Introduction
In patients with advanced primary or recurrent
gynecologic (1), urologic or rectal
cancers without metastatic disease, extensive aggressive surgery
such as pelvic exenteration may be necessary for curative intent
treatment (2).
Brunschwig was the first to describe pelvic
exenteration in the 1940s (3).
Exenteration was initially considered as a palliative procedure for
patients with extensive pelvic cancers, with an extremely high
perioperative mortality rate of 23%. After significant progress
related to patient selection, improvements in operative techniques
and intensive care, the mortality rate has decreased to 3-9% as
documented in more recent studies (4,5), and a
5-year survival rate between 20 and 60% (6-14)
with a good quality of life (15).
Primary pelvic exenteration is considered as a
first-line radical surgical procedure for patients with advanced
pelvic malignancies, prior to any oncological treatment. It may be
performed in patients with stage IVa gynecological cancers
(1), tumor-associated urogenital or
rectogenital fistulas, and in some cases, when the histology of the
disease (soft tissue sarcomas, neuroendocrine tumors) predicts
chemoradiation therapy resistance (13). This procedure may also be performed
in rare malignant conditions such as synchronous pelvic cancers
(16).
The procedure may be classified as total (removal of
the tumor together with the uterus, vagina, urinary bladder and
rectum), anterior (bladder, uterus and vagina) and posterior
(rectum, uterus and vagina). In relation to the levator ani muscle,
the procedure is classified as supralevatorian, infralevatorian or
infralevatorian with vulvectomy. The surgical procedure includes an
exenterative phase followed by a reconstructive phase consisting of
a continent or incontinent urinary diversion, definitive end
colostomy or low rectal anastomosis or and vaginal and pelvic floor
reconstruction (17,18).
The aim of the present study was to analyze our
primary pelvic exenteration experience from a single institution in
patients with locally advanced primary pelvic malignancies.
Materials and methods
This study is a retrospective analysis of all
patients who underwent primary pelvic exenteration for advanced
pelvic cancer in a tertiary university hospital in Târgu Mures,
Romania. The study was approved by the Ethics Medical Committee of
the University Hospital of Târgu Mureș (protocol code 27227,
10/03/2020). Twenty-three primary exenterations were performed
between August 2011 and July 2020. Informed consent was obtained
from every case, and all patients were evaluated by the
anesthesiology team in order to evaluate their medical condition to
support a complex surgical intervention. All procedures were
performed with a curative intent, not for palliation purposes. None
of the patients submitted to primary pelvic exenteration received
neoadjuvant treatment. The exenteration was the first therapeutic
approach. For exclusion of all oncological contraindications for
pelvic exenteration and assessment of operability, the preoperative
work-up included a mandatory transvaginal or transrectal echography
plus magnetic resonance imaging (MRI) or computed tomography (CT).
All patients proposed for a total or anterior exenteration
underwent cystoscopy, or a colonoscopy for total or posterior
exenteration. Among the 27 patients identified, pelvic exenteration
was abandoned in 4 patients due to oncological contraindications
encountered during surgery: Pelvic sidewall involvement of the
tumor with extension to the bony structures and involvement of the
neurovascular structures of the sciatic foramen in 1 patient, or
the detection of peritoneal carcinomatosis in 3 cases, which had
not been described in the preoperative imaging work-up. Data were
collected from the medical records and consisted of patient
demographics, types of malignancy, details on intraoperative
management provided, postoperative complications and follow-up. The
presence of postoperative complications were assessed according to
the Clavien-Dindo scale (19).
Statistical analysis
Statistical analysis was performed using the SPSS
21.0 statistical package (IBM Corp.). Quantitative variables are
presented as mean and median, while qualitative and categorical
variables are expressed both as integer and percentage values.
Survival curve was calculated using the Kaplan-Meier method.
