Introduction
Amyloidosis is a disease characterized by
extracellular deposition of misfolded protein and involves multiple
organs and tissue with widely varying clinical features (1). Thus, due to the wide range of
symptoms, amyloidosis is difficult to rapidly diagnose. Several
studies have reported amyloidosis with involvement of the
gastrointestinal tract or peritoneum (2-10),
but reports of amyloidosis involving both organs are rare. The
present study reported a rare case of primary amyloidosis (AL)
simultaneously involving the gastrointestinal tract, mesentery and
omentum.
Case report
A 66-year-old male presented with repeated diarrhoea
and abdominal distension for nine months and a 2-day history of
bloody stool. The patient underwent laparotomy 1 month previously
for adhesive ileus, which did not relieve his symptoms. The
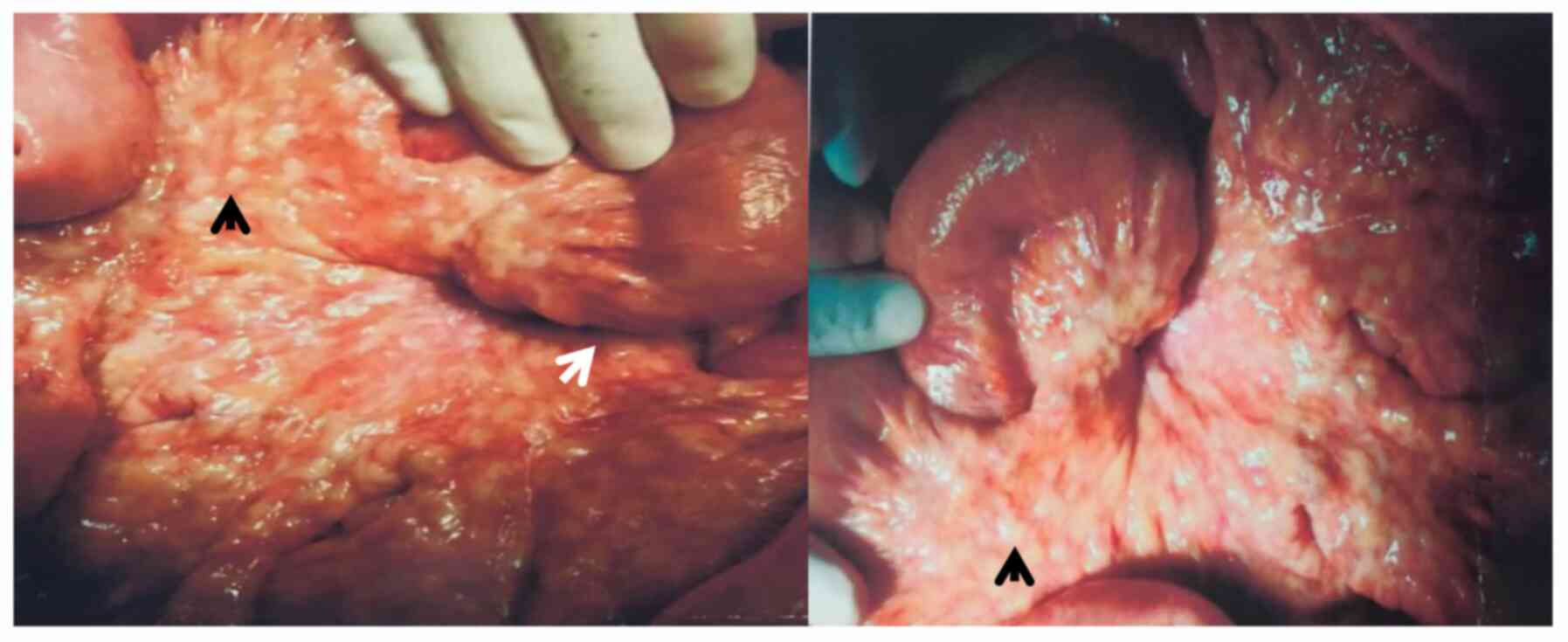
operation revealed multiple nodules in the mesentery of the small
intestine and extensive peritoneal adhesions (Fig. 1). Post-operative pathology indicated
hyaline degeneration and fibrinoid necrosis with inflammatory cell
infiltration. The patient had lost 30 kg of body weight over nine
months. He had no history of tuberculosis or rheumatoid arthritis
and no family history of hereditary diseases.
On physical examination, the patient's body
temperature, blood pressure, respiratory rate, pulse and BMI were
36.8˚C, 110/78 mmHg, 19 bpm, 84 bpm and 17.2 kg/m2,
respectively. Palpation of the abdomen indicated multiple hard
nodules of varying sizes in the abdominal wall as well as abdominal
tenderness and mild rebound tenderness. Laboratory tests revealed
the following (Table I):
Haemoglobin 80 g/l, C-reactive protein 13.5 mg/l, erythroid
sedimentation rate 28 mm/h, procalcitonin 0.12 ng/ml, globulin 16
g/l, albumin 26 g/l and CA125 187 U/ml. The patient's level of
urinary immunoglobulin kappa light chain was 456 mg/l; the
immunoglobulin lambda light chain level was normal. Serum
immunoglobulin kappa and lambda light chain values were 8.31 and
2.56 g/l, respectively, serum immunoglobulin lambda light chain was
decreased. According to bone marrow biopsy, the proportion of
plasma cells in the bone marrow was slightly increased. T-SPOT
tuberculosis-specific enzyme-linked immunospot assay and
tuberculosis antibody tests were negative. Antinuclear antibody and
antineutrophil cytoplasmic antibody spectra were also negative.
Ultrasound examination of the abdomen demonstrated colonic wall
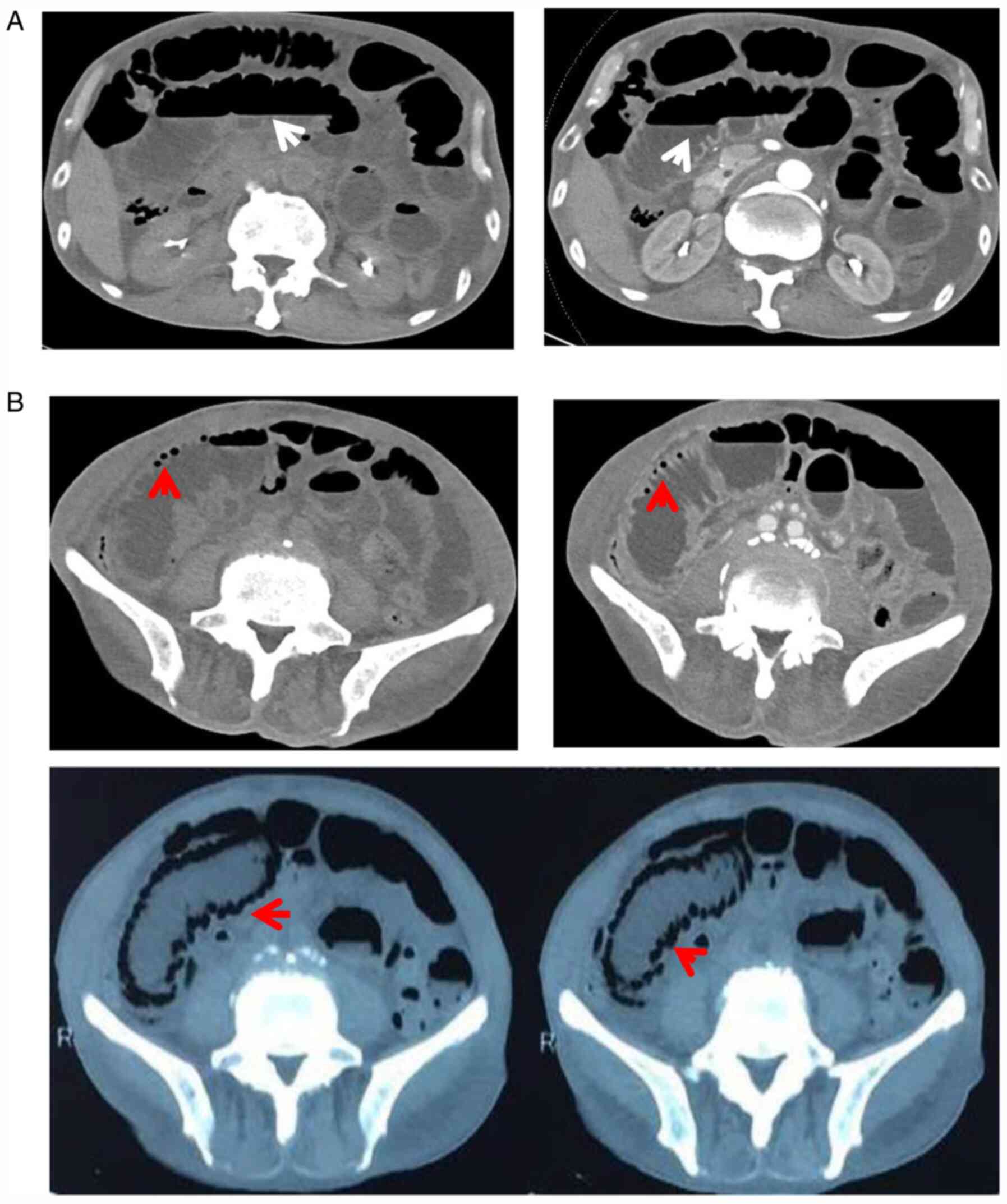
thickening and stiffness. Abdominal contrast-enhanced CT revealed
peritoneum and mesentery thickening, as well as distension of the
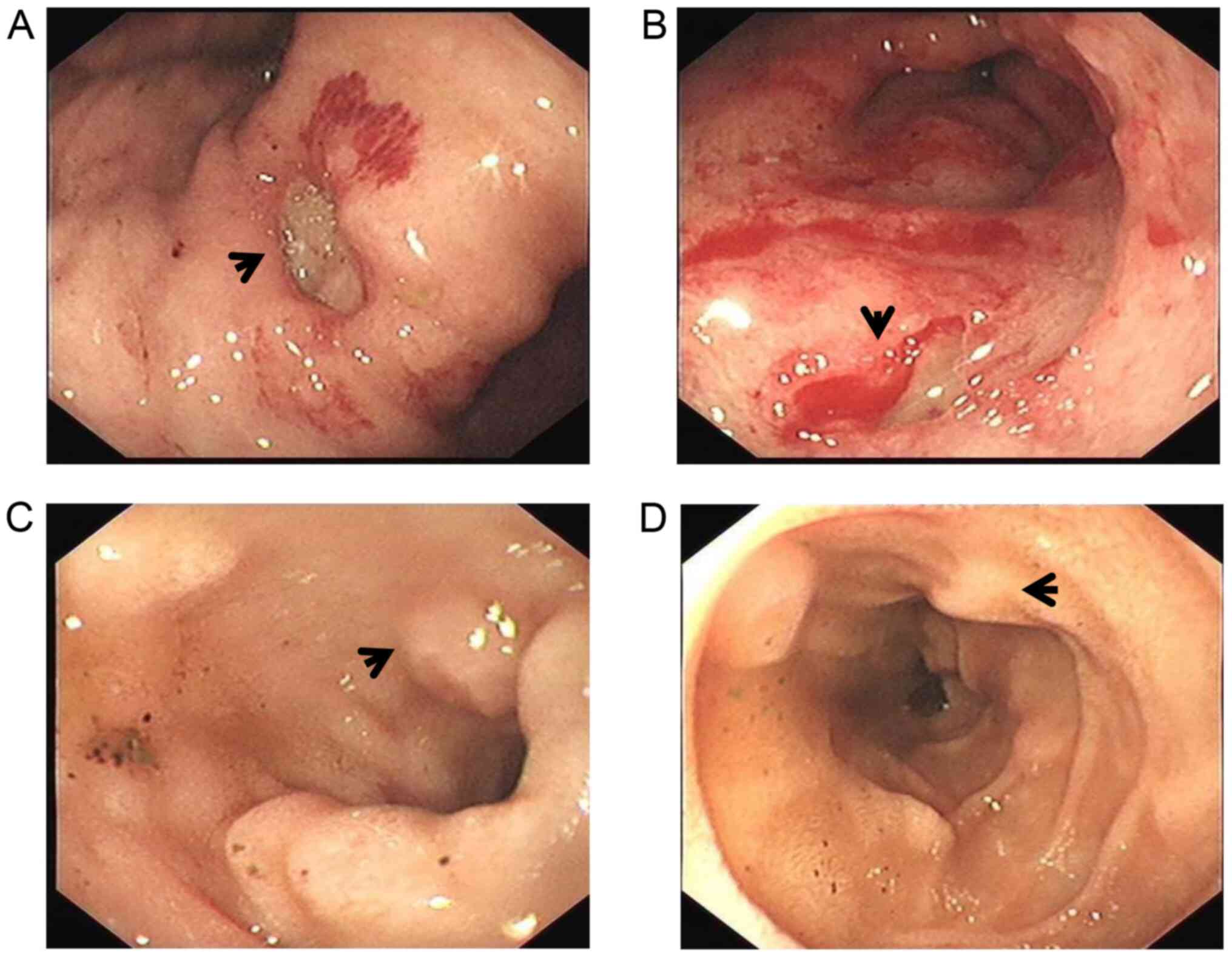
small bowel with pneumatosis intestinalis (Fig. 2). Multiple gastric ulcers were
observed on initial upper gastrointestinal endoscopy (Fig. 3), and histological examination
revealed superficial gastritis with gland hyperplasia. At another
hospital, enteroscopy performed one month previously revealed
multiple protuberant lesions in the colon. On double-balloon
enteroscopy via the oral route, duodenal and upper jejunal mucosa
swelling (Fig. 4), as well as
villous atrophy, were detected. Histological examination indicated
