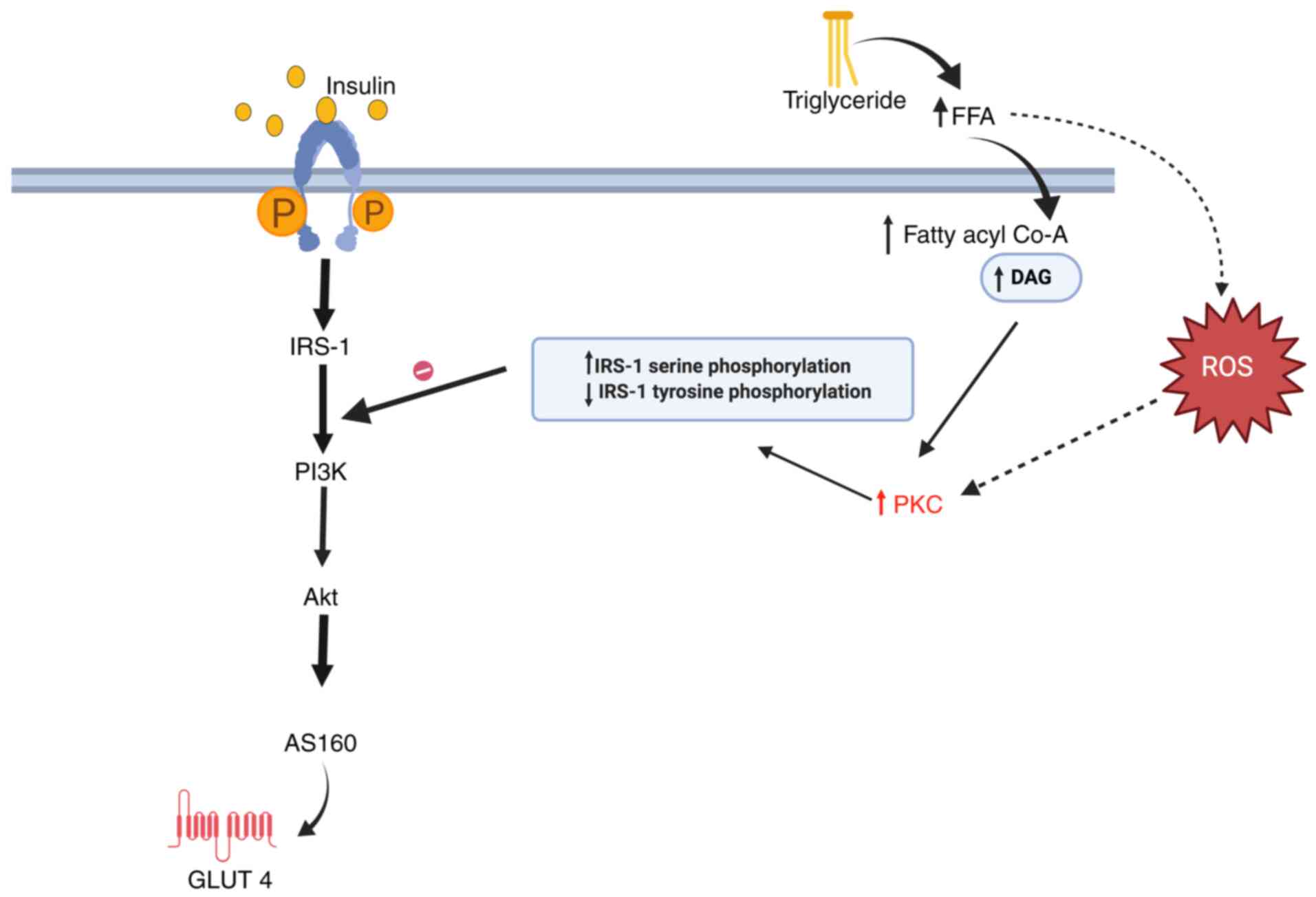 | Figure 2Effect of TGs and FFAs on the insulin
signalling pathway. Elevated TG levels increase FFAs, which leads
to accumulation of DAG and fatty acyl Co-A along with increased
ROS. All of this together activates PKC, which interrupts the
insulin signalling pathway (schematic created with BioRender.com). IRS, insulin receptor substrate;
AS160, Akt substrate of 160 kDa; Glut-4, glucose transporter 4;
FFA, free fatty acid; ROS, reactive oxygen species; P, phosphate;
PKC, protein kinase C; DAG, diacylglycerol; Co-A, coenzyme A; FFA,