Results
Epidemiological and preoperative
clinical characteristics
Over a period of 9 years, 23 patients underwent
primary exenteration for locally advanced stage pelvic cancers. The
median patient age at the time of surgery was 55 years (range,
43-72 years). The origin of the primary tumor included stage IVa
cervical cancer (11 cases, 48.9%), stage IVa endometrial cancer (1
case, 4.3%), stage IVa vaginal cancer (6 cases, 26%), stage IIIb
bladder cancer (3 cases, 13%), stage IIIc rectal cancer (1 case,
4.3%) and undifferentiated pelvic sarcoma (1 case, 4.3%) (Table I). Ten out of the 17 patients with
stage IVa cervical or vaginal cancer had already developed a
vesico-vaginal (8 women) or recto-vaginal (2 women) fistula at the
moment of surgery; also, the patient with stage IIIc rectal cancer
had developed a recto-vaginal fistula.
 | Table IDemographic characteristics and
intraoperative details of the patients undergoing
exenterations. |
Table I
Demographic characteristics and
intraoperative details of the patients undergoing
exenterations.
|
Characteristics/intraoperative
details | | Data values |
|---|
| Mean age (range) in
years | | 53.5 (43-72) |
| Origin of malignancy,
n (%) | Stage/histological
type | |
|
Cervical | Iva/squamous cell
carcinoma | 11 (48.9%) |
|
Endometrial |
Iva/adenocarcinoma | 1 (4.3%) |
|
Vaginal | Iva/squamous cell
carcinoma | 6 (26%) |
|
Bladder | IIIb/urothelial
carcinoma | 3 (13%) |
|
Rectum |
IIIc/adenocarcinoma | 1 (4.3%) |
|
Undifferentiated
pelvic sarcoma, n (%) | | 1 (4.3%) |
| Type of exenteration
regarding topography, n (%) | | |
|
Total | | 7 (30.5%) |
|
Anterior | | 13 (56.5%) |
|
Posterior | | 3 (13%) |
| Type of exenteration
regarding the levator ani muscle, n (%) | | |
|
Supralevatorian | | 13 (56.5%) |
|
Infralevatorian | | 10 (43.5%) |
|
Infralevatorian
with vulvectomy | | 5 (21.7%) |
| Type of urinary tract
reconstruction, n (%) | | |
|
Non-continent
urinary conduit type Bricker | | 20 (87%) |
| Type of bowel
reconstruction, n (%) | | |
|
Colostomy | | 7 (30.5%) |
|
Colorectal
anastomosis | | 3 (13%) |
| Length of surgery
(min), median (range) | | 364 (270-560) |
| Estimated blood loss
(ml), median (range) | | 600 (300-2,100) |
| Transfusion volumes
(ml), median (range) | | 700 (0-1,800) |
| Hospital stay after
PE (days), median (range) | | 20 (11-75) |
Procedures and complications of pelvic
exenteration
As the type of exenterative procedure was related to
the tumor involvement of pelvic organs, 7 (30.5%) patients required
total exenteration, 13 (56.5%) procedures were anterior and 3 (13%)
were posterior exenterations. Regarding the levator ani muscle,
with the aim to obtain tumor-free resection margins, 13 (56.5%)
pelvic exenterations were supralevatorian, 10 (43.5%)
infralevatorian, and 5 (21.7%) were infralevatorian with
vulvectomy.
Among the 10 patients with total or posterior pelvic
exenterations, a low rectal anastomosis was performed in 3 cases
and in 7 patients an end definitive colostomy was conducted due to
insufficient unaffected rectal stump. Urinary diversion procedures
were performed for all patients who underwent a total or anterior
exenteration, tailoring a Bricker's ileal (in 15 patients) or
sigmoid (in 5) incontinent conduit (20), technically easier to perform
compared to other urinary diversion procedures and also associated
with lower rates of postoperative complications. The option for an
ileal or sigmoid urinary conduit after total exenteration is
dependent on the remaining length of the sigmoid colon and on the
avoidance of an unnecessary ileal anastomosis needed for the ileal
conduit. In all anterior exenterations, an ileal conduit was
performed. All ureteric-enteral anastomoses were adjusted on
‘double J’ ureteral stents in order to prevent a subsequent
stenosis. The median length of surgery (364 min), median estimated
blood loss (610 ml) and the need for transfusion in our series are
documented in Table I.
All of the patients were maintained in the intensive
care unit for more than 4 days for close monitoring due to the
complexity of the procedure and for postoperative therapy as
antithrombotic prophylaxis, total parenteral nutrition,
prophylactic antibiotic treatment and intravenous albumin
administration.
