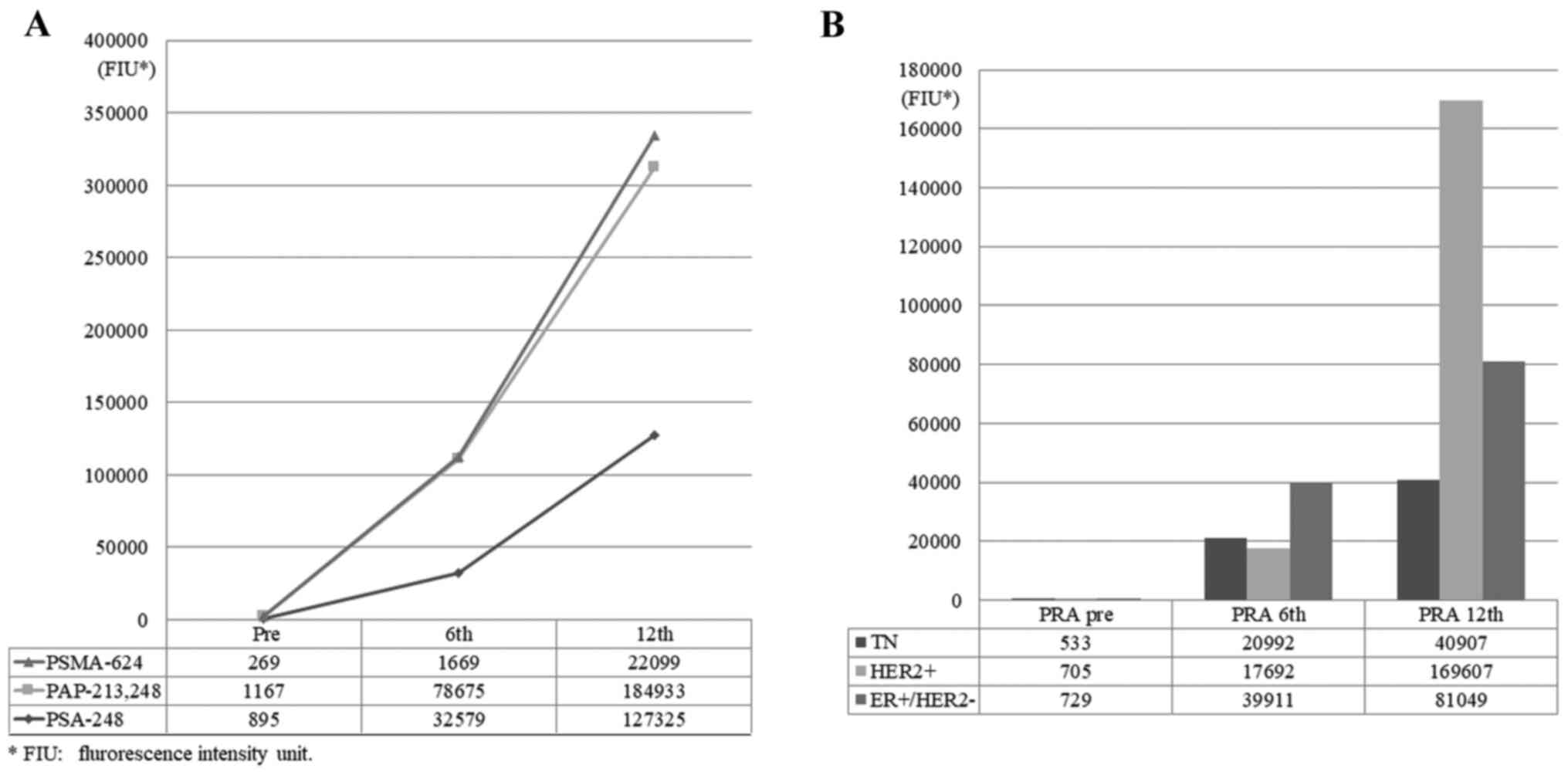
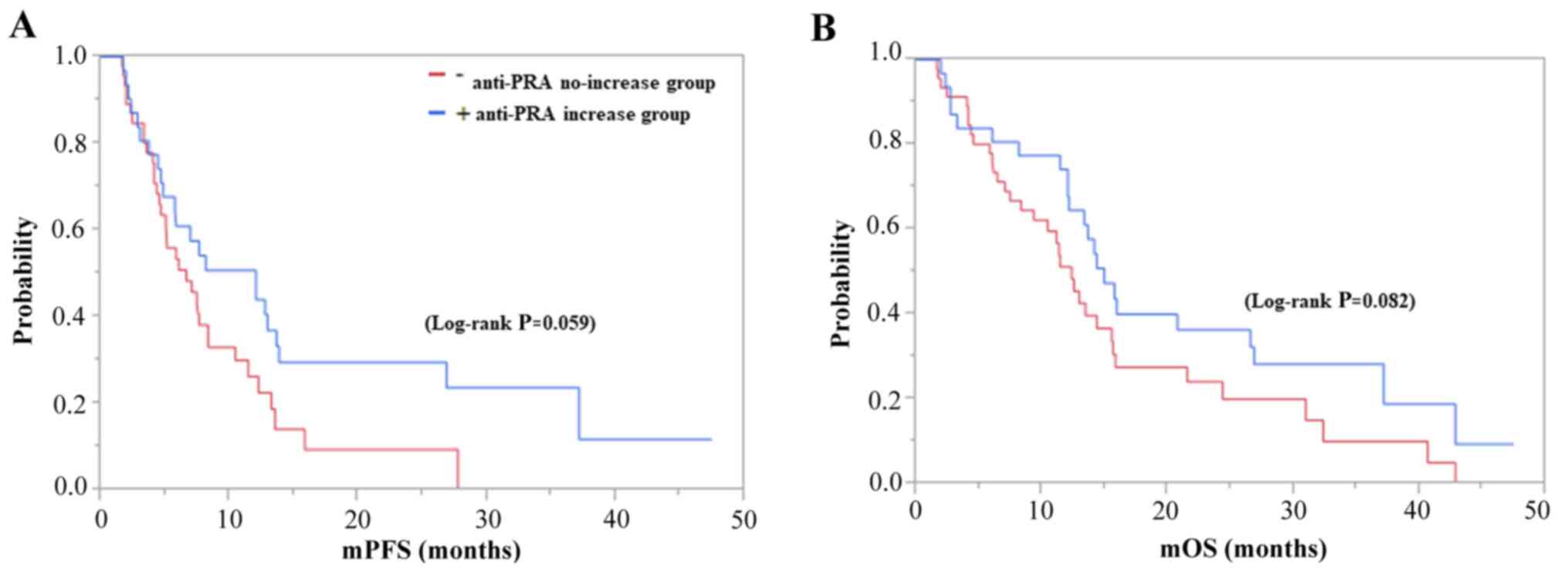
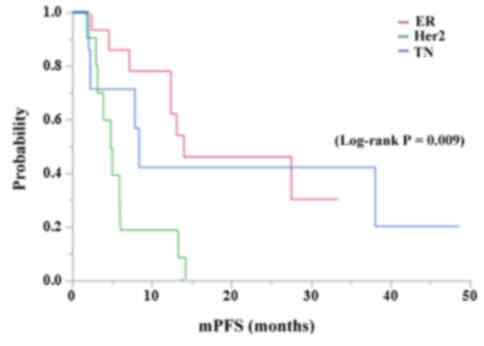
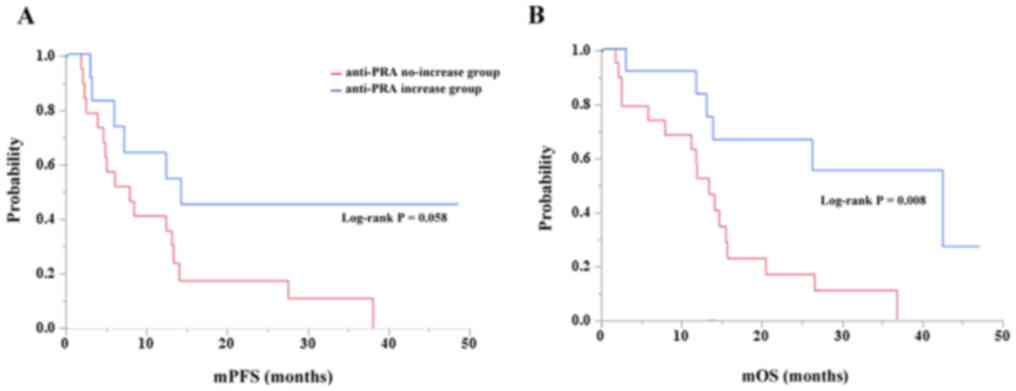
|
1
|
Yu H, Diamandis EP, Levesque M, Giai M,
Roagna R, Ponzone R, Sismondi P, Monne M and Croce CM: Prostate
specific antigen in breast cancer, benign breast disease and normal
breast tissue. Breast Cancer Res Treat. 40:171–178. 1996.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Black MH and Diamandis EP: The diagnostic
and prognostic utility of prostate-specific antigen for diseases of
the breast. Breast Cancer Res Treat. 59:1–14. 2000.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Mannello F and Gazzanelli G:
Prostate-specific antigen (PSA/hK3): A further player in the field
of breast cancer diagnostics? Breast Cancer Res. 3:238–243.
2001.PubMed/NCBI View
Article : Google Scholar
|
|
4
|
Alanen KA, Kuopio T, Collan YU, Kronqvist
P, Juntti L and Nevalainen TJ: Immunohistochemical labelling for
prostate-specific antigen in breast carcinomas. Breast Cancer Res
Treat. 56:169–176. 1999.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Kantoff PW, Schuetz TJ, Blumenstein BA,
Glode LM, Bilhartz DL, Wyand M, Manson K, Panicali DL, Laus R,
Schlom J, et al: Overall survival analysis of a phase II randomized