chronic mucositis.
 | Table ILaboratory data. |
Table I
Laboratory data.
| Item | Result | Normal values |
|---|
| Hemoglobin (g/l) | 80 | 120-160 |
| Fecal occult
blood | +++ | Negative |
| Urinary
immunoglobulin kappa light chain (mg/l) | 456.0 | 0-18.5 |
| Urinary
immunoglobulin lambda light chain (mg/l) | <50.0 | 0-50 |
| Serum immunoglobulin
kappa light chain (g/l) | 8.31 | 6.29-13.5 |
| Serum immunoglobulin
lambda light chain (g/l) | 2.56 | 3.13-7.23 |
| C-reactive protein
(mg/l) | 13.5 | 0-8 |
| PCT (ng/ml) | 0.12 | 0-0.05 |
| ESR (mm/h) | 28 | 0-15 |
| Albumin (g/l) | 26 | 35-55 |
| Globulin (g/l) | 16 | 20-30 |
| CA125 (U/ml) | 187.00 | 0-35 |
| T-SPOT.TB and
tuberculosis antibody | Negative | Negative |
| ANCAs and ANAs | Negative | Negative |
As the patient's symptoms were not relieved by our
treatment or explained by the results of the examinations, repeated
upper gastrointestinal endoscopy was performed, which revealed
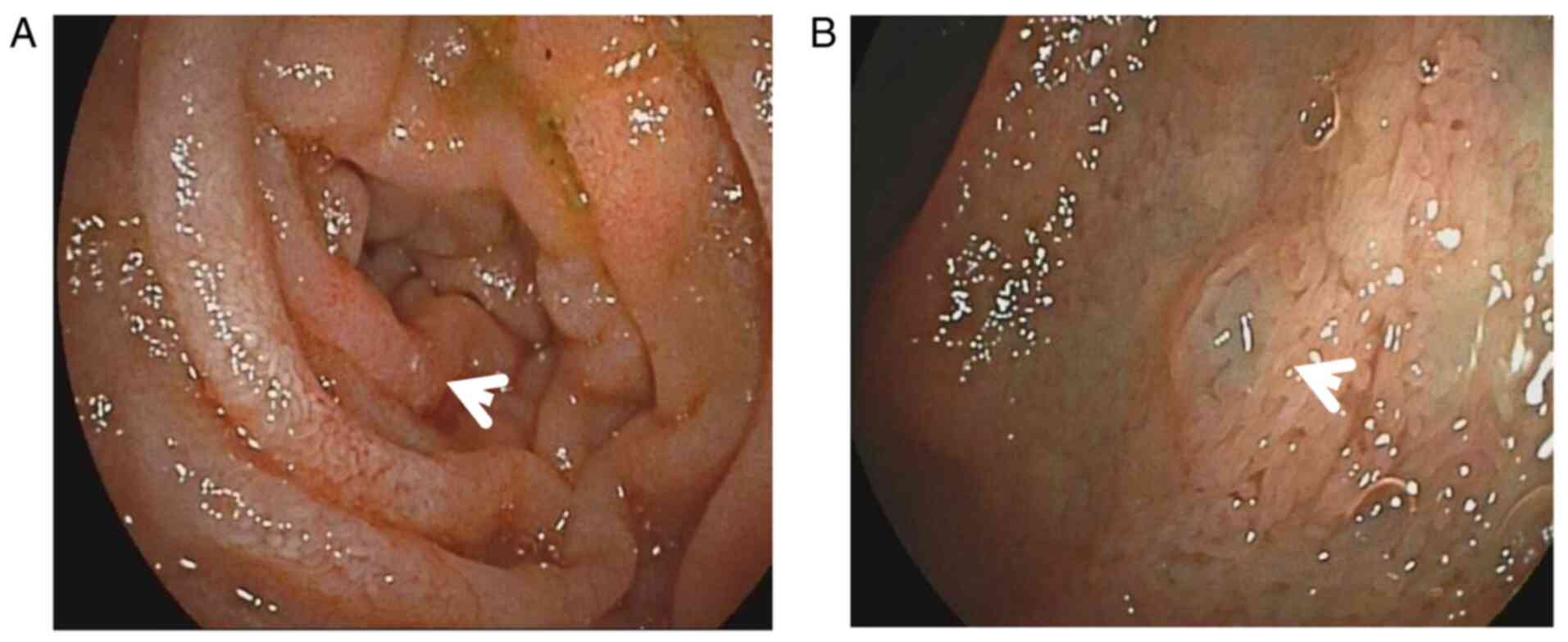
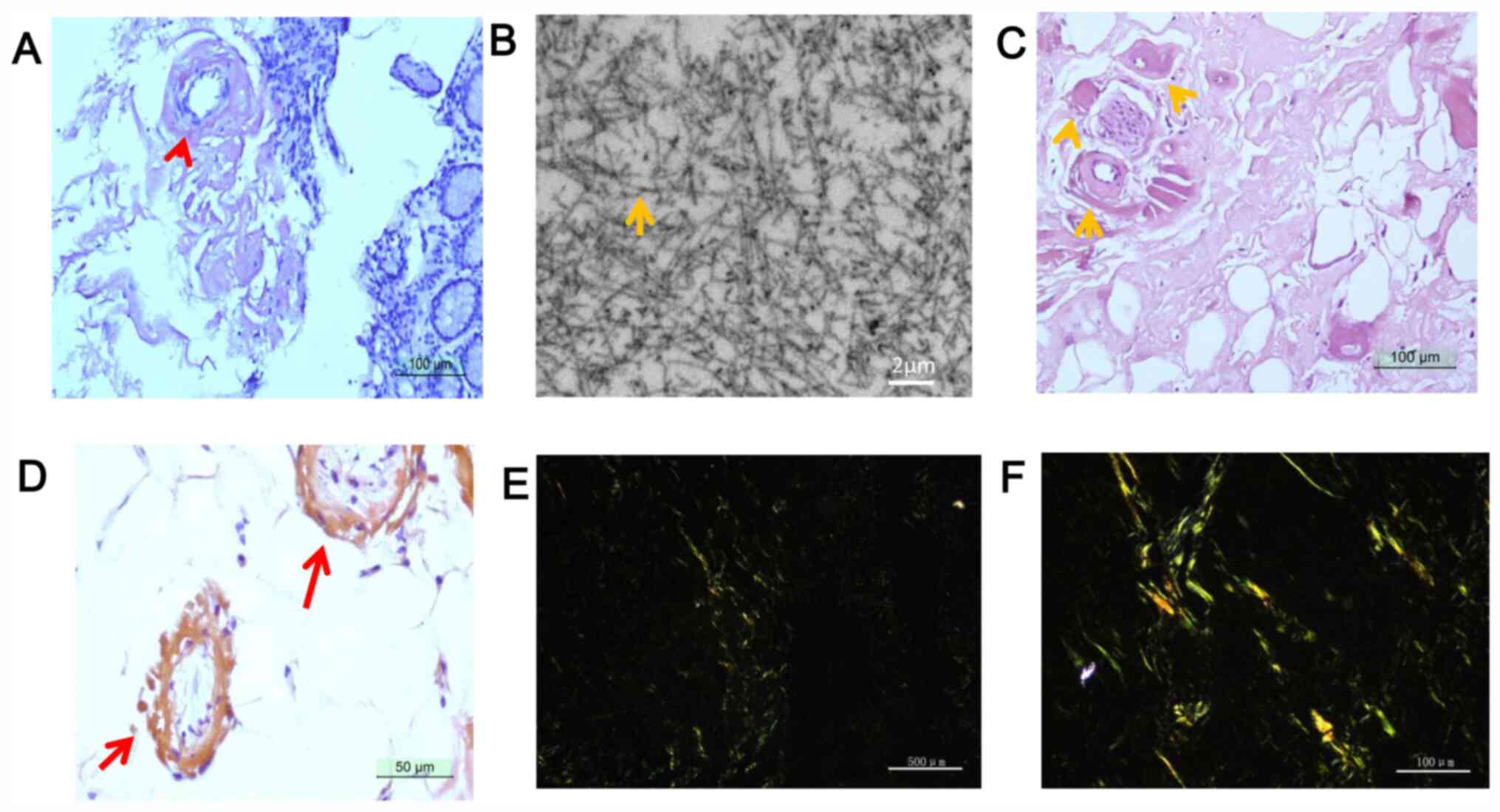
multiple gastric ulcers that bled easily upon contact. Congo red
staining of gastric mucosa biopsy specimens confirmed the presence
of amyloid deposits (Fig. 5). In
addition, non-branching fibrils were observed in the gastric mucosa
by electron microscopy. In mesentery biopsy specimens that were
acquired during surgery, hyaline degeneration of arteriole walls
was revealed by haematoxylin-eosin staining and those samples were
positive on Congo red staining (Fig.
5). According to all available information and examinations of
this patient, a diagnosis of AL involving the gastrointestinal
tract and peritoneum was made. The patient was subsequently treated
with corticosteroids and thalidomide. Regrettably, the patient
responded poorly to treatment and died from massive haemorrhage of
the gastrointestinal tract one month later.
Discussion
Amyloidosis is characterized by abnormal
extracellular deposition of diverse types of amyloid proteins
within various organs. Congo red staining is a common, effective
test used to differentiate amyloid deposits from other protein
deposits (1). Furthermore,
nonbranching fibrils may be observed under electron microscopy in
amyloidotic tissue (11). There are
two major types of amyloidosis (12). The most common form is AL, which is
associated with abnormal deposition of monoclonal immunoglobulin
light chain (13,14) produced by a plasma cell clone. AL is
also associated with plasma cell dyscrasias. The second most common
form is secondary amyloidosis, which is associated with abnormal
deposition of serum amyloid protein A. This form typically occurs
as a result of chronic inflammatory diseases or infections. In the
patient of the present study, urinary Igκ was significantly
increased, the proportion of plasma cells in the bone marrow was
slightly increased and multiple organs were involved; therefore,
this patient was diagnosed with AL.
