Introduction
Preeclampsia (PE) is a predominant
pregnancy-specific vascular disorder of increasing incidence, which
is also a major cause of maternal and infant morbidity and
mortality (1). PE affects 5-7% of
all pregnant women and is responsible for >70,000 maternal and
500,000 fetal mortality worldwide every year (2). PE is characterized by new-onset
hypertension and proteinuria after 20 weeks of pregnancy (3). Individuals with PE and their children
are at a higher risk of developing severe cardiovascular
complications and metabolic disorders (4). Clinical and pathological studies
previously confirmed that placental dysplasia, excessive maternal
inflammation and endothelial dysfunction are central to the
pathogenesis of PE (2,5). Accumulating evidence has also shown
that the etiology of PE involves poor placentation due to
inadequate trophoblast invasion (6,7).
Therefore, it is imperative to perform preclinical experiments in
trophoblasts to elucidate the mechanism underlying PE
pathogenesis.
It has been previously indicated that long
non-coding RNAs (lncRNAs) can serve active roles in numerous
cellular processes, including regulation of gene expression, and
post-transcriptional and epigenetic modifications in the
development of most organs, such as the placenta (8,9).
Genome-wide lncRNA expression profile studies in PE placentas using
microarray have highlighted a number of differentially expressed
lncRNAs in placentas with PE compared with in normal placenta
(10,11). In particular, lncRNAs, including
H19, metastasis associated lung adenocarcinoma transcript 1 and HOX
antisense intergenic RNA, have all been reported to regulate the
pathogenesis of PE (12-14).
An existing review outlined that lncRNAs have an impact on both the
occurrence and development of PE through changes in the biological
functions of trophoblasts, immune regulation, epigenetic regulation
and energy metabolism (15). The
aim of the present study was to identify new lncRNAs to explore the
potential mechanism of PE. Brain cytoplasmic RNA 1 (BCYRN1) is a
brain-specific lncRNA with a confirmed regulatory role in dendritic
translation in neurons (16). It
has also been reported that BCYRN1 is expressed in a variety of
cancer cells, such as colon cancer and esophageal cancer cells,
which can regulate the expression of various proteolytic enzymes
and promote tumor migration and invasion (17,18).
Since BCYRN1 can promote the proliferation and migration of airway
smooth muscle cells (19,20), it may also affect the biological
function of non-cancer cells. Trophoblasts have been recognized to
share histological and behavioral characteristics with cancer cells
(21). It has been widely reported
that insufficient trophoblast infiltration is a key cause of PE
(22). As such, the expression and
activation of proteolytic enzymes is required for the invasion of
ectotrophoblast cells into the decidua and subsequent initiation of
vascular remodeling (23,24). However, whether lncRNA BCYRN1 can
exert regulatory effects on trophoblast cells warrants further
investigation. Therefore, the present study aimed to identify the
expression profile and clinical relevance of the lncRNA BCYRN1 in
placentas with PE, to explore the effect of lncRNA BCYRN1 on the
biological physiology of HTR-8/SVneo trophoblasts and their
possible underlying mechanism.
Materials and methods
Study subjects
A total of 30 patients with mild PE (MPE group) and
30 patients with severe PE (SPE group) who underwent cesarean
section at the Affiliated Hospital of North Sichuan Medical College
(Nanchong, China) between April 2017 and April 2019 were recruited.
In addition, 30 healthy pregnant women who delivered via cesarean
section were selected as the control group (Normal group). All
pregnancies were singleton and delivered via cesarean section.
Those with pregnancy-related or other complications, including
gestational diabetes mellitus, primary hypertension and chronic
nephritis, were excluded. The diagnostic criteria of SPE according
to the 8th edition of Obstetrics and Gynecology published by the
People's Medical Publishing House were used (25). The specific diagnostic criteria for
PE were as follows: i) ≥2 blood pressure rises [systolic blood
pressure (SBP) ≥140 mmHg or diastolic blood pressure (DBP) ≥90
mmHg] after 20 weeks of gestation at an interval time of ≥6 h; and
ii) 24-h urine protein ≥300 mg. Patients with PE who met ≥1 of the
following criteria were diagnosed as SPE: i) SBP ≥60 mmHg or DBP
≥110 mmHg; ii) platelet count <100x109/l; iii) liver
function damage (serum transaminase concentration >2 times of
the upper limit of the normal range (~0-40 U/l)); iv) renal
function impairment, such as the 24-h urine protein ≥2 g, 24-h
urine volume <400 ml or serum creatinine >106 µmol/l; v)
pulmonary edema; vi) persistent headache, visual impairment or
other abnormalities of central nervous system; vii) heart failure;
viii) fetal growth restriction or oligohydramnios and placental
abruption; and ix) intravascular hemolysis, including anemia,
jaundice and increased lactate dehydrogenase levels. Patients who
were not diagnosed with SPE were considered MPE. The present study
was approved by the Academic Ethics Committee of the Affiliated
Hospital of North Sichuan Medical College and informed consent was
obtained from all individuals.
Sample collection
Placental tissues of the enrolled individuals were
obtained 15 min after delivery. Placental biopsies (2x2x2 cm) were
collected at 3, 6, 8 and 12 o'clock on the maternal surface and at
the central position to avoid the placental margin, calcification,
hemorrhage and necrosis. The placental tissues were washed with
PBS, frozen in liquid nitrogen and stored at -80˚C for mRNA and
protein extraction after the blood in the tissue was removed with
sterile filter paper. The diagnosis of preeclampsia and grading in
this study was not based on pathological tissues, but on clinical
symptoms and indicators. Therefore, hematoxylin and eosin staining
was not performed on the placental tissues.
Cell culture
HTR-8/SVneo cells, (American Type Culture
Collection), which are frequently used to study the function of
trophoblasts, were generated by using freshly isolated extravillous
cytotrophoblasts from first trimester placenta and transfected with
a plasmid containing the simian virus 40 large T antigen (26). It contains two populations, one of
epithelial and one of mesenchymal origin (27). HTR-8/SVneo cells were seeded in a
25-cm2 flask and cultured in RPMI-1640 (Thermo Fisher
Scientific Inc.) containing 10% fetal bovine serum (FBS;
PAN-Biotech GmbH), 100 U/ml streptomycin and 100 U/ml penicillin at
37˚C and 5% CO2. After the HTR-8/SVneo cells grew to
~75% confluency, the cells were detached with 1 ml 0.25% trypsin
(Sinopharm Chemical Reagent Co., Ltd.) and passaged at a ratio of
1:2. The cells in the logarithmic growth phase were selected for
subsequent experiments.