Upon final pathology report, clear resection margins
were achieved only in 19 out of 23 patients (86.2%). All 5 patients
with positive margins were sent for adjuvant chemotherapy.
No major intraoperative complications occurred.
Postoperative complications were characterized using the
Clavien-Dindo classification (19).
Seven patients (30.5%) experienced early complications and 1
patient presented with late complication, respectively (Table II). Among the early complications,
one Clavien-Dindo grade V was registered, a patient 46 years of
age, referred to the hospital for stage IIIB bladder cancer. This
patient underwent an anterior supralevatorian exenteration, with no
intraoperative complications, but experienced sudden death on the
16th postoperative day due to a pulmonary embolism after home
discharge. Four patients experienced Clavien-Dindo grade IIIb
complications: Enteric fistulas-3 ileal fistulas with peritonitis
and one entero-vaginal fistula, all necessitating re-laparotomies
and ileum re-anastomosis. Two patients who underwent
infralevatorian exenteration with vulvectomy developed a perineal
wound infection with tissue necrosis, necessitating prolonged local
treatment (Clavien-Dindo grade II). Only one patient has
experienced a late complication: A ureteric-enteral stenosis solved
finally by a unilateral permanent percutaneous nephrostomy.
 | Table IIEarly and late complications and
survival outcomes of the patients (n=23) after pelvic
exenteration. |
Table II
Early and late complications and
survival outcomes of the patients (n=23) after pelvic
exenteration.
| Complications | | Total study group
data [n (%)] |
|---|
| Early
complications | | 7 (30.5) |
|
Clavien-Dindo
grade II | Perineal wound
infection | 2 (8.7) |
|
Clavien-Dindograde
IIIb | Bowel fistula | 4 (17.4) |
|
Clavien-Dindograde
V | Pulmonary
embolism | 1 (4.3) |
| Late
complications | | |
|
Urostomy
stenosis | | 1 (4.3) |
| Survival
outcomes | | |
|
Alive, free
of disease | | 15 (65.2) |
|
Deceased | | 8 (34.8) |
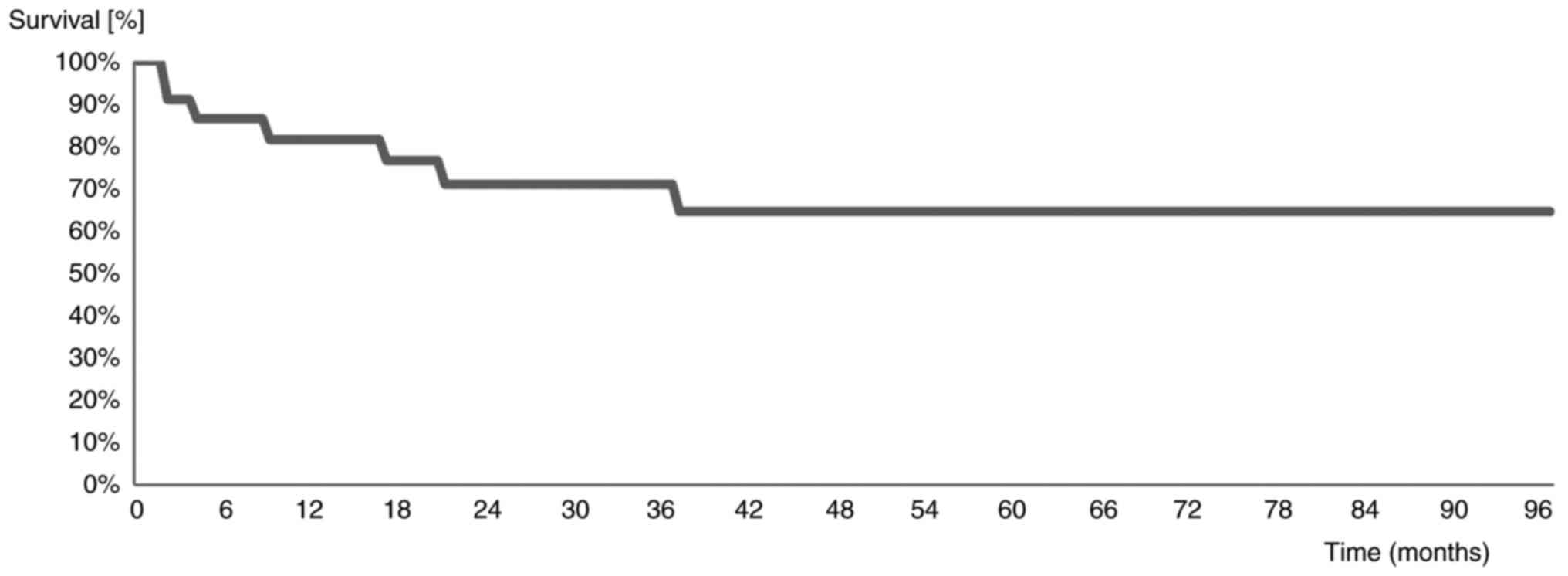
Survival outcomes
Over a median follow-up period of 35 months, 8
(34.8%) patients died. The median overall survival (OS) was 33
months (range, 1-96 months) (Fig.
