1. Introduction
Crohn's disease (CD) is an immune-mediated chronic
recurrent, systemic disease characterized by gastrointestinal
inflammation (1). CD is a complex
disease, which occurs due to genetic and environmental factors
(2). Accumulating evidence has
indicated that bacteria have a major role in the pathogenesis of
inflammatory bowel disease (IBD) (3). Intestinal bacteria and normal
intestinal microbiota may cause damage to the intestinal barrier
and induce inflammation in susceptible hosts, which indicates that
all bacteria may develop into potential pathogens (4). Changes in microbiota composition and
metabolism may lead to enhanced immune response, epithelial
dysfunction and increased mucosal permeability (5). Sartor (4,5)
performed extensive research on CD in different rodent models. He
established at least 11 different models of genetically engineered
mice and rats, induced enteritis models of indomethacin, guinea
pigs fed carrageenan, a sulfated red seaweed extract, and cotton
top tamarins a primate model of colitis (5). These observations in animal models
demonstrated that intestinal flora imbalance is a key factor in the
development of intestinal inflammation and IBD.
The incidence of CD in industrialized countries has
been steadily increasing over the past 70 years. In Europe, the
incidence of CD is estimated to be 12.7/100,000 individuals per
year (6). The clinical
manifestations are abdominal pain, fever, intestinal obstruction
and hematochezia or mucinous diarrhea, which mainly affect the
gastrointestinal tract and severely compromise the quality of life
of affected patients. Since CD is characterized as a discontinuous
or segmental inflammatory reaction of the total intestinal wall, it
is easy to penetrate into adjacent organs, tissues or skin
(7). Therefore, attention should be
paid to the formation of fistula in addition to the common
complications caused by intestinal obstruction (7).
Anal fistula is the most common perianal lesion
observed in CD (8). Panés and
Rimola (9) summarized the current
classification of perianal fistula in their review article.
Perianal fistula is a common complication of CD and it is estimated
to affect 26-28% of patients within the first two decades following
diagnosis (10). In recent years,
local injections of mesenchymal stem cells (MSCs) have achieved
promising results in terms of fistula closure (11). However, the lack of clinical safety
and effectiveness data remains a major constraint to the
development of novel treatments or therapeutic strategies. The
pathogenesis of the disease may be investigated and novel treatment
methods may be explored by establishing specific animal models and
by further studying the safety and effectiveness of different
treatment schemes in animals.
2. Literature search methodology
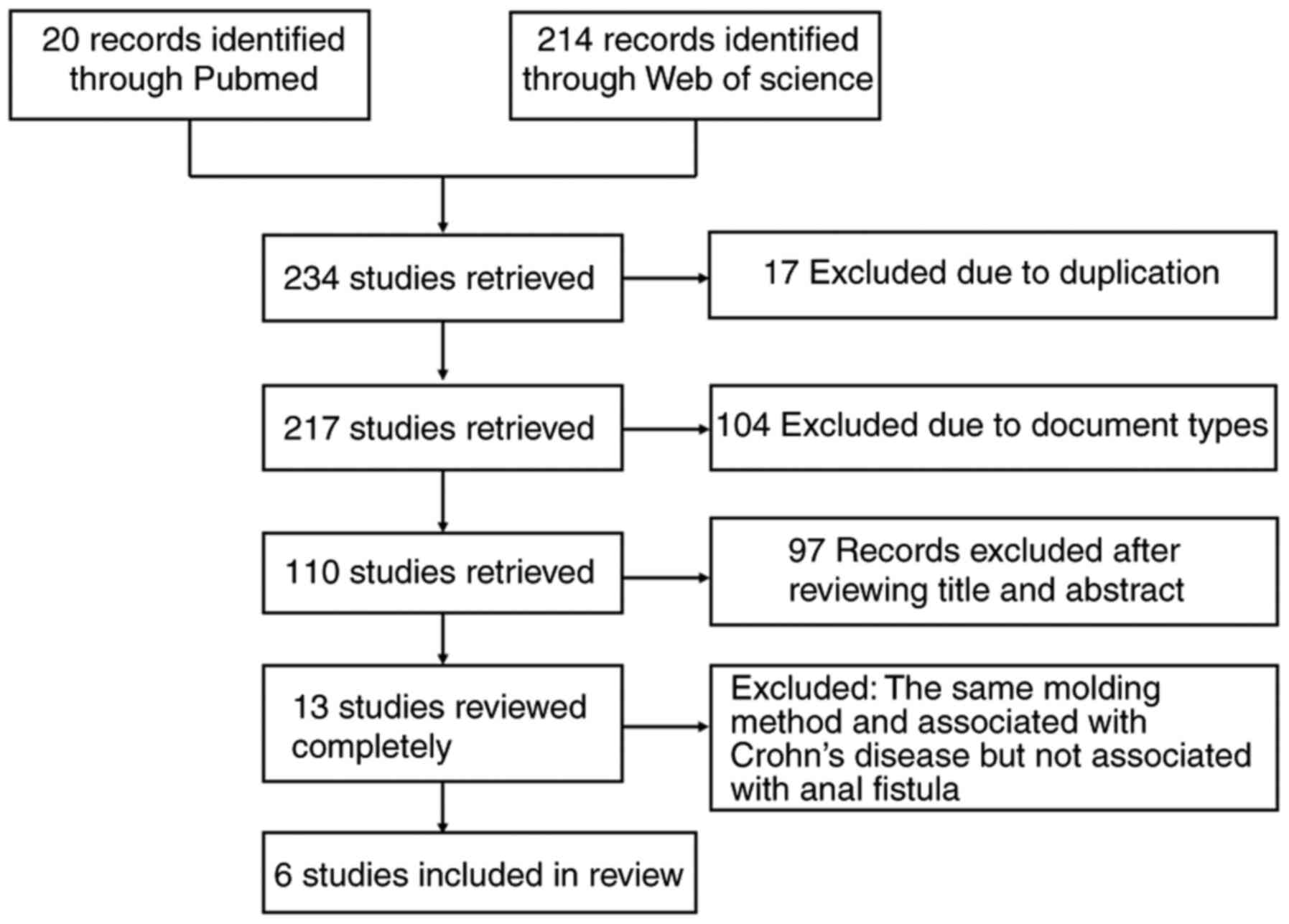
This study was performed according to the PRISMA
guidelines. The search included articles published in PubMed and
Web of Science. Specific search terms were included, such as
‘Crohn's disease AND fistula AND animal model’. All relevant
articles up to December 31, 2020 were included by reviewing the
titles and abstracts regarding animal models of CD with anal
fistula. In addition, the reference lists of the relevant articles
were also screened. The detailed literature screening process is
depicted in Fig. 1. In the initial
literature search, 234 citations were identified. These articles