Cell transfection
Sequences for small interfering (si)RNAs against
BCYRN1 (forward, 5'-UUGCUUUGAGGGAAGUUAC-3' and reverse,
5'-GUAACUUCCCUCAAAGCAATT-3'), negative control (NC) siRNA (forward,
5'-UUCUCCGAACGUGUCACGU-3' and reverse, 5'-ACGUGACACGUUCGGAGAA-3'),
the recombinant plasmid pcDNA3.1-BCYRN1 and pcDNA3.1 empty vector
were purchased from Shanghai GenePharma Co., Ltd. HTR-8/SVneo cells
were placed in 96-well plates (5x103 cells/well) and
transfected using Lipofectamine® 3000 (Thermo Fisher
Scientific, Inc.) when the cells reached 70-90% confluence. Cells
were assigned into the si-BCYRN1 (2 µl), si-NC, pcDNA3.1-BCYRN1
(1,000 ng) and pcDNA3.1 groups. At 12 h post-siRNA-transfection or
8 h post-vector-transfection, the medium was refreshed and cells
were cultured further at 37˚C with 5% CO2. Untransfected
cells were used as the control group. Total RNA was extracted at 48
h post-transfection and BCYRN1 expression was verified via reverse
transcription quantitative polymerase chain reaction (RT-qPCR). In
addition, HTR-8/SVneo cells transfected with pcDNA3.1-BCYRN1 for 24
h were incubated in the medium containing the Wnt signaling
inhibitor XAV939 (2 µmol/l; cat. no. HY-15147; MedChemExpress) at
37˚C for 24 h, whilst the medium supplemented with dimethyl
sulfoxide (DMSO) was used as a control.
Cell Counting Kit-8 (CCK-8) assay
HTR-8/SVneo cells were plated into 96-well plates at
5,000 cells/well and placed in 5% CO2 at 37˚C. In total,
20 µl CCK-8 (Nanjing Jiancheng Bioengineering Institute) detection
solution was added to each well at 37˚C for 1 h. Absorbance values
at 450 nm were detected in each well at 0, 24, 48, 72 and 96 h,
respectively, using an automatic microplate reader (Bio-Rad 680;
Bio-Rad Laboratories, Inc.).
Flow cytometry
The original culture medium of HTR-8/SVneo cells was
first discarded before 1.5 ml 0.25% trypsin without EDTA was added
to each plate. After washing with PBS, 1x105 cells were
collected and cultured in the dark with 5 µl Annexin V-APC and 5 µl
PI staining solution (Nanjing Keygen Biotech Co., Ltd.) for 15 min
at room temperature, followed by detection using a flow cytometer
(MoFloAstrios EQ; Beckman Coulter, Inc.). Annexin V-APC (+) PI (-)
cells were defined as early apoptotic cells whereas Annexin Ⅴ-APC
(+) PI (+) cells were defined as late apoptotic cells. The flow
cytometry data were analyzed using FlowJo software 8.7.1 (FlowJo
LLC). The apoptotic rate was calculated as (early apoptotic cells +
late apoptotic cells)/total cells x100%.
Transwell assays
HTR-8/SVneo cells were resuspended in serum-free
RPMI-1640 medium and seeded into the upper chamber of the Transwell
chamber (8-µm pore size; Corning, Inc.) precoated with Matrigel (BD
Biosciences; Matrigel was diluted at 1:8 and added in the Transwell
chamber until fully solidified at 37˚C for 12 h) in a single layer
with 5x104 cells/well. RPMI-1640 medium with 10% FBS was
placed into the lower chamber. Cells were cultured for 48 h at 5%
CO2 at 37˚C. The cells were then fixed for 15 min with
95% ethanol at room temperature and stained for 20 min with 0.1%
crystal violet at room temperature. Three visual fields were
arbitrarily selected for each well under a light microscope
(magnification, x100) to count the number of invasive cells for
statistical analysis. Transwell chambers without Matrigel were used
to measure cell migration in the same manner.
Matrigel-based tube formation
assay
The serum-free RPMI-1640 culture medium was mixed
with Matrigel at 1:1 in an ice box overnight. This mixture was then
placed into 24-well plates at 150 µl/well at 37˚C for 30 min for
solidification. HTR-8/SVneo cells were detached using trypsin,
resuspended and plated into Matrigel-precoated 24-well plates at
1x104 cells/well with 500 µl RPMI-1640 medium and 5%
FBS. After 4 h incubation at 37˚C, tube formation was observed
using a contrast microscope (magnification, x100). Three visual
fields were arbitrarily selected per well to count the total number
of bifurcation points forming >3 lumens.
RT-qPCR
Total RNA was extracted from placental tissues or
HTR-8/SVneo cells using TRIzol® reagent (Thermo Fisher
Scientific, Inc.). The concentration and purity of the extracted
RNA were determined using a UV spectrophotometer (Nano Photometer;
IMPLEN GmbH). In total, 10 µl total RNA was used for RT with 5X
All-In One RT MasterMix (Applied Biological Materials, Inc.).
Reverse transcription reaction conditions were as follows: 25˚C for
10 min, 42˚C for 15 min and 85˚C for 5 min. The cDNA was produced
after the reaction and stored at -20˚C. Using the consequent cDNA
as a template, EvaGreen 2X qPCR MasterMix-Low Rox (cat. no.
E824483-4 Shanghai Macklin Biochemical Co., Ltd.) was used for
qPCR. The total reaction volume was 20 µl. PCR amplification
conditions were as follows: Denaturation at 95˚C for 10 min,
followed by 40 cycles of denaturation at 95˚C for 15 sec,
denaturation at 95˚C for 1 min, annealing/extension at 60˚C for 30
sec and denaturation at 60˚C for 15 sec. The relative expression of
BCYRN1 was normalized to that of GAPDH and calculated using the
2-ΔΔCq method (28). The
primer sequences were synthesized by Sangon Biotech Co., Ltd.
(Table I). Each sample was
performed three times independently.
 | Table IPrimer sequences used for reverse
transcription-quantitative PCR. |
Table I
Primer sequences used for reverse
transcription-quantitative PCR.
| Gene | Forward, 5'-3' | Reverse, 5'-3' |
|---|
| lncRNA BCYRN1 |
TCAGAGCGACAATTTGAGATC |
GCAGTAGCAGCAGCATTTC |
| GAPDH |
GATTGTTGCCATCAACGACC |
GTGCAGGATGCATTGCTGAC |
Western blot analysis
Total protein was harvested from the placental
tissues or HTR-8/SVneo cells using RIPA buffer (Beyotime Institute
of Biotechnology), and the protein concentration was measured using
a bicinchoninic acid protein quantitative kit (Sangon Biotech Co.,
Ltd.). A total of 50 µg protein/lane was separated using 7.5%
SDS-PAGE. The protein was then transferred onto nitrocellulose
membranes (EMD Millipore). After blocking using 5% skim milk at
room temperature for 2 h, the membranes were probed with primary
rabbit anti-human polyclonal antibodies against Wnt1 (cat. no.
ab63934; 1:1,000; Abcam), β-catenin (cat. no. ab16051; 1:1,000;
Abcam) and GAPDH (cat. no. ab9485; 1:1,000; Abcam) at 4˚C
overnight, followed by incubation for 2 h at room temperature with
horseradish peroxidase-labeled goat anti-rabbit IgG secondary
antibodies (cat. no. HS-GR-HRP-500; 1:10,000; Shijiazhuang No.4
Pharmaceutical Hanlin Biotechnology Co., Ltd.). Finally, enhanced
chemiluminescence reagent (Immobilon Western Chemilum HRP
Substrate; cat. no. WBKLS0100; EMD Millipore) was added for
visualizing the membranes, before the density of bands was
quantified using a Gel-Pro Analyzer 4.0 software (Media
Cybernetics, Inc.). Each sample was tested three times
independently.