free fatty acid. |
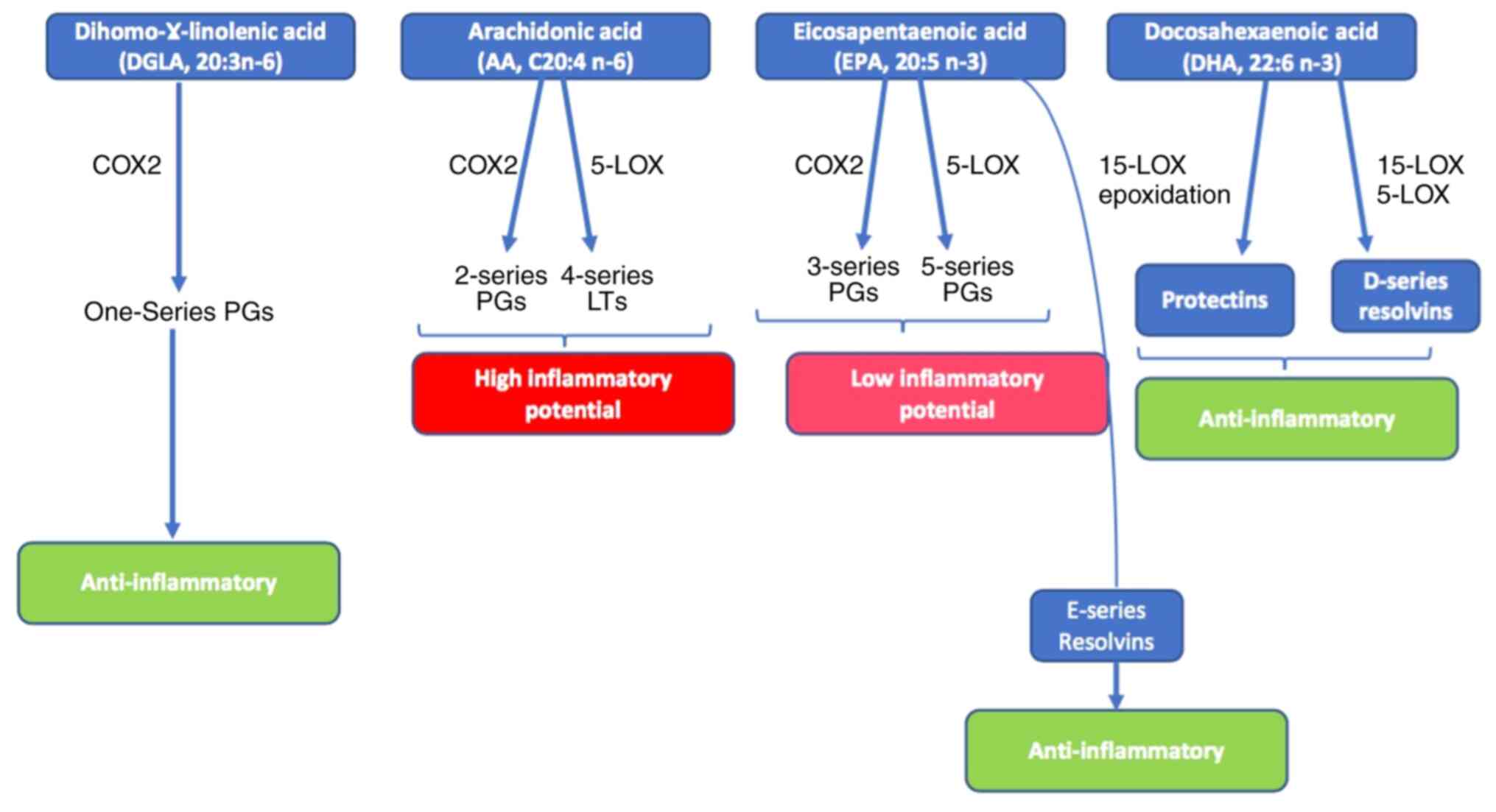
3. n-3 and n-6 PUFA bioactive mediators
Adipocytes and pancreatic β-cells produce a major
group of pro-inflammatory mediators, the eicosanoids. Oxidation of
n-3 and n-6 PUFAs [eicosapentaenoic acid (EPA), arachidonic acid
(AA) and the linolenic acid (LA) derivative dihomo-γ-LA] release
major eicosanoids, including prostaglandins (PGs), leukotrienes
(LTs), thromboxanes and lipoxins. These may be either
pro-inflammatory or anti-inflammatory (28,29).
