|
1
|
Winsper C: The aetiology of borderline
personality disorder (BPD): Contemporary theories and putative
mechanisms. Curr Opin Psychol. 21:105–110. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dumpa V and Bhandari V: Surfactant,
steroids and non-invasive ventilation in the prevention of BPD.
Semin Perinatol. 42:444–452. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Mir IN, Chalak LF, Brown LS, Johnson-Welch
S, Heyne R, Rosenfeld CR and Kapadia VS: Impact of multiple
placental pathologies on neonatal death, bronchopulmonary
dysplasia, and neurodevelopmental impairment in preterm infants.
Pediatr Res. 87:885–891. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Maturu P, Wei-Liang Y, Androutsopoulos VP,
Jiang W, Wang L, Tsatsakis AM and Couroucli XI: Quercetin
attenuates the hyperoxic lung injury in neonatal mice: Implications
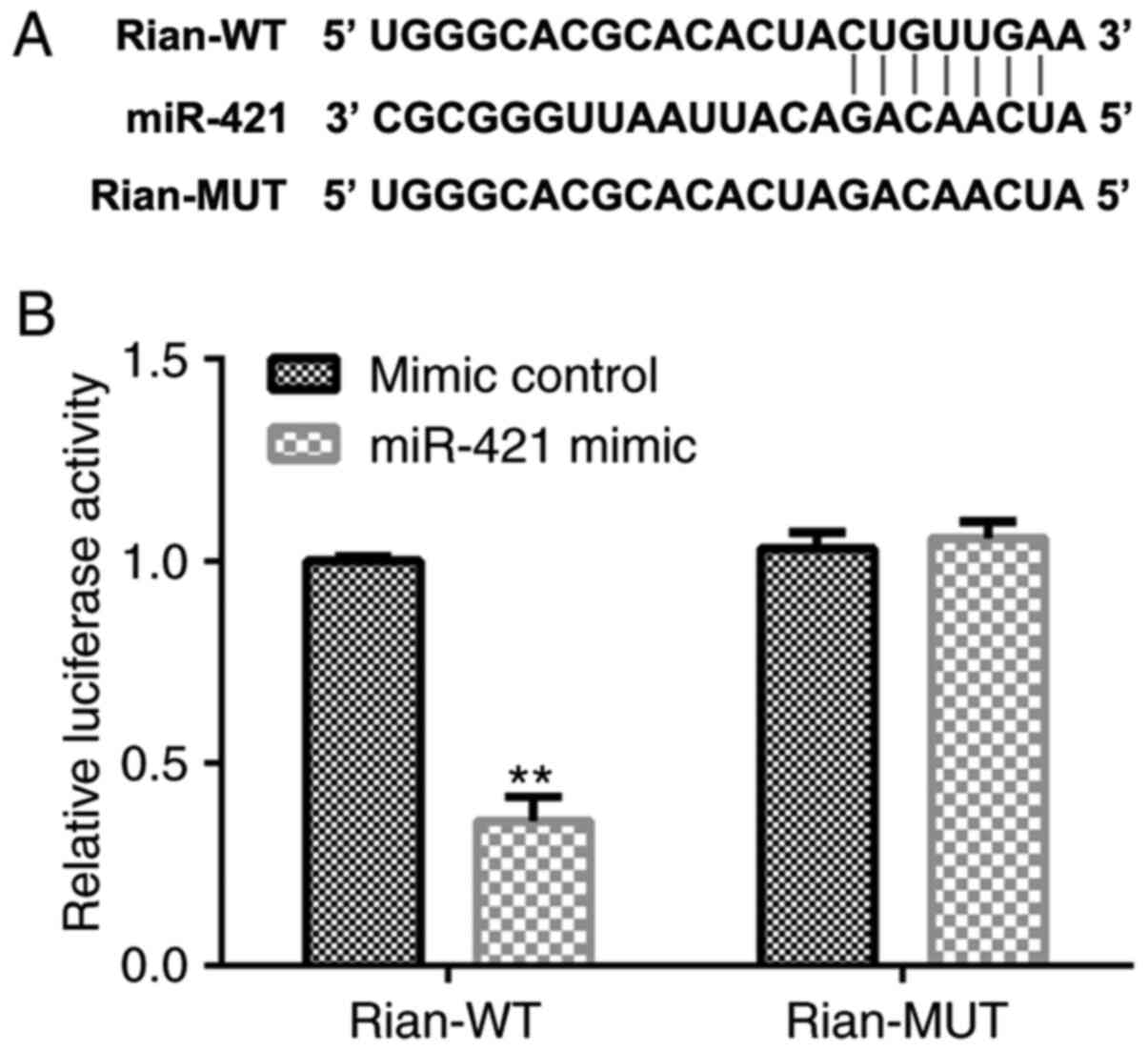
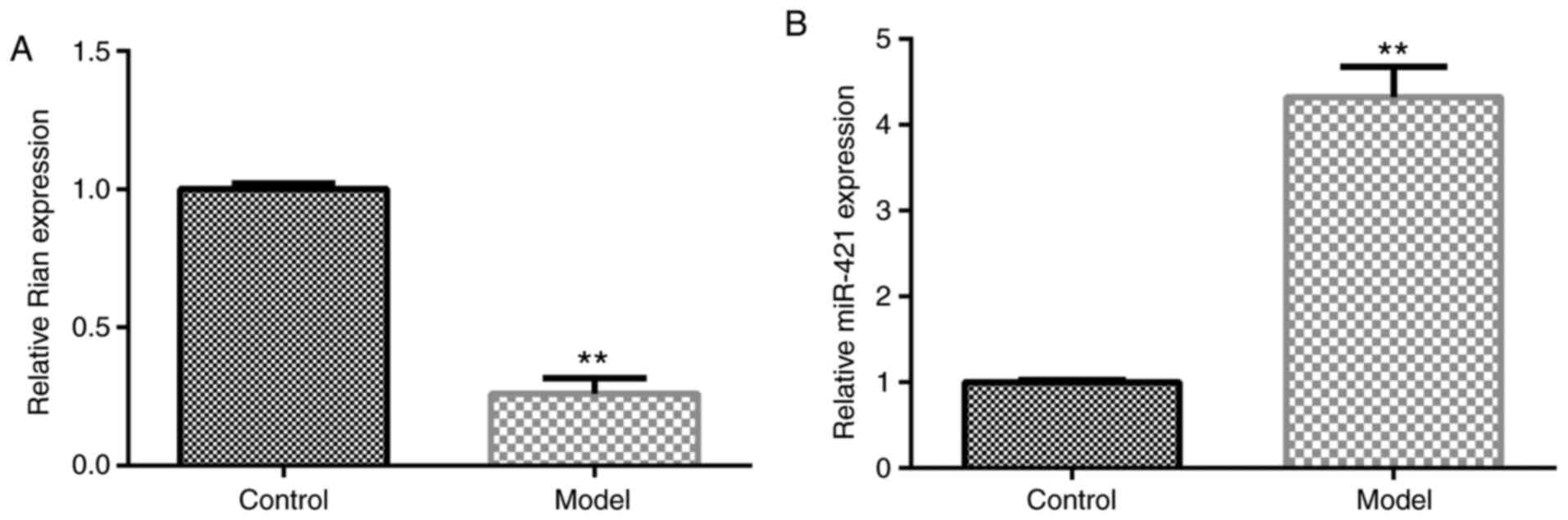
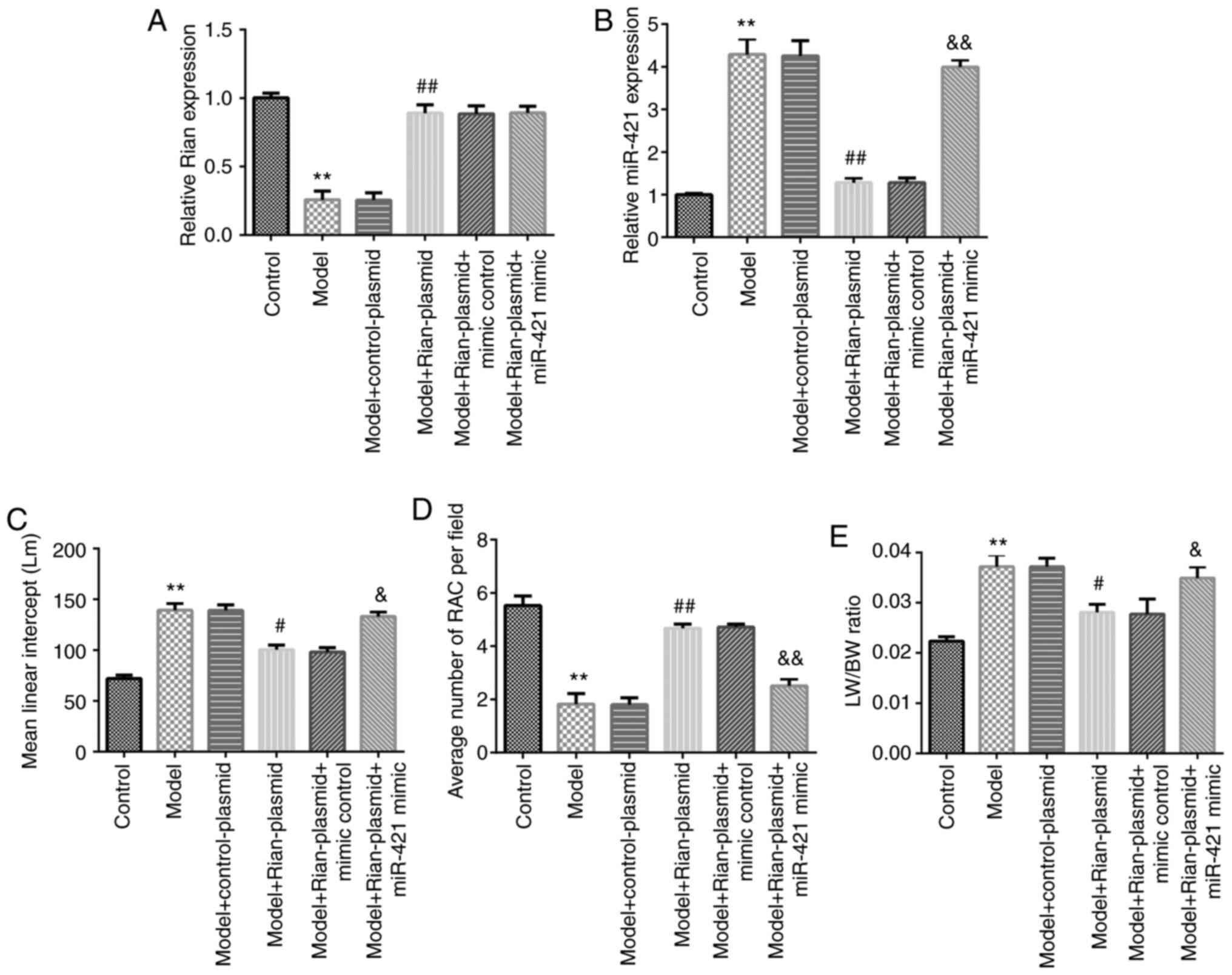
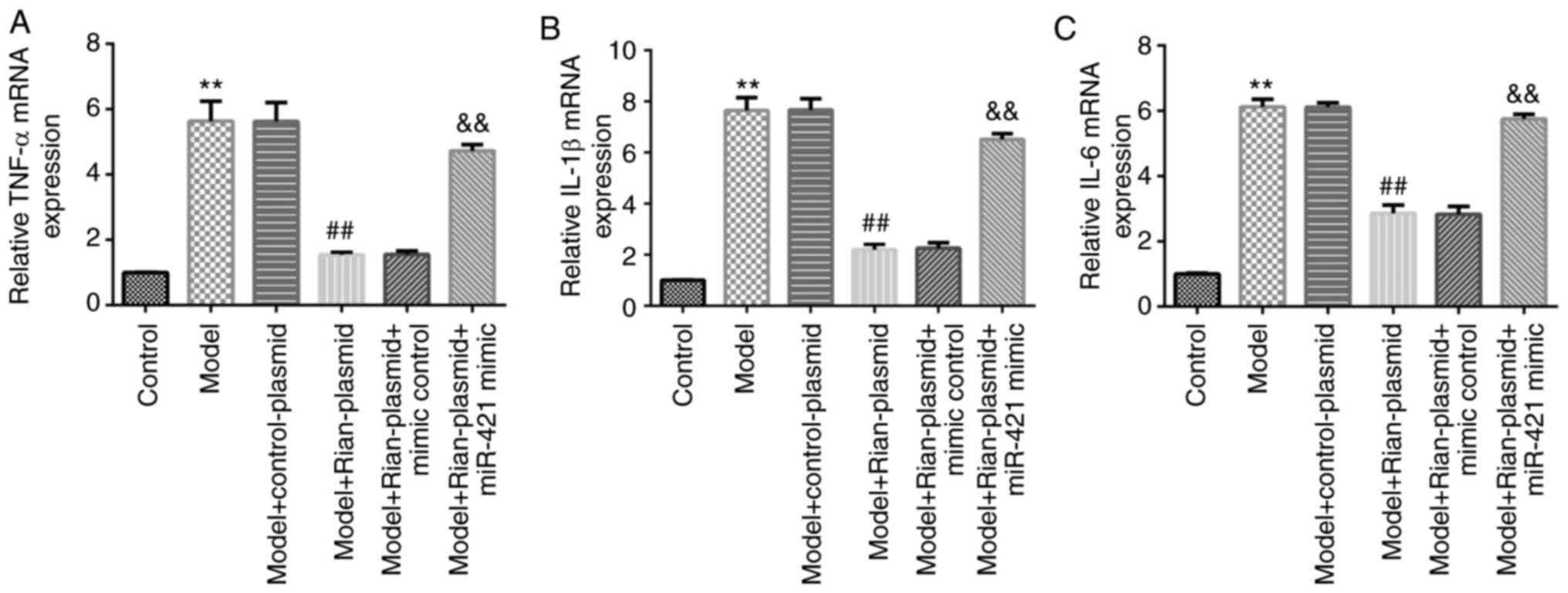
for Bronchopulmonary dysplasia (BPD). Food Chem Toxicol. 114:23–33.
2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Bashir RA, Bhandari V, Vayalthrikkovil S,
Rabi Y, Soraisham A, Tang S, Al Awad E and Lodha A:
Chorioamnionitis at birth does not increase the risk of
neurodevelopmental disability in premature infants with
bronchopulmonary dysplasia. Acta Paediatr. 105:e506–e512.
2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
De Paepe ME, Mao Q, Chao Y, Powell JL,
Rubin LP and Sharma S: Hyperoxia-induced apoptosis and Fas/FasL
expression in lung epithelial cells. Am J Physiol Lung Cell Mol
Physiol. 289:L647–L659. 2005.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kallapur GS and Jobe AH: Contribution of
inflammation to lung injury and development. Arch Dis Child Fetal
Neonatal Ed. 91:F132–F135. 2006.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Fiaturi N, Russo JW, Nielsen HC and
Castellot JJ Jr: CCN5 in alveolar epithelial proliferation and
differentiation during neonatal lung oxygen injury. J Cell Commun
Signal. 12:217–229. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Ratner V, Slinko S, Utkina-Sosunova I,
Starkov A, Polin RA and Ten VS: Hypoxic stress exacerbates
hyperoxia-induced lung injury in a neonatal mouse model of
bronchopulmonary dysplasia. Neonatology. 95:299–305.
2009.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zhu Y, Fu J, Yang H, Pan Y, Yao L and Xue
X: Hyperoxia-induced methylation decreases RUNX3 in a newborn rat
model of bronchopulmonary dysplasia. Respir Res.
16(75)2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mai LJ, Fu XX, He G, Zhao EN and Xue M:
Effect of asiaticoside on hyperoxia-induced bronchopulmonary
dysplasia in neonatal rats and related mechanism. Zhongguo Dang Dai
Er Ke Za Zhi. 22:71–76. 2020.PubMed/NCBI View Article : Google Scholar : (In Chinese).
|
|
12
|
Hu Y, Xie L, Yu J, Fu H, Zhou D and Liu H:
Inhibition of microRNA-29a alleviates hyperoxia-induced
bronchopulmonary dysplasia in neonatal mice via upregulation of
GAB1. Mol Med. 26(3)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Zhang T, Chen J, Wu H, Pan W, Yang X, Li
Y, Liu M and Huang Y: Improved survival and survival without
bronchopulmonary dysplasia in very low birth weight infants after
active perinatal care. Niger J Clin Pract. 23:980–987.
2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Salviano-Silva A, Lobo-Alves SC, Almeida
RC, Malheiros D and Petzl-Erler ML: Besides pathology: Long
non-coding RNA in cell and tissue homeostasis. Noncoding RNA.
4(3)2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Bijkerk R, Au YW, Stam W, Duijs JMGJ,
Koudijs A, Lievers E, Rabelink TJ and van Zonneveld AJ: Long
non-coding RNAs Rian and Miat mediate myofibroblast formation in
kidney fibrosis. Front Pharmacol. 10(215)2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Gu T, He H, Xing Y, Liu Q, Gu N, Kenkichi
S, Jiang H and Wu Q: Expression of non-coding RNA AB063319 derived
from Rian gene during mouse development. J Mol Histol. 42:105–112.
2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yao P, Li YL, Chen Y, Shen W, Wu KY and Xu
WH: Overexpression of long non-coding RNA Rian attenuates cell
apoptosis from cerebral ischemia-reperfusion injury via
Rian/miR-144-3p/GATA3 signaling. Gene. 737(144411)2020.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Zhong L, Jia J and Ye G:
Rian/miR-210-3p/Nfkb1 feedback loop promotes hypoxia-induced cell
apoptosis in myocardial infarction through deactivating the
PI3K/Akt signaling pathway. J Cardiovasc Pharmacol. 76:207–215.
2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Liu B, Li J and Cairns MJ: Identifying
miRNAs, targets and functions. Brief Bioinform. 15:1–19.
2014.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Dupont C, Kappeler L, Saget S, Grandjean V
and Levy R: Role of miRNA in the transmission of metabolic diseases
associated with paternal diet-induced obesity. Front Genet.
10(337)2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Chen J, Wu L, Sun Y, Yin Q, Chen X, Liang
S, Meng Q, Long H, Li F, Luo C and Xiao X: Mir-421 in plasma as a
potential diagnostic biomarker for precancerous gastric lesions and
early gastric cancer. PeerJ. 7(e7002)2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ren Z, He M, Shen T, Wang K, Meng Q, Chen
X, Zhou L, Han Y, Ji C, Liu S and Fu Q: MiR-421 promotes the
development of osteosarcoma by regulating MCPIP1 expression. Cancer
Biol Ther. 21:231–240. 2020.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Yin Y, Xu L, Chang Y, Zeng T, Chen X, Wang
A, Groth J, Foo WC, Liang C, Hu H and Huang J: N-Myc promotes
therapeutic resistance development of neuroendocrine prostate
cancer by differentially regulating miR-421/ATM pathway. Mol
Cancer. 18(11)2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Chen L, Tang Y, Wang J, Yan Z and Xu R:
miR-421 induces cell proliferation and apoptosis resistance in
human nasopharyngeal carcinoma via downregulation of FOXO4. Biochem
Biophys Res Commun. 435:745–750. 2013.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yuan HS, Xiong DQ, Huang F, Cui J and Luo
H: MicroRNA-421 inhibition alleviates bronchopulmonary dysplasia in
a mouse model via targeting Fgf10. J Cell Biochem. 120:16876–16887.
2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Park MS, Rieger-Fackeldey E, Schanbacher
BL, Cook AC, Bauer JA, Rogers LK, Hansen TN, Welty SE and Smith CV:
Altered expressions of fibroblast growth factor receptors and
alveolarization in neonatal mice exposed to 85% oxygen. Pediatr
Res. 62:652–657. 2007.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Li JH, Liu S, Zhou H, Qu LH and Yang JH:
starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA
interaction networks from large-scale CLIP-Seq data. Nucleic Acids
Res. 42 (Database Issue):D92–D97. 2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Clément T, Salone V and Rederstorff M:
Dual luciferase gene reporter assays to study miRNA function.