The mesentery is a double layer of peritoneum that
suspends the small and large bowel from the posterior abdominal
wall, preventing these organs from collapsing into the pelvis
(15). The mesentery, which
contains vessels, nerves, lymphoid tissue, adipose tissue, fibrous
tissue and macrophages (16),
supplies the intestinal tract with nutrients and is involved in
immune defence. These anatomical and functional features suggest
that the mesentery acts as an organ (16). Furthermore, mesenteric abnormalities
have been indicated to be involved in numerous diseases. The
abdominal distension of the patient of the present study was likely
due to amyloid deposition in the mesentery. Several studies have
reported that the clinical presentation of amyloidosis involves the
mesentery (6,7,9). In
the present case, mesentery amyloidosis was detected, which
manifested as multiple nodules and extensive peritoneal adhesions;
such a presentation has not been previously reported likely due to
its rarity, to the best of our knowledge. Therefore, in the future,
Congo red staining should be considered in similar situations.
Pneumatosis intestinalis is characterized by gas
pockets in the wall of the small or large bowel (17); however, the pathogenesis of this
condition remains elusive. One study speculated that pneumatosis
intestinalis is due to disruption of intestinal mucosal integrity
by amyloid deposits, leading to infiltration of luminal gas or
gas-producing bacteria (18).
Although gastrointestinal amyloidosis is a rare cause of
pneumatosis intestinalis, several studies have reported that
amyloidosis involving the intestinal tract may present as
pneumatosis intestinalis (17,19).
Indeed, the patient of the present study had pneumatosis
intestinalis. These results indicate that pneumatosis intestinalis
may be a sign of amyloidosis involving the gastrointestinal tract,
which may help accelerate the diagnosis of amyloidosis when it
involves multiple organs and pneumatosis intestinalis is
present.
The principle of the treatment of AL is to eliminate
the underlying abnormal plasma cells by chemotherapy associated
with haematopoietic stem cell transplantation. Corticosteroids have
been indicated to be useful in the treatment of patients with AL
experiencing refractory diarrhoea and protein loss enteropathy
(20). Accordingly, the patient of
the present study was treated with corticosteroids and thalidomide,
which temporarily relieved his symptoms.
In summary, the present study reported on a rare
case of AL simultaneously involving the gastrointestinal tract,
mesentery and omentum. The detailed endoscopic and laparotomy
findings for this case were also presented. Early diagnosis of AL
is important, as most cases are diagnosed at a late stage and have
poor prognosis. In patients with unexplained gastrointestinal
symptoms, particularly those with multisystemic involvement, Congo
red staining is recommended.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All datasets used and/or analysed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
GD and ZS conceptualized and designed the case
study. MQ interpreted CT images and performed data collection. GD
and QG analysed and interpreted the data. GD and ZS drafted and
critically revised the manuscript. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
The study protocol conformed to the ethical
guidelines of the Declaration of Helsinki and was approved by the
Ethics Review Committee of The First People's Hospital of Yunnan
Province (Kunming, China). Written informed consent was obtained
from the legal guardian of the patient.
Patient consent for publication
The legal guardian of the patient provided consent
for the publication of this case report.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Hazenberg BP, van Gameren II, Bijzet J,
Jager PL and van Rijswijk MH: Diagnostic and therapeutic approach
of systemic amyloidosis. Neth J Med. 62:121–128. 2004.PubMed/NCBI
|
|
2
|
Asakura K, Yanai S, Nakamura S, Kawaski K,
Eizuka M, Ishida K, Sugai T, Ueda M, Yamashita T, Ando Y and
Matsumoto T: Endoscopic findings of small-bowel lesions in familial
amyloid polyneuropathy: A case report. Medicine (Baltimore).
95(e2896)2016.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wang Z, Huang C and Ji F: Primary
amyloidosis mimicking Crohn's disease: A case report. Int J Clin
Exp Med. 8:16137–16139. 2015.PubMed/NCBI
|
|
4
|
Siau K, Elzubeir A, Cooper SC and Iqbal T:
Amyloidosis: An unusual cause of upper gastrointestinal bleeding.
BMJ Case Rep. 2016(bcr2016217653)2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Groisman GM and Cohen HI: Small intestinal
amyloidosis: A rare cause of diverticular disease. Case Rep Pathol.
2014(362835)2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Vanhoenacker F, Vanwambeke K and Jacomen
G: Amyloidosis: An unusual cause of mesenteric, omental and lymph
node calcifications. JBR-BTR. 97:283–286. 2014.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Coulier B, Montfort L, Doyen V and Gielen
I: MDCT findings in primary amyloidosis of the greater omentum and
mesentery: A case report. Abdom Imaging. 35:88–91. 2010.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Horger M, Vogel M, Brodoefel H, Schimmel H
and Claussen C: Omental and peritoneal involvement in systemic
amyloidosis: CT with pathologic correlation. AJR Am J Roentgenol.
186:1193–1195. 2006.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Halm U, Berr F, Tannapfel A, Klöppel R,
Secknus R and Mössner J: Primary amyloidosis of the mesentery and
the retroperitoneum presenting with lymphedema. Am J Gastroenterol.
93:2299–2300. 1998.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Poturoğlu S, Mungan Z, Boztaş G,
Kaymakoglu S, Ozdil S, Akyüz F, Aksoy N, Kamali S, Pinarbasi B,
Cevikbaş U and Okten A: Peritoneal amyloidosis caused by familial
mediterranean fever. J Gastroenterol Hepatol. 20:325–326.
2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sipe JD and Cohen AS: Review: History of
the amyloid fibril. J Struct Biol. 130:88–98. 2000.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Sipe JD, Benson MD, Buxbaum JN, Ikeda S,
Merlini G, Saraiva MJ and Westermark P: Nomenclature 2014: Amyloid
fibril proteins and clinical classification of the amyloidosis.
Amyloid. 21:221–224. 2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Freudenthaler S, Hegenbart U, Schönland S,
Behrens HM, Krüger S and Röcken C: Amyloid in biopsies of the
gastrointestinal tract-a retrospective observational study on 542
patients. Virchows Arch. 468:569–577. 2016.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Cowan AJ, Skinner M, Seldin DC, Berk JL,
Lichtenstein DR, O'Hara CJ, Doros G and Sanchorawala V: Amyloidosis
of the gastrointestinal tract: A 13-year, single-center, referral
experience. Haematologica. 98:141–146. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Patel RR and Planche K: Applied peritoneal
anatomy. Clin Radiol. 68:509–520. 2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Coffey JC and O'Leary DP: The mesentery:
Structure, function, and role in disease. Lancet Gastroenterol
Hepatol. 1:238–247. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Raghunathan V, Louis D and Wirk B:
Gastrointestinal tract amyloidosis presenting with pneumatosis
intestinalis. J Clin Med Res. 9:654–658. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Khalil PN, Huber-Wagner S, Ladurner R,
Kleespies A, Siebeck M, Mutschler W, Hallfeldt K and Kanz KG:
Natural history, clinical pattern, and surgical considerations of
pneumatosis intestinalis. Eur J Med Res. 14:231–239.
2009.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Khalid F, Kaiyasah H, Binfadil W, Majid M,
Hazim W and ElTayeb Y: Pneumatosis intestinalis due to
gastrointestinal amyloidosis: A case report & review of
literature. Int J Surg Case Rep. 23:29–32. 2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Gertz MA: How to manage primary
amyloidosis. Leukemia. 26:191–198. 2012.PubMed/NCBI View Article : Google Scholar
|