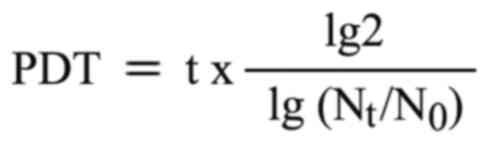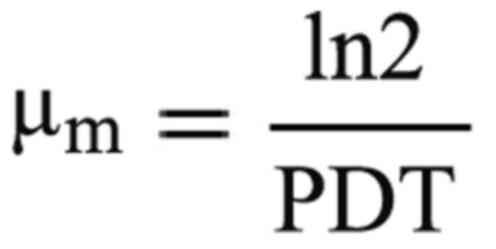Introduction
Diabetes is a chronic disease that occurs when the
pancreas is no longer able to provide insulin or when the body
cannot make use of the insulin it produces (1). According to data from the
International Diabetes Federation, ~463 million adults suffered
from diabetes in 2019 (https://www.idf.org). Approximately 10% of them had
type 1 diabetes (TID). However, there are still no effective
methods for the treatment of the disease. Individuals with TID
depend on daily insulin injections to maintain their blood glucose
levels. In recent years, the transplantation of stem cells with
multi-directional differentiation potential to replace the islet
cells has become a novel treatment strategy (2). Clinical trials with mesenchymal stem
cell (MSC)-based therapies are currently limited (3). A study using intravenous infusion of
bone marrow MSCs (BMMSCs) demonstrated improvement of C-peptide at
one year (4), which suggested the
potential of MSCs in the treatment of diabetes. The present study
first compared the differentiation potential of three types of MSC
into islet β-like cells in vitro.
These three types of MSC were umbilical cord-derived
MSCs (UCMSCs), dental pulp MSCs (DPSCs) and menstrual blood-derived
MSCs (MENSCs), which were isolated from the Wharton's jelly tissue
of the human umbilical cord (5),
deciduous teeth (6) and menstrual
blood (7), respectively. The three
MSC types share advantages in their clinical application, such as
convenient harvesting procedures, high proliferation,
multi-directional differentiation capacity and low immunogenicity
(8). Studies have indicated that
UCMSCs may be induced to form cartilage, bone, adipose and nerve
cells (9). Numerous studies
assessed the differentiation potency of UCMSCs into pancreatic
cells in vitro, which proved their differentiation ability
(10). In the present study, UCMSCs
were used as a judgement standard to compare the differentiation
ability of DPSCs and MENSCs. DPSCs are one of the novel types of
MSCs that have been proposed for tissue regeneration to repair bone
defects (11). DPSCs are obtained
from human exfoliated deciduous teeth, having the characteristics
of being easy to obtain with little damage caused to donors
(12). It has been demonstrated
that MENSCs have the ability to differentiate into osteogenic,
neurogenic and chondrogenic cell lineages (13), which prompts the potential of MENSCs
for use in regenerative medicine research. The isolation of MENSCs
features reproducibility and no damage to donors, which means the
source of MENSCs is rich and they are easy to obtain (7). Previous studies addressed the
differentiation ability of the three types of MSC, particularly
MENSCs. In addition to comparing the differentiation ability of
three types of MSC, it was explored whether MENSCs are able to
differentiate into islet cells as a novel clinical application.
As for the methodology, the previous differentiation
methods were improved (14), and in
combination with the results of the experiments, an optimized
differentiation method was established. Collagen coating is used to
promote the proliferation and differentiation ability of cells
(15), among the different
collagens, rat tail collagen is easy to obtain and mostly used
(16,17). In this procedure, 12-well cell
culture plates were pre-incubated with rat tail collagen, which
improved the differentiation ability in comparison with the
previous method without collagen coating.
The biological characteristics of these MSCs,
including proliferation, survival and differentiation capacities,
were assessed in former studies (18). Other biological characteristics,
particularly the evaluation of differentiation ability into
pancreatic β-like cells, were performed in the present study.
Although several types of MSC were reported to be able to
differentiate into pancreatic β-like cells in vitro,
including BMMSCs, adipose-derived MSCs (ADSCs) and UCMSCs, the
curative effect was not ideal in vivo (19-21).
The purpose of the present study was to screen novel sources of
MSCs for the treatment of diabetes and to provide a new scope for
pre-clinical trials in vivo. The present study will aid in
future decision-making for choosing suitable seed cells for the
treatment of TID in clinical trials.
Materials and methods
Isolation and culture of MENSCs,
UCMSCs and DPSCs
Isolation of MENSCs, UCMSCs and DPSCs was performed
at the Cell Culture & Bioprocess Engineering Laboratory,
Shanghai Jiaotong University (Shanghai, China). Relevant materials
and samples were provided by Shanghai Kun'ai Biological Technology
Co., Ltd., (Shanghai, China). The present study was approved by the
Ethics Committee of the School of Pharmacy, Shanghai Jiaotong
University (Shanghai, China).
UCMSCs
Umbilical cord tissues were collected from a
35-year-old mother and full-term fetus after caesarean section in
Handan Central Hospital (Handan, China) in March 2014. The donor
had a physical examination to rule out infectious diseases. The
tissues were stored at 4˚C and transported to Shanghai. The
isolation of UCMSCs was performed in the laboratory of Shanghai
Jiao Tong University. Collected umbilical cord tissues were washed
with PBS several times to remove red blood cells. After Wharton's
jelly tissues were removed from umbilical cords, they were chopped
into small pieces and placed on 55-mm2 plates at 37˚C
for 2 h. The tissues adhered to the plates to avoid suspension of
the pieces when medium was added. Then the plates were covered with
α-MEM medium (Gibco; Thermo Fisher Scientific, Inc.) containing 15%
FBS (HyClone; Cytiva).
MENSCs
Menstrual blood was obtained from a 39-year-old
female without abnormal discharge or infection by sterile Diva Cup
on the second day of flow in October 2014 at the donor's home
(Shanghai, China). The donor was tested for infectious diseases,
blood chemistry indicators and ultrasound examinations to ensure
health. The isolation of MENSCs was performed in the laboratory of
Shanghai Jiao Tong University. The collected menstrual blood (5 ml)
was mixed with an equal volume of PBS, 0.2 ml amphotericin B
(Invitrogen; Thermo Fisher Scientific, Inc.), 0.2 ml streptomycin
(Invitrogen; Thermo Fisher Scientific, Inc.) and 0.1 ml
EDTA-Na2 (Invitrogen; Thermo Fisher Scientific, Inc.) at
4˚C within 24 h of collection. Density gradient centrifugation was
applied twice at 200 x g at 4˚C for 10 min, followed by discarding
the supernatant and adding fresh medium containing 15% FBS for cell
culture.
DPSCs
Dental pulp tissues were obtained from shed
deciduous teeth. The tissues were donated from a 15-year-old male
in April 2014 at the donor's home (Shanghai, China). The isolation
of DPSCs was operated in the laboratory. The dental pulp tissues
were gently separated from the root and the crown. Then, the
tissues were minced and dissociated in 0.3% collagenase type I
(cat. no. SCR103; Sigma-Aldrich; Merck KgaA) at 37˚C for 1 h.
Subsequently, dissociation was terminated by adding α-MEM with 10%
FBS, followed by centrifugation at 100 x g at 4˚C for 5 min. To the
sediment, α-MEM containing 15% FBS was added to re-suspend the
cells and single cell suspensions were cultured as primary MSCs in
6-well plates at a concentration of 1x105
cells/well.
The suspension density of isolated MENSCs, UCMSCs
and DPSCs primary culture cells was adjusted to 2x106
cells/ml. These cells were cultured in 6-well cell culture plates.
Cells that failed to stick to the walls were discarded after 48 h.
The medium was changed every two days. The morphology of growing
cells was observed under an inverted phase contrast microscope
(Olympus Corporation).
Cell surface markers on MENSCs, UCMSCs
and DPSCs
MENSCs, UCMSCs and DPSCs at passage 4 (P4) were
trypsinized with 0.25% TrypLE (Invitrogen; Thermo Fisher
Scientific, Inc.), washed twice with PBS (pH 7.4) and suspended in
PBS at a concentration of 5x106 cells/ml. The sample was
incubated with fluorescein isothiocyanate-conjugated monoclonal
antibody CD14 (cat. no. 11-0149-42), CD45 (cat. no. 11-9459-42),
isotype control (cat. no. 11-4714-41) (all 1:200; all from
eBioscience; Thermo Fisher Scientific, Inc.), or incubated with
phycoerythrin-conjugated monoclonal antibody CD29 (cat. no.
