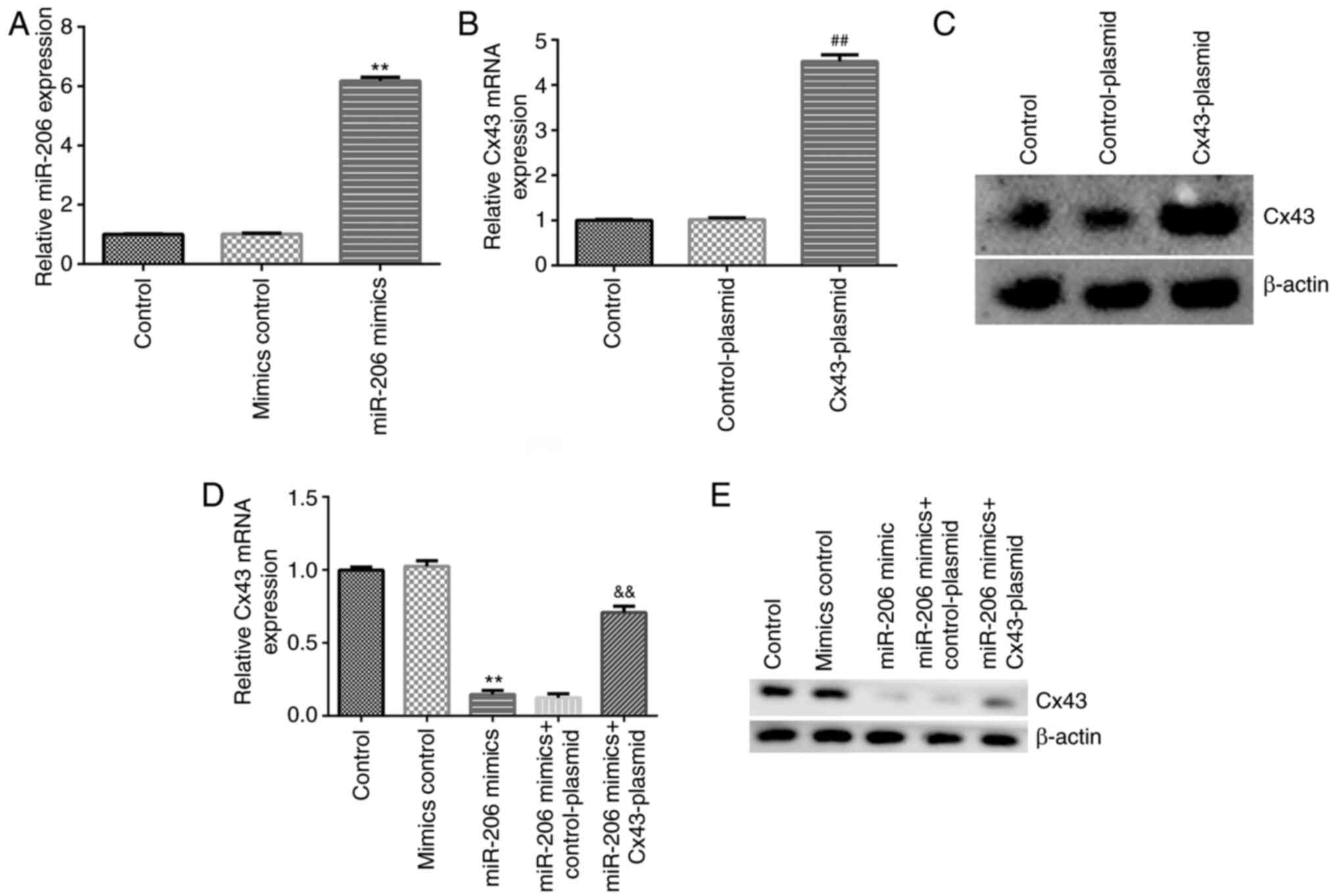
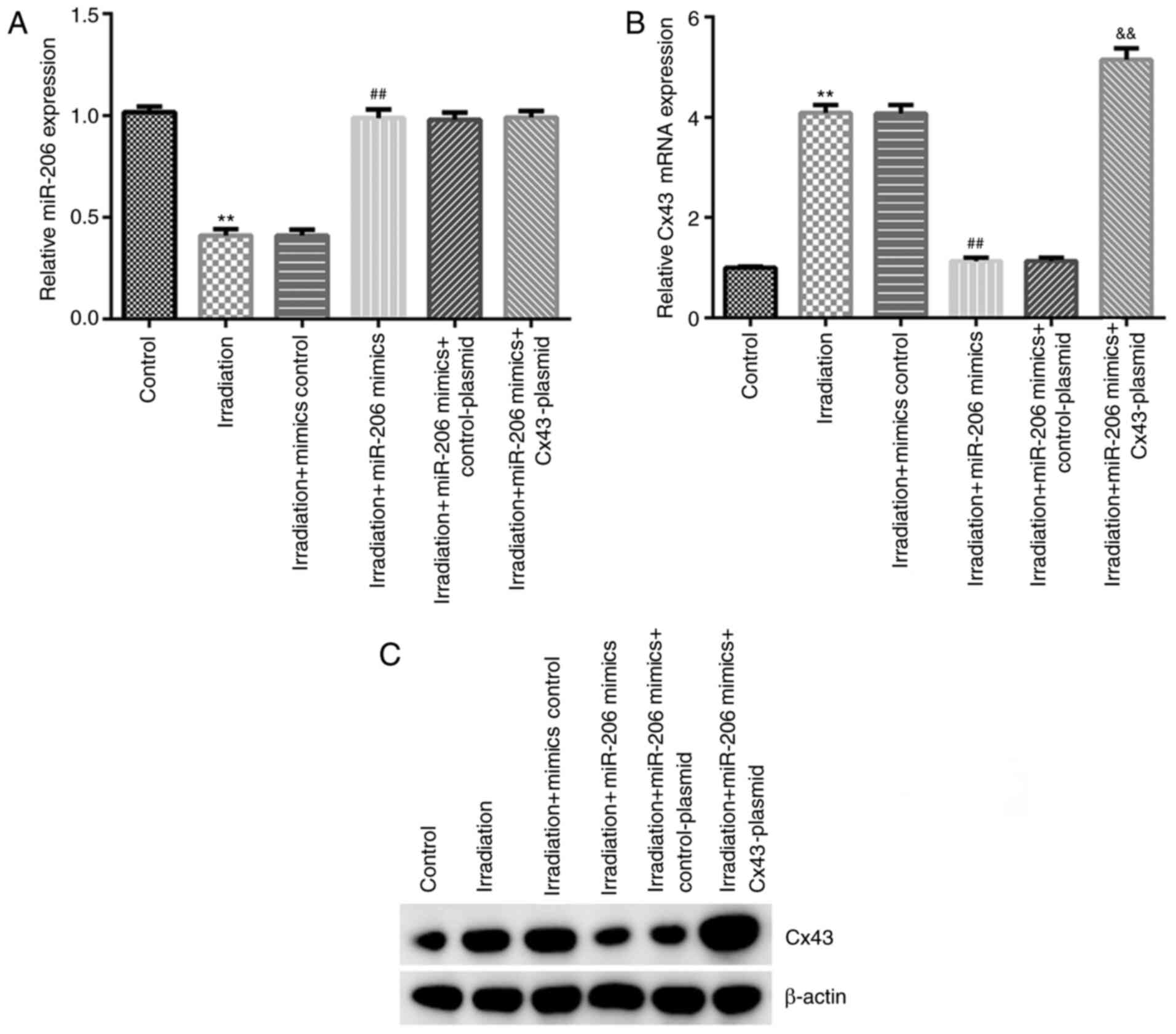