Statistical analysis
All data were processed using SPSS 21.0 (IBM Corp.)
and GraphPad Prism 6.0 (GraphPad Software, Inc.). All experiments
were repeated three times. The measurement data were presented as
the mean ± standard deviation and analyzed using one-way
(comparison of three or more groups with only one independent
variable) or two-way (comparison of three or more groups with two
independent variables) analysis of variance followed by Tukey's
multiple comparisons test. Pearson correlation analysis was used to
analyze the correlation between two continuous variables.
Categorical data were expressed as number of cases and Fisher's
exact test was used to assess the association when the expected
frequency was <5. Kendall's τ-b correlation analysis was used to
observe the correlation between the frequency of neonatal asphyxia
and BCYRN1 expression. P<0.05 was considered to indicate a
statistically significant difference.
Results
Clinical baseline characteristics of
enrolled population
There was no significant difference in age and
gestational weeks in the individuals among the three groups, whilst
the body mass index of the MPE group was significantly higher
compared with that in the Normal group (Table II). SBP, DBP and 24-h urine protein
in the MPE and SPE groups were significantly higher compared with
those in the Normal group (P<0.01; Table II). In addition, SBP, DBP and 24-h
urine protein were significantly higher in the SPE group compared
with in the MPE group (P<0.01; Table II). There was no significant
difference in the incidence of neonatal asphyxia among the three
groups.
 | Table IIComparison of baseline clinical
characteristics. |
Table II
Comparison of baseline clinical
characteristics.
| Parameter | Normal | Mild
preeclampsia | Severe
preeclampsia |
|---|
| Number of cases,
n | 30 | 30 | 30 |
| Age, years | 32.62±4.47 | 31.76±5.13 | 34.08±4.19 |
| Gestation,
weeks | 35.88±3.24 | 35.74±3.23 | 35.54±2.59 |
| Body mass index at
early pregnancy, kg/m2 | 24.93±1.75 |
26.48±2.03a | 25.35±2.26 |
| Systolic blood
pressure, mmHg | 113.50±8.71 |
152.72±9.89b |
164.63±16.30b,c |
| Diastolic blood
pressure, mmHg | 70.97±7.03 |
93.78±7.82b |
105.86±11.31b,c |
| 24 h urine protein,
g | 0.207±0.087 |
1.487±0.476b |
3.767±0.725b,c |
| Fetal birth weight,
kg | 3.318±0.352 |
2.972±0.227b |
2.426±0.325b,c |
| Neonatal asphyxia,
nd | 0 | 1 | 5 |
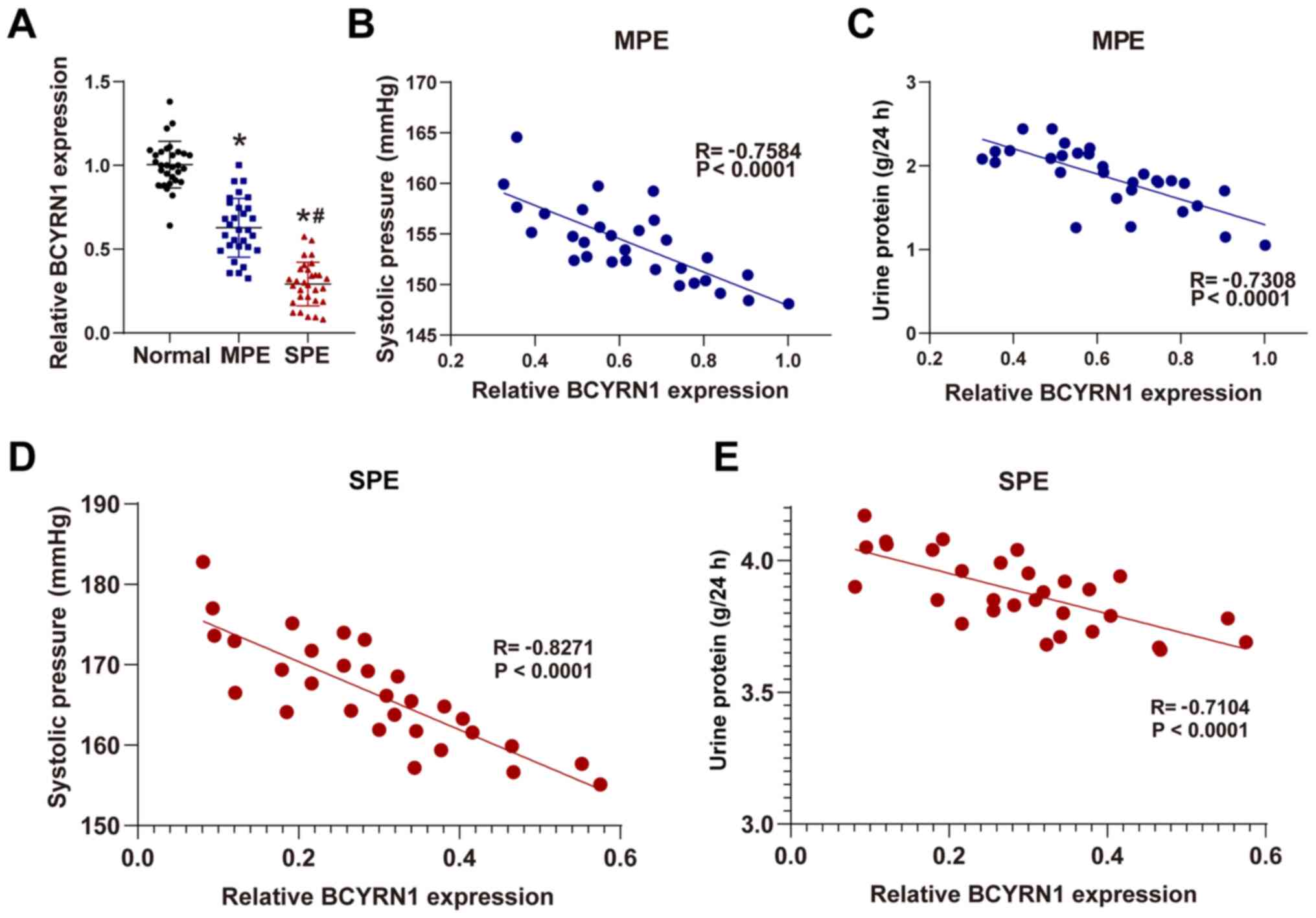
lncRNA BCYRN1 expression is decreased
in placenta samples of patients with PE and correlated with
clinical features of PE
BCYRN1 expression in the placental tissue from the
MPE and SPE groups was significantly reduced compared with in the
Normal group, whilst BCYRN1 expression in the SPE group was
significantly lower compared with in the MPE group (all P<0.05;
Fig. 1A). BCYRN1 expression was
downregulated in the placental tissues of patients with PE, which
decreased further with progressive aggravation of the disease.
The correlation and association between BCYRN1
expression and the clinical characteristics of pregnant women and
newborns was analyzed further. BCYRN1 expression in placental
tissues was negatively correlated with admission SBP and 24-h urine
protein (P<0.05; Fig. 1B-E).
However, relative BCYRN1 expression was not correlated with
neonatal birth weight or incidences of neonatal asphyxia
(P>0.05) (data not shown).
Overexpression of lncRNA BCYRN1
promotes trophoblast cell proliferation and inhibits apoptosis
Trophoblast cells are an important component of the
placental tissue, and participate in placental development and
angiogenesis, where the aberrant dysfunction of trophoblast cells
is closely associated with PE pathogenesis (2). HTR-8/SVneo cells were transfected with
either si-BCYRN1 or pcDNA3.1-BCYRN1, following which cell viability
and apoptosis of HTR-8/SVneo cells were analyzed and compared.