555443), CD34 (cat. no. 550761), CD44 (cat. no. 550989) (all 1:50;
all from BD Biosciences), CD90 (cat. no. 555596), isotype control
(cat. no. 550617) (both 1:100; both from BD Biosciences) for 30 min
at 4˚C in the dark according to the manufacturer's recommendation.
Finally, cells were washed twice with PBS and then analyzed with a
standard FACSAria flow cytometer (BD Biosciences) and results were
evaluated with CellQuest Pro software v1.0.2 (BD Biosciences).
Growth kinetics of MENSCs, UCMSCs and
DPSCs
MENSCs, UCMSCs and DPSCs with 90% confluency were
dissociated with TrypLE. Based on the cell count, 104
cells/well were seeded in 24-well cell culture plates (Corning
Inc.) and cultured at 37˚C with 5% CO2 (v/v) and
saturated humidity. After 24 h of incubation, each of the three
cell types were dissociated in the three wells, stained with the
same amount of trypan blue (Gibco; Thermo Fisher Scientific, Inc.)
and the cell number was then counted with an automatic cell counter
(Countstar). These procedures were repeated for the three wells
every two days. The medium was changed every two days. Finally, to
plot the growth curves for MENSCs, UCMSCs and DPSCs, the culture
time was displayed on the abscissa, while the mean number of cells
was presented on the ordinate (18).
The population doubling time (PDT) was calculated
for these cells according to the following equation:
where t is the culture time, lg is log10,
Nt the harvested cell number and N0 the cell
number at the beginning.
The maximum specific cell growth rate
(µm) was calculated using the PDT value as follows:
where ln is loge.
Multi-differentiation ability of
MENSCs, UCMSCs and DPSCs
For adipogenic differentiation, MENSCs, UCMSCs and
DPSCs were cultured in Dulbecco's modified Eagle's medium (DMEM;
Gibco; Thermo Fisher Scientific, Inc.) supplemented with high
glucose, 10% fetal bovine serum (FBS; HyClone; Cytiva), 1 mM
dexamethasone, 0.5 mM methyl-isobutyl-xanthine, 10 mg/ml insulin
and 100 mM indomethacin (all from Sigma-Aldrich; Merck KGaA) for 4
weeks. The control groups were cultured in DMEM supplemented with
high glucose and 10% FBS. At the end of the incubation, adipogenic
differentiation was assayed with 0.3% Oil red O (Sigma-Aldrich;
Merck KGaA) staining for lipid droplets for 1 h at 37˚C. After
observation with an inverted phase-contrast microscope, the stained
cells were eluted with 500 µl isopropyl alcohol per well for 5 min
at room temperature and then quantified by determining the
absorbance at 510 nm using a microplate reader (Thermo Fisher
Scientific, Inc.).
For osteogenic differentiation, MENSCs, UCMSCs and
DPSCs were cultured in DMEM supplemented with high glucose, 10%
FBS, 0.1 mM dexamethasone, 10 mM β-glycerolphosphate and 50 mM
ascorbic acid (all from Sigma-Aldrich; Merck KGaA) for 3 weeks. The
control groups were cultured in high-glucose DMEM just containing
10% FBS. At the end of the incubation, osteogenic differentiation
was assayed with 0.1% Alizarin red S (Sigma-Aldrich; Merck KGaA)
staining for calcium deposition for 1 h at 37˚C. After observation
with an inverted phase-contrast microscope, the stained cells were
eluted with 500 µl 10% cetylpyridinium chloride per well
(Sigma-Aldrich; Merck KGaA) for 10 min at room temperature and then
quantified by measuring the absorbance at 562 nm using a microplate
reader.
Production of pancreatic β-like cells
from MENSCs, UCMSCs and DPSCs using three-step method
Study of the role of inductive substances (16,22),
analysis of previous studies reporting differentiation methods from
mesenchymal into pancreatic cells and combination with the
preliminary experimental data resulted in the design of a
differentiation method (14,23),
the three-step induction system, to differentiate MENSCs, UCMSCs
and DPSCs into islet cells in vitro.
MENSCs, UCMSCs and DPSCs at P3 that grew to 70%
confluency were dissociated, the concentration was adjusted to
105 cells/ml and cells were seeded in 12-well cell
culture plates. The plates were pre-coated with type 1 rat-tail
collagen (cat. no. C7661; Sigma-Aldrich; Merck KGaA). The induction
procedure was then followed:
Step 1
Following incubation overnight, the medium was
changed to high-glucose DMEM containing 10% FBS and 10-6
mol/l retinoic acid (Sigma-Aldrich; Merck KGaA) to induce
differentiation for 48 h. The medium was then changed to
high-glucose DMEM only with 10% FBS, followed by culture for 24
h.
Step 2
Acetic acid (0.1 M) was used to dissolve type 1 rat
tail collagen to a 5 mg/ml solution. A total of 200 µl rat tail
collagen solution, was mixed with 798 µl DMEM medium and 12 µl 0.1
M NaOH. A total of 30 µl of this solution was added to each well of
12-well plates, and placed at 37˚C for 20 min. The cells were then
dissociated with TrypLE and the concentration of the cell
suspension was adjusted to 105 cells/ml. The cells were
seeded in new 12-well cell culture plates pre-incubated with type 1
rat-tail collagen. MENSCs, UCMSCs and DPSCs were cultured in the
induction medium consisting of low-glucose DMEM, 10% FBS, 10 mmol/l
nicotinamide, 20 ng/ml epidermal growth factor and 50 ng/ml basic
fibroblast growth factor (all from Sigma-Aldrich; Merck KGaA) for 7
days.
Step 3
The medium of the second step was removed and
changed to low-glucose DMEM containing exendin 4 (Sigma-Aldrich;
Merck KGaA) and 10% FBS to continue induction for another 7
days.
After differentiation, MSCs were stained with
dithizone (DTZ; Sigma-Aldrich; Merck KGaA). DTZ is a zinc-chelating
agent known to stain pancreatic β-cells due to their high zinc
content (24). Images were captured
with an inverted phase-contrast microscope (Olympus Corporation)
and analyzed with ImageJ software v2.0 (National Institutes of
Health) imitating the way of immunohistochemistry to obtain
quantitative statistics (25).
Furthermore, the staining intensity of these images was graded from
1-5 (the weakest stained as 1 and the strongest stained as 5),
determined in a double-blinded way by three independent
investigators, followed by statistical evaluation (26).
Immunofluorescence staining
Following differentiation, the MSCs were fixed with
4% paraformaldehyde for 20 min, permeabilized with PBS containing
0.3% Triton-X and then blocked with PBS containing 5% goat serum
(cat. no. G9023; Sigma-Aldrich; Merck KgaA) for 45 min at room
temperature. Cells were incubated with rabbit monoclonal
anti-insulin primary antibody (1:200; cat. no. ab181547; Abcam) at
4˚C overnight. The three types of cells were then washed with PBS
and stained with the tetramethylrhodamine (TRITC)-conjugated goat
anti-rabbit secondary antibody (1:300; cat. no. A16101; Invitrogen;
Thermo Fisher Scientific, Inc.) at room temperature for 1 h in the
dark. Cell nuclei were stained using 4,6-diamidino-2-phenylindole
(DAPI; Invitrogen; Thermo Fisher Scientific, Inc.). Images of cells
were captured using fluorescence microscopy (Leica
Microsystems).
Reverse-transcription quantitative
(RT-q)PCR
To detect the ability for differentiation, several
representative genes, INSULIN, glucose transporter 2 (GLUT-2) and
neurogenin 3 (NGN-3), were analyzed through RT-qPCR. Total RNA was
isolated from both induced cells and control cells using TRIzol
reagent (Biyuntian) following the manufacturer's protocol. The RNA
concentration and quality were assessed with a NanoDrop One
spectrophotometer (Thermo Fisher Scientific, Inc.). Subsequently,
the RT reaction was performed using the RT reagent kit with gDNA
Eraser (Takara Bio, Inc.) according to the manufacturer's protocol.
Real-time PCR was performed with the primers presented in Table I in a StepOne Plus System (Thermo
Fisher Scientific, Inc.). According to the manufacturer's protocol
for the FastStart Universal SYBR-Green Master (cat. no.
04913914001; Roche Diagnostics), the PCR mixture was prepared and
the PCR parameters were set. The thermocycling conditions use were
as follows: 95˚C for 10 min, followed by 40 cycles at 95˚C for 15
sec and at 60˚C for 60 sec. Melting curve analysis was performed at
the end of RT-qPCR. The results were calculated through
2-∆∆Cq method and normalized to GAPDH and the control
group (27).
 | Table IPrimer sequences designed
forquantitative PCR detection of genes to determine the
differentiation ability from stem cells into pancreatic β-like
cells. |
Table I
Primer sequences designed
forquantitative PCR detection of genes to determine the
differentiation ability from stem cells into pancreatic β-like
cells.
| Gene name | Forward primer
(5'-3') | Reverse primer
(5'-3') |
|---|
| GAPDH |
ATGGGGAAGGTGAAGGTCG |
GGGGTCATTGATGGCAACAATA |
| INSULIN |
AGCATCTGCTCCCTCTACCA |
TGCTGGTTCAAGGGCTTTAT |
| GLUT-2 |
CAGCTGTCTTGTGCTCTGCTTGT |
GCCGTCATGCTCACATAACTCA |
| NGN-3 |
AAGAGCGAGTTGGCACTGAGC |
CGTACAAGCTGTGGTCCGC |
Quantitative insulin secretion
assay
After differentiation, the induction medium was
removed and the cells were washed twice with PBS. New medium with
high or low glucose was added and the supernatants were collected
after glucose stimulation for 4 h. The concentration of insulin in
the supernatants collected from the three types of MSCs in high or
low glucose medium was determined using the Human Insulin ELISA kit
(cat. no. KAQ1251; Invitrogen; Thermo Fisher Scientific, Inc.). The
experiments were performed following the according to the
manufacturer's protocol of the ELISA kit. Absorbance was read at
450 nm. The standard curve was constructed using the absorbance of
the standard sample.
Statistical analysis
All values were expressed as the mean ± standard
deviation. Comparisons between two groups were analyzed with the
unpaired Student's t-test. Comparisons between more than two groups
were analyzed with one-way analysis of variance (ANOVA) followed by
least-significant differences post-hoc tests for equal variances
assumed and Tamhane's T2 post-hoc tests if no equal variances were
assumed. A two-way ANOVA with Tukey's post-hoc test was used for
comparisons between two factors. For non-parametric data, a
Kruskal-Wallis test with Dunn's post-hoc test was used for grading
scale analysis. P<0.05 was considered to indicate statistical
significance. All analyses were performed with SPSS 23.0 (IBM
Corporation).
Results
Morphology of MENSCs, UCMSCs and
DPSCs
Most of the primary MENSCs, UCMSCs and DPSCs adhered
to the culture plates within 24 h after seeding and exhibited a
polygonal or round morphology. The cells presented with pseudopodia
and displayed similar spindle-shaped morphology for ~2 days of
culture (Fig. 1A).
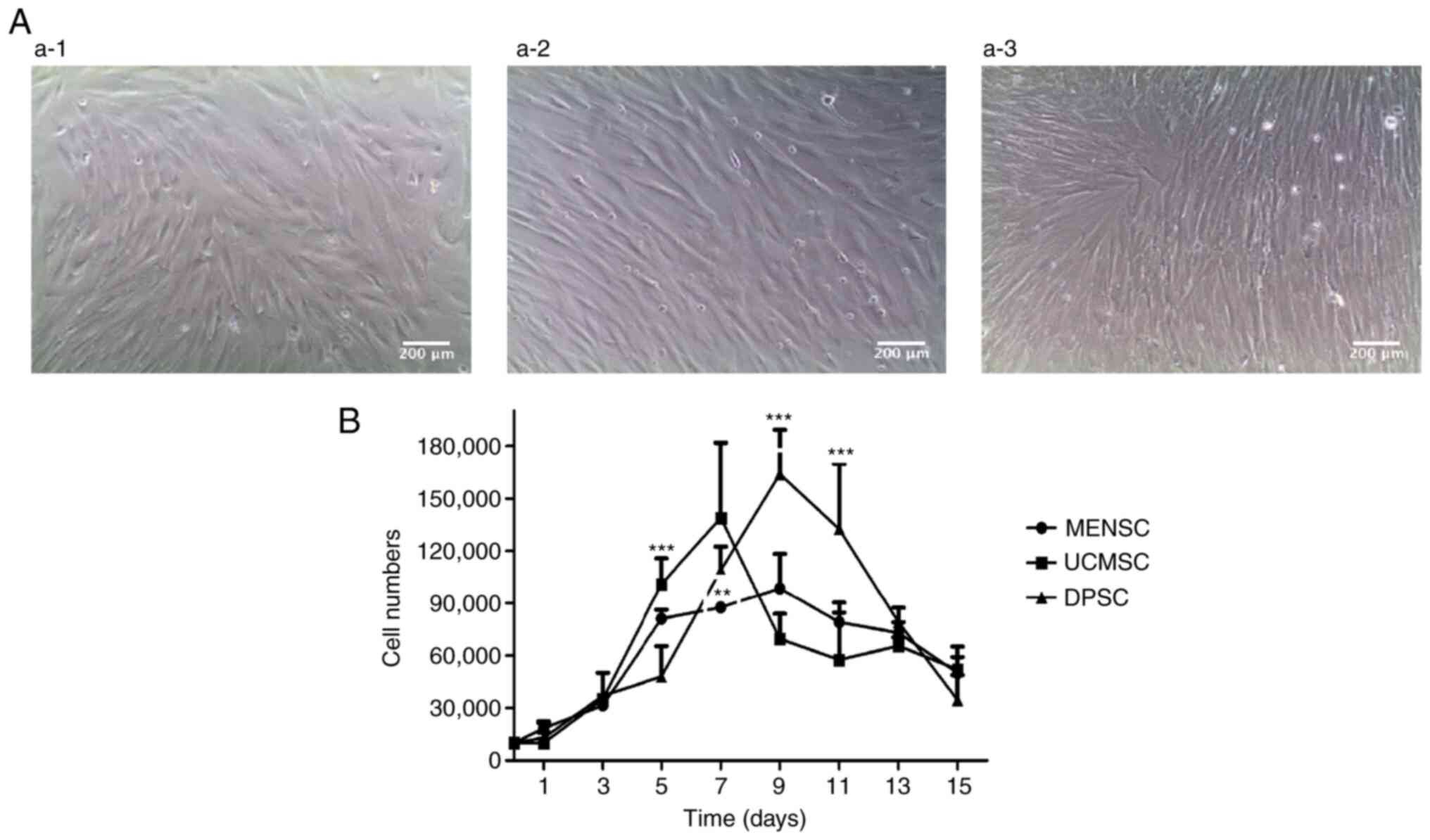 | Figure 1Basic characteristics of the three
types of MSC. (A) Morphology of (a-1) P4 MENSCs, (a-2) P4 UCMSCs
and (a-3) P4 DPSCs scultured ex vivo (magnification x100;
scale bar, 200 µm). (B) Growth curves of MENSCs, UCMSCs and DPSCs.
Initial cell seeding: 1.0x104 cells/cm2.
Values are expressed as the mean ± standard deviation of results
from three independent experiments. **P<0.01,
***P<0.001. Significances indicate comparison among
three types of MSCs, at the same time point. MENSCs, menstrual
blood-derived MSCs; UCMSCs, umbilical cord-derived MSCs; DPSCs,
dental pulp MSCs; MSC, mesenchymal stem cell; P4, passage 4; d,
days. |
Expression of cell surface markers on
MENSCs, UCMSCs and DPSCs
The expression of the surface receptor molecules
CD14, CD29, CD34, CD44, CD45 and CD90 on MENSCs, UCMSCs and DPSCs
was determined through flow cytometry. The results indicated that
MENSCs, UCMSCs and DPSCs were all positive for CD29, CD44 and CD90
and negative for CD14, CD34 and CD45, which were consistent with
the identification criteria for MSCs (28). The flow cytometry results are
provided in Fig. S1. Statistical
analysis indicated no significant difference in the surface marker
profiles of these three types of cells (P>0.05).
Growth curves of MENSCs, UCMSCs and
DPSCs
Growth curves of MENSCs, UCMSCs and DPSCs were drawn
(Fig. 1B) and the PDT was
calculated and presented in Table
II. The PDT of UCMSCs was the lowest among the three types of
MSC, followed by that of DPSCs and then MENSCs. The growth curves
also indicated that UCMSCs had the fastest and MENSCs had the
slowest proliferation. Specifically, in the first three days, the
growth trend of the three types of MSC was similar. At day 7,
UCMSCs reached a maximum number of cells, while MENSCs and DPSCs
were almost at the maximum at day 9. After 9 days, the number of
the three cell types decreased continuously until day 15, which is
related to the contact inhibition induced by the full confluency
(29) (Fig. 1B).
 | Table IICell populationdoubling time and the
largest growth rate of MENSCs, UCMSCs and DPSCs. |
Table II
Cell populationdoubling time and the
largest growth rate of MENSCs, UCMSCs and DPSCs.
| Cell type | Population doubling
time (h) | Maximum specific
cell growth rate (s-1) |
|---|
| MENSC-P4 | 65.40±12.96 | 0.0106 |
| UCMSC-P6 | 45.17±4.22 | 0.0156 |
| DPSC-P5 | 53.81±2.64 | 0.0130 |
Multi-differentiation capabilities of
MENSCs, UCMSCs and DPSCs
As indicated in Fig.
2, the three cell types were able to efficiently differentiate
into osteocytes and adipocytes. The osteogenesis of UCMSCs was much
more prominent and efficient than that of DPSCs and MENSCs. When
Alizarin red S staining was performed at the end of week 3, most of
the UCMSCs stained red, followed by DPSCs and MENSCs in terms of
quantity (Fig. 2A and B). The quantitative results are provided
in Fig. 2E. The absorbance of the
induced groups of all these MSCs was stronger than that of the
control groups, which indicated that all the treated MSCs
differentiated into osteoblasts. The absorbance of UCMSCs was
stronger than that of MENSCs and DPSCs, which was consistent with
the observations in the staining images.
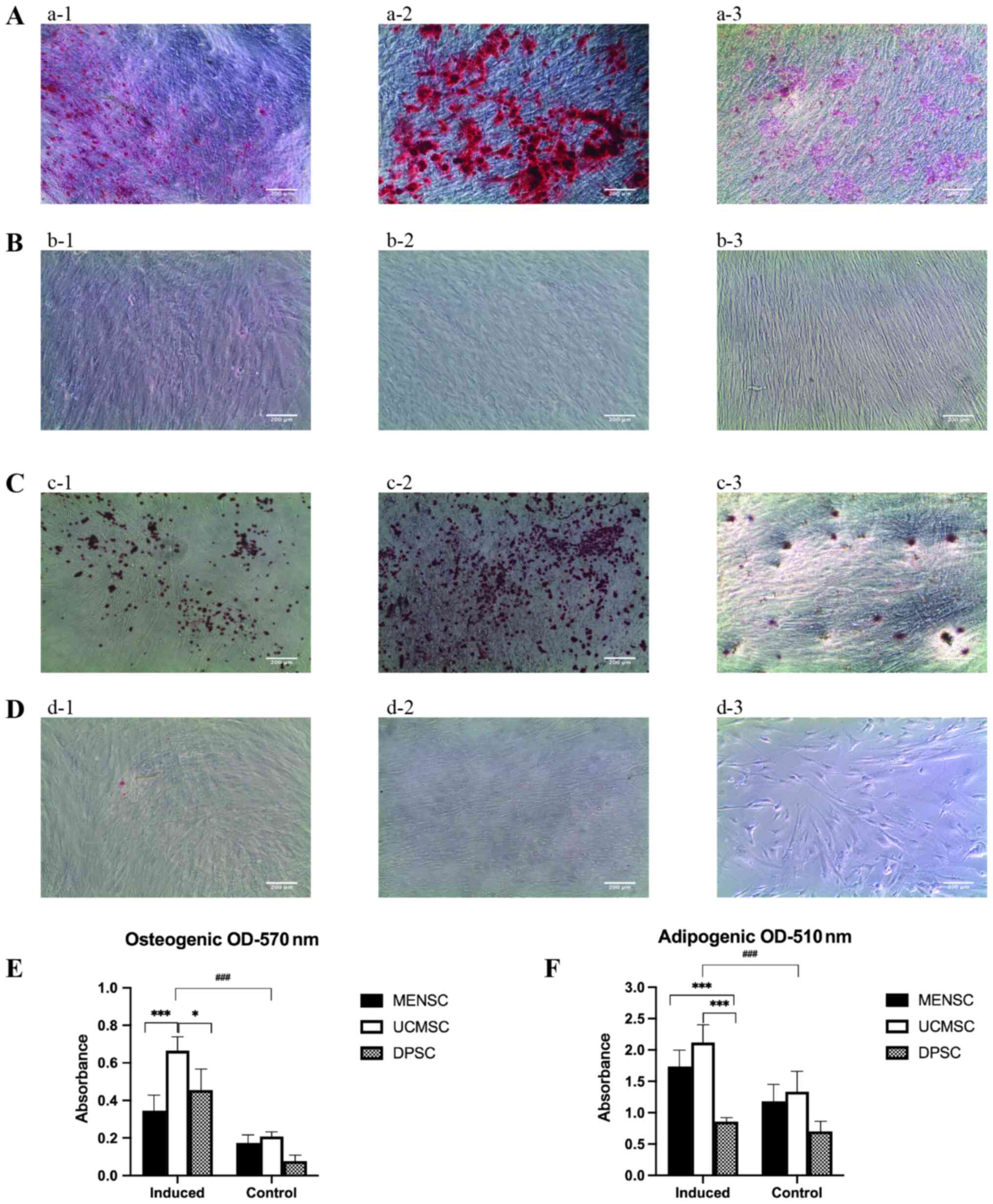 | Figure 2Osteogenic and adipogenic
differentiation of the three types of MSC. (A) Osteogenesis
differentiation of P4 (a-1) MENSCs, (a-2) UCMSCs and (a-3) DPSCs
safter Alizarin red S staining. (B) Control groups for osteogenesis
differentiation of P4 (b-1) MENSCs, (b-2) UCMSCs and (b-3) DPSCs
after Alizarin red S staining. (C) Adipogenesis differentiation of
P4 (c-1) MENSCs, (c-2) UCMSCs and (c-3) DPSCs safter Oil red O
staining. (D) Control groups for adipogenesis differentiation of
(d-1) MENSCs, (d-2) UCMSCs and (d-3) DPSCs after Oil red O staining
(magnification x100; scale bar, 200 µm). (E) Semiquantitative
results of osteogenic differentiation. (F) Semiquantitative results
of adipogenic differentiation. *P<0.05,
***P<0.001; ###P<0.001 vs. control.
MENSCs, menstrual blood-derived MSCs; UCMSCs, umbilical
cord-derived MSCs; DPSCs, dental pulp MSCs; MSC, mesenchymal stem
cell; OD, optical density; P4, passage 4. |
For adipogenic induction, UCMSCs exhibited the best
differentiation capability among the three types of MSC. The Oil
red O staining for adipocytes was more prominent for UCMSCs
compared with that of MENSCs and DPSCs (Fig. 2C and D). The quantitative results are presented
in Fig. 2F. The absorbance of the
induced groups of all of these MSCs was stronger than that of the
control groups, which indicated that all of these MSCs
differentiated into adipocytes. The absorbance of UCMSCs and MENSCs
was stronger than that of DPSCs.
Pancreatic β-like cell differentiation
ability of MENSCs, UCMSCs and DPSCs Quantitative analysis of DTZ
staining
After induction, MSCs were subjected to staining
with DTZ and then observed under the microscope. Stained sample
islet cells were red (Fig. 3A and
B), as the induced islet cells
contain zinc ions that combine with dithizone, producing a palm red
color. MENSCs exhibited the most obvious DTZ staining, while UCMSCs
and DPSCs did not stain as well as MENSCs. The quantitative
staining percentage of MENSCs was higher than that of UCMSCs and
DPSCs (Fig. 3C). The results of the
graded analysis suggested that the grade of staining of MENSCs was
higher than that of UCMSCs and DPSCs and the results were
consistent with those quantified with ImageJ (Fig. 3D).