PGs and LTs derived from AA are pro-inflammatory, whereas PGs and
LTs derived from EPA are anti-inflammatory (Fig. 3) (16,30,31).
4. FAs and T2DM
A number of physiological pathways are affected by
SFAs (32,33). Hepatic de novo lipogenesis
occurs in response to a high dietary intake of carbohydrates or
total calories, thereby increasing the endogenous levels of SFAs
(32,33). According to the Chinese philosophy
concept, palmitic and oleic acid were described as the ‘Yin’ and
‘Yang’ of FAs by Palomer et al (34). Increased palmitic acid levels in
subjects with diabetes may be explained via the enhancement of
deleterious complex lipid synthesis, impairment of cellular
organelle function and promotion of receptor-mediated inflammation
(35,36). The rate of FA delivery to
non-adipose tissues (liver, muscle, heart and pancreatic islets)
depends on the increased plasma non-esterified FA (NEFA) content,
promoting lipotoxicity and lipoapoptosis. These conditions raise
intracellular palmitic acid levels above their mitochondrial
oxidation limit and are converted to harmful complex FA-derived
lipids, such as DAG and ceramide.
A high intracellular DAG content activates PKC
isoforms, followed by phosphorylation of insulin serine residue 1,
attenuating the insulin signalling pathway. Pro-inflammatory
signalling cascade inhibitor and ceramide synthesis are, in turn,
activated by PKC isoforms. Furthermore, ceramide also inhibits
mitochondrial β-oxidation of FAs, induces endoplasmic reticulum
(ER) stress and activates the NACHT, LRR and PYD domains-containing
protein 3 inflammasome, a major instigator of inflammation
(37). The perturbation of ER
homeostasis by increased NEFA levels affects the lipid composition
of ER due to lipid dysregulation, resulting in ER stress, which
affects calcium signalling and attenuation of protein translation.
Eventually, these modifications cause metabolic dysregulation and
T2DM.
As mentioned above, according to the Chinese
philosophy concept, if palmitate is the ‘Yin’ of FAs in T2DM, oleic
acid is the ‘Yang’, as it elicits beneficial effects on insulin
sensitivity (38). These beneficial
effects of oleic acid are mediated by several mechanisms. Oleic
acid reduces leukotriene B4 (LTB4) levels and increases insulin
sensitivity (39). Oleic acid
elicits anti-inflammatory effects by increasing the levels of the
anti-inflammatory cytokine IL-10 and adiponectin and reducing the
levels of pro-inflammatory cytokines (interleukin-6, tumour
necrosis factor-α), induces macrophage polarization and reduces the
secretion of LTB4(38). FFA
receptor 4 or G protein-coupled receptor (GPR)120 may also mediate
these effects of oleic acid.
Unsaturated FAs have a key role in normal tissue
function as major components of membrane bilayers, determining
factors of cell structure and function and modulators of gene
expression. Unsaturated FAs may be MUFAs or PUFAs (38). A previous study demonstrated that,
in subjects with high fasting TG levels, MUFAs buffer β-cell
hyperactivity and IR (40).
LA and α-LA (ALA) are essential FAs, as the human
body lacks the desaturase enzymes catalysing their endogenous
production and they may only be obtained from dietary sources
(32,36). The relative FA levels in the plasma,
serum or erythrocyte membrane reflect the rate of conversion of
different FAs catalysed by key enzymes, such as FA desaturases
(FADS) and elongases (33,34).
ALA is converted into the long-chain omega-3
polyunsaturated FA EPA or docosahexaenoic acid (DHA) (41). Preclinical data suggested that
omega-3 improves insulin signalling (41). The proposed mechanism of how omega-3
improves insulin sensitivity is that omega-3 PUFAs attenuate ER
stress, enhance FA β-oxidation in mitochondria and uncouple
mitochondria, causing reduction in lipid buildup and reactive
oxygen species accumulation (38).