Compared with the si-NC group, BCYRN1 expression was significantly
decreased in the si-BCYRN1 group, whereas BCYRN1 expression in the
pcDNA3.1-BCYRN1 group was significantly elevated compared with that
in the pcDNA3.1-NC group (all P<0.05; Fig. 2A), suggesting that the transfection
was effective. Compared with the si-NC group, the viability of
trophoblast cells was reduced in the si-BCYRN1 group, but was
increased at 72 h in the pcDNA3.1-BCYRN1 group compared with the
pcDNA3.1 group (all P<0.05; Fig.
2B). Similarly, the apoptosis rate of the si-BCYRN1 group was
also significantly elevated compared with the si-NC group, whilst
that of the pcDNA3.1-BCYRN1 group was significantly decreased
compared with the pcDNA3.1 group (P<0.05; Fig. 2C).
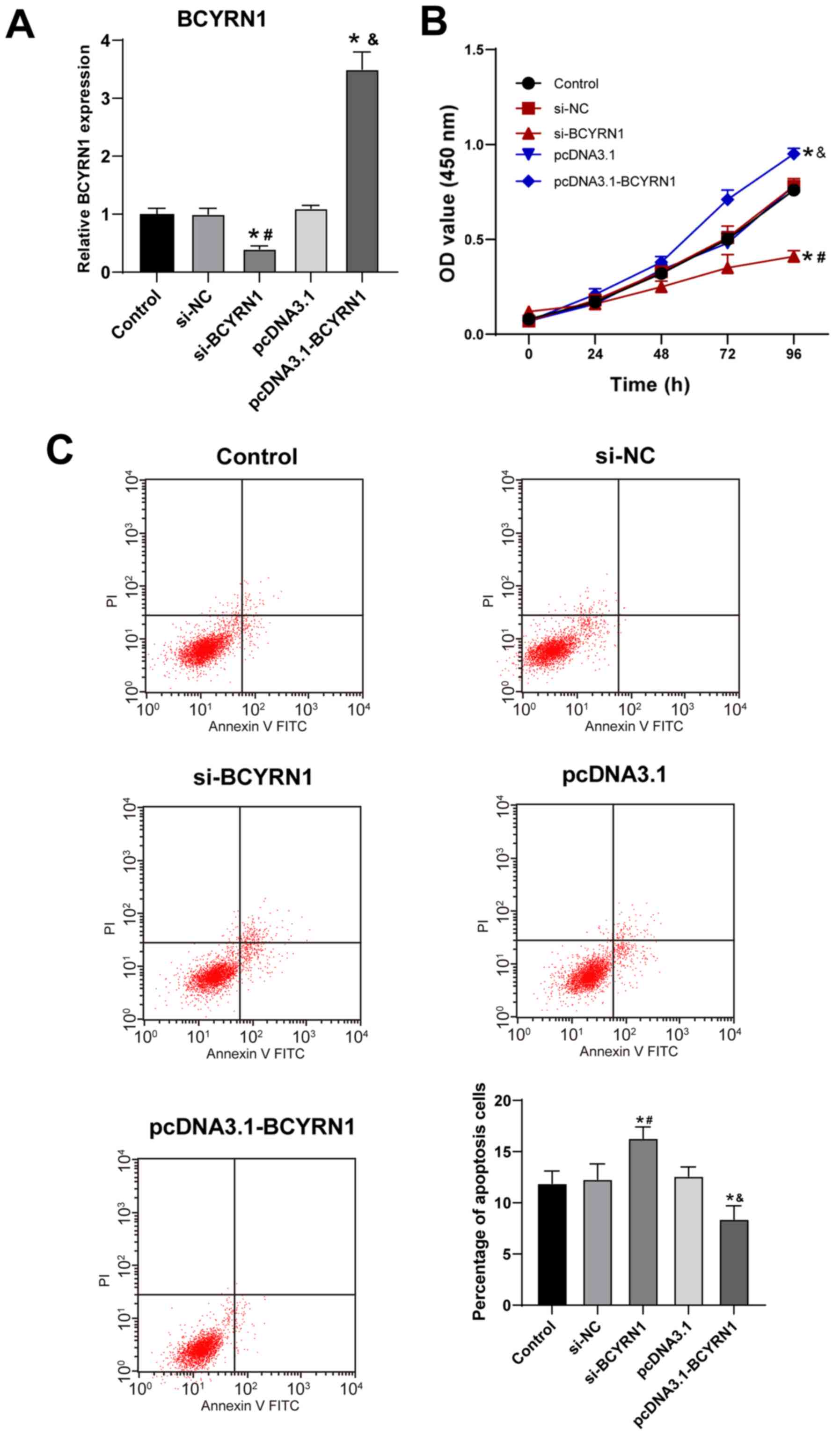 | Figure 2Overexpression of long non-coding RNA
BCYRN1 promotes trophoblast cell viability and inhibits apoptosis.
(A) After 48 h transfection, BCYRN1 expression was detected by
reverse transcription-quantitative PCR. (B) Cell viability was
detected by Cell Counting Kit-8 assay. (C) Apoptosis rate was
detected by flow cytometry. Cell experiments were performed three
times. The data are expressed as the mean ± standard deviation.
Data in panels A and C were analyzed by one-way ANOVA, and data in
panel B were analyzed by two-way ANOVA, followed by Tukey's
multiple comparisons test. *P<0.05 vs. control group;
#P<0.05 vs. si-NC group; &P<0.05
vs. pcDNA3.1 group. BCYRN1, brain cytoplasmic RNA 1; PE,
preeclampsia; NC, negative control, si, small interfering; OD,
optical density; PI, propidium iodide. |
Overexpression of lncRNA BCYRN1
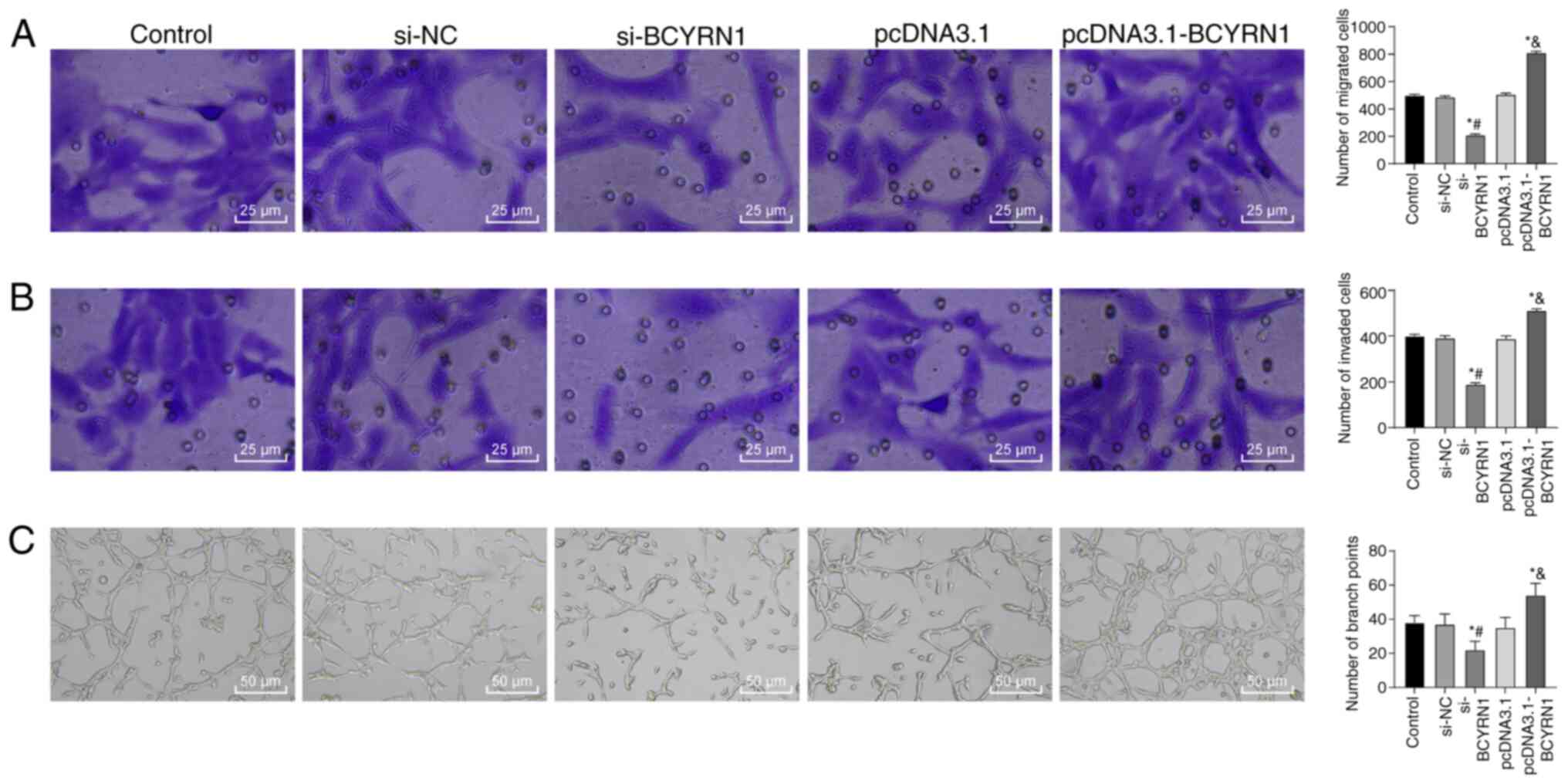
promotes trophoblast cell migration and invasion
The migration, invasion and tube-forming abilities
of trophoblast cells are crucial to the establishment of
maternal-fetal circulation (29).
Reduced invasive ability of trophoblast cells and subsequent
abnormal remodeling of the spiral artery are important features of
PE (30). The number of migratory
HTR-8/SVneo cells in the si-BCYRN1 group was significantly
decreased compared with that in the si-NC group, whilst the number
of migratory cells in the pcDNA3.1-BCYRN1 group was significantly
increased compared with the pcDNA3.1 group (all P<0.05; Fig. 3A). Compared with the si-NC group,
the number of invasive cells was significantly decreased in the
si-BCYRN1group, but significantly increased in the pcDNA3.1-BCYRN1
group compared with the pcDNA3.1 group (all P<0.05; Fig. 3B). Compared with the si-NC group,
the number of bifurcation points in the si-BCYRN1 group was
significantly decreased, whilst that of the pcDNA3.1-BCYRN1 group
was significantly enhanced compared with the pcDNA3.1 group (all
P<0.05; Fig. 3C).
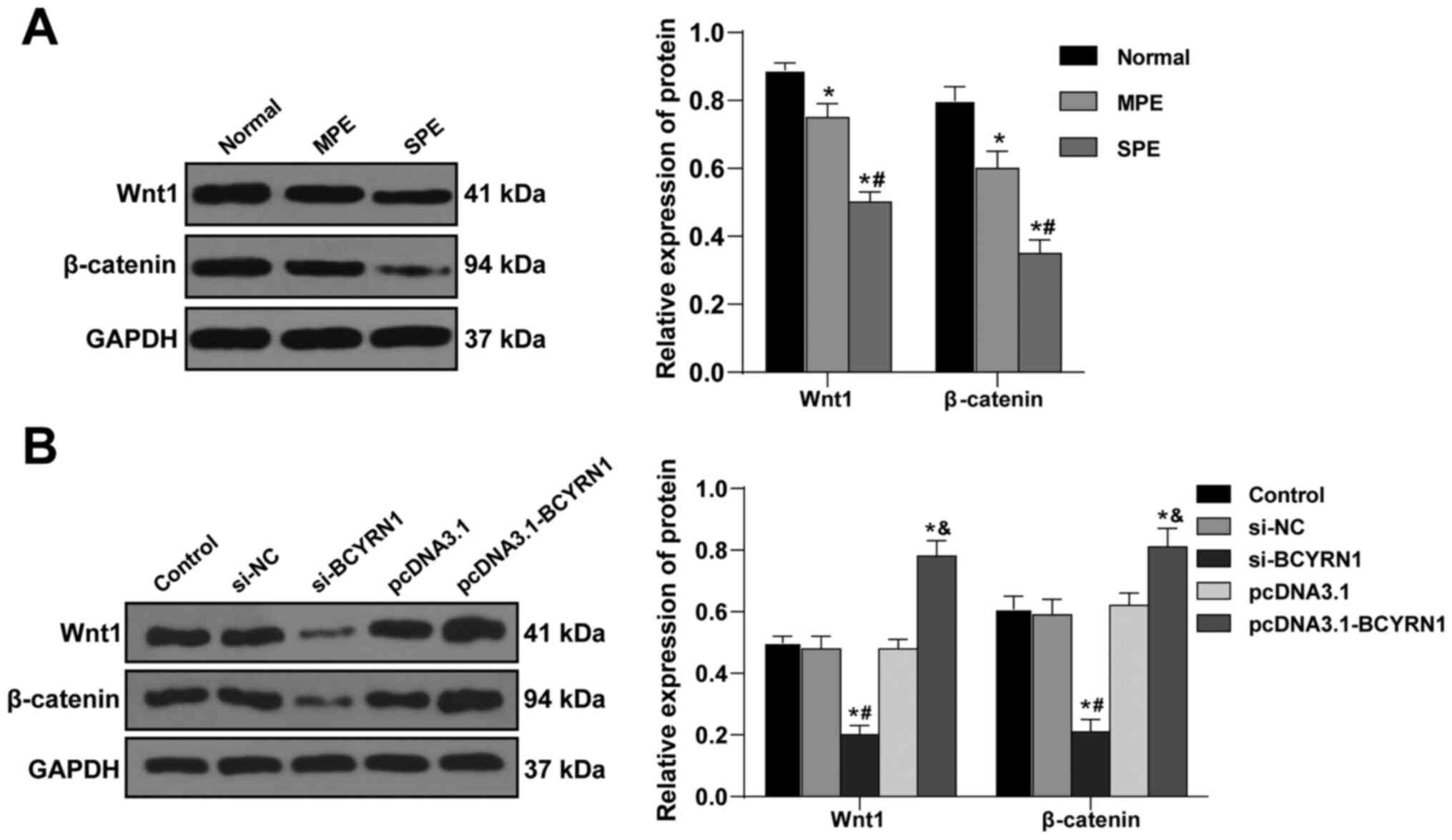
lncRNA BCYRN1 activates the
Wnt/β-catenin axis in trophoblast cells
The Wnt/β-catenin pathway can regulate the
proliferation and apoptosis of trophoblast cells, and is pivotal
for trophoblast cell invasion and implantation into the endometrium
(31). It was hypothesized that
lncRNA BCYRN1 may regulate the Wnt/β-catenin signaling axis in
trophoblast cells. The levels of Wnt1 and β-catenin in the
placental tissues from patients with SPE were significantly lower
compared with those in the Normal group and patients with MPE (all
P<0.05; Fig. 4A). Furthermore,
the levels of Wnt1 and β-catenin were significantly reduced in the
si-BCYRN1 group compared with those in the si-NC group, but were
elevated in the pcDNA3.1-BCYRN1 group compared with in the pcDNA3.1
group (all P<0.05; Fig. 4B).