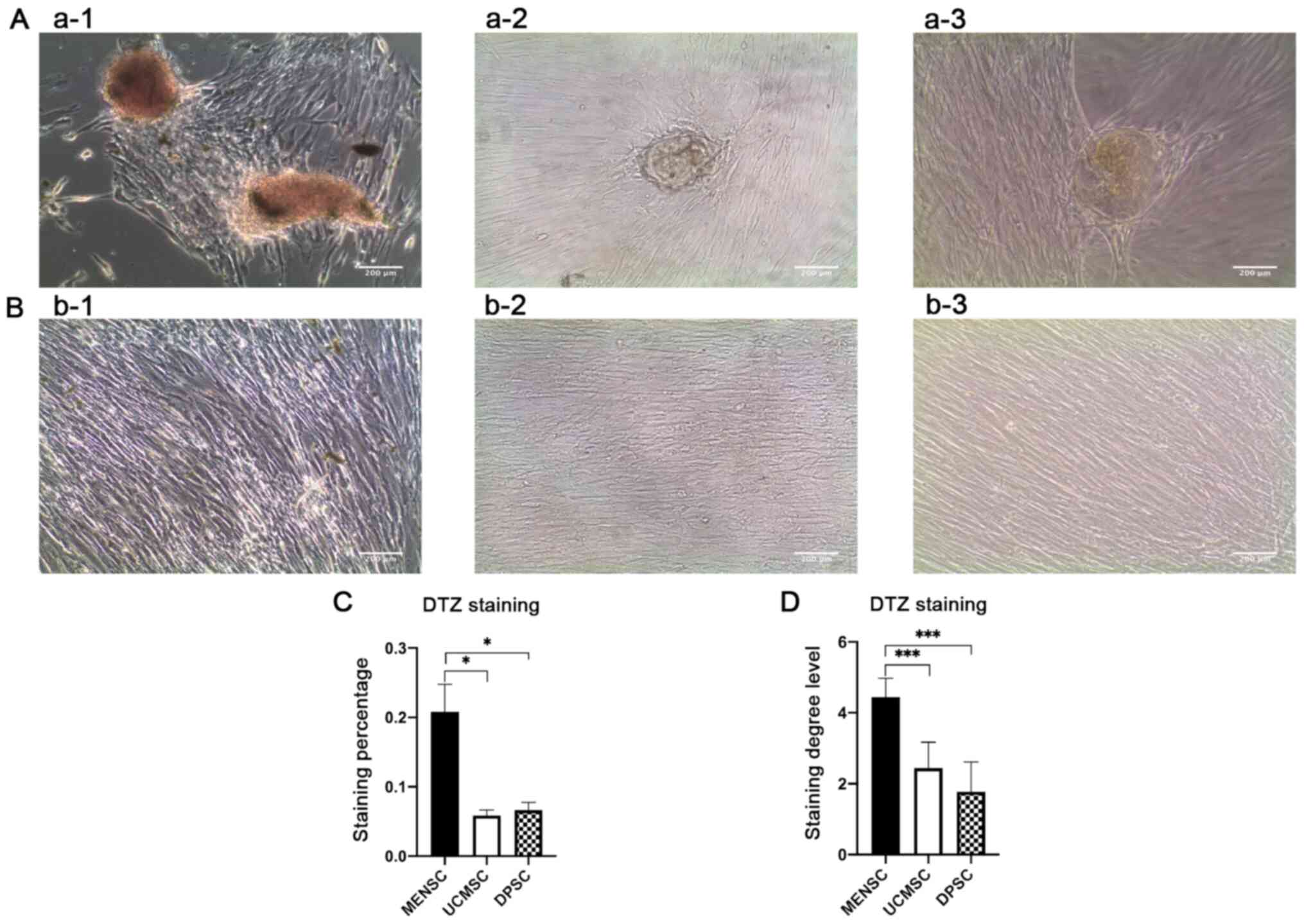 | Figure 3Results of differentiation into
pancreatic β-like cells. (A) DTZ staining of P4 (a-1) MENSCs, (a-2)
UCMSCs and (a-3) DPSCs. (B) The DTZ staining results of
undifferentiated groups of P4 (b-1) MENSCs, (b-2) UCMSCs and (b-3)
DPSCs (magnification x100; scale bar, 200 µm). (C) Quantitative
results of the DTZ staining. The percentage of the whole
microscopic imageswas determined with ImageJ software, imitating
immunohistochemistry. (D) Quantitative results of DTZ staining with
grading from 1-5. *P<0.05, ***P<0.001.
MENSCs, menstrual blood-derived MSCs; UCMSCs, umbilical
cord-derived MSCs; DPSCs, dental pulp MSCs; MSC, mesenchymal stem
cell; DTZ, dithizone; P4, passage 4. |
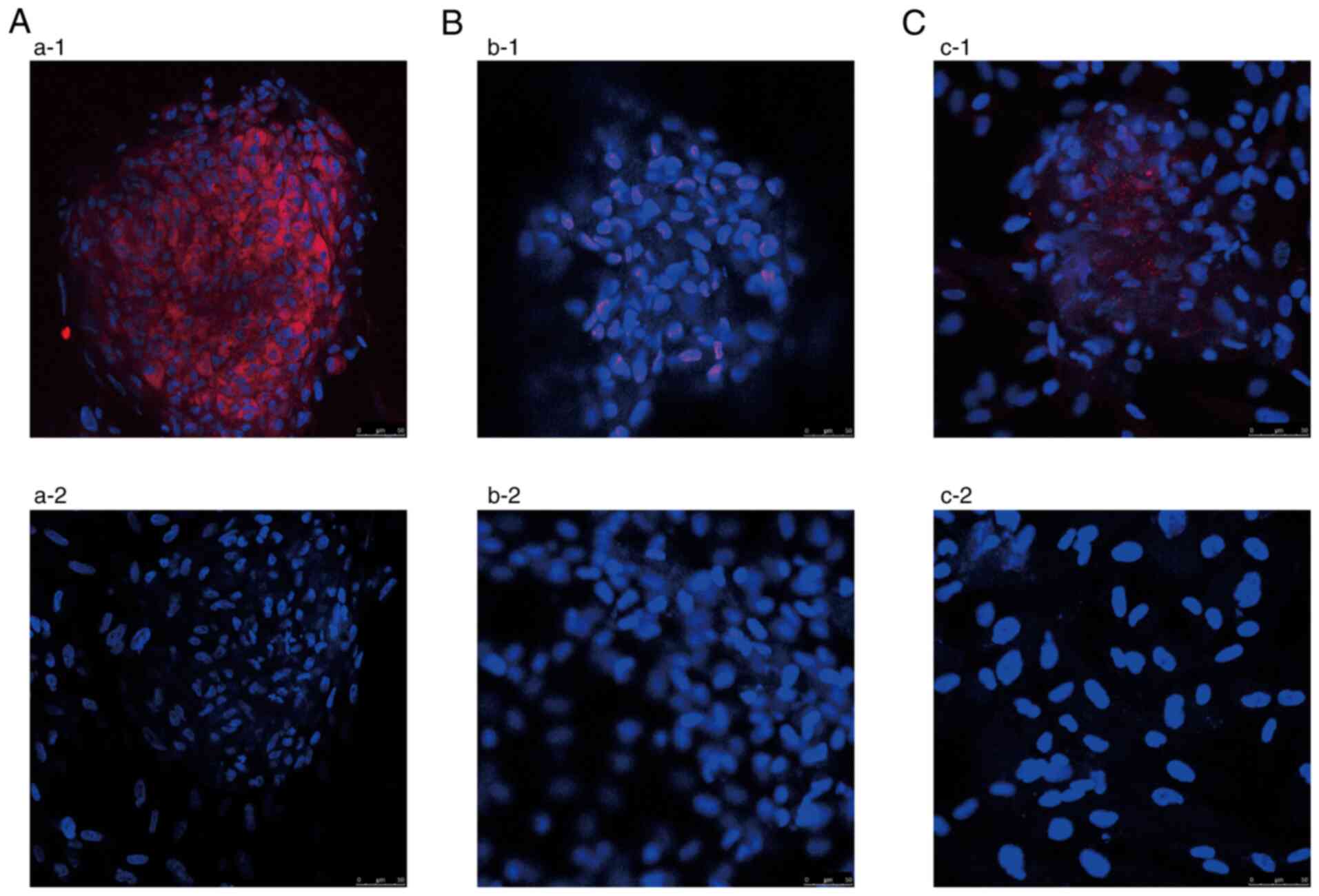
Insulin expression by laser immune
confocal microscopy
After the three-step induction, the MENSCs tested
positive for insulin expression, while the UCMSCs were negative for
insulin and the DPSCs exhibited weak expression (Fig. 4). DAPI-stained nuclei appeared blue
and cells that expressed insulin appeared red. It was proved that
the induced MENSCs obtained a synthetic insulin function. The
ability of MENSCs to produce insulin was the best, followed by that
of DPSCs and then that of UCMSCs.