1). The 2-year and 5-year survival rates were 72 and 66%,
respectively.
Discussion
Although pelvic exenteration is originally intended
as a palliative procedure, currently it is performed with curative
intent for the treatment of pelvic disease (rectal, cervical,
endometrial, vaginal, bladder and soft-tissue sarcoma) (13). The most important parameter in the
evaluation of risk related to the operative procedure is the
mortality rate. Since the initial description, mortality rates have
improved from higher than 30% to more acceptable rates of 0 to 10%
with 5-year OS varying between 20 and 60%, despite the high
morbidity rate (5-14,21-24).
In the present study, there was one perioperative death (4.3%) due
to a sudden pulmonary embolism, despite prophylactic anticoagulant
protocol during hospitalization and home discharge and patient
early mobilization.
The role of pelvic exenteration for pelvic
recurrences after gynecologic, urinary or rectal cancers for
patients previously irradiated, when no other therapeutic options
with curative intent are available, is well established. In
contrast, there is a continued debate between oncologic surgeons
and medical oncologists or radiotherapists regarding primary pelvic
exenterations. The evidence is scarce and only a few studies on
this topic have been published (1,2,7-11,13,14).
In our study, 11 out of the 23 patients (47.8%) with primary pelvic
exenterations had fistulas at the moment of surgery, a condition
that inevitably alters the quality of life of these patients and
that will not be solved by oncologic treatment. The 5-year OS after
pelvic exenterations which ranges between 20 and 60% in all
studies, when the oncological indications and contraindications are
respected (5-14,22-26),
is similar or higher compared to the OS after chemoradiotherapy for
these advanced pelvic cancers, considered separately. Kramer et
al (25) reported that 22% of
his patients who underwent radiochemotherapy for locally advanced
cervical cancer with curative intent developed fistulas, and the
5-year survival rate was 18%. Moore et al (26) reported that fistulas appeared as a
complication of radiochemotherapy in 48% of his patients.
For all our primary pelvic exenteration patients, a
5-year survival probability of 43% was calculated, which is similar
to the rates found in other studies on exenteration (6-14,27).
Also, the morbidity after exenteration was comparable: In our
study, 4 patients (17%) had to endure a second surgery due to bowel
fistulas. Other early minor complications in our series included
perineal wound infection in 2 (8.7%) patients. Late postoperative
complications were noted in one case (4.3%), presenting an urostomy
stenosis. The relatively high 5-year OS and low morbidity after the
procedure are strong arguments in favor of primary pelvic
exenteration. The current series supports the increasing number of
studies regarding the role of pelvic exenteration for selected
patients with locally advanced primary pelvic malignancies
(28).
In light of the associated morbidities, the aim of
any exenterative surgery must include the achievement of tumor-free
margins. Existing tumors that are fixed to the lower pelvic side
wall have long been regarded as a contraindication for pelvic
exenterations, but the role of laterally extended pelvic resection
(LEER) in the surgical treatment of pelvic malignancies has been
confirmed by some reports (29,30).
The only contraindication for LEER is the involvement of the
sciatic nerve (31), but these
pelvic side wall resections are technically difficult and may be
associated with increased risks because of frequent anatomic
anomalies (31).
Completeness of the tumor resection was the only
variable with a significant impact on survival according to Zoucas
et al (32). In our series,
clear resection margins were achieved in only 82.6% of the
patients. The inferior resection line (urethra, vagina and rectum)
has been proven to be the weak point for the majority of patients
with a positive microscopic resection line and this has to be
pushed downwards as much as necessary to obtain clear margins.
An important element of every pelvic exenteration
procedure, affecting the duration of surgery, the frequency and
type of complication, and the postoperative quality of life, is the
method used for urinary and/or fecal diversion. The Bricker
procedure remains the most performed technique for urinary
diversion (20). In our study
group, this method was applied in all patients with anterior or
total exenteration. But, in recent years, the low rectal
anastomosis and orthotopic continent urinary diversions are more
commonly used after pelvic exenterations, mainly for patients more
fit for a prolonged surgery and without tumor involvement of the
bladder neck or lower rectum. These surgical techniques avoid the
need of external stomas and, subsequently, the patient quality of
life is significantly improved (33).
The learning curve for pelvic exenterations is long
for the entire involved team (gynecologic oncologist surgeon,
anesthesiologist, radiologist, urologist). Comparing the early
period when pelvic exenteration was implemented in our department,
in recent years, the tendency is to lower significantly the
operative time, the blood transfusion volume and the complication
rate. After more than 80 pelvic exenterations (not only primary)
already performed by our team, the shift towards implementation of
Enhanced Recovery After Surgery (ERAS) protocols before, during and
after pelvic exenteration has contributed to better outcomes for
these extremely fragile patients with advanced pelvic malignancies,
but this issue will be the subject of another future paper.
Major biases of our study are the retrospective
nature of the analysis, the heterogeneity of the advanced pelvic
cancers and the relatively small number of patients. Yet, these
factors are present in the majority of series reported in the
literature in regards to pelvic exenteration. Despite these
limitations, our study has contributed to the evidence that primary
pelvic exenteration is a feasible surgical option for selected
patients with locally advanced pelvic malignancies accompanied by
acceptable long-term outcomes. It is imperative to adopt a
multidisciplinary approach when performing such technically
demanding operations to achieve better outcomes for the patients.
In the near future, considering the new data regarding the safety
of minimally invasive surgery in the treatment of cervical cancer
(34), the exenterative and
partially the reconstructive phase might be performed by
laparoscopic or robotic surgery.
Primary pelvic exenteration for locally advanced
pelvic malignancies is accompanied by considerable morbidity, but
with acceptable OS. The eligibility of patients for this radical
surgical approach should be assessed by careful patients'
selection, preoperative counseling and should be carried out only
in surgical centers with well trained staff.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data that support the findings of this study are
available from the corresponding author (MG), upon reasonable
request.
Authors' contributions
MG contributed to the conception and design of the
study, analysis and interpretation of the patient data. MEC
contributed to the conception and design of the study and was the
leading surgeon for all the surgical procedures described in our
report. ALC, SLK and MS contributed to the acquisition of the
patient data, the analysis and interpretation of the data of the
study and the writing of the article. AAM and NB supervised the
work and revised the article, contributed to the drafting of the
work and its critical revision for important intellectual content.
All authors read and approved the final manuscript and agree to be
accountable for all aspects of the research in ensuring that the
accuracy or integrity of any part of the work are appropriately
investigated and resolved.
Ethics approval and consent to
participate
The study was conducted according to the guidelines
of the Declaration of Helsinki, and approved by the Ethics Medical
Committee of the University Hospital of Târgu Mureș (protocol code
27227, 10/03/2020).
Patient consent for publication
This manuscript does not contain case details,
personal information or images that may enable an individual to be
identified.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Ungar L, Palfalvi L and Novak Z: Primary
pelvic exenteration in cervical cancer patients. Gynecol Oncol. 111
(Suppl 2):S9–S12. 2008.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Maggioni A, Roviglione G, Landoni F,
Zanagnolo V, Peiretti M, Colombo N, Bocciolone L, Biffi R, Minig L
and Morrow CP: Pelvic exenteration: Ten-year experience at the
European institute of oncology in Milan. Gynecol Oncol. 114:64–68.