were examined and six studies were finally selected for inclusion
in the present review. The inclusion criterion was that the content
was required to be relevant to animal experiments on CD with anal
fistula. The exclusion criteria were as follows: i) Simple anal
fistula without any direct association with CD; and ii) repetitive
modeling methods, referring to the same modeling method being used
in different studies. A total of 234 related studies were
retrieved, examined and extracted. After review of the abstract and
full-text, 228 articles were excluded. A total of six articles were
summarized in accordance with the inclusion criteria. The species
of each experimental model and the modeling methods from each study
were characterized. The modeling methods and characteristics are
discussed below.
3. Establishment of body surface fistula
model associated with CD in rats
Ryska et al (12) studied rats using a modified cecal
anastomosis described by Bültmann et al (13). Ryska et al (12) performed the following operations on
rats: The animals underwent a short (2-2.5 cm) median laparotomy
following back fixation. The blind area of the cecum was
subcutaneously pulled through the fascia and muscular layer. The
cecum was exposed to the side of the laparotomy through a
subcutaneous tunnel (2.5 ml/3 cm), simulating the fistula. The
intestine was opened and the edge was fixed to the skin without
adopting the valgus position. This simulated an external opening.
The rats remained at that state for 4 weeks and all fistulas were
verified by fecal secretion. The probes were able to pass through
the fistula freely using X-ray angiography (12).
Ryska et al (12) used this method to establish a rat
model of anal fistula and to observe the efficacy and distribution
of adipose-derived mesenchymal stem cells (ADSCs) that included
bioluminescent molecules. It was concluded that local application
of ADSCs significantly improved the fistula closure rate in this
animal model and may provide a novel method for the treatment of CD
with anal fistula. Considering that the fistula in this model was
artificially created without the background of chronic intestinal
inflammation, a certain localized type of disease must be taken
into account (10). The data
revealed the lack of a complete and accurate clinical correlation
between modified cecostomy and the incidence of CD with anal
fistula (14). Therefore, the
simulated lesion was closer to the external orifice of the perianal
fistula. This study provided evidence supporting the use of ADSCs
in the treatment of perianal CD-related fistula.
4. Establishment of anal fistula model
following enteritis induced by 2,4,6-trinitrobenzensulfonic acid
(TNBS) in rats
In 2020, Flacs et al (15) induced proctitis in rats by rectal
enema using TNBS. After seven days, the sphincter fistula was
established using a surgical line. The anal sphincter was pierced
into the external orifice to form the fistula. The external mouth
was located ~1 cm from the anus. Subsequently, the needle was
removed from the catheter and the surgical line was inserted into
the fistula along the catheter. TNBS was injected twice a week
until it was removed on the 28th day. Each rat was examined by
pelvic magnetic resonance imaging (MRI) prior to and after 7 days
of suture extraction. The rats were then sacrificed and the fistula
was examined by pathological evaluation.
Flacs et al (15) demonstrated that this preclinical
model was optimal. A persistent fistula was confirmed by MRI and
its pathological examination indicated acute and chronic
inflammation, granulation tissue, fibrosis, epithelialization and
proctitis adjacent to the rectum (16). This repeatable preclinical model may
be used to evaluate the effectiveness of innovative treatment for
CD with perianal fistula. It also revealed significant inflammatory
activity in the fistula and emphasized the difference between
common and CD-induced anal fistula (14). In short, this model is able to
successfully reproduce the imaging and pathological characteristics
of CD with anal fistula. This simple and repeatable preclinical
model may be used to evaluate the efficacy of innovative treatments
or strategies for CD with anal fistula.
TNBS is a hapten that induces a direct type 1
T-helper cell response (17). The
latter involves the adaptive immune response for the induction of
colitis. The use of the TNBS reagent for the induction of colitis
is associated with certain limitations. A major limitation of this
model is the lack of sufficient immunopathological relevance of
using a chemically-induced, self-limiting model of colonic
injury-induced inflammation. However, it is an optimal method for
inducing CD-associated enteritis due to its operating success and
repeatability. The use of spontaneously formed or genetic animal
models should also be considered.
5. Spontaneous fistula perianal disease in
the senescence-accelerated mouse (SAM)P1/YitFc strain
Rivera-Nieves et al (18) established a strain of SAMP1/Yit mice
at the University of Virginia in 2003. The phenotypic and immune
characteristics were systematically analyzed at the age of 4, 10,
40 and >60 weeks. The distinctive characteristics between the
SAMP1/YitFc substrain and its parent SAMP1/Yit Japanese strain have
been previously described (19). In
addition, it was reported that perianal inflammation developed into
fistula in a group of mice, in which superficial perianal ulcers
formed fistula bundles similar to those noted in CD-associated
human perineal fistula. This was the first report of an animal
model of IBD at that time.
Rivera-Nieves et al (18) further provided a spontaneous model
of terminal ileitis similar to CD. In addition, since inbreeding of
SAMP1/YitFc mice has been continuously performed for over 20
generations from their original ancestors, genetic variations, by
mutation and/or genetic drift, may have occurred. Rivera-Nieves
et al (18) provided
detailed characteristics of the clinical, pathological and
histological progression of a mouse model of spontaneous
discontinuous chronic ileitis characterized by focal fibrosis and
perianal fistula. This model may provide a unique tool for further
understanding the pathogenesis of chronic intestinal inflammation
of CD and developing novel methods for the treatment of its serious
complications. However, only 5% of the mice developed anal fistula
lesions, which limits the use of the model for the investigation of
the clinical manifestations of CD.
6. Spontaneous enterocutaneous fistulae in
mice following transplantation of human fetal small intestine
Bruckner et al (20) transplanted human fetal intestines
into mice as a novel platform to assess inflammation by
enterocutaneous fistulas (21). The
authors reported successful transplantation of human fetal small
intestines from week 12 to 18 of gestation to the back of mice
(age, 12-16 weeks) with severe combined immune deficiency.
Histopathological and immunohistochemical analyses of the xenostomy
graft indicated that the epithelial cells of the mucosa were
spindle-shaped and formed fistulas to the chronic non-healing wound
in the skin of the host mice covered with the graft. The data
indicated that ~17% of the fully developed enterocutaneous fistulas
were spontaneously localized in the subcutaneous human intestinal
xenograft, which displayed striking histopathological similarity
with CD fistulas (22-26).
A lack of IL-10 gene expression in the inflammatory response of
fully developed human intestinal xenografts to lumen bacteria and
systemic lipopolysaccharides was also reported (27). It is assumed that the human
intestinal xenografts lacking IL-10 gene expression exhibited
inflammation and fistula.
Scharl et al (26) developed a replicable model of
spontaneous enterocutaneous fistula with similar morphology to that
of CD fistula. However, the practicability of their CD-associated
fistula xenograft model requires further verification. Development
of fistula did not occur in all xenografts, suggesting that its
induction may be artificial, which does not accurately reflect the
pathological process of patients with CD.
Mice, which have a strong reproductive capacity and
a short reproductive cycle may be used to establish a variety of
gene-modified mouse models and to spontaneously induce
fistula-associated disease (28,29).
However, due to the long disease cycle and low incidence rate, the
practicability of this model is limited.
7. Improved model of mechanically induced
porcine ARF
Dryden et al (30) established a model of a larger, more
invasive fistula based on Buchanan et al (31), which resembled a clinical fistula.
The original technique was improved by guiding the 14G venous
catheter through the skin incision at different positions (90˚,
180˚ and 270˚, respectively) in the perineum. The catheter was
inserted through the muscle of the anal sphincter (confirmed by
palpation) and the angle was adjusted following penetration of the
rectal mucosa by the catheter. The latter was subsequently removed
and a guide wire was passed through the catheter cavity.
Subsequently, a 20-ml F dilator was passed through the sphincter
above the guide wire. The 4-mm silicon wire was fixed for 4 weeks
through the channel behind the expander. Fistula formation was
confirmed by MRI and microbiological evaluation. After 28 days, the
fat around the aorta was extracted and processed into stromal
vessels. Stromal vascular fraction (SVF) was labeled with primary
ADSCs by membrane staining and subsequently injected into the
fistula or stents. The untreated fistula was injected with fibrin
glue. The healing of the animals was observed at the 2nd and 4th
week and they were subsequently euthanized to evaluate the healing
of the fistula by histological examination. This study made a
preliminary evaluation of isolated adipose mesenchymal primary cell
therapy for ARF and evaluated the role of bioabsorbable scaffolds
in cell therapy. It was suggested that SVF transmitted through
bioabsorbable scaffolds promotes faster healing than SVF or fibrin
glue alone. It was concluded that stent technology may improve the
cure rate of cell therapy for CD-associated ARF.
The model has several characteristics that limit its
applicability to human fistulostomy disease, such as the comparison
of the mechanical nature of its origin with the spontaneous
inflammatory etiology of the disease in humans and the treatment of
model fistulostomy without intervention. An additional limitation
is the distinction between SVF-induced healing and natural healing
at the time prior to the initiation of natural healing, which
rarely occurs in human fistulas, particularly those caused by CD.
In contrast to these findings, surgical treatments may achieve
higher temporary closure rates, whereas they also have a propensity
to injure the anal sphincter and induce incontinence (32,33).
The authors of these studies suggested that the local anatomy of
pigs was similar to that of humans and that the anal fistula model
of large animals confirmed by MRI and histology may be further used
for investigations regarding the diagnosis and treatment of anal
fistula. However, this model is associated with major limitations
with regard to animal feeding, cost, size and repeatability.
The local anatomy of pigs is similar to that of
humans. The large animal model of perianal disease confirmed by
MRI, histology and pathology may be used to study the diagnosis and
therapeutic strategies for CD with anal fistula. Pig models have
been used to simulate the development of CD with anal fistula
(30,34). The pigs were eventually euthanized
to obtain tissue samples and pathological specimens. Compared with
the sacrificing of animals, the application of endoscopy in
research studies is considered a minimally invasive and more humane
method, along with assessing the intestinal condition of the
animals and obtaining tissue specimens (35-37).
8. Canine model of human fistulizing CD
Ferrer et al (38) reported that canine anal furunculosis
(CAF) is a chronic progressive inflammatory disease characterized
by the formation of perianal fistula in dogs. Dogs may naturally
develop an immune disease, which has several characteristics in
common with human CD-induced fistula (39,40).
These studies used spontaneous perianal fistula as a CD model of
fistulostomy. Human embryonic stem cell (hESC)-derived MSCs were
injected into each fistula at the same time to treat CAF. The
therapeutic potential and safety of intracerebral injection of stem
cells from different sources were evaluated by recording the number
and depth of the fistulas at 6 months following treatment and the
average daily dose of cyclosporin required to prevent injury in the
dogs. The results indicated that hESC-MSCs were well tolerated. At
1 month after surgery, the expression levels of serum IL-2 and IL-6
were decreased. The data indicated that the inflammatory cytokines
IL-2 and IL-6 were associated with the development of CD (41). A previous study demonstrated the
safety and therapeutic potential of hESC-MSCs in large animal
models (38). This study for Canine
model of human fistulizing CD was the first preliminary evidence
indicating the use of pluripotent stem cell-derived therapy for the
treatment of fistulas in a large animal model (38). The treatment of spontaneous canine
fistula with expanded hESCs supports the concept that canine models
may be used to evaluate cell therapy. However, this was an open
trial and, as a result, the efficacy was not compared with any
placebo group. In the absence of a predictable time of onset or the
inability to easily induce fistula, this model has limited
applicability to preclinical studies.
Dogs exhibit high similarity with humans in terms of
the anal muscles, glands and biological functions, and they are
considered as one of the few animal species with the type of anal
gland suitable for inducing anal fistula formation. Due to the
complex animal ethics and experimental methodology, only a small
number of studies have been performed using canine models. It has
been suggested that endoscopy should be used in experiments with
large animals, as it is simple and may reduce the level of trauma
to animals (35,37). Furthermore, endoscopy may be used
repeatedly to obtain a more accurate assessment of the disease
status.
9. Discussion
Several issues regarding perianal CD have remained
to be thoroughly addressed. The majority of the causes of anal
fistula originate from anorectal abscess, whereas certain cases of
anal fistula involve inflammatory diseases of the anorectal region,
such as CD and tuberculosis (42).
Previous studies have indicated that two mechanisms appear to have
a major role in the development of CD: Epithelial-to-mesenchymal
transition and matrix-remodeling enzymes (43). The expression levels of matrix
metalloproteinase (MMP) enzymes have exhibited a significant
increase following the development of fistula in CD (43). This effect has been notably observed
with regard to MMP3 protein and mRNA levels (44). Clinical observations have indicated
that antibiotic therapy and fecal diversion may benefit the
management of perianal fistulizing CD, suggesting that the gut
microbiota may also be a pathogenic contributor (45). Investigation of the pathogenesis of
CD is challenging due to the lack of relevant animal models and the
difficulty in obtaining human tissue samples that may accurately
reflect the different stages of fistula formation. It has been
indicated that certain patients with serious cavity diseases have
never had an anal fistula, whereas certain cavity and perianal
lesions exhibited inconsistent reactions, indicating that this type
of complication may involve specific factors and requires more
effective treatment (9).
The present review has certain limitations. First,
the literature search included only animal models of fistula
formation. As a result, other animal models of CD-associated
inflammation were omitted or did not meet the inclusion criteria.
However, it must be considered that fistula formation in animal
models does not fully reflect the condition in humans. In addition,
it is well known that the risk of fistula formation increases with
the duration of the disease. Therefore, animal models are either
not monitored long enough to observe their long-term condition, or
only a limited number of animals develop the disease, which is an
accurate reflection of the clinical condition in humans. In
addition, animal models of spontaneous fistula formation have been
previously reported. Furthermore, clinicians have limited knowledge
of the diagnosis and treatment of emerging fistulas. The inclusion
criteria described in the present study also impose several
limitations. Therefore, it is suggested to develop more specific
animal models that represent the corresponding clinical
condition.
A total of six studies included animal models of CD
with anal fistula, of which two model types were used. Of these,
one study comprised large animal models, such as pigs and dogs.
These are generally considered more similar to humans and may be
used to simulate the development of various clinical diseases in
humans. However, the experimental conditions used in these animals
do not fully represent the clinical condition of CD in humans
(46,47). The second type of animal model
involved immunodeficient mice. Immunodeficiency may exhibit an
inherited predisposition to CD, which may be used to assess the
mechanism of perianal CD (19).
However, due to their physiological structure, the use of endoscopy
in these small animal models to visually assess intestinal
morphology is difficult, which limits the overall feasibility of
evaluating the disease. Optimal pathological tissue specimens may
only be obtained by sacrificing the animals. Therefore, these are
not considered ideal models for anorectal diseases. However,
medium-sized animals, such as rabbits, may be used to assess CD and
perianal disease. Although a rabbit model that resembles human CD
perianal fistula is yet to be established, several studies have
examined experimental colitis in rabbits (38). The rabbit is considered a
medium-to-large size animal. Rabbits are easy to maintain and
rabbit models are cost-effective and have satisfactory
repeatability and operability. A variety of preclinical rabbit
models have been widely reported (48-50).
In these models, the auxiliary examinations of the human body, such
as ultrasound, CT, MRI and endoscopy, are able to be successfully
simulated. It is worth emphasizing that rabbit models allow the use
of endoscopy, which is valuable (51). An accumulating number of studies
have confirmed that rabbit models of immune deficiency were
successfully established (52,53).
However, additional experimental data are required to support and
confirm the utility of rabbits in preclinical models of perianal
fistulizing CD.
It remains necessary, albeit challenging, to
establish a suitable preclinical model of CD with anal fistula.
This model requires to be assessed with regard to safety,
repeatability and clinical similarity to humans. Perianal disease
treatment technology may be used for CD. However, an ideal animal
model is required. The optimal animal model should include the
genetically-mediated development of CD with anal fistula. However,
an ideal model for preclinical research is difficult to be
established due to the long experimental period required.
Therefore, the evaluation and identification of a relatively
practical and reliable animal model is required to study CD with
anal fistula.
10. Conclusion
In conclusion, a total of six existing preclinical
studies of perianal fistulizing CD that focused on intestinal
inflammation or fistula were discussed in the present review. Of
note, although considerable progress in research on animal models
of perianal fistulizing CD has been made, the extrapolation of the
basic research data to the clinical setting requires additional
study.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
Data sharing is not applicable.