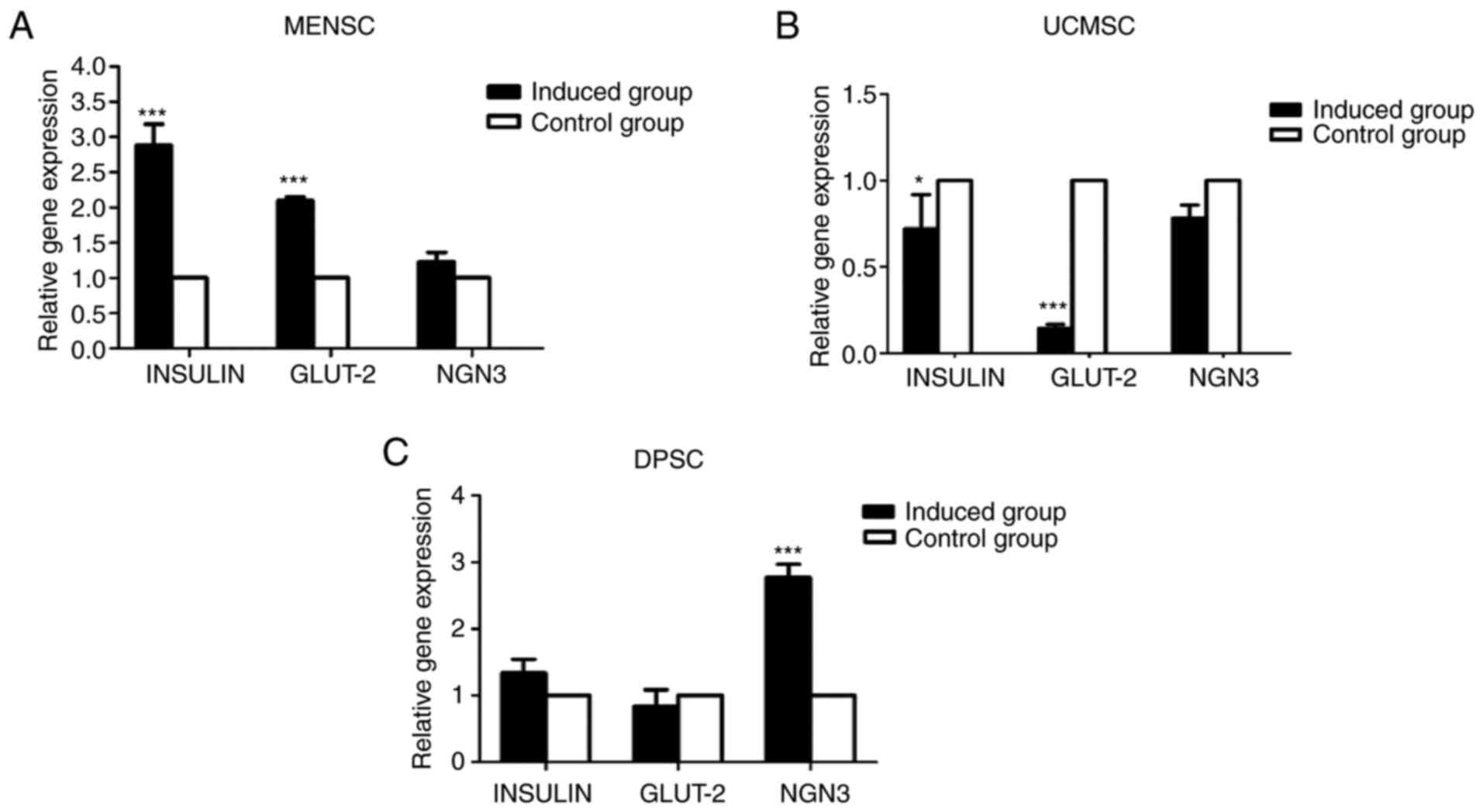
Expression of genes characteristic for
islet cells
The expression of islet-related genes was analyzed
by using RT-qPCR (Fig. 5).
The iconic genes INSULIN, GLUT-2 and NGN-3 were detected.
Subsequent to differentiation, the expression of INSULIN and GLUT-2
in MENSCs was significantly higher than that in the control group.
The expression of NGN3 was slightly higher in the induced group
with no significance. Furthermore, the expression of INSULIN and
GLUT-2 in UCMSCs after induction was lower than that in the control
group. The expression of NGN3 was not altered after induction. The
amount of NGN-3 expressed in the DPSCs was much higher than that in
the control group (P<0.01). The average INSULIN expression of
the induced group was slightly higher than that of the control
group, while GLUT-2 expression of the induced group was lower.
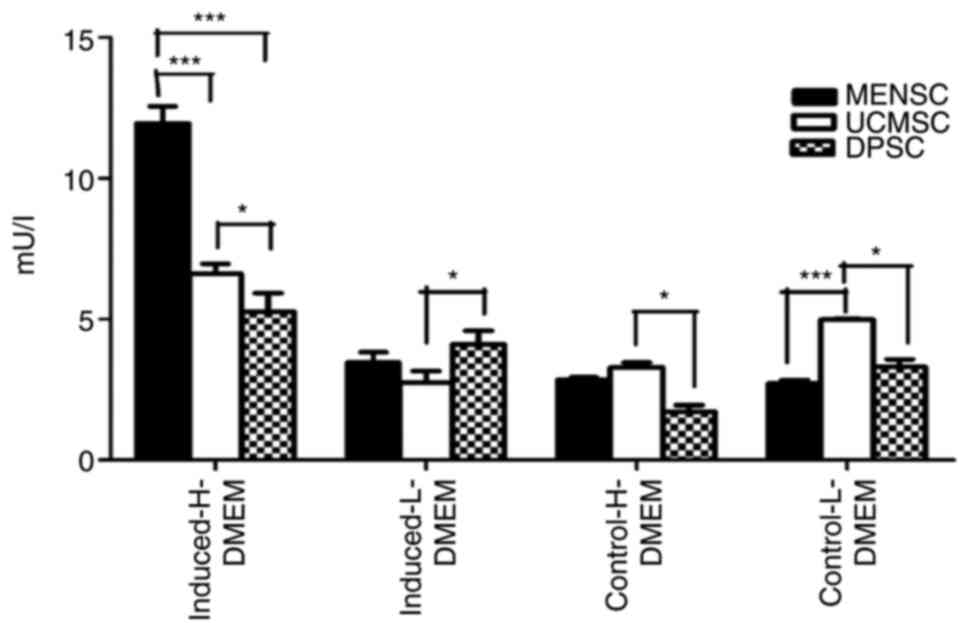
Insulin secretion by ELISA
The insulin secretion for the three types of adult
MSCs varied according to the different glucose concentrations in
the medium (Fig. 6). The level of
insulin secreted by MENSCs induced in vitro into islet cells
exposed to high glucose concentration was 11.95±0.61 mU/l. For
UCMSCs induced in the presence of high glucose, the level of
secreted insulin was 6.61±0.37 mU/l and that of DPSCs was 5.26±0.66
mU/l. When the glucose concentration was 5.5 mmol/l, the insulin
secretion of induced MENSCs was 3.46±0.29 mU/l, while that of
UCMSCs was 2.75±0.39 mU/l and that of DPSCs was 4.11±0.42 mU/l.
Additional insulin was not secreted after low glucose stimulation
both in induced and control MSCs, which indicated that the
secretion of insulin was dependent on glucose stimulation in the
medium. According to the results, it was observed that induced MSCs
secreted higher concentration of extracellular insulin compared
with control MSCs when exposed to high glucose concentration of 25
mmol/l. When induced MSCs were exposed to high glucose
concentration, MENSCs secreted the highest amount of insulin,
followed by UCMSCs and then DPSCs. Based on these results, MENSCs
were more suitable than UCMSCs and DPSCs as seed cell candidates
for the induction of islet cells in vitro.
Discussion
In the present study, the ability of three types of
MSC to differentiate into pancreatic β-like cells in vitro
was compared. Other studies reported the use of diverse methods to
differentiate MSCs toward a pancreatic cell lineage (30-33).
