Introduction
Cardiovascular disease (CVD) is the leading cause of
death worldwide while atherosclerotic coronary artery disease (CAD)
is mainly involved (1). After
performing percutaneous coronary intervention (PCI), patients are
still at risk of developing new stenosis, such as intra-stent
restenosis (ISR). The treatment of patients with ISR represents an
important clinical problem and is still considered a challenge
(2-4).
Despite the proven safety and efficacy of drug-eluting stents (DES)
in patients undergoing PCI, bare-metal stents (BMS) are still
widely used as well, mainly because of their reduced cost and
concerns about a debatable increased risk of bleeding associated
with prolonged dual antiplatelet therapy after DES (5,6). In
addition, neoatherosclerosis is associated more often with 1st
generation DES than with BMS and occurs several months/years
following PCI, while atherosclerosis in native coronary arteries
develops over decades (7).
The incidence of ISR is still significant when
considering either DES or BMS for patients following PCI (8,9),
mainly because inflammatory responses after PCI lead to abnormal
neointimal healing and thus generate a higher risk of unfavorable
outcomes (7). This suggests that
the type of stent is only one factor to consider when searching for
additional promoters of ISR. In fact, the results of previous
research conclude that the factors associated with ISR after PCI
have not been clearly defined. Thus, the present study aimed to
detect the clinical, biological, imagistic and procedural factors
associated with ISR.
Patients and methods
Patient selection and study
design
The design of our study was observational,
cross-sectional, over a 2-year period, from a single high-volume
PCI center. A total of 235 consecutive patients who were diagnosed
with angina pectoris or acute coronary syndrome (myocardial
infarction with or without ST elevation) treated by PCI, were
included. Our study population was divided into 2 groups:
Experimental group (119 patients) that presented ISR documented by
coronary angiography (>50% stenosis of a previously stented
segment) and the control group (116 patients) without angiographic
ISR, but with different other culprit lesions or no significant
angiographic stenosis.
Patients were eligible for the study if they were
≥18 years and presented with a diagnosis of angina pectoris or
acute myocardial infarction previously treated by stent
implantation. Patients were not eligible for the study if they
refused or abandoned treatment, if they did not report for the
control visit or if they were part of a vulnerable category (e.g.,
pregnant women, patients in coma). The study was approved by the
University of Medicine and Pharmacy ‘Grigore T. Popa’ Iasi Research
Ethics Committee, and all subjects had initially agreed and signed
an informed consent in order to take part in this study.
Clinical and paraclinical
characteristics
Diagnosis of ISR was documented by coronary
angiography in patients with suggestive coronary symptoms and
ischemic changes in non-invasive or invasive paraclinical
investigations. All patients underwent clinical and laboratory
examination, providing a large number of variables that could be
considered risk factors for ISR: i) clinical variables: age, sex,
smoking, hypertension, diabetes, obesity, chronic kidney disease
(CKD; creatinine clearance <60 ml/min), acute renal failure,
thrombolysis in myocardial infarction (TIMI) score; ii)
paraclinical variables: left ventricular ejection fraction (LVEF),
albuminuria, LDL cholesterol, erythrocyte sedimentation rate (ESR),
creatinine clearance, uric acid, C-reactive protein (CRP),
fibrinogen; iii) coronary anatomical variables: left main (LM),
left anterior descending (LAD), left circumflex artery (LCX), right
coronary artery (RCA); and iv) variables depending on the
procedure: type, length and diameter of stent and pressure under
which the stent was implanted.
Statistical analysis
Data analysis was performed using SPSS 20.0
(Statistical Package for the Social Sciences; IBM Corp.).
Comparison between the 2 groups of patients was performed using
Chi-square test for the categorical data and one-way ANOVA and
Student's t-test for continuous data. When a normal distribution
was not present for continuous variables, Mann
Whitney/Kruskal-Wallis tests were used. In order to estimate the
strength of the association between risk factors and outcome (with
or without ISR) relative risk (RR) with 95% confidence interval
(CI) was used in the statistical analysis of the data. A P-value
<0.05 was considered statistically significant.
Results
The mean age of the patients was 61.84±11.12 years
(63.08 years, ISR group; 60.57 years, non-ISR group; P=0,084).
Factors associated with ISR are presented in Tables I and II. Smoking, hypertension, diabetes, high
CRP levels, CKD and TIMI score were found to be significantly
associated with ISR in this group of patients. All these factors
influenced the risk via a directly proportional relationship.
Regarding the endothelial dysfunction markers, the cut-off values
were calculated in order to establish the risk associated with
them: ESR=30 mm/h, uric acid=5 mg/dl, creatinine clearance=5
ml/min, CRP=2 mg/dl, fibrinogen=400 mg%. Analyzing both study
groups, approximately 44% of the patients had an LDL value >100
mg/dl.
 | Table ISummary of the general factors
associated with ISR. |
Table I
Summary of the general factors
associated with ISR.
| Variable | ISR group (n=119) n
(%) | Non-ISR group (n=116)
n (%) | P-value | RR | 95% CI |
|---|
| Sex | | | | | |
|
Male | 83(70) | 81(70) | 0.989 | | |
|
Female | 36(30) | 34(30) | 0.522 | | |
| Age ≥60 years | 77 (64.7) | 64 (55.2) | 0.125 | | |
| Current smokers | 74 (62.2) | 44 (37.9) | 0.001 | 1.63 | 1.25-2.13 |
| Hypertension | 72 (62.1) | 38 (31.9) | 0.001 | 1.86 | 1.41-2.45 |
| Diabetes
mellitus | 68 (57.1) | 31 (26.7) | 0.001 | 1.83 | 1.42-2.36 |
| Obesity | 35 (29.4) | 27 (23.3) | 0.285 | 1.16 | 0.89-1.52 |
| LDL cholesterol
>70 mg/dl | 91 (76.5) | 93 (80.2) | 0.596 | 1.05 | 0.95-1.20 |
| ESR >30 mm/h | 48 (40.3) | 37 (31.9) | 0.178 | 1.19 | 0.93-1.53 |
| Uric acid >5
mg/dl | 30 (71.4) | 23 (60.5) | 0.303 | 1.27 | 0.79-2.07 |
| Creatinine clearance
<75 ml/min/1.73 m2 | 27 (44.3) | 31 (47.7) | 0.699 | 0.93 | 0.65-1.34 |
| CRP >2 mg/dl | 94 (87.0) | 83 (77.6) | 0.050 | 1.44 | 0.93-2.24 |
| Fibrinogen >400
mg/dl | 84 (70.6) | 72 (62.6) | 0.195 | 1.20 | 0.90-1.60 |
| Albuminuria | 16 (13.4) | 19 (16.4) | 0.528 | 0.89 | 0.60-1.31 |
| CKD | 43 (36.1) | 11 (9.5) | 0.001 | 1.90 | 1.53-2.36 |
| Acute renal
failure | 7 (5.9) | 5 (4.3) | 0.583 | 1.16 | 0.71-1.91 |
 | Table IISummary table of the specific factors
associated with ISR. |
Table II
Summary table of the specific factors
associated with ISR.
| Variable | ISR group (n=119)
(%) | Non-ISR group
(n=116) (%) | P-value |
|---|
| Site of lesion | | | |
|
LAD | 47.9 | 50.9 | NS |
|
RCA | 28.6 | 35.3 | NS |
|
LCX | 22.7 | 13.8 | NS |
|
LM | 0.8 | 0.0 | NS |
| Clinical diagnosis
before stent implantation | | | |
|
MI right
ventricle | 0.8 | 3.4 | NS |
|
MI
posterior-inferior-lateral | 5.0 | 3.4 | NS |
|
MI
antero-lateral | 8.4 | 6.9 | NS |
|
MI
anterior | 31.1 | 29.3 | NS |
|
MI
inferior | 21.0 | 16.4 | NS |
|
Angina
pectoris | 33.6 | 40.5 | NS |
| TIMI score | | | |
|
TIMI 1 | 2.5 | 0.0 | 0.001 |
|
TIMI 2 | 19.3 | 3.4 | 0.001 |
|
TIMI 3 | 78.2 | 96.6 | 0.001 |
| Ejection fraction
(EF) | | | |
|
EF
<40% | 52.9 | 54.3 | NS |
|
EF=40-49% | 31.9 | 22.4 | NS |
|
EF
>50% | 15.1 | 23.3 | NS |
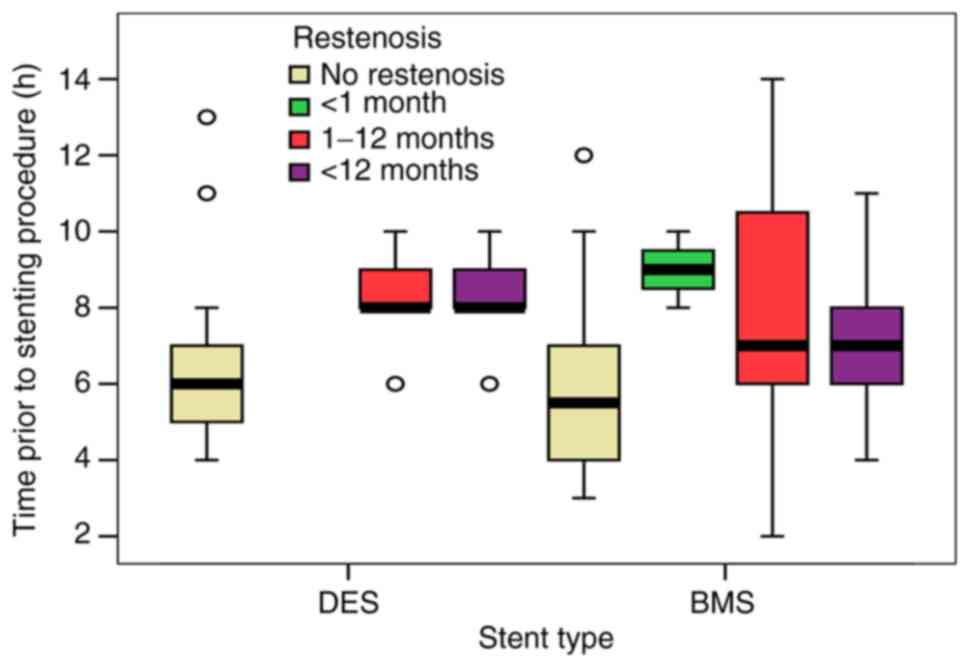
Our study showed a 60.9% use of BMS with a more
significant frequency in patients with ISR (73.1 vs. 48.3%,
P=0.001), suggesting the high importance that should be given to
the type of stent used for PCI. Comparing the 2 types of stent, the
data for DES implantation were more consistent and well-described.
Assessing the presence of restenosis events in patients that
suffered stent implantation in the first 8 h after myocardial
infarction, BMS implantation was directly linked to restenosis
events over a period of 1-12 months and also >12 months, while
DES implantation showed no significant restenosis events over the
follow-up period (Fig. 1).
Furthermore, early ISR (during the first month after PCI) was
observed only in patients with BMS regardless of the time the stent
was implemented, followed by a higher incidence of ISR in patients
with BMS vs. DES after a 1-month period (1-12 months: 27.3 vs.
14.1%; >12 months: 31.5 vs. 20.7%) (Table III).
 | Table IIICorrelation between the type of stent
and restenosis. |
Table III
Correlation between the type of stent
and restenosis.
| | Restenosis | |
|---|
| | No restenosis | <1 month | 1-12 months | >2 months | Total |
|---|
| Stent type | | | | | |
|
DES, N | 60 | 0 | 13 | 19 | 92 |
|
% stent
type | 65.2% | 0% | 14.1% | 20.7% | 100% |
|
BMS, N | 56 | 3 | 39 | 45 | 143 |
|
% stent
type | 39.1% | 2.1% | 27.3% | 31.5% | 100% |
| Total | | | | | |
|
N | 116 | 3 | 52 | 64 | 235 |
|
% stent
type | 49.4% | 1.3% | 22.1% | 27.2% | 100% |
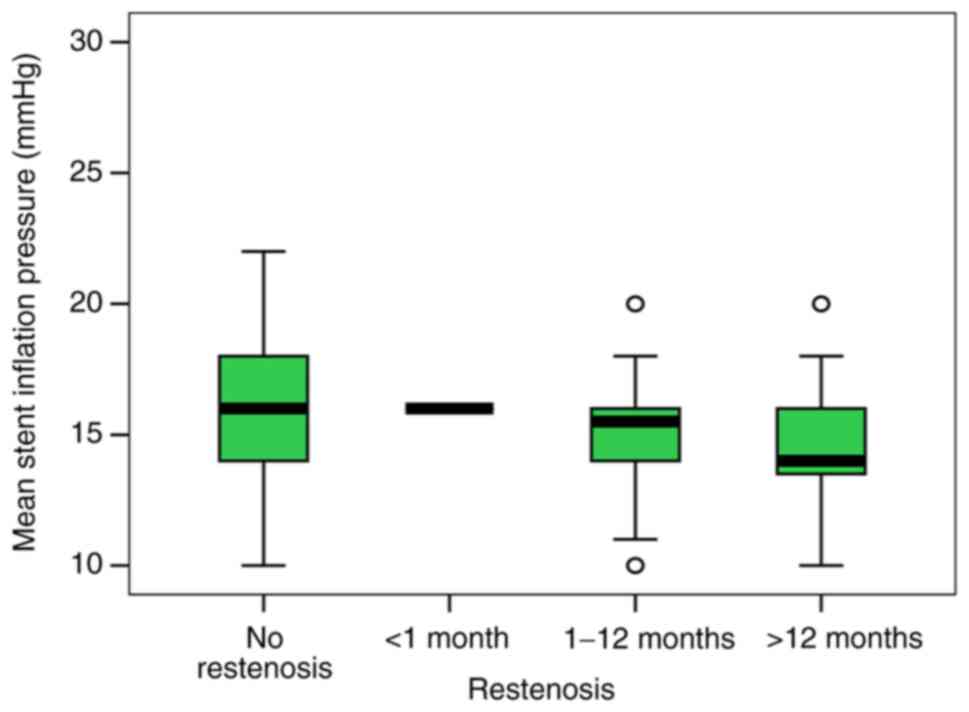
Furthermore, our study aimed to identify if there is
a correlation between restenosis and various specific variables:
The pressure under which the stent was deployed, the diameter of
the stent or the length of the stent. The mean stent inflation
pressure was significantly lower in patients with BMS implantation.
Thus, the ISR group presented a lower pressure when compared to the
control group (14.47 vs. 16.14 mmHg, P=0.004). Although the mean
stent inflation pressure did not correlate with the time course of
restenosis, the level was slightly lower in patients with ISR after
12 months compared to patients with no restenosis (14.65 vs. 15.56;
P=0.628) (Fig. 2).
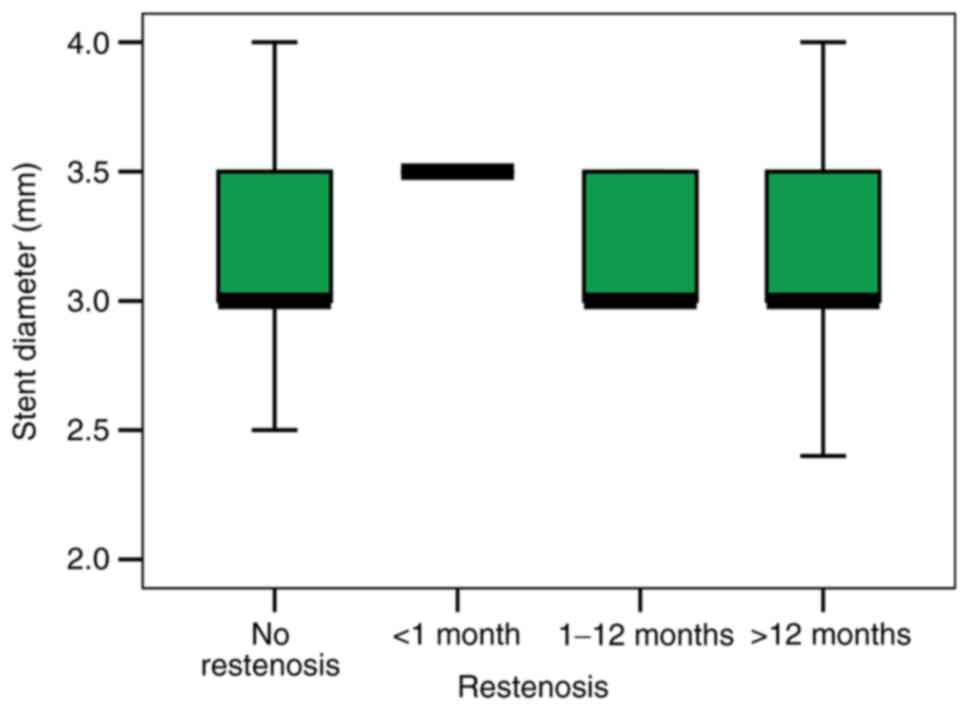
Moreover, an increased stent diameter used for PCI
in our patients did not resonate with a high ISR incidence (mean
stent diameter was 3.24 mm in patients without ISR vs. 3.22 mm in
patients with ISR; P=0.810) (Fig.
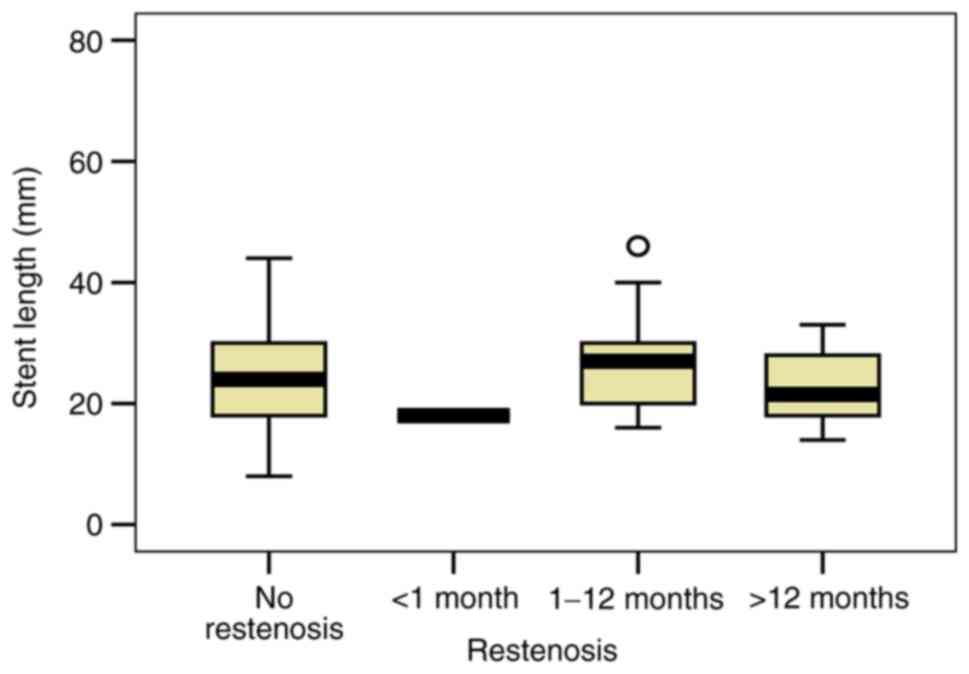
3). Analyzing the length of the stent, our study revealed a
slight ISR increase in patients with complex coronary lesions that
implied longer stents (mean length 24.98 mm in patients without ISR
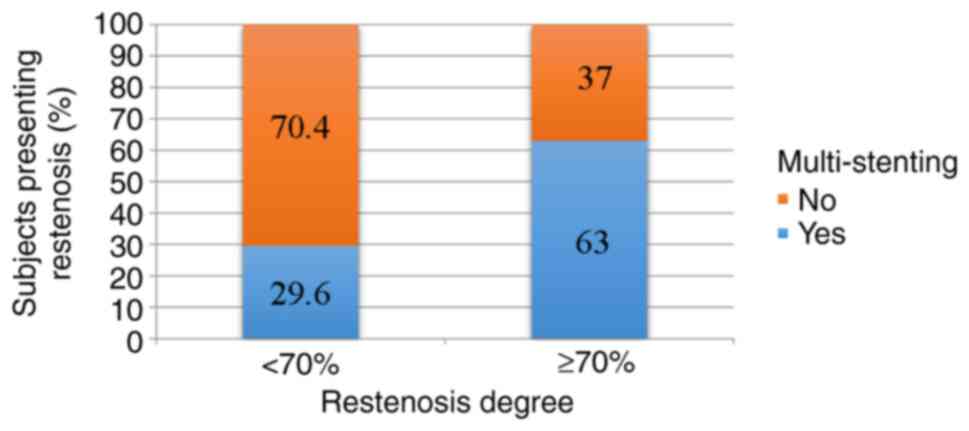
vs. 25.22 mm in patients with ISR; P=0.311) (Fig. 4). Also, the correlation between
multi-stenting and restenosis degree >70% was statistically
significant (63 vs. 29.6%; P=0.004), indicating an estimated 2
times higher risk (RR=2.13; 95% CI: 1.17-3.88) (Fig. 5).
Discussion
The main finding of the present study is that ISR in
patients following PCI is associated with smoking, hypertension,
type 2 diabetes, CKD, TIMI score, the type of stent, low inflating
stent pressure and multi-stenting. Patients were screened for
clinical and paraclinical characteristics, coronary-lesion related
factors and stent-related factors. Studies are controversial
concerning the risk of ISR in patients after PCI, thus further
research in larger cohort studies is needed.
Smoking continues to represent a major health risk
factor with a significant contribution to cardiovascular morbidity
and mortality. A recent meta-analysis performed on 141 cohort
studies and 55 study reports concluded that smoking one cigarette
per day carries around half of the risk than for those smoking 20
cigarettes per day (10). In our
study, smoking represents an important risk factor that leads to
CAD and also to post-PCI ISR.
Our results demonstrated that the estimated risk
induced by arterial hypertension was higher in patients without ISR
(RR=1.86; 95% CI: 1.41-2.45; P=0.001). Our findings are in
accordance with other studies that have identified a positive
correlation between hypertension and ISR. In a retrospective study
that included 289 patients, Wihanda et al identified
hypertension as a risk factor associated with ISR in patients
following PCI (11). Moreover,
Mohan and Dhall found a significant and positive correlation
between hypertension and ISR (12).
There is still a lack of clarity in describing the
exact mechanism that promotes the risk of ISR in patients with
diabetes, but a recent animal laboratory study revealed that
insulin and, moreover, insulin receptors are primarily responsible
for the accelerated intimal hyperplasia in diabetes which is
directly linked to the restenosis phenomenon. These results are
surprising, considering multiple previous studies that imply a more
important effect of another factor, the insulin-like growth
factor-1(13). The
physiopathological mechanism presented in the literature and the
higher incidence of diabetes in our patients with ISR compared to
those without confirm the inclusion of diabetes mellitus in the
group of risk factors for ISR.
The endothelial dysfunction responsible for ISR is
determined by an inflammatory status in patients with CAD.
C-reactive protein (CRP) is recognized as an important marker for
systemic inflammation and for predicting cardiovascular events,
therefore it can be used in primary and secondary prevention. The
cut-off value for CRP in our study was set at 2 mg/dl. A study on
1, 234 patients undergoing DES implantation showed that high levels
of CRP (>2 mg/dl) were detected in 38% of patients at baseline
and in 23.6% during late phase, both stages associated with a
higher risk for major cardiac adverse events (MACE). Moreover, high
CRP level in the late phase was a better predictor of MACE compared
to the CRP level at baseline (14).
Our findings are relatively similar with the current literature
data. High CRP levels suggest a chronic inflammation that persists
even after revascularization.
Another known marker of endothelial dysfunction that
registered high levels in our patients with ISR is the uric acid
level. Hyperuricemia might inhibit endothelial nitric oxide
synthesis and stimulate the secretion of inflammatory cytokines,
leading to neointimal hyperplasia associated with a high risk of
restenosis (15). The cut-off value
for uric acid is different among studies, usually between 6 and 10
mg/dl. A recent analysis of 21,386 individuals identified a
prognostic cut-off value of 5.34 mg/dl for all heart failure and
4.89 mg/dl for fatal heart failure (16). Therefore, the values remain
debatable. In our study, the cut-off value was established at 5
mg/dl. Even though we observed an increased incidence of elevated
uric acid levels in patients with ISR, the correlation was not
statistically significant. However, the value of uric acid
correlated well with the ISR incidence, in accordance with the
literature data (17,18).
Endothelial dysfunction is also considered a
complication in patients with CKD. Modifications at the vascular
level influence the evolution after coronary revascularization. Our
study defines a statistically significant correlation between CKD
and ISR. Data in the literature have also identified a causal
relationship between these two variables (19,20).
Another important factor in the result of the
initial procedure and in the evaluation of the risk of unfavorable
events in the future is the TIMI score. Results after the initial
angiography showed a higher incidence of patients with suboptimal
results, especially in patients that presented ISR afterwards
(P=0.001). Restenosis at 1-12 months was predominantly represented
by patients with TIMI 2 score (52.2%), while restenosis in a period
>12 months was predominantly represented by patients with TIMI 3
score (66.7%), but the results were not statistically significant
(P=0.184). These results suggest that late restenosis occurs
without an association with initial coronarography and initial
stent angioplasty.
The advantages of DES over BMS in preventing ISR
have been presented in the literature and are also confirmed in our
study. Zbinden et al showed a significant higher risk of ISR
in segments with a BMS compared to segments with a DES (5.4 vs.
0.76% after 2 years) in 2,278 patients (21). In addition, a systematic review
concerning the treatment of coronary ISR confirmed a higher rate of
ISR after BMS implantation (20-35%) vs. DES implantation (5-10%)
(22). In our analysis, BMS
presented an associated risk of ISR approximately 2 times higher as
compared to DES.
Inflation pressure during stent implantation is
correlated to angiographic lumen improvement and stent extension,
but the direction of this correlation has not been yet established.