This suggests that lncRNA BCYRN1 can promote the activation of the
Wnt/β-catenin axis in trophoblast cells.
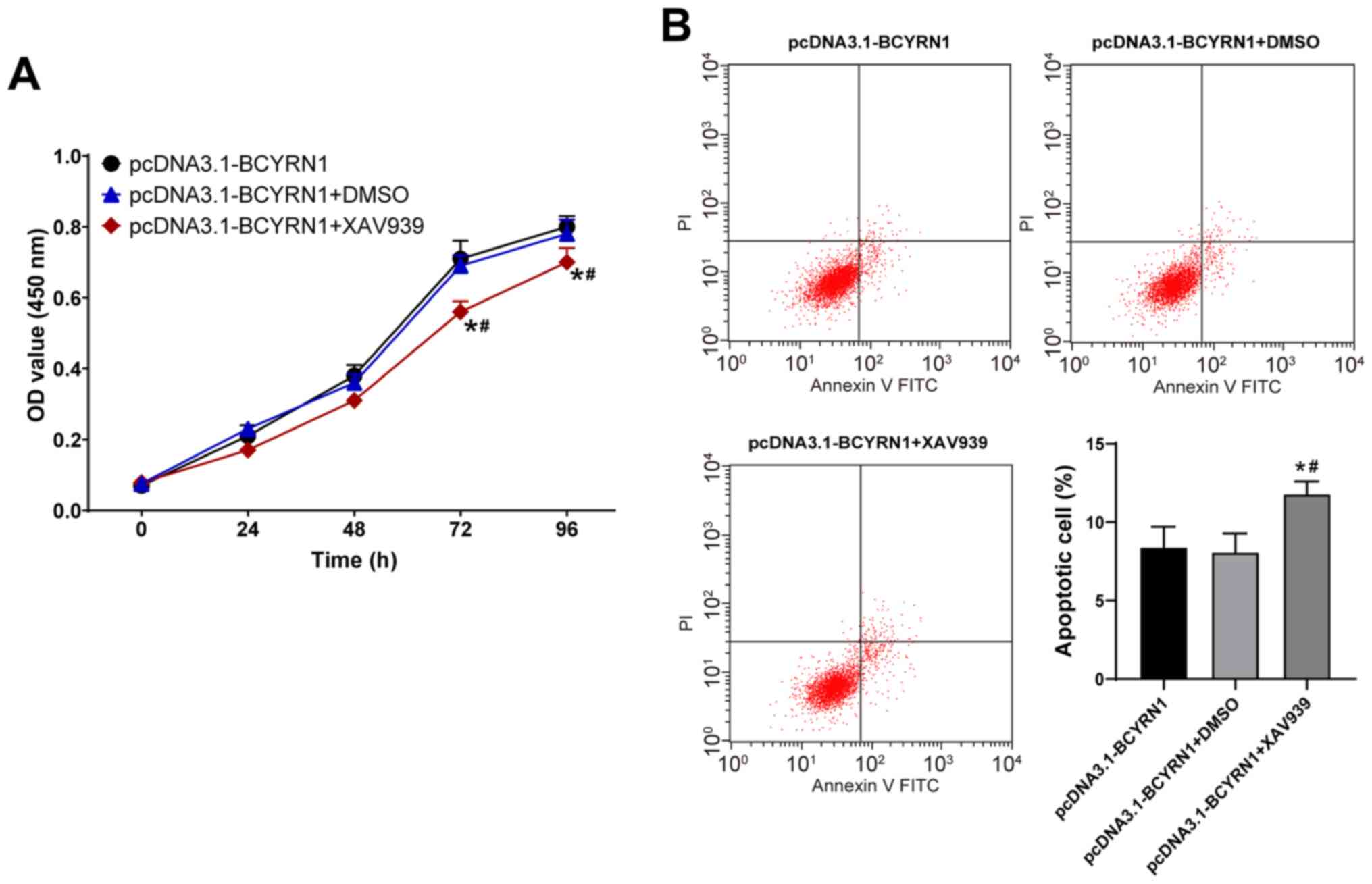
lncRNA BCYRN1 promotes trophoblast
proliferation and blocks apoptosis by activating the Wnt/β-catenin
axis
To investigate whether lncRNA BCYRN1 affects the
viability and apoptosis of trophoblast cells by activating the
Wnt/β-catenin pathway, the Wnt pathway inhibitor XAV939 or DMSO was
applied to treat HTR8/SVneo cells overexpressing BCYRN1. Compared
with the pcDNA3.1-BCYRN1 group, the viability of the
pcDNA3.1-BCYRN1 + XAV939 group was significantly reduced at 72 and
96 h (P<0.05; Fig. 5A), whilst
the apoptosis rate was enhanced (P<0.05; Fig. 5B). These results suggested that
XAV939 can attenuate the effect of BCYRN1 overexpression on
HTR8/SVneo cell viability and apoptosis.
Discussion
Early onset of PE has been reported to be the result
of poor placental implantation, leading to an inflammatory cascade
resulting in high blood pressure (32). To the best of our knowledge, the
present study was the first to determine the expression profile of
lncRNA BCYRN1 in placental tissues with PE and to explore the
potential regulatory effects of BCYRN1 on the physiology of
trophoblast cells. The expression level of BCYRN1 was found to be
downregulated in placenta tissues with PE; a similar reduction in
trophoblast cells also suppressed the migration and invasion of
trophoblasts. BCYRN1 may therefore be involved in the development
of placental abnormalities during the early stages of PE. These
results suggested that the pathogenesis of PE may be associated
with epigenetic changes.
The present study demonstrated that BCYRN1
expression was reduced in the placental tissue from patients with
PE. Additionally, BCYRN1 expression in placental tissues from
patients with MPE was lower compared with that of SPE. This
suggested that BCYRN1 expression is downregulated in the placental
tissues of patients with PE, and is decreased further as the
disease worsens, suggesting that BCYRN1 may be associated with the
severity of PE, which may have clinical relevance. Importantly,
BCYRN1 expression in placental tissues was negatively correlated
with admission SBP and 24-h urine protein levels. The incidence of
pregnancy adverse effects increases in patients with elevated 24-h
proteinuria (33). These results
provided epidemiological evidence that reduced expression of lncRNA
BCYRN1 is correlated with increased adverse pregnancy effects.
However, the sample size in the present study was small, and
multi-center and large-scale experiments are required to verify
this finding. Additionally, the biological sample used in the
present study was placental tissues after delivery. In future
studies, the expression of BCYRN1 in maternal serum should also be
measured.