In addition, a previous study reported the positive impact of these
omega-3 FAs on mitofusin 2, which is involved in mitochondrial
dynamics homeostasis and mitochondria-associated membrane integrity
maintenance (41).
EPA and DHA regulate insulin sensitivity via Akt
phosphorylation, activation of AMP-activated protein kinase (a
treatment target for T2DM) and activation of peroxisome
proliferator-activated receptor (PPAR)-γ (42). Several studies have demonstrated the
role of EPA and DHA in preventing lipotoxicity and insulin
sensitivity restoration (43-48).
Omega-3 FAs also modulate pancreatic β-cell insulin secretion by
exerting a direct effect on the lipid raft function and structure,
and indirectly by inhibition of the expression of pro-inflammatory
mediators in adipose tissue and the promotion of adipokine
production (48). By contrast,
omega-6 FAs are pro-inflammatory, as the product of omega-6
desaturation, AA, produces inflammatory cytokines and
eicosanoids.
Omega-3 PUFAs inhibit inflammatory cytokine and
eicosanoid production from AA and induce adipokine production from
adipose tissue, and directly affect β-cell function by binding to
PPARs, GPR40 and GPR120, thereby promoting insulin secretion
(48). It remains elusive how
exactly these PUFAs affect glucose metabolism (49). It has also been reported that a
defect in the activity of D6 and D5 desaturases, the key enzymes of
the PUFA desaturation pathway, may be a key factor in the
development of IR (48), with
ensuing public health implications.
Overall, the role of the increase in the FFA content
of the body in IR may be explained with the insulin binding to its
receptor. Under normal conditions, binding of insulin to its
receptor in the adipose tissue cell membrane triggers an
intracellular signal that suppresses the activity of
hormone-sensitive lipase (HSL), an intracellular enzyme found in
adipocytes, which hydrolyses lipids such as TG.
When an individual is insulin-resistant, this
intracellular signalling is suppressed, thereby increasing HSL
activity, which hydrolyses TG to glycerol and FFAs, which are then
released into the circulation and accumulate in the liver. These
FAs are taken up by the hepatocytes and are channelled to their
secretory pathways. Due to IR, esterification increases. The enzyme
lipoprotein lipase in the blood vessels hydrolyses monoglycerides
and FFAs. This process continues, thereby increasing the FA content
(49).
Omega-3 and omega-6 FAs continuously compete for the
desaturation enzymes. ALA is preferred over LA by both FADS1 and
FADS2(50). Therefore, maintaining
an optimal balance between omega-6 and omega-3 FAs in the diet is
crucial for human health, as the physiological state is
anti-inflammatory (51). Unbalanced
omega-6/omega-3 ratio in favour of omega-6 PUFAs is highly
prothrombotic and pro-inflammatory, contributing to the prevalence
of atherosclerosis, obesity and diabetes (51). As previously reported, the target
ratio of omega-6/omega-3 should be 1:1 to 2:1(51). Although the Indian diet is low in
fat when compared to the western diet (52), the incidence of coronary artery
disease and T2DM are on the increase in this population. As per the
previous report by Mani and Kurpad (52) although the Indian diet is rich in
PUFAs, the ratio is in favour of omega-6 FAs, since the most
abundant PUFA in the majority of plant products is LA. Therefore,
the LA intake is high, irrespective of the type of vegetable oil
used in a typical Indian diet (52). Therefore, dietary FA interventions
specifically aimed at maintaining an optimal omega-6/omega-3 ratio
may overcome the risk of developing T2DM.
5. Conclusion
Taken together, the present review indicated that
the desaturation pathway appears to be highly important for
maintaining lipid homeostasis in the human body, the key to which
may be ensuring a healthy omega-6/omega-3 ratio. This suggests
that, in T2DM research, lipidomics may represent a useful approach
to improve the understanding of the effect of different lipids on
the risk of developing T2DM.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Not applicable.
Authors' contributions
SSS and SK wrote, revised and edited the article.