controlled trial of a Poxviral-based PSA-targeted immunotherapy in
metastatic castration-resistant prostate cancer. J Clin Oncol.
28:1099–1105. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Gulley JL, Madan RA, Tsang KY, Jochems C,
Marté JL, Farsaci B, Tucker JA, Hodge JW, Liewehr DJ, Steinberg SM,
et al: Immune impact induced by PROSTVAC (PSA-TRICOM), a
therapeutic vaccine for prostate cancer. Cancer Immunol Res.
2:133–141. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Takahashi R, Toh U, Iwakuma N, Takenaka M,
Otsuka H, Furukawa M, Fujii T, Seki N, Kawahara A, Kage M, et al:
Feasibility study of personalized peptide vaccination for
metastatic recurrent triple-negative breast cancer patients. Breast
Cancer Res. 16(R70)2014.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Sasada T, Komatsu N, Suekane S, Yamada A,
Noguchi M and Itoh K: Overcoming the hurdles of randomised clinical
trials of therapeutic cancer vaccines. Eur J Cancer. 46:1514–1519.
2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Terasaki M, Shibui S, Narita Y, Fujimaki
T, Aoki T, Kajiwara K, Sawamura Y, Kurisu K, Mineta T, Yamada A and
Itoh K: Phase I trial of a personalized peptide vaccine for
patients positive for human leukocyte antigen-A24 with recurrent or
progressive glioblastoma multiforme. J Clin Oncol. 29:337–344.
2011.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Yanagimoto H, Shiomi H, Satoi S, Mine T,
Toyokawa H, Yamamoto T, Tani T, Yamada A, Kwon AH, Komatsu N, et
al: A phase II study of personalized peptide vaccination combined
with gemcitabine for non-resectable pancreatic cancer patients.
Oncol Rep. 24:795–801. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Hattori T, Mine T, Komatsu N, Yamada A,
Itoh K, Shiozaki H and Okuno K: Immunological evaluation of
personalized peptide vaccination in combination with UFT and UZEL
for metastatic colorectal carcinoma patients. Cancer Immunol
Immunother. 58:1843–1852. 2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Noguchi M, Mine T, Komatsu N, Suekane S,
Moriya F, Matsuoka K, Yutani S, Shichijo S, Yamada A, Toh U, et al:
Assessment of immunological biomarkers in patients with advanced
cancer treated by personalized peptide vaccination. Cancer Biol
Ther. 10:1266–1279. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Komatsu N, Shichijo S, Nakagawa M and Itoh
K: New multiplexed flow cytometric assay to measure anti-peptide
antibody: A novel tool for monitoring immune responses to peptides
used for immunization. Scand J Clin Lab Invest. 64:535–545.
2004.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Matsueda S, Komatsu N, Kusumoto K, Koga S,
Yamada A, Kuromatsu R, Yamada S, Seki R, Yutani S, Shichijo S, et
al: Humoral immune responses to CTL epitope peptides from
tumor-associated antigens are widely detectable in humans: A new
biomarker for overall survival of patients with malignant diseases.
Dev Comp Immunol. 41:68–76. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Gulley JL, Arlen PM, Bastian A, Morin S,
Marte J, Beetham P, Tsang KY, Yokokawa J, Hodge JW, Ménard C, et
al: Combining a recombinant cancer vaccine with standard definitive
radiotherapy in patients with localized prostate cancer. Clin
Cancer Res. 11:3353–3362. 2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Noguchi M, Koga N, Moriya F and Itoh K:
Immunotherapy in prostate cancer: Challenges and opportunities.
Immunotherapy. 8:69–77. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yu H, Levesque MA, Clark GM and Diamandis
EP: Prognostic value of prostate-specific antigen for women with
breast cancer: A large United States cohort study. Clin Cancer Res.
4:1489–1497. 1998.PubMed/NCBI
|
|
18
|
Takenaka M, Seki N, Toh U, Hattori S,
Kawahara A, Yamaguchi T, Koura K, Takahashi R, Otsuka H, Takahashi
H, et al: FOXP3 expression in tumor cells and tumor-infiltrating
lymphocytes is associated with breast cancer prognosis. Mol Clin
Oncol. 1:625–632. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Okabe M, Toh U, Iwakuma N, Saku S, Akashi
M, Kimitsuki Y, Seki N, Kawahara A, Ogo E, Itoh K and Akagi Y:
Predictive factors of the tumor immunological microenvironment for
long-term follow-up in early stage breast cancer. Cancer Sci.
108:81–90. 2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Sakamoto S, Matsueda S, Takamori S, Toh U,
Noguchi M, Yutani S, Yamada A, Shichijo S, Yamada T, Suekane S, et
al: Immunological evaluation of peptide vaccination for cancer
patients with the HLA-A11(+) or -A33(+) allele. Cancer Sci.
108:598–603. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Disis ML, Wallace DR, Gooley TA, Dang Y,
Slota M, Lu H, Coveler AL, Childs JS, Higgins DM, Fintak PA, et al:
Concurrent trastuzumab and HER2/neu-specific vaccination in
patients with metastatic breast cancer. J Clin Oncol. 27:4685–4692.
2009.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Higgins M, Curigliano G, Dieras V, Kuemmel
S, Kunz G, Fasching PA, Campone M, Bachelot T, Krivorotko P, Chan
S, et al: Safety and immunogenicity of neoadjuvant treatment using
WT1-immunotherapeutic in combination with standard therapy in
patients with WT1-positive Stage II/III breast cancer: A randomized
phase I study. Breast Cancer Res Treat. 162:479–488.
2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Isla Larrain MT, Colussi AG, Demichelis
SO, Barbera A, Creton A, Segal-Eiras A and Croce MV: Humoral immune
response against tumoral mucin 1 (MUC1) in breast cancer patients.
Int J Biol Markers. 28:318–325. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Clive KS, Tyler JA, Clifton GT, Holmes JP,
Ponniah S, Peoples GE and Mittendorf EA: The GP2 peptide: A
HER2/neu-based breast cancer vaccine. J Surg Oncol. 105:452–458.
2012.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mittendorf EA, Clifton GT, Holmes JP,
Schneble E, van Echo D, Ponniah S and Peoples GE: Final report of
the phase I/II clinical trial of the E75 (nelipepimut-S) vaccine
with booster inoculations to prevent disease recurrence in
high-risk breast cancer patients. Ann Oncol. 25:1735–1742.
2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Disis ML, Goodell V, Schiffman K and
Knutson KL: Humoral epitope-spreading following immunization with a
HER-2/neu peptide based vaccine in cancer patients. J Clin Immunol.
24:571–578. 2004.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Disis ML, Schiffman K, Guthrie K, Salazar
LG, Knutson KL, Goodell V, dela Rosa C and Cheever MA: Effect of
dose on immune response in patients vaccinated with an her-2/neu
intracellular domain protein-based vaccine. J Clin Oncol.
22:1916–1925. 2004.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Kawano K, Tsuda N, Matsueda S, Sasada T,
Watanabe N, Ushijima K, Yamaguchi T, Yokomine M, Itoh K, Yamada A
and Kamura T: Feasibility study of personalized peptide vaccination
for recurrent ovarian cancer patients. Immunopharmacol
Immunotoxicol. 36:224–236. 2014.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Kawano K, Tsuda N, Waki K, Matsueda S,
Hata Y, Ushijima K, Itoh K, Yamada A and Kamura T: Personalized
peptide vaccination for cervical cancer patients who have received
prior platinum-based chemotherapy. Cancer Sci. 106:1111–1117.
2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zarghami N, Grass L and Diamandis EP:
Steroid hormone regulation of prostate-specific antigen gene
expression in breast cancer. Br J Cancer. 75:579–588.
1997.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yu H, Diamandis EP, Zarghami N and Grass
L: Induction of prostate specific antigen production by steroids
and tamoxifen in breast cancer cell lines. Breast Cancer Res Treat.
32:291–300. 1994.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Zarghami N and Diamandis EP: Detection of
prostate-specific antigen mRNA and protein in breast tumors. Clin
Chem. 42:361–366. 1996.PubMed/NCBI
|
|
33
|
Hu R, Dawood S, Holmes MD, Collins LC,
Schnitt SJ, Cole K, Marotti JD, Hankinson SE, Colditz GA and Tamimi
RM: Androgen receptor expression and breast cancer survival in
postmenopausal women. Clin Cancer Res. 17:1867–1874.
2001.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Vera-Badillo FE, Templeton AJ, de Gouveia
P, Diaz-Padilla I, Bedard PL, Al-Mubarak M, Seruga B, Tannock IF,
Ocana A and Amir E: Androgen receptor expression and outcomes in
early breast cancer: A systematic review and meta-analysis. J Natl
Cancer Inst. 106(djt319)2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Bianchini G and Gianni L: The immune
system and response to HER2-targeted treatment in breast cancer.
Lancet Oncol. 15:e58–e68. 2014.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Bianchini G, Pusztai L, Pienkowski T, Im
YH, Bianchi GV, Tseng LM, Liu MC, Lluch A, Galeota E, Magazzù D, et
al: Immune modulation of pathologic complete response after
neoadjuvant HER2-directed therapies in the NeoSphere trial. Ann
Oncol. 26:2429–2436. 2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Soliman H: Immunotherapy strategies in the
treatment of breast cancer. Cancer Control. 20:17–21.
2013.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Gober HJ, Kistowska M, Angman L, Jenö P,
Mori L and De Libero G: Human T cell receptor gammadelta cells
recognize endogenous mevalonate metabolites in tumor cells. J Exp
Med. 197:163–168. 2003.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Hewitt RE, Lissina A, Green AE, Slay ES,
Price DA and Sewell AK: The bisphosphonate acute phase response:
rapid and copious production of proinflammatory cytokines by
peripheral blood gd T cells in response to aminobisphosphonates is
inhibited by statins. Clin Exp Immunol. 139:101–111.
2005.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Cheng ML and Fong L: Effects of
RANKL-targeted therapy in immunity and cancer. Front Oncol.
3(329)2014.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Tan W, Zhang W, Strasner A, Grivennikov S,
Cheng JQ, Hoffman RM and Karin M: Tumour-infiltrating regulatory T
cells stimulate mammary cancer metastasis through RANKL-RANK
signalling. Nature. 470:548–553. 2011.PubMed/NCBI View Article : Google Scholar
|