In a non-randomized study on 136 patients undergoing PCI with BMS,
a high inflation pressure was associated with unfavorable results
on long term that included higher rates of MACE and target lesion
revascularization (TLR). Higher inflation pressure was associated
with an increased risk of ISR and TLR vs. low inflation pressure
(71 vs. 16%, respectively 27 vs. 8%) (23). However, in a randomized study, there
were no significant differences concerning the risk of ISR when
using low or high inflation pressure during stent implementation
(24). Another study analyzed
moderate to high balloon inflation pressure during PCI and found no
measurable improvement in late outcome (25). Finally, a recent retrospective study
on over 90,000 stent implementations suggests that a low and a very
high pressure elevates the risk of ISR (26). Our results support the findings of
this last study. Low pressure was reported in our group of patients
with ISR as well as in the group with BMS. Furthermore, our study
described a positive correlation between multi-stenting usage and
ISR risk. A total of 20.2% of patients from the group with ISR
presented 2 or more stents at the region where restenosis was
documented vs. 3.4% in the group without ISR (P=0.001).
Our retrospective study has a number of limitations.
Firstly, the small sample size analyzed may overestimate the
magnitude of an association or even induce false-positive results.
The patients were not divided into ischemic or angina subgroups.
However, considering the large amount of data gathered for the
analysis, this study also offers an overview of variables that need
to be taken into account when establishing correlations with ISR.
Secondly, the economical factor has a high influence in the
treatment decision of the baseline CAD diagnosis, the study
reporting an increased number of BMS implantations.
In conclusion, smoking, hypertension, diabetes
mellitus, high CRP levels, CKD, TIMI score, stent type, low
pressure for stent implantation and multi-stenting are factors
associated with ISR in patients following PCI. Thus, a close
follow-up should be targeted in these patients.
Acknowledgements
We would like to thank the Cardiac Catheterisation
Laboratory and the Intensive Coronary Care Unit of ‘George I.M.
Georgescu’ Institute of Cardiovascular Diseases, Iasi.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
DMA and CAG developed the study concept and design.
DMA collected the data and created the database. IIC, LM, AA and
CAG performed the literature research and contributed to the
introduction, results and discussion sections. OM and IM conducted
the statistical analysis and created the images and tables, with
assistance from DMA and CAG. All authors were involved in drafting
and finalizing the manuscript. All authors read and approved the
final manuscript for publication.
Ethics approval and consent to
participate
The study was approved by the University of Medicine
and Pharmacy ‘Grigore T. Popa’ Iasi Research Ethics Committee and
all subjects had initially agreed and signed an informed consent in
order to take part in this study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Lozano R, Naghavi M, Foreman K, Lim S,
Shibuya K, Aboyans V, Abraham J, Adair T, Aggarwal R, Ahn SY, et
al: Global and regional mortality from 235 causes of death for 20
age groups in 1990 and 2010: A systematic analysis for the global
burden of disease study. Lancet. 380:2095–2128. 2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Alfonso F, Byrne RA, Rivero F and Kastrati
A: Current treatment of in-stent restenosis. J Am Coll Cardiol.
63:2659–2673. 2014.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Alfonso F: Treatment of drug-eluting stent
restenosis the new pilgrimage: Quo vadis? J Am Coll Cardiol.
55:2717–2720. 2010.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Rao G, Sheth S and Grines C: Percutaneous
coronary intervention: 2017 in review. J interv Cardiol.
31:117–128. 2018.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Baschet L, Bourguignon S, Marque S,
Durand-Zaleski I, Teiger E, Wilquin F and Levesque K:
Cost-effectiveness of drug-eluting stents versus bare-metal stents
in patients undergoing percutaneous coronary intervention. Open
Heart. 3(e000445)2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Neupane S, Khawaja O, Edla S, Singh H,
Othman H, Bossone E, Yamasaki H, Rosman HS, Eggebrecht H and Mehta
RH: Meta-analysis of drug eluting stents compared with bare metal
stents in high bleeding risk patients undergoing percutaneous
coronary interventions. Catheter Cardiovasc Interv. 94:98–104.
2019.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ochijewicz D, Tomaniak M, Opolski G and
Kochman J: Inflammation as a determinant of healing response after
coronary stent implantation. Int J Cardiovasc Imaging. 37:791–801.
2021.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Stone GW and Kirtane AJ: Bare metal and
drug-eluting coronary stents. In: Topol EJ, Teristein PS (ed)
Textbook of interventional cardiology 6th ed., Philadelphia:
Saunders Elsevier pp171-196, 2012.
|
|
9
|
Her AY and Shin ES: Current management of
in-stent restenosis. Korean Circ J. 48:337–349. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hackshaw A, Morris JK, Boniface S, Tang JL
and Milenković D: Low cigarette consumption and risk of coronary
heart disease and stroke: Meta-analysis of 141 cohort studies in 55
study reports. BMJ. 360(j5855)2018.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Wihanda D, Alwi I, Yamin M, Shatri H and
Mudjaddid E: Factors associated with In-stent restenosis in
patients following percutaneous coronary intervention. Acta Med
Indones. 47:209–215. 2015.PubMed/NCBI
|
|
12
|
Mohan S and Dhall A: A comparative study
of restenosis rates in bare metal and drug-eluting stents. Int J
Angiol. 19:e66–e72. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Li Q, Fu J, Xia Y, Qi W, Ishikado A, Park
K, Yokomizo H, Huang Q, Cai W, Rask-Madsen C, et al: Homozygous
receptors for insulin and not IGF-1 accelerate intimal hyperplasia
in insulin resistance and diabetes. Nat Commun.
10(4427)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Shiba M, Itaya H, Iijima R and Nakamura M:
Influence of late vascular inflammation on long-term outcomes among
patients undergoing implantation of drug eluting stents: Role of
C-Reactive protein. J Am Heart Assoc. 5(e003354)2016.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Kim SY, Guevara JP, Kim KM, Choi HK,
Heitjan DF and Albert DA: Hyperuricemia and coronary heart disease:
A systematic review and meta-analysis. Arthritis Care Res
(Hoboken). 62:170–180. 2010.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Muiesan ML, Salvetti M, Virdis A, Masi S,
Casiglia E, Tikhonoff V, Barbagallo CM, Bombelli M, Cicero AFG,
Cirillo M, et al: Serum uric acid, predicts heart failure in a
large Italian cohort: Search for a cut-off value the uric acid
right for heart health study. J Hypertens. 39:62–69.
2021.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wu Y and Fu X: Comprehensive analysis of
predictive factors for rapid angiographic stenotic progression and
restenosis risk in coronary artery disease patients underwent
percutaneous coronary intervention with drug-eluting stents
implantation. J Clin Lab Anal. 33(e22666)2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Joo HJ, Jeong HS, Kook H, Lee SH, Park JH,
Hong SJ, Yu CW and Lim DS: Impact of hyperuricemia on clinical
outcomes after percutaneous coronary intervention for in-stent
restenosis. BMC Cardiovasc Disord. 18(114)2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lambert ND, Sacrinty MT, Ketch TR, Turner
SJ, Santos RM, Daniel KR, Applegate RJ, Kutcher MA and Sane DC:
Chronic kidney disease and dipstick proteinuria are risk factors
for stent thrombosis in patients with myocardial infarction. Am
Heart J. 157:688–694. 2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Gao WD, Ma M, Zhang GX, Zhang XF and Sun
G: First-generation versus second-generation drug-eluting stents in
patients with chronic kidney disease: A systematic review and
meta-analysis. Postgrad Med. 131:43–51. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zbinden R, von Felten S, Wein B, Tueller
D, Kurz DJ, Reho I, Galatius S, Alber H, Conen D, Pfisterer M, et
al: Impact of stent diameter and length on in-stent restenosis
after DES vs BMS implantation in patients needing large coronary
stents-A clinical and health-economic evaluation. Cardiovasc Ther.
35:19–25. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Pleva L, Kukla P and Hlinomaz O: Treatment
of coronary in-stent restenosis: A systematic review. J Geriatr
Cardiol. 15:173–184. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Uretsky BF, Rosanio S, Lerakis S, Wang FW,
Smiley M, Stouffer GA, Tocchi M and Estella P: A prospective
evaluation of angiography-guided coronary stent implantation with
high versus very high balloon inflation pressure. Am Heart J.
140:804–812. 2000.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Dirschinger J, Kastrati A, Neumann FJ,
Boekstegers P, Elezi S, Mehilli J, Schühlen H, Pache J, Alt E,
Blasini R, et al: Influence of balloon pressure during stent
placement in native coronary arteries on early and late
angiographic and clinical outcome: A randomized evaluation of
high-pressure inflation. Circulation. 100:918–923. 1999.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mattos LA, Sousa AG, Chaves A, Feres F,
Pinto I, Tanajura L, Centemero M, Abizaid A, Seixas AC, Abizaid A,
et al: Influence of balloon pressure inflation in patients
undergoing primary coronary stent implantation during acute
myocardial infarction: A quantitative coronary angiography
analysis. Arq Bras Cardiol. 80:250–268. 2003.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Fröbert O, Sarno G, James SK, Saleh N and
Lagerqvist B: Effect of stent inflation pressure and
Post-Dilatation on the outcome of coronary artery intervention. A
report of more than 90,000 stent implantations. PLoS One.
8(e56348)2013.PubMed/NCBI View Article : Google Scholar
|