A three-step method to effectively induce MENSCs, UCMSCs and DPSCs
to differentiate into pancreatic β-like cells in vitro was
developed. As described by Veres et al (34), the stem cell-derived β (SC-β) cell
protocol led to stem cell differentiation into six stages. These
six stages were divided into two major steps during differentiation
(35), formation of pancreatic
progenitors and maturation of insulin-expressing SC-β cells. Based
on this flow, the protocols were improved according to the
preliminary conditions tested in the present study results of our
previous study. It was observed that all of the different MSC types
were similar in cell surface markers and basic biological
characteristics. Following the application of the three-step
differentiation method, MENSCs had a strong ability to form
spherical shapes. The formation of spherical cells in UCMSCs and
DPSCs was less than that of MENSCs. Differences were not only
observed in terms of morphology, but also in the staining behavior.
DTZ staining was evident in MENSCs, whereas UCMSCs and DPSCs were
evidently not stained. In the present study, the expression of
insulin was also analyzed through confocal laser microscopy and the
results indicated that the production of insulin by MENSCs was much
higher than that in DPSCs and UCMSCs. As specific genes of
pancreatic β-cells, INSULIN, GLUT-2 and NGN-3 were then analyzed.
The expression of the three genes in MENSCs was positive compared
to that in the undifferentiated control group. According to the
results, MENSCs had a stronger ability to differentiate into
pancreatic cells than DPSCs and UCMSCs, which means that MENSCs are
a novel suitable source for the curative treatment of TID. It was
preliminarily concluded that MENSCs have potential for the
treatment of TID. However, further studies, such as those using
flow cytometry and additional gene expression analyses, are
required to confirm this conclusion. Flow cytometry may be applied
to quantitatively study the efficiency of differentiation and the
percentage of insulin-positive cells after induction (34). Flow cytometry is also beneficial to
the qualitative analysis of the differentiation potency of the
three types of MSC. NGN-3, GLUT-2, pancreatic and duodenal homeobox
1 (PDX-1), INSULIN, glucagon and somatostatin are iconic genes for
the differentiation of pancreatic cells (36). The present study explored the
changes in the expression of NGN-3, INSULIN and GLUT2. The
expression of PDX-1, glucagon and somatostatin remain to be
explored using RT-qPCR or confocal laser microscopy to strengthen
the results of the present study.
Previous studies have demonstrated the
differentiation ability of MENSCs, UCMSCs and DPSCs (9,37,38).
To the best of our knowledge, the present study was the first to
compare the ability of the three stem cell types to differentiate
into pancreatic β-like cells using the same differentiation
protocol in the same article.
In the present study, the use of collagen coating
enhanced the ability of MSCs to form pancreatic β-like cells. It
was previously reported that the collagen/hyaluronic acid scaffold
was able to enhance the differentiation ability of ADSCs into
insulin-producing cells (39).
Sefcik et al (17) utilized
collagen to enhance the osteogenic differentiation ability of ADSCs
in vitro.
In the present study, it was speculated that the
different differentiation abilities of the MSCs were due to the
physiological location of these cells. Suman et al (40) concluded that MSCs are types of germ
layer specific multipotent stem cells (MPSCs), which can give rise
to monopotent stem cells, and monopotent stem cells differentiate
into one adult cell lineage. The pancreas split belongs to the
endoderm (41), and endodermal
MPSCs tend to differentiate into monopotent stem cells for
pancreas, lung and liver cell lineages. The location of the
endometrium where the MENSCs are collected is close to endoderm in
physiological characteristics (42). It was reported that UCMSCs may
express a low amount of ectodermal and endodermal markers, and the
majority of the cells express mesodermal markers after cultivation
for several days (43). The dental
pulp begins in the molar pulp and is derived from the ectoderm
originated from migrating neural crest cells (44). According to previous studies, the
formation of the definitive endoderm (DE) lineage was essential but
not necessary for the differentiation of pancreatic cells, which
have the ability to produce insulin and react to high glucose
concentrations (45). Based on the
results, the MENSCs from the endoderm had a stronger ability to
form pancreatic β-like cells than UCMSCs and DPSCs without the
formation of the DE lineage, which indicated that MENSCs are a more
suitable source for TID treatment. However, it remains elusive
whether the location germ layers where MSCs are derived from serve
a role in their differentiation potency into pancreatic cells and
this remains to be explored.
In conclusion, in the present study, the
differentiation capability of MENSCs, UCMSCs and DPSCs into
pancreatic β-like cells was compared. The MENSCs had the best
differentiation ability into pancreatic beta-like cells than UCMSCs
and DPSCs. The results demonstrated that the use of MENSCs as a
potential new source of progenitor cells for application in the
treatment of diabetes was more effective than the use of UCMSCs and
DPSCs in vitro.
Supplementary Material
Flow cytometry results for MENSCs,
UCMSCs and DPSCs for CD14, CD29, CD34, CD44, CD45 and CD90
according to the criteria for MSCs. Black areas represent isotype
control group and white areas represent monoclonal antibody
combination group. MENSCs, menstrual blood-derived MSCs; UCMSCs,
umbilical cord-derived MSCs; DPSCs, dental pulp MSCs; MSC,
mesenchymal stem cell.
Acknowledgements
Not applicable.
Funding
Funding: This project was supported by the National Natural
Science Foundation of China (grant. no. 81871122).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YTC conceived the present study and supervised it.
YFM, ZJW and YTC designed and initiated the experiments. YFM, ZJW,
JG and YY performed the experiments. YFM, ZJW, JG, YY, FLZ, HJR and
NMQ analyzed the data. YFM and FLZ revised the manuscript. ZJW, NMQ
and YTC confirmed the authenticity of the raw data. All authors
provided input for the paper writing and discussion during the
whole period. All authors have read and approved the final
manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from each
donor and the present study was approved by the Ethics Committee at
the School of Pharmacy, Shanghai Jiaotong University (Shanghai,
China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Stumvoll M, Goldstein BJ and van Haeften
TW: Type 2 diabetes: Principles of pathogenesis and therapy.
Lancet. 365:1333–1346. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Senior PA and Pettus JH: Stem cell
therapies for type 1 diabetes: Current status and proposed road map
to guide successful clinical trials. Diabet Med. 36:297–307.
2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Pixley JS: Mesenchymal stem cells to treat
type 1 diabetes. Biochim Biophys Acta Mol Basis Dis.
1866(165315)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Carlsson PO, Schwarcz E, Korsgren O and Le
Blanc K: Preserved β-cell function in type 1 diabetes by
mesenchymal stromal cells. Diabetes. 64:587–592. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Troyer DL and Weiss ML: Concise review:
Wharton's jelly-derived cells are a primitive stromal cell
population. Stem Cells. 26:591–599. 2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Novais A, Lesieur J, Sadoine J, Sliman L,
Baroukh B, Saubaméa B, Schmitt A, Vital S, Poliard A, Hélary C, et
al: Priming dental pulp stem cells from human exfoliated deciduous
teeth with fibroblast growth factor-2 enhances mineralization
within tissue-engineered constructs implanted in craniofacial bone
defects. Stem Cells Transl Med. 8:844–857. 2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Liu Y, Niu R, Li W, Lin J, Stamm C,
Steinhoff G and Ma N: Therapeutic potential of menstrual
blood-derived endometrial stem cells in cardiac diseases. Cell Mol
Life Sci. 76:1681–1695. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Asahara T, Kalka C and Isner JM: Stem cell
therapy and gene transfer for regeneration. Gene Ther. 7:451–457.
2000.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Brown C, McKee C, Bakshi S, Walker K,
Hakman E, Halassy S, Svinarich D, Dodds R, Govind CK and Chaudhry
GR: Mesenchymal stem cells: Cell therapy and regeneration
potential. J Tissue Eng Regen Med. 13:1738–1755. 2019.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Shivakumar SB, Bharti D, Subbarao RB, Park
JM, Son YB, MUllah I, Choe YH, Lee HJ, Park BW, Lee SL and Rho GJ:
Pancreatic endocrine-like cells differentiated from human umbilical
cords wharton's jelly mesenchymal stem cells using small molecules.