Both authors have read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Boden G and Shulman GI: Free fatty acids
in obesity and type 2 diabetes: Defining their role in the
development of insulin resistance and β-cell dysfunction. Eur J
Clin Invest. 32 (Suppl 3):14–23. 2002.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Boden G: Effects of free fatty acids (FFA)
on glucose metabolism: Significance for insulin resistance and type
2 diabetes. Exp Clin Endocrinol Diabetes. 111:121–124.
2003.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Boden G: Obesity, insulin resistance and
free fatty acids. Curr Opin Endocrinol Diabetes Obes. 18:139–143.
2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Risérus U, Willett WC and Hu FB: Dietary
fats and prevention of type 2 diabetes. Prog Lipid Res. 48:44–51.
2009.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Shevchenko A and Simons K: Lipidomics:
Coming to grips with lipid diversity. Nat Rev Mol Cell Biol.
11:593–598. 2010.PubMed/NCBI View
Article : Google Scholar
|
|
6
|
Gutch M, Kumar S, Razi SM, Gupta KK and
Gupta A: Assessment of insulin sensitivity/resistance. Indian J
Endocrinol Metab. 19:160–164. 2015.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Garaulet M, Pérez-Llamas F, Pérez-Ayala M,
Martínez P, de Medina FS, Tebar FJ and Zamora S: Site-specific
differences in the fatty acid composition of abdominal adipose
tissue in an obese population from a Mediterranean area: Relation
with dietary fatty acids, plasma lipid profile, serum insulin, and
central obesity. Am J Clin Nutr. 74:585–591. 2001.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Pietiläinen KH, Róg T, Seppänen-Laakso T,
Virtue S, Gopalacharyulu P, Tang J, Rodriguez-Cuenca S, Maciejewski
A, Naukkarinen J, Ruskeepää AL, et al: Association of lipidome
remodeling in the adipocyte membrane with acquired obesity in
humans. PLoS Biol. 9(e1000623)2011.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Borkman M, Storlien LH, Pan DA, Jenkins
AB, Chisholm DJ and Campbell LV: The relation between insulin
sensitivity and the fatty-acid composition of skeletal-muscle
phospholipids. N Engl J Med. 328:238–244. 1993.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Ståhlman M, Pham HT, Adiels M, Mitchell
TW, Blanksby SJ, Fagerberg B, Ekroos K and Borén J: Clinical
dyslipidemia is associated with changes in the lipid composition
and inflammatory properties of apolipoprotein- B containing
lipoproteins from women with type 2 diabetes. Diabetologia.
55:1156–1166. 2012.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wang Y, Botolin D, Xu J, Christian B,
Mitchell E, Jayaprakasam B, Nair MG, Peters JM, Busik JV, Olson LK,
et al: Regulation of hepatic fatty acid elongase and desaturase
expression in diabetes and obesity. J Lipid Res. 47:2028–2041.
2006.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Huo T, Cai S, Lu X, Sha Y, Yu M and Li F:
Metabonomic study of biochemical changes in the serum of type 2
diabetes mellitus patients after the treatment of metformin
hydrochloride. J Pharm Biomed Anal. 49:976–982. 2009.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Markgraf DF, Al-Hasani H and Lehr S:
Lipidomics-Reshaping the Analysis and Perception of Type 2
Diabetes. Int J Mol Sci. 17(1841)2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Wong G, Barlow CK, Weir JM, Jowett JB,
Magliano DJ, Zimmet P, Shaw J and Meikle PJ: Inclusion of plasma
lipid species improves classification of individuals at risk of
type 2 diabetes. PLoS One. 8(e76577)2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Rhee EP, Cheng S, Larson MG, Walford GA,
Lewis GD, McCabe E, Yang E, Farrell L, Fox CS, O'Donnell CJ, et al:
Lipid profiling identifies a triacylglycerol signature of insulin
resistance and improves diabetes prediction in humans. J Clin
Invest. 121:1402–1411. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
16
|
Vargas E and Sepulveda CMA: Biochemistry,
Insulin Metabolic Effects. In: StatPearls. Stat Pearls Publishing,
Treasure Island, FL, 2019. https://www.ncbi.nlm.nih.gov/books/NBK525983/.
Accessed April 21, 2019.
|
|
17
|
Betts GJ, Desaix P, Johnson E, Korol O,
Kruse D, Poe B, et al: Human Anatomy and Physiology. OpenStax
College, 2013.
|
|
18
|
Röder PV, Wu B, Liu Y and Han W:
Pancreatic regulation of glucose homeostasis. Exp Mol Med.
48(e219)2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Tokarz VL, MacDonald PE and Klip A: The
cell biology of systemic insulin function. J Cell Biol.
217:2273–2289. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Samuel VT and Shulman GI: Mechanisms for
insulin resistance: Common threads and missing links. Cell.
148:852–871. 2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
No authors listed. Diabetes is more
prevalent among the urban poor: A summary of the findings of the
ICMR-INDIAB Study. Curr Med Issues. 15:243–244. 2017.
|
|
22
|
Mohan V and Pradeepa R: Epidemiology of
Diabetes in different regions of India. Health Administrator XXII
1,. 2:1–18. 2009.
|
|
23
|
Tandon N, Anjana RM, Mohan V, Kaur T,
Afshin A, Ong K, Mukhopadhyay S, Thomas N, Bhatia E, Krishnan A, et
al: The increasing burden of diabetes and variations among the
states of India: The Global Burden of Disease Study 1990-2016 India
State-Level Disease Burden Initiative Diabetes Collaborators.
Lancet Glob Health. 6:1352–1362. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chawla A, Chawla R and Jaggi S:
Microvasular and macrovascular complications in diabetes mellitus:
Distinct or continuum? Indian J Endocrinol Metab. 20:546–551.
2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yajnik CS, Fall CH, Coyaji KJ, Hirve SS,
Rao S, Barker DJ, Joglekar C and Kellingray S: Neonatal
anthropometry: The thin-fat Indian baby. The Pune Maternal
Nutrition Study. Int J Obes Relat Metab Disord. 27:173–180.
2003.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Unnikrishnan RI, Rema M, Pradeepa R, Deepa
M, Shanthirani CS, Deepa R and Mohan V: Prevalence and risk factors
of diabetic nephropathy in an urban south Indian population. The
Chennai Urban Rural Epidemiology Study (CURES-45). Diabetes Care.
30:2019–2024. 2007.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Tan SY and Merchant J: Frederick Banting
(1891-1941): Discoverer of insulin. Singapore Med J. 58:2–3.
2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Pradeepa R, Rema M, Vignesh J, Deepa M,
Deepa R and Mohan V: Prevalence and risk factors for diabetic
neuropathy in an urban south Indian population: The Chennai Urban
Rural Epidemiology Study (CURES-55). Diabet Med. 25:407–412.
2008.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Mohan V, Deepa R, Rani SS and Premalatha
G: Chennai Urban Population Study (CUPS No.5). Prevalence of
coronary artery disease and its relationship to lipids in a
selected population in south India: The Chennai Urban Population
Study (CUPS No. 5). J Am CollCardiol. 38:682–687. 2001.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Premalatha G, Shanthirani CS, Deepa R,
Markovitz J and Mohan V: Prevalence and risk factors of peripheral
vascular disease in a selected south Indian population the Chennai
Urban Population Study (CUPS). Diabetes Care. 23:1295–1300.
2000.PubMed/NCBI View Article : Google Scholar
|
|
31
|
White MG, Shaw JA and Taylor R: Type 2
Diabetes: The Pathologic Basis of Reversible β-Cell Dysfunction.
Diabetes Care. 39:2080–2088. 2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Andersson-Hall U, Carlsson NG, Sandberg AS
and Holmäng A: Circulating Linoleic Acid is Associated with
Improved Glucose Tolerance in Women after Gestational Diabetes.
Nutrients. 10(1629)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Ertunc ME and Hotamisligil GS: Lipid
signaling and lipotoxicity in metaflammation: Indications for
metabolic disease pathogenesis and treatment. J Lipid Res.
57:2099–2114. 2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Palomer X, Pizarro-Delgado J, Barroso E
and Vázquez-Carrera M: Palmitic and Oleic Acid: The Yin and Yang of
Fatty Acids in Type 2 Diabetes Mellitus. Trends Endocrinol Metab.