Methods Mol Biol. 1296:187–198. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Chen X, Zhang X and Pan J: Effect of
montelukast on bronchopulmonary dysplasia (BPD) and related
mechanisms. Med Sci Monit. 25:1886–1893. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chen X, Peng W, Zhou R, Zhang Z and Xu J:
Montelukast improves bronchopulmonary dysplasia by inhibiting
epithelial-mesenchymal transition via inactivating the TGF-β1/Smads
signaling pathway. Mol Med Rep. 22:2564–2572. 2020.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Stark A, Dammann C, Nielsen HC and Volpe
MV: A pathogenic relationship of bronchopulmonary dysplasia and
retinopathy of prematurity? A review of angiogenic mediators in
both diseases. Front Pediatr. 6(125)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Kalikkot TR, Guaman MC and Shivanna B:
Bronchopulmonary dysplasia: A review of pathogenesis and
pathophysiology. Respir Med. 132:170–177. 2017.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Principi N, Di Pietro GM and Esposito S:
Bronchopulmonary dysplasia: Clinical aspects and preventive and
therapeutic strategies. J Transl Med. 16(36)2018.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Hwang JS and Rehan VK: Recent advances in
bronchopulmonary dysplasia: Pathophysiology, prevention, and
treatment. Lung. 196:129–138. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
van Rossem MC, van de Loo M, Laan BJ, de
Sonnaville ES, Tamminga P, van Kaam AH and Onland W: Accuracy of
the diagnosis of bronchopulmonary dysplasia in a referral-based
health care system. J Pediatr. 167:540–544.e1. 2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Abman SH, Collaco JM, Shepherd EG, Keszler
M, Cuevas-Guaman M, Welty SE, Truog WE, McGrath-Morrow SA, Moore
PE, Rhein LM, et al: Interdisciplinary care of children with severe
bronchopulmonary dysplasia. J Pediatr. 181:12–28. 2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Cai Q, Wang S, Jin L, Weng M, Zhou D, Wang
J, Tang Z and Quan Z: Long non-coding RNA GBCDRlnc1 induces
chemoresistance of gallbladder cancer cells by activating
autophagy. Mol Cancer. 18(82)2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Gong X, Qiu J, Qiu G and Cai C:
Adrenomedullin regulated by miRNA-574-3p protects premature infants
with bronchopulmonary dysplasia. Biosci Rep.
40(BSR20191879)2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Wang J, Yin J, Wang X, Liu H, Hu Y, Yan X,
Zhuang B, Yu Z and Han S: Changing expression profiles of mRNA,
lncRNA, circRNA and miRNA in lung tissue reveal the
pathophysiological of bronchopulmonary dysplasia (BPD) in mouse
model. J Cell Biochem. 120:9369–9380. 2019.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Sun Z, Shi K, Yang S, Liu J, Zhou Q, Wang
G, Song J, Li Z, Zhang Z and Yuan W: Effect of exosomal miRNA on
cancer biology and clinical applications. Mol Cancer.
17(147)2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Xu B, Liu J, Xiang X, Liu S, Zhong P, Xie
F, Mou T and Lai L: Expression of miRNA-143 in pancreatic cancer
and its clinical significance. Cancer Biother Radiopharm.
33:373–379. 2018.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Hanna Y, Laliberté C, Ben Fadel N, Lemyre
B, Thébaud B, Barrowman N, Bijelic V, Hoey L and Katz SL: Effect of
oxygen saturation targets on the incidence of bronchopulmonary
dysplasia and duration of respiratory supports in extremely preterm
infants. Paediatr Child Health. 25:173–9. 2020.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kaneko M, Sato M, Ogasawara K, Imamura T,
Hashimoto K, Momoi N and Hosoya M: Serum cytokine concentrations,
chorioamnionitis and the onset of bronchopulmonary dysplasia in
premature infants. J Neonatal Perinatal Med. 10:147–155.
2017.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Dias-Freitas F, Metelo-Coimbra C and
Roncon-Albuquerque RJ: Molecular mechanisms underlying hyperoxia
acute lung injury. Respir Med. 119:23–28. 2016.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Yin R, Yuan L, Ping L and Hu L: Neonatal
bronchopulmonary dysplasia increases neuronal apoptosis in the
hippocampus through the HIF-1alpha and p53 pathways. Respir Physiol
Neurobiol. 220:81–87. 2016.PubMed/NCBI View Article : Google Scholar
|