J Cell Physiol. 234:3933–3947. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Pedroni ACF, Sarra G, de Oliveira NK,
Moreira MS, Deboni MCZ and Marques MM: Cell sheets of human dental
pulp Stem cells for future application in bone replacement. Clin
Oral Investig. 23:2713–2721. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Liu JJ, Yu F, Sun Y, Jiang B, Zhang W,
Yang J, Xu GT, Liang A and Liu S: Concise reviews: Characteristics
and potential applications of human dental tissue-derived
mesenchymal stem cells. Stem Cells. 33:627–638. 2015.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Patel AN, Park E, Kuzman M, Benetti F,
Silva FJ and Allickson JG: Multipotent menstrual blood stromal stem
cells: Isolation, characterization, and differentiation. Cell
Transplant. 17:303–311. 2008.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shivakumar SB, Lee HJ, Son YB, Bharti D,
Ock SA, Lee SL, Kang YH, Park BW and Rho GJ: In vitro
differentiation of single donor derived human dental mesenchymal
stem cells into pancreatic beta cell-like cells. Biosci Rep.
39(BSR20182051)2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Linh NTB, Abueva CDG, Jang DW and Lee BT:
Collagen and bone morphogenetic protein-2 functionalized
hydroxyapatite scaffolds induce osteogenic differentiation in human
adipose-derived stem cells. J Biomed Mater Res Part B Appl
Biomater. 108:1363–1371. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Bilic-Curcic I, Kalajzic Z, Wang L and
Rowe DW: Origins of endothelial and osteogenic cells in the
subcutaneous collagen gel implant. Bone. 37:678–687.
2005.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Sefcik LS, Neal RA, Kaszuba SN, Parker AM,
Katz AJ, Ogle RC and Botchwey EA: Collagen nanofibres are a
biomimetic substrate for the serum-free osteogenic differentiation
of human adipose stem cells. J Tissue Eng Regen Med. 2:210–220.
2008.PubMed/NCBI View
Article : Google Scholar
|
|
18
|
Ren H, Sang Y, Zhang F, Liu Z, Qi N and
Chen Y: Comparative analysis of human mesenchymal stem cells from
umbilical cord, dental pulp, and menstrual blood as sources for
cell therapy. Stem Cells Int. 2016(3516574)2016.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Skyler JS, Fonseca VA, Segal KR and
Rosenstock J: MSB-DM003 Investigators. Allogeneic mesenchymal
precursor cells in type 2 diabetes: A randomized,
placebo-controlled, dose-escalation safety and tolerability pilot
study. Diabetes Care. 38:1742–1749. 2015.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Trivedi HL, Thakkar UG, Vanikar AV and
Dave SD: Treatment of polyglandular autoimmune syndrome type 3
using co-transplantation of insulin-secreting mesenchymal stem
cells and haematopoietic stem cells. BMJ Case Rep.
2011(bcr0720114436)2011.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liu X, Zheng P, Wang X, Dai G, Cheng H,
Zhang Z, Hua R, Niu X, Shi J and An Y: A preliminary evaluation of
efficacy and safety of Wharton's jelly mesenchymal stem cell
transplantation in patients with type 2 diabetes mellitus. Stem
Cell Res Ther. 5(57)2014.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Hoveizi E and Mohammadi T: Differentiation
of endometrial stem cells into insulin-producing cells using
signaling molecules and zinc oxide nanoparticles, and
three-dimensional culture on nanofibrous scaffolds. J Mater Sci
Mater Med. 30(101)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Santamaria X, Massasa EE, Feng YZ, Wolff E
and Taylor HS: Derivation of insulin producing cells from human
endometrial stromal stem cells and use in the treatment of murine
diabetes. Mol Ther. 19:2065–2071. 2011.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Shiroi A, Yoshikawa M, Yokota H, Fukui H,
Ishizaka S, Tatsumi K and Takahashi Y: Identification of
insulin-producing cells derived from embryonic stem cells by
zinc-chelating dithizone. Stem Cells. 20:284–292. 2002.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Shu J, Dolman GE, Duan J, Qiu GP and Ilyas
M: Statistical colour models: An automated digital image analysis
method for quantification of histological biomarkers. Biomed Eng
Online. 15(46)2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Gibson-Corley KN, Olivier AK and Meyerholz
DK: Principles for valid histopathologic scoring in research. Vet
Pathol. 50:1007–1015. 2013.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Dominici M, Le Blanc K, Mueller I,
Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A,
Prockop D and Horwitz E: Minimal criteria for defining multipotent
mesenchymal stromal cells. The international society for cellular
therapy position statement. Cytotherapy. 8:315–317. 2006.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Zhou YF, Bosch-Marce M, Okuyama H,
Krishnamachary B, Kimura H, Zhang L, Huso DL and Semenza GL:
Spontaneous transformation of cultured mouse bone marrow-derived
stromal cells. Cancer Res. 66:10849–10854. 2006.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Van Pham P, Nguyen PTM, Nguyen ATQ, Pham
VM, Bui ANT, Dang LTT, Nguyen KG and Phan N: Improved
differentiation of umbilical cord blood-derived mesenchymal stem
cells into insulin-producing cells by PDX-1 mRNA transfection.
Differentiation. 87:200–208. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Duruksu G and Aciksari A: Guiding the
differentiation direction of pancreatic islet-derived stem cells by
glycated collagen. Stem Cells Int. 3(6143081)2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Gao F, Wu Y, Wen H, Zhu W, Ren H, Guan W
and Tian X: Multilineage potential research on pancreatic
mesenchymal stem cells of bovine. Tissue Cell. 56:60–70.
2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Phadnis SM, Joglekar MV, Dalvi MP,
Muthyala S, Nair PD, Ghaskadbi SM, Bhonde RR and Hardikar AA: Human
bone marrow-derived mesenchymal cells differentiate and mature into
endocrine pancreatic lineage in vivo. Cytotherapy. 13:279–293.
2011.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Veres A, Faust AL, Bushnell HL, Engquist
EN, Kenty JHR, Harb G, Poh YC, Sintov E, Gürtler M, Pagliuca FW, et
al: Charting cellular identity during human in vitro beta-cell
differentiation. Nature. 569:368–373. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Manaph NPA, Sivanathan KN, Nitschke J,
Zhou XF, Coates PT and Drogemuller CJ: An overview on small
molecule-induced differentiation of mesenchymal stem cells into
beta cells for diabetic therapy. Stem Cell Res Ther.
10(18)2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Chandra V, Swetha G, Phadnis S, Nair PD
and Bhonde RR: Generation of pancreatic hormone-expressing
islet-like cell aggregates from murine adipose tissue-derived stem
cells. Stem Cells. 27:1941–1953. 2009.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Chen LJ, Qu JJ and Xiang C: The
multi-functional roles of menstrual blood-derived stem cells in
regenerative medicine. Stem Cell Res Ther. 10(10)2019.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xing YX, Zhang YP, Wu X, Zhao B, Ji YW and
Xu X: A comprehensive study on donor-matched comparisons of three
types of mesenchymal stem cells-containing cells from human dental
tissue. J Periodont Res. 54:286–299. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Khorsandi L, Khodadadi A, Nejad-Dehbashi F
and Saremy S: Three-dimensional differentiation of adipose-derived
mesenchymal stem cells into insulin-producing cells. Cell Tissue
Res. 361:745–753. 2015.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Suman S, Domingues A, Ratajczak J and
Ratajczak MZ: Potential clinical applications of stem cells in
regenerative medicine. Adv Exp Med Biol. 1201:1–22. 2019.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Bastidas-Ponce A, Scheibner K, Lickert H
and Bakhti M: Cellular and molecular mechanisms coordinating
pancreas development. Development. 144:2873–2888. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Lin J, Xiang D, Zhang JL, Allickson J and
Xiang C: Plasticity of human menstrual blood stem cells derived
from the endometrium. J Zhejiang Univ Sci B. 12:372–380.
2011.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Bahmanpour S, Khozani TT and Tazangi FR:
Evaluation of the capability of the Wharton's jelly mesenchymal
stem cell aggregates to express the markers of three germ cell
lineages. Arch Iran Med. 22:85–90. 2019.PubMed/NCBI
|
|
44
|
Lan X, Sun Z, Chu C, Boltze J and Li S:
Dental pulp stem cells: An attractive alternative for cell therapy
in ischemic stroke. Front Neurol. 10(824)2019.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Baetge EE: Production of beta-cells from
human embryonic stem cells. Diabetes Obes Metab. 10:186–194.
2008.PubMed/NCBI View Article : Google Scholar
|