Authors' contributions
SL, KZ, YG and EW made substantial contributions to
the conception and design, acquisition of data and analysis and
interpretation of data. SL KZ, YG and EW performed the literature
search. JH was involved in drafting the manuscript, revising it
critically for important intellectual content and gave final
approval of the version to be published. SL and JH confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Schwartz DA, Loftus EV Jr, Tremaine WJ,
Panaccione R, Harmsen WS, Zinsmeister AR and Sandborn WJ: The
natural history of fistulizing Crohn's disease in olmsted county,
minnesota. Gastroenterology. 122:875–880. 2002.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Verstockt B, Smith KG and Lee JC:
Genome-wide association studies in Crohn's disease: Past, present
and future. Clin Transl Immunology. 7(e1001)2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Balfour Sartor R: Bacteria in Crohn's
disease: Mechanisms of inflammation and therapeutic implications. J
Clin Gastroenterol. 41 (Suppl 1):S37–S43. 2007.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Balfour Sartor R: Enteric microflora in
IBD: Pathogens or commensals? Inflamm Bowel Dis. 3:230–235.
1997.PubMed/NCBI
|
|
5
|
Sartor RB: Microbial influences in
inflammatory bowel diseases. Gastroenterology. 134:577–594.
2008.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Lahat A, Assulin Y, Beer-Gabel M and
Chowers Y: Endoscopic ultrasound for perianal Crohn's disease:
Disease and fistula characteristics, and impact on therapy. J
Crohns Colitis. 6:311–316. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Zhu P, Chen Y, Gu Y, Chen H, An X, Cheng
Y, Gao Y and Yang B: Analysis of clinical characteristics of
perianal Crohn's disease in a single-center. Zhonghua Wei Chang Wai
Ke Za Zhi. 19:1384–1388. 2016.PubMed/NCBI(In Chinese).
|
|
8
|
Molodecky NA, Soon IS, Rabi DM, Ghali WA,
Ferris M, Chernoff G, Benchimol EI, Panaccione R, Ghosh S, Barkema
HW and Kaplan GG: Increasing incidence and prevalence of the
inflammatory bowel diseases with time, based on systematic review.
Gastroenterology. 142:46–54.e42, e30. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Panés J and Rimola J: Perianal fistulizing
Crohn's disease: Pathogenesis, diagnosis and therapy. Nat Rev
Gastroenterol Hepatol. 14:652–664. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hellers G, Bergstrand O, Ewerth S and
Holmström B: Occurrence and outcome after primary treatment of anal
fistulae in Crohn's disease. Gut. 21:525–527. 1980.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Garcia-Olmo D, Herreros D, Pascual I,
Pascual JA, Del-Valle E, Zorrilla J, De-La-Quintana P,
Garcia-Arranz M and Pascual M: Expanded adipose-derived stem cells
for the treatment of complex perianal fistula: A phase II clinical
trial. Dis Colon Rectum. 52:79–86. 2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Ryska O, Serclova Z, Mestak O, Matouskova
E, Vesely P and Mrazova I: Local application of adipose-derived
mesenchymal stem cells supports the healing of fistula: Prospective
randomised study on rat model of fistulising Crohn's disease. Scand
J Gastroentero. 52:543–550. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Bültmann O, Philipp C, Ladeburg M and
Berlien HP: Creation of a caecostoma in mice as a model of an
enterocutaneous fistula. Res Exp Med (Berl). 198:215–228.
1998.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Panés J, García-Olmo D, Van Assche G,
Colombel JF, Reinisch W, Baumgart DC, Dignass A, Nachury M,
Ferrante M, Kazemi-Shirazi L, et al: Expanded allogeneic
adipose-derived mesenchymal stem cells (Cx601) for complex perianal
fistulas in Crohn's disease: A phase 3 randomized, double-blind
controlled trial. Lancet. 388:1281–1290. 2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Flacs M, Collard M, Doblas S, Zappa M,
Cazals-Hatem D, Maggiori L, Panis Y, Treton X and Ogier-Denis E:
Preclinical model of perianal fistulizing Crohn's disease. Inflamm
Bowel Dis. 26:687–696. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Soker G, Gulek B, Yilmaz C, Kaya O, Arslan
M, Dilek O, Gorur M, Kuscu F and İrkorucu O: The comparison of CT
fistulography and MR imaging of perianal fistulae with surgical
findings: A case-control study. Abdom Radiol (NY). 41:1474–1483.
2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Camoglio L, Te Velde AA, de Boer A, ten
Kate FJ, Kopf M and van Deventer SJ: Hapten-induced colitis
associated with maintained Th1 and inflammatory responses in
IFN-gamma receptor-deficient mice. Eur J Immunol. 30:1486–1495.
2000.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Rivera-Nieves J, Bamias G, Vidrich A,
Marini M, Pizarro TT, McDuffie MJ, Moskaluk CA, Cohn SM and
Cominelli F: Emergence of perianal fistulizing disease in the
SAMP1/YitFc mouse, a spontaneous model of chronic ileitis.