2009.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Brunschwig A: Complete excision of pelvic
viscera for abdominal carcinoma; a one-stage abdominoperineal
operation with end colostomy and bilateral ureteral implantation
into the colon above the colostomy. Cancer. 1:177–183.
1948.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Berek JS, Howe C, Lagasse LD and Hacker
NF: Pelvic exenteration for recurrent gynecologic malignancy:
Survival and morbidity analysis of the 45-year experience at UCLA.
Gynecol Oncol. 99:153–159. 2005.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Forner DM and Lampe B: Intestinal
complications after pelvic exenterations in gynecologic oncology.
Int J Gynecol Cancer. 19:958–962. 2009.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Khoury-Collado F, Einstein MH, Bochner BH,
Alektiar KM, Sonoda Y, Abu-Rustum NR, Brown CL, Gardner GJ, Barakat
RR and Chi DS: Pelvic exenteration with curative intent for
recurrent uterine malignancies. Gynecol Oncol. 124:42–47.
2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Fotopoulou C, Neumann U, Kraetschell R,
Schefold JC, Weidemann H, Lichtenegger W and Sehouli J: Long-term
clinical outcome of pelvic exenteration in patients with advanced
gynecological malignancies. J Surg Oncol. 101:507–512.
2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Schmidt AM, Imesch P, Fink D and Egger H:
Indications and long-term clinical outcomes in 282 patients with
pelvic exenteration for advanced or recurrent cervical cancer.
Gynecol Oncol. 125:604–609. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Soper JT, Berchuck A, Creasman WT and
Clarke-Pearson DL: Pelvic exenteration: Factors associated with
major surgical morbidity. Gynecol Oncol. 35:93–98. 1989.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Anthopoulos AP, Manetta A, Larson JE,
Podczaski ES, Bartholomew MJ and Mortel R: Pelvic exenteration: A
morbidity and mortality analysis of a seven-year experience.
Gynecol Oncol. 35:219–223. 1989.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Eisenkop SM and Spirtos NM: Procedures
required to accomplish complete cytoreduction of ovarian cancer: Is
there a correlation with ‘biological aggressiveness’ and survival?
Gynecol Oncol. 82:435–441. 2001.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Marnitz S, Köhler C, Müller M, Behrens K,
Hasenbein K and Schneider A: Indications for primary and secondary
exenterations in patients with cervical cancer. Gynecol Oncol.
103:1023–1030. 2006.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ferenschild FT, Vermaas M, Verhoef C,
Ansink AC, Kirkels WJ, Eggermont AM and de Wilt JH: Total pelvic
exenteration for primary and recurrent malignancies. World J Surg.
33:1502–1508. 2009.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hawighorst-Knapstein S, Fusshoeller C,
Franz C, Trautmann K, Schmidt M, Pilch H, Schoenefuss G, Knapstein
PG, Koelbl H, Kelleher DK and Vaupel P: The impact of treatment for
genital cancer on quality of life and body image-results of a
prospective longitudinal 10-year study. Gynecol Oncol. 94:398–403.
2004.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Höckel M and Dornhöfer N: Pelvic
exenteration for gynaecological tumours: Achievements and
unanswered questions. Lancet Oncol. 7:837–847. 2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Căpîlna ME, Rusu SC, Laczko C, Szabo B and
Marian C: Three synchronous primary pelvic cancers-a case report.
Eur J Gynaecol Oncol. 36:216–218. 2015.PubMed/NCBI
|
|
17
|
PelvExCollaborative. Pelvic exenteration
for advanced nonrectal pelvic malignancy. Ann Surg. 270:899–905.
2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Chiva LM, Lapuente F, González-Cortijo L,
González-Martín A, Rojo A, García JF and Carballo N: Surgical
treatment of recurrent cervical cancer: State of the art and new
achievements. Gynecol Oncol. 110 (3 Suppl 2):S60–S66.