At present, the clinical consensus is that PE is
caused by poor placental formation during early pregnancy (2). During placental implantation under PE,
trophoblast cells lack invasiveness, which leads to the incomplete
remodeling of the spiral artery, thereby causing placental ischemia
and hypoxia, oxidative stress and systemic inflammatory responses
(2). The present study focused on
the biological behavior of trophoblast cells, namely trophoblast
migration and invasion, which is an early pathological process in
placental dysplasia (2).
HTR-8/SVneo cells have been used extensively to study the
biological activity of trophoblasts in vitro (26). Therefore, HTR-8/SVneo cells were
used in the present study to estimate the in vitro role of
BCYRN1 in PE. After transfection of HTR-8/SVneo cells, the
viability, migratory, invasive and tube-forming capabilities of
trophoblast cells in the si-BCYRN1 group were impaired, whilst the
opposite effects were observed in the pcDNA3.1-BCYRN1 group. The
migration, invasion and tube formation by trophoblasts into the
endometrium and vascular system are key steps during placenta
formation, in addition to contributing to the pathogenesis of PE
(24,29,34,35).
The present study revealed that the apoptosis rate in the si-BCYRN1
group was higher, whilst that in the pcDNA3.1-BCYRN1 group was
decreased. Decreased trophoblast proliferation and invasion, and
increased apoptosis constitute the main underlying causes of PE
(30,36). These previous findings supported the
data from the present study that BCYRN1 overexpression increased
trophoblast cell proliferation, migration, invasion and
tube-forming abilities whilst blocking apoptosis to alleviate PE
progression. However, the effect of BCYRN1 on the expression of
enzymes in trophoblast cells, the effect of BCYRN1 on ischemia,
hypoxia and inflammatory factors in placental tissues and potential
targets for the treatment of PE all require further study.
Early in pregnancy, enhanced Wnt/β-catenin signaling
is a prerequisite for the proper implantation and invasion of
trophoblast cells (37). The
Wnt/β-catenin axis regulates the apoptosis and invasion of
trophoblast cells and is essential for placentation (32). It was therefore speculated in the
present study that lncRNA BCYRN1 may regulate the Wnt/β-catenin
pathway in trophoblast cells. The levels of Wnt1 and β-catenin
proteins in the si-BCYRN1 and the pcDNA3.1-BCYRN1 groups were
decreased and elevated, respectively. To verify if lncRNA BCYRN1
exerts the biological effects on trophoblast cells by evoking the
Wnt/β-catenin axis, the Wnt pathway inhibitor XAV939 was used to
treat HTR8/SVneo cells overexpressing BCYRN1. Compared with that in
the pcDNA3.1-BCYRN1 group, cell viability in the pcDNA3.1-BCYRN1 +
XAV939 group was inhibited, whilst the apoptosis rate was enhanced.
Consistently, downregulated activity of the Wnt pathway may result
in trophoblast dysfunction, which can contribute to the
pathogenesis of PE (38). Results
from the present study suggested that lncRNA BCYRN1 can promote the
activation of the Wnt/β-catenin axis in trophoblast cells.
In conclusion, the present study indicated that
BCYRN1 increased trophoblast viability and prevented apoptosis by
activating the Wnt/β-catenin pathway to potentially delay or block
the onset of PE. The expression pattern of lncRNA BCYRN1 in
placental tissues from patients with PE was detected to explore the
regulatory mechanism of lncRNA BCYRN1 on the biological behavior of
trophoblasts. In the present study, the classical Wnt pathway was
found to regulate cell behavior downstream of BCYRN1, though the
non-β-catenin-dependent Wnt pathway has not been studied. In
addition, whether BCYRN1 participates in PE processes in other
ways, such as by absorbing microRNA sponging, requires further
study. Further studies will be performed to determine the role of
BCYRN1 in the biological function of trophoblast cells from the
perspective of epigenetics and transcriptomics.
Acknowledgements
Not applicable.
Funding
Funding: This work was funded by the Research Center for the
Development of Primary Health Care in Sichuan Province in 2019,
North Sichuan Medical College (grant no. SWFZ19-Y-35), the ‘Sichuan
Grassroots Health and Health Development Research Team’, a
high-level Social Science Research Team of Sichuan Province
[Sichuan Social Union Issued (grant no. 2017 No. 43)], the 2019
Municipal School-School Cooperative Scientific Research Project in
Nanchong (North Sichuan Medical College; grant nos. 19SXHZ0291 and
19SXHZ0435) and Nanchong Municipal Applied Technology Research and
Development Fund Project in 2020 (grant no. 20YFZJ0099).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LC designed the study and performed the experiments.
LC, QS and BF collected the data. LC, QS and YC analyzed the data.
LC, BF prepared the manuscript. LC and YC confirm the authenticity
of all the raw data. All authors read and approved the final
manuscript.
Ethics approval and consent to
participate
The present study was approved by the Academic
Ethics Committee of the Affiliated Hospital of North Sichuan
Medical College (Nanchong, China) and informed consent was obtained
from all individuals.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Dymara-Konopka W, Laskowska M and
Oleszczuk J: Preeclampsia-current management and future approach.
Curr Pharm Biotechnol. 19:786–796. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Rana S, Lemoine E, Granger JP and
Karumanchi SA: Preeclampsia: Pathophysiology, challenges, and
perspectives. Circ Res. 124:1094–1112. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Malik A, Jee B and Gupta SK: Preeclampsia:
Disease biology and burden, its management strategies with
reference to India. Pregnancy Hypertens. 15:23–31. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Suvakov S, Bonner E, Nikolic V, Jerotic D,
Simic TP, Garovic VD, Lopez-Campos G and Mcclements L: Overlapping
pathogenic signalling pathways and biomarkers in preeclampsia and
cardiovascular disease. Pregnancy Hypertens. 20:131–136.
2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Cheng SB and Sharma S: Preeclampsia and
health risks later in life: An immunological link. Semin
Immunopathol. 38:699–708. 2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Chen Q, Jiang S, Liu H, Gao Y, Yang X, Ren
Z, Gao Y, Xiao L, Hu H, Yu Y, et al: Association of lncRNA
SH3PXD2A-AS1 with preeclampsia and its function in invasion and
migration of placental trophoblast cells. Cell Death Dis.
11(583)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Li Q, Wang T, Huang S, Zuo Q, Jiang Z,
Yang N and Sun L: lncRNA MALAT1 affects the migration and invasion
of trophoblast cells by regulating FOS expression in early-onset
preeclampsia. Pregnancy Hypertens. 21:50–57. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Apicella C, Ruano CSM, Mehats C, Miralles
F and Vaiman D: The role of epigenetics in placental development
and the etiology of preeclampsia. Int J Mol Sci.
20(2837)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Mcaninch D, Roberts CT and Bianco-Miotto
T: Mechanistic insight into long noncoding RNAs and the placenta.
Int J Mol Sci. 18(1371)2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
He X, He Y, Xi B, Zheng J, Zeng X, Cai Q,
Ouyang Y, Wang C, Zhou X, Huang H, et al: lncRNAs expression in
preeclampsia placenta reveals the potential role of lncRNAs
contributing to preeclampsia pathogenesis. PLoS One.