29:178–190. 2018.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Wu HT, Chen W, Cheng KC, Ku PM, Yeh CH and
Cheng JT: Oleic acid activates peroxisome proliferator-activated
receptor δ to compensate insulin resistance in steatotic cells. J
Nutr Biochem. 23:1264–1270. 2012.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Simopoulos AP: The impact of the Bellagio
Report on healthy agriculture, healthy nutrition, healthy people:
Scientific and policy aspects and the International Network of
Centers for Genetics, Nutrition and Fitness for Health. J
Nutrigenet Nutrigenomics. 7:191–211. 2014.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Linus Pauling Institute: Essential Fatty
acids. https://lpi.oregonstate.edu/mic/other-nutrients/essential-fatty-acids#membrane-structure-function.
Accessed May, 2019.
|
|
38
|
Lopez S, Bermudez B, Ortega A, Varela LM,
Pacheco YM, Villar J, Abia R and Muriana FJ: Effects of meals rich
in either monounsaturated or saturated fat on lipid concentrations
and on insulin secretion and action in subjects with high fasting
triglyceride concentrations. Am J Clin Nutr. 93:494–499.
2011.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Bhaswant M, Poudyal H and Brown L:
Mechanisms of enhanced insulin secretion and sensitivity with n-3
unsaturated fatty acids. J Nutr Biochem. 26:571–584.
2015.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lepretti M, Martucciello S, Burgos Aceves
MA, Putti R and Lionetti L: Omega-3 Fatty Acids and Insulin
Resistance: Focus on the Regulation of Mitochondria and Endoplasmic
Reticulum Stress. Nutrients. 10(350)2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Day EA, Ford RJ and Steinberg GR: AMPK as
a therapeutic target for treating metabolic diseases. Trends
Endocrinol Metab. 28:545–560. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Pérez-Matute P, Pérez-Echarri N, Martínez
JA, Marti A and Moreno-Aliaga MJ: Eicosapentaenoic acid actions on
adiposity and insulin resistance in control and high-fat-fed rats:
Role of apoptosis, adiponectin and tumour necrosis factor-alpha. Br
J Nutr. 97:389–398. 2007.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Wang X and Chan CB: n-3 polyunsaturated
fatty acids and insulin secretion. J Endocrinol. 224:R97–R106.
2015.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Rimoldi OJ, Finarelli GS and Brenner RR:
Effects of diabetes and insulin on hepatic delta6 desaturase gene
expression. Biochem Biophys Res Commun. 283:323–326.
2001.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Mansouri V, Javanmard SH, Mahdavi M and
Tajedini MH: Association of Polymorphism in Fatty Acid Desaturase
Gene with the Risk of Type 2 Diabetes in Iranian Population. Adv
Biomed Res. 7(98)2018.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Kim OY, Lim HH, Yang LI, Chae JS and Lee
JH: Fatty acid desaturase (FADS) gene polymorphisms and insulin
resistance in association with serum phospholipid polyunsaturated
fatty acid composition in healthy Korean men: Cross-sectional
study. Nutr Metab (Lond). 8(24)2011.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Yary T, Voutilainen S, Tuomainen TP,
Ruusunen A, Nurmi T and Virtanen JK: Serum n-6 polyunsaturated
fatty acids, ∆5- and ∆6-desaturase activities, and risk of incident
type 2 diabetes in men: The Kuopio Ischaemic Heart Disease Risk
Factor Study. Am J Clin Nutr. 103:1337–1343. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Vessby B, Ahrén B, Warensjö E and
Lindgärde F: Plasma lipid fatty acid composition, desaturase
activities and insulin sensitivity in Amerindian women. Nutr Metab
Cardiovasc Dis. 22:176–181. 2012.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Das UN: A defect in the activity of Delta6
and Delta5 desaturases may be a factor predisposing to the
development of insulin resistance syndrome. Prostaglandins Leukot
Essent Fatty Acids. 72:343–350. 2005.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Mathias RA, Pani V and Chilton FH: Genetic
Variants in the FADS Gene: Implications for Dietary Recommendations
for Fatty Acid Intake. Curr Nutr Rep. 3:139–148. 2014.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Wu Y, Ding Y, Tanaka Y and Zhang W: Risk
factors contributing to type 2 diabetes and recent advances in the
treatment and prevention. Int J Med Sci. 11:1185–1200.
2014.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Mani I and Kurpad AV: Fats and fatty acids
in Indian diets: Time for serious introspection. Indian J Med Res.
144:507–514. 2016.PubMed/NCBI View Article : Google Scholar
|