2008.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Dindo D, Demartines N and Clavien PA:
Classification of surgical complications: A new proposal with
evaluation in a cohort of 6336 patients and results of a survey.
Ann Surg. 240:205–213. 2004.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Bricker EM: Bladder substitution after
pelvic evisceration. Surg Clin North Am. 30:1511–1521.
1950.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Căpîlna ME, Moldovan B and Szabo B: Pelvic
exenteration-our initial experience in 15 cases. Eur J Gynaecol
Oncol. 36:142–145. 2015.PubMed/NCBI
|
|
22
|
Bacalbasa N, Balescu I, Vilcu M, Neacsu A,
Dima S, Croitoru A and Brezean I: Pelvic exenteration for locally
advanced and relapsed pelvic malignancies-an analysis of 100 cases.
In Vivo. 33:2205–2210. 2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Lewandowska A, Szubert S, Koper K, Koper
A, Cwynar G and Wicherek L: Analysis of long-term outcomes in 44
patients following pelvic exenteration due to cervical cancer.
World J Surg Onc. 18(234)2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Goldberg JM, Piver MS, Hempling RE, Aiduk
C, Blumenson L and Recio FO: Improvements in pelvic exenteration:
Factors responsible for reducing morbidity and mortality. Ann Surg
Oncol. 5:399–406. 1998.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Kramer C, Peschel RE, Goldberg N, Kohorn
EI, Chambers JT, Chambers SK and Schwartzet PE: Radiation treatment
of FIGO stage IVA carcinoma of the cervix. Gynecol Oncol.
32:323–326. 1989.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Moore KN, Gold MA, McMeekin DS and Zorn
KK: Vesicovaginal fistula formation in patients with stage IVA
cervical carcinoma. Gynecol Oncol. 106:498–501. 2007.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Forner DM and Lampe B: Exenteration as a
primary treatment for locally advanced cervical cancer: Long-term
results and prognostic factors. Am J Obstet Gynecol. 205:148.e1–e6.
2011.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Ahmadi N, Tan KK, Solomon MJ, Al-Mozany N
and Carter J: Pelvic exenteration for primary and recurrent
gynecologic malignancies is safe and achieves acceptable long-term
outcomes. J Gynecol Surg. 30:255–259. 2014.
|
|
29
|
Höckel M, Horn LC and Einenkel J:
(Laterally) extended endopelvic resection: Surgical treatment of
locally advanced and recurrent cancer of the uterine cervix and
vagina based on ontogenetic anatomy. Gynecol Oncol. 127:297–302.
2012.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Li L, Ma SQ, Tan XJ, Zhong S and Wu M:
Pelvic exenteration for recurrent and persistent cervical cancer.
Chin Med J (Engl). 131:1541–1548. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Căpîlna ME, Szabo B, Rusu SC, Becsi J,
Moldovan B, Neagoe RM and Muhlfay G: Anatomical variations of the
obturator veins and their surgical implications. Eur J Gynaecol
Oncol. 38:263–265. 2017.PubMed/NCBI
|
|
32
|
Zoucas E, Frederiksen S, Lydrup ML,
Månsson W, Gustafson P and Alberius P: Pelvic exenteration for
advanced and recurrent malignancy. World J Surg. 34:2177–2184.
2010.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Andikyan V, Khoury-Collado F, Gerst SR,
Talukdar S, Bochner BH, Sandhu JS, Abu-Rustum N, Sonoda Y, Barakat
RR and Chi DS: Anterior pelvic exenteration with total vaginectomy
for recurrent or persistent genitourinary malignancies: Review of
surgical technique, complications, and outcome. Gynecol Oncol.
126:346–350. 2012.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Chiva L, Zanagnolo V, Querleu D,
Martin-Calvo N, Arévalo-Serrano J, Căpîlna ME, Fagotti A,
Kucukmetin A, Mom C, Chakalova G, et al: SUCCOR study: An
international European cohort observational study comparing
minimally invasive surgery versus open abdominal radical
hysterectomy in patients with stage IB1 cervical cancer. Int J
Gynecol Cancer. 30:1269–1277. 2020.PubMed/NCBI View Article : Google Scholar
|