8(e81437)2013.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Long W, Rui C, Song X, Dai X, Xue X, Lu Y,
Shen R, Li J, Li J and Ding H: Distinct expression profiles of
lncRNAs between early-onset preeclampsia and preterm controls. Clin
Chim Acta. 463:193–199. 2016.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Harati-Sadegh M, Kohan L, Teimoori B,
Mehrabani M and Salimi S: The effects of placental long noncoding
RNA H19 polymorphisms and promoter methylation on H19 expression in
association with preeclampsia susceptibility. IUBMB Life.
72:413–425. 2020.PubMed/NCBI View
Article : Google Scholar
|
|
13
|
Mohammadpour-Gharehbagh A, Jahantigh D,
Saravani M, Harati-Sadegh M, Maruie-Milan R, Teimoori B and Salimi
S: Impact of HOTAIR variants on preeclampsia susceptibility based
on blood and placenta and in silico analysis. IUBMB Life.
71:1367–1381. 2019.PubMed/NCBI View
Article : Google Scholar
|
|
14
|
Wu HY, Wang XH, Liu K and Zhang JL: lncRNA
MALAT1 regulates trophoblast cells migration and invasion via
miR-206/IGF-1 axis. Cell Cycle. 19:39–52. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yang X and Meng T: Long noncoding RNA in
Preeclampsia: Transcriptional noise or innovative indicators?
Biomed Res Int. 2019(5437621)2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sosinska-Zawierucha P, Zawierucha P,
Breborowicz A and Barciszewski J: Prediction of secondary and
tertiary structures of human BC200 RNA (BCYRN1) based on
experimental and bioinformatic cross-validation. Biochem J.
475:2727–2748. 2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Huang W, Zhou R, Mao L, Deng C and Dang X:
Esophageal cancer related gene-4 inhibits the migration and
proliferation of oral squamous cell carcinoma through BC200
lncRNA/MMP-9 and -13 signaling pathway. Cell Signal.
62(109327)2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wu K, Xu K, Liu K, Huang J, Chen J, Zhang
J and Zhang N: Long noncoding RNA BC200 regulates cell growth and
invasion in colon cancer. Int J Biochem Cell Biol. 99:219–225.
2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhang XY, Tang XY, Ma LJ, Guo YL, Li XS,
Zhao LM, Tian CJ, Cheng DJ, Chen ZC and Zhang LX: Schisandrin B
down-regulated lncRNA BCYRN1 expression of airway smooth muscle
cells by improving miR-150 expression to inhibit the proliferation
and migration of ASMC in asthmatic rats. Cell Prolif.
50(e12382)2017.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhang XY, Zhang LX, Tian CJ, Tang XY, Zhao
LM, Guo YL, Cheng DJ, Chen XL, Ma LJ and Chen ZC: lncRNAs BCYRN1
promoted the proliferation and migration of rat airway smooth
muscle cells in asthma via upregulating the expression of transient
receptor potential 1. Am J Transl Res. 8:3409–3418. 2016.PubMed/NCBI
|
|
21
|
Xie D, Zhu J, Liu Q, Li J, Song M, Wang K,
Zhou Q, Jia Y and Li T: Dysregulation of HDAC9 represses
trophoblast cell migration and invasion through TIMP3 activation in
Preeclampsia. Am J Hypertens. 32:515–523. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zou AX, Chen B, Li QX and Liang YC:
MiR-134 inhibits infiltration of trophoblast cells in placenta of
patients with preeclampsia by decreasing ITGB1 expression. Eur Rev
Med Pharmacol Sci. 22:2199–2206. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Hannon T, Innes BA, Lash GE, Bulmer JN and
Robson SC: Effects of local decidua on trophoblast invasion and
spiral artery remodeling in focal placenta creta-an
immunohistochemical study. Placenta. 33:998–1004. 2012.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Nawrocki B, Polette M, Marchand V, Maquoi
E, Beorchia A, Tournier JM, Foidart JM and Birembaut P:
Membrane-type matrix metalloproteinase-1 expression at the site of
human placentation. Placenta. 17:565–572. 1996.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Perl M, Lomas-Neira J, Venet F, Chung CS
and Ayala A: Pathogenesis of indirect (secondary) acute lung
injury. Expert Rev Respir Med. 5:115–126. 2011.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Graham CH, Hawley TS, Hawley RG,
Macdougall JR, Kerbel RS, Khoo N and Lala PK: Establishment and
characterization of first trimester human trophoblast cells with
extended lifespan. Exp Cell Res. 206:204–211. 1993.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Abou-Kheir W, Barrak J, Hadadeh O and
Daoud G: HTR-8/SVneo cell line contains a mixed population of
cells. Placenta. 50:1–7. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) Method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Ji L, Brkic J, Liu M, Fu G, Peng C and
Wang YL: Placental trophoblast cell differentiation: Physiological
regulation and pathological relevance to preeclampsia. Mol Aspects
Med. 34:981–1023. 2013.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Alasztics B, Kukor Z, Panczel Z and Valent
S: The pathophysiology of preeclampsia in view of the two-stage
model. Orv Hetil. 153:1167–1176. 2012.PubMed/NCBI View Article : Google Scholar : (In Hu).
|
|
31
|
Zhang Z, Wang X, Zhang L, Shi Y, Wang J
and Yan H: Wnt/β-catenin signaling pathway in trophoblasts and
abnormal activation in preeclampsia (Review). Mol Med Rep.
16:1007–1013. 2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Ogge G, Chaiworapongsa T, Romero R,
Hussein Y, Kusanovic JP, Yeo L, Kim CJ and Hassan SS: Placental
lesions associated with maternal underperfusion are more frequent
in early-onset than in late-onset preeclampsia. J Perinat Med.
39:641–652. 2011.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Bouzari Z, Javadiankutenai M, Darzi A and
Barat S: Does proteinura in preeclampsia have enough value to
predict pregnancy outcome? Clin Exp Obstet Gynecol. 41:163–168.
2014.PubMed/NCBI
|
|
34
|
Kokkinos MI, Murthi P, Wafai R, Thompson
EW and Newgreen DF: Cadherins in the human
placenta-epithelial-mesenchymal transition (EMT) and placental
development. Placenta. 31:747–755. 2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang Y, Cao L, Jia J, Ye L, Wang Y, Zhou
B and Zhou R: CircHIPK3 is decreased in preeclampsia and affects
migration, invasion, proliferation, and tube formation of human
trophoblast cells. Placenta. 85:1–8. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Fu G, Ye G, Nadeem L, Ji L, Manchanda T,
Wang Y, Zhao Y, Qiao J, Wang YL, Lye S, et al: MicroRNA-376c
impairs transforming growth factor-β and nodal signaling to promote
trophoblast cell proliferation and invasion. Hypertension.
61:864–872. 2013.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Van Der Horst PH, Wang Y, Van Der Zee M,
Burger CW and Blok LJ: Interaction between sex hormones and
WNT/beta-catenin signal transduction in endometrial physiology and
disease. Mol Cell Endocrinol. 358:176–184. 2012.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Zhang Z, Li H, Zhang L, Jia L and Wang P:
Differential expression of β-catenin and Dickkopf-1 in the third
trimester placentas from normal and preeclamptic pregnancies: A
comparative study. Reprod Biol Endocrinol. 11(17)2013.PubMed/NCBI View Article : Google Scholar
|