Gastroenterology. 124:972–982. 2003.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Matsumoto S, Okabe Y, Setoyama H, Takayama
K, Ohtsuka J, Funahashi H, Imaoka A, Okada Y and Umesaki Y:
Inflammatory bowel disease-like enteritis and caecitis in a
senescence accelerated mouse P1/Yit strain. Gut. 43:71–78.
1998.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Bruckner RS, Nissim-Eliraz E, Marsiano N,
Nir E, Shemesh H, Leutenegger M, Gottier C, Lang S, Spalinger MR,
Leibl S, et al: Transplantation of human intestine into the mouse:
A novel platform for study of inflammatory enterocutaneous
fistulas. J Crohns Colitis. 13:798–806. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Savidge TC, Morey AL, Ferguson DJ, Fleming
KA, Shmakov AN and Phillips AD: Human intestinal development in a
severe-combined immunodeficient xenograft model. Differentiation.
58:361–371. 1995.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Scharl M, Huber N, Lang S, Fürst A, Jehle
E and Rogler G: Hallmarks of epithelial to mesenchymal transition
are detectable in Crohn's disease associated intestinal fibrosis.
Clin Transl Med. 4(1)2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Scharl M and Rogler G: Pathophysiology of
fistula formation in Crohn's disease. World J Gastrointest
Pathophysiol. 5:205–212. 2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Scharl M, Frei P, Frei SM, Biedermann L,
Weber A and Rogler G: Epithelial-to-mesenchymal transition in a
fistula-associated anal adenocarcinoma in a patient with
long-standing Crohn's disease. Eur J Gastroenterol Hepatol.
26:114–118. 2014.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Frei SM, Hemsley C, Pesch T, Lang S, Weber
A, Jehle E, Rühl A, Fried M, Rogler G and Scharl M: The role for
dickkopf-homolog-1 in the pathogenesis of Crohn's
disease-associated fistulae. PLoS One. 8(e78882)2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Scharl M, Weber A, Fürst A, Farkas S,
Jehle E, Pesch T, Kellermeier S, Fried M and Rogler G: Potential
role for SNAIL family transcription factors in the etiology of
Crohn's disease-associated fistulae. Inflamm Bowel Dis.
17:1907–1916. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Nissim-Eliraz E, Nir E, Shoval I, Marsiano
N, Nissan I, Shemesh H, Nagy N, Goldstein AM, Gutnick M, Rosenshine
I, et al: Type three secretion system-dependent microvascular
thrombosis and ischemic enteritis in human gut xenografts infected
with enteropathogenic Escherichia coli. Infect Immun.
85:e00558–17. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Demeyer A, Van Nuffel E, Baudelet G,
Driege Y, Kreike M, Muyllaert D, Staal J and Beyaert R:
MALT1-deficient mice develop atopic-like dermatitis upon aging.
Front Immunol. 10(2330)2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Peintner L, Venkatraman A, Waeldin A,
Hofherr A, Busch T, Voronov A, Viau A, Kuehn EW, Köttgen M and
Borner C: Loss of PKD1/polycystin-1 impairs lysosomal activity in a
CAPN (calpain)-dependent manner. Autophagy. 1–17. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Dryden GW, Boland E, Yajnik V and Williams
S: Comparison of stromal vascular fraction with or without a novel
bioscaffold to fibrin glue in a porcine model of mechanically
induced anorectal fistula. Inflamm Bowel Dis. 23:1962–1971.
2017.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Buchanan GN, Sibbons P, Osborn M, Bartram
CI, Ansari T, Halligan S and Cohen CR: Experimental model of
fistula-in-ano. Dis Colon Rectum. 48:353–356. 2005.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Rehg KL, Sanchez JE, Krieger BR and Marcet
JE: Fecal diversion in perirectal fistulizing Crohn's disease is an
underutilized and potentially temporary means of successful
treatment. Am Surg. 75:715–718. 2009.PubMed/NCBI
|
|
33
|
Galandiuk S, Kimberling J, Al-Mishlab TG
and Stromberg AJ: Perianal Crohn disease: Predictors of need for
permanent diversion. Ann Surg. 241:796–801. 2005.PubMed/NCBI View Article : Google Scholar
|
|
34
|
A Ba-Bai-Ke-Re MM, Chen H, Liu X and Wang
YH: Experimental porcine model of complex fistula-in-ano. World J
Gastroentero. 23:1828–1835. 2017.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Radefeld K, Papp S, Havlicek V, Morrell
JM, Brem G and Besenfelder U: Endoscopy-mediated intratubal
insemination in the cow-development of a novel minimally invasive
AI technique. Theriogenology. 115:117–123. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Sobel DS: Upper respiratory tract
endoscopy in the cat: A minimally invasive approach to diagnostics
and therapeutics. J Feline Med Surg. 15:1007–1017. 2013.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Salavati M, Pérez-Accino J, Tan YL, Liuti
T, Smith S, Morrison L and Salavati Schmitz S: Correlation of
minimally invasive imaging techniques to assess intestinal mucosal
perfusion with established markers of chronic inflammatory
enteropathy in dogs. J Vet Intern Med. 35:162–171. 2021.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Ferrer L, Kimbrel EA, Lam A, Falk EB, Zewe
C, Juopperi T, Lanza R and Hoffman A: Treatment of perianal
fistulas with human embryonic stem cell-derived mesenchymal stem
cells: A canine model of human fistulizing Crohn's disease. Regen
Med. 11:33–43. 2016.PubMed/NCBI View Article : Google Scholar
|
|
39
|
House AK, Gregory SP and Catchpole B:
Pattern-recognition receptor mRNA expression and function in canine
monocyte/macrophages and relevance to canine anal furunuclosis. Vet
Immunol Immunopathol. 124:230–240. 2008.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Massey J, Short AD, Catchpole B, House A,
Day MJ, Lohi H, Ollier WE and Kennedy LJ: Genetics of canine anal
furunculosis in the German shepherd dog. Immunogenetics.
66:311–324. 2014.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Patterson AP and Campbell KL: Managing
anal furunculosis in dogs. Compend Contin Educ Vet. 27:339–355.
2005.
|
|
42
|
Sandborn WJ, Fazio VW, Feagan BG and
Hanauer SB: American Gastroenterological Association Clinical
Practice Committee. AGA technical review on perianal Crohn's
disease. Gastroenterology. 125:1508–1530. 2003.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Parks AG, Gordon PH and Hardcastle JD: A
classification of fistula-in-ano. Br J Surg. 63:1–12.
1976.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Kirkegaard T, Hansen A, Bruun E and
Brynskov J: Expression and localisation of matrix
metalloproteinases and their natural inhibitors in fistulae of
patients with Crohn's disease. Gut. 53:701–709. 2004.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Seow-Choen F, Hay AJ, Heard S and Phillips
RK: Bacteriology of anal fistulae. Br J Surg. 79:27–28.
1992.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Mazzoni M, Caremoli F, Cabanillas L, de
Los Santos J, Million M, Larauche M, Clavenzani P, De Giorgio R and
Sternini C: Quantitative analysis of enteric neurons containing
choline acetyltransferase and nitric oxide synthase
immunoreactivities in the submucosal and myenteric plexuses of the
porcine colon. Cell Tissue Res. 383:645–654. 2021.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Zhang Y, Li F, Wang H, Yin C, Huang J,
Mahavadi S, Murthy KS and Hu W: Immune/Inflammatory response and
hypocontractility of rabbit colonic smooth muscle After
TNBS-induced colitis. Dig Dis Sci. 61:1925–1940. 2016.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Murai K, Hamamoto S, Okuma T, Kageyama K,
Yamamoto A, Ogawa S, Nota T, Sohgawa E, Jogo A and Miki Y: Survival
benefit of radiofrequency ablation with intratumoral cisplatin
administration in a rabbit VX2 lung tumor model. Cardiovasc
Intervent Radiol. 44:475–481. 2021.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Schilling AL, Moore J, Kulahci Y, Little
SR, Rigatti LH, Wang EW and Lee SE: Evaluating inflammation in an
obstruction-based chronic rhinosinusitis model in rabbits. Int
Forum Allergy Rhinol. 11:807–809. 2021.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Boissady E, Kohlhauer M, Lidouren F,
Hocini H, Lefebvre C, Chateau-Jouber S, Mongardon N, Deye N, Cariou
A, Micheau P, et al: Ultrafast hypothermia selectively mitigates
the early humoral response after cardiac arrest. J Am Heart Assoc.
9(e017413)2020.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Huang J, Shuang J, Xiong G, Wang X, Zhang
Y, Tang X, Fan Z, Shen Y, Song H and Liu Z: Establishing a rabbit
model of malignant esophagostenosis using the endoscopic
implantation technique for studies on stent innovation. J Transl
Med. 12(40)2014.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Hashikawa Y, Hayashi R, Tajima M, Okubo T,
Azuma S, Kuwamura M, Takai N, Osada Y, Kunihiro Y, Mashimo T and
Nishida K: Generation of knockout rabbits with X-linked severe
combined immunodeficiency (X-SCID) using CRISPR/Cas9. Sci Rep.
10(9957)2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Song J, Wang G, Hoenerhoff MJ, Ruan J,
Yang D, Zhang J, Yang J, Lester PA, Sigler R, Bradley M, et al:
Bacterial and pneumocystis infections in the lungs of gene-knockout
rabbits with severe combined immunodeficiency. Front Immunol.
9(429)2018.PubMed/NCBI View Article : Google Scholar
|