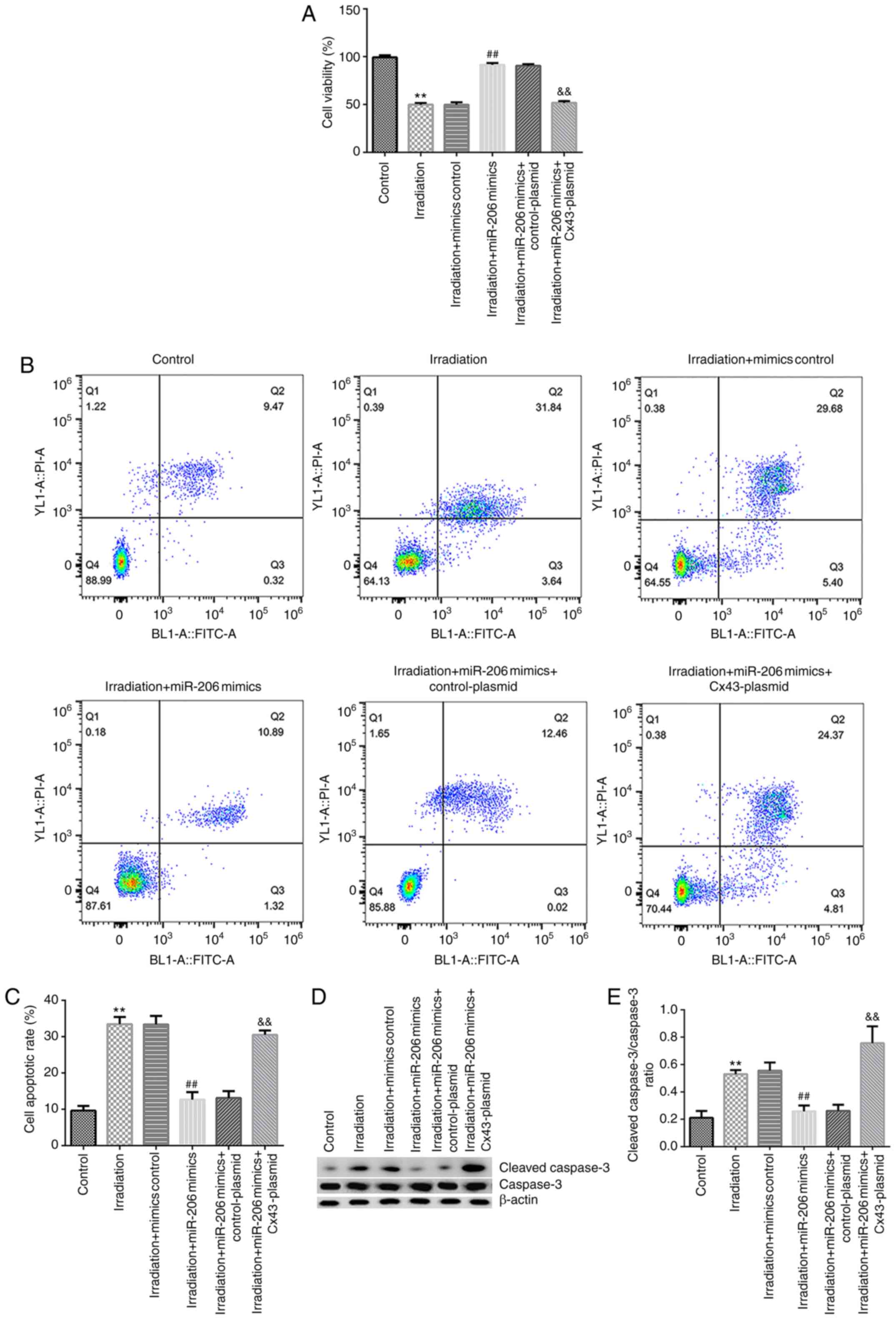
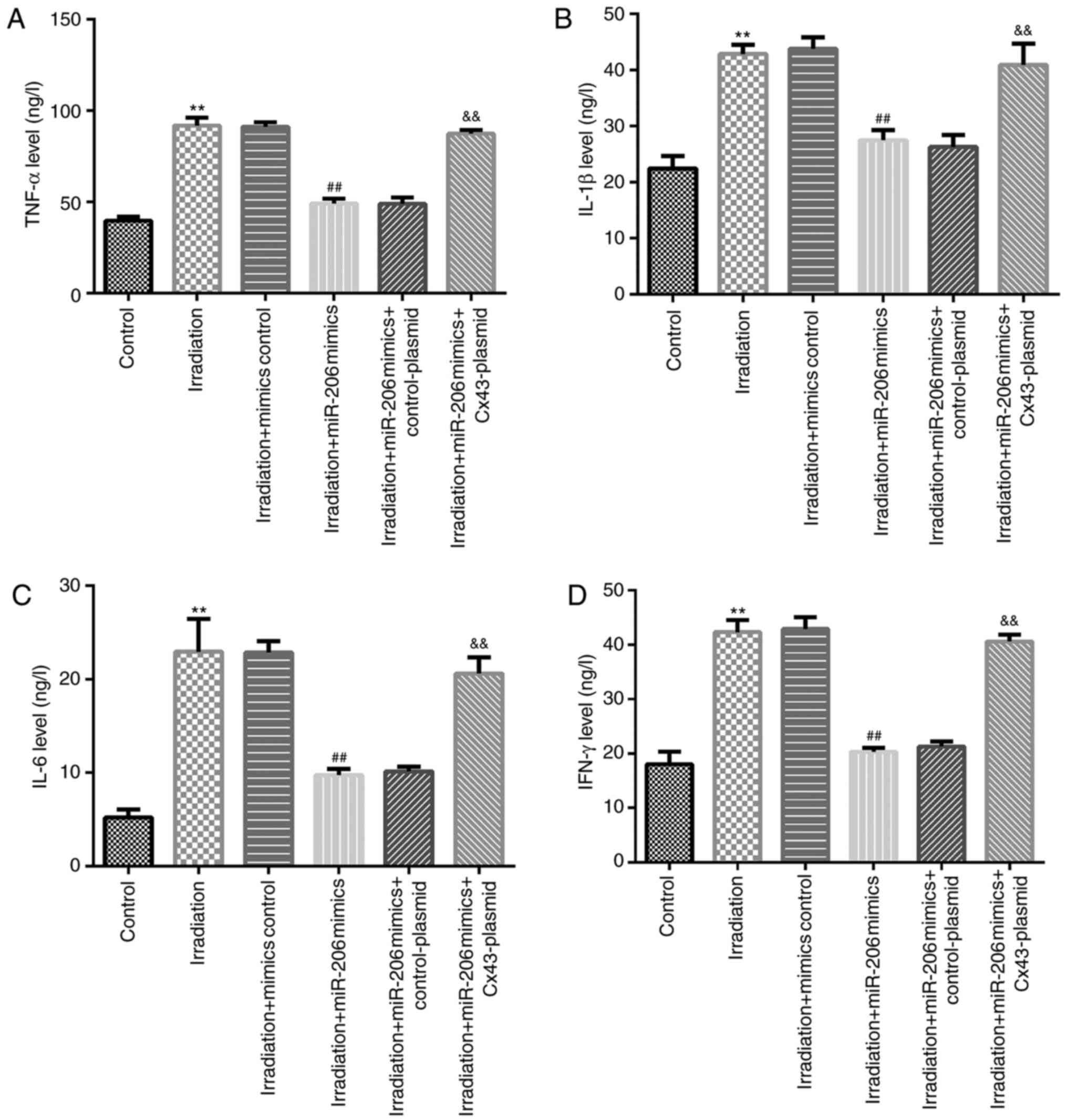
|
1
|
Wisniewski T, Winiecki J, Makarewicz R and
Zekanowska E: The effect of radiotherapy and hormone therapy on
osteopontin concentrations in prostate cancer patients. J BUON.
25:527–530. 2020.PubMed/NCBI
|
|
2
|
Alcântara-Silva TR, de Freitas-Junior R,
Freitas NM, de Paula Junior W, da Silva DJ, Machado GD, Ribeiro MK,
Carneiro JP and Soares LR: Music therapy reduces
radiotherapy-induced fatigue in patients with breast or
gynecological cancer: A Randomized Trial. Integr Cancer Ther.
17:628–635. 2018.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Agrawal V, Benjamin KT and Ko EC:
Radiotherapy and Immunotherapy Combinations for Lung Cancer. Curr
Oncol Rep. 23(4)2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Brada M and Jankowska P: Radiotherapy for
pituitary adenomas. Endocrinol Metab Clin North Am. 37:263–275.
2008.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Dahl O, Dale JE and Brydøy M: Rationale
for combination of radiation therapy and immune checkpoint blockers
to improve cancer treatment. Acta Oncol. 58:9–20. 2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yamanaka R, Abe E, Sato T, Hayano A and
Takashima Y: Secondary intracranial tumors following radiotherapy
for pituitary adenomas: A systematic review. Cancers (Basel).
9(E103)2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Xiang B, Zhu X, He M, Wu W, Pang H, Zhang
Z, Yang Y, Li Y, Wang Y, Wang Y, et al: Pituitary dysfunction in
patients with intracranial germ cell tumors treated with
radiotherapy. Endocr Pract. 26:1458–1468. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kuan FC, Lee KD, Huang SF, Chen PT, Huang
CE, Wang TY and Chen MC: Radiotherapy is associated with an
accelerated risk of ischemic stroke in oral cavity cancer survivors
after primary surgery. Cancers (Basel). 12(E616)2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Schultz C, Dehghani F, Hubbard GB, Thal
DR, Struckhoff G, Braak E and Braak H: Filamentous tau pathology in
nerve cells, astrocytes, and oligodendrocytes of aged baboons. J
Neuropathol Exp Neurol. 59:39–52. 2000.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Doron H, Amer M, Ershaid N, Blazquez R,
Shani O, Lahav TG, Cohen N, Adler O, Hakim Z, Pozzi S, et al:
Inflammatory activation of astrocytes facilitates melanoma brain
tropism via the CXCL10-CXCR3 signaling axis. Cell Rep.
28:1785–1798.e6. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Tsunemi T, Ishiguro Y, Yoroisaka A, Valdez
C, Miyamoto K, Ishikawa K, Saiki S, Akamatsu W, Hattori N and
Krainc D: Astrocytes protect human dopaminergic neurons from
alpha-synuclein accumulation and propagation. J Neurosci.
40:8618–8628. 2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Nielsen BS, Hansen DB, Ransom BR, Nielsen
MS and MacAulay N: Connexin hemichannels in astrocytes: An
assessment of controversies regarding their functional
characteristics. Neurochem Res. 42:2537–2550. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Pei H, Zhai C, Li H, Yan F, Qin J, Yuan H,
Zhang R, Wang S, Zhang W, Chang M, et al: Connexin 32 and connexin
43 are involved in lineage restriction of hepatic progenitor cells
to hepatocytes. Stem Cell Res Ther. 8(252)2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Biendarra-Tiegs SM, Clemens DJ, Secreto FJ
and Nelson TJ: Human induced pluripotent stem cell-derived
non-cardiomyocytes modulate cardiac electrophysiological maturation
through Connexin 43-mediated cell-cell interactions. Stem Cells
Dev. 29:75–89. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ma JW, Ji DD, Li QQ, Zhang T and Luo L:
Inhibition of connexin 43 attenuates oxidative stress and apoptosis
in human umbilical vein endothelial cells. BMC Pulm Med.
20(19)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang X, Feng L, Xin M, Hao Y, Wang X,
Shang P, Zhao M, Hou S, Zhang Y, Xiao Y, et al: Mechanisms
underlying astrocytic connexin-43 autophagy degradation during
cerebral ischemia injury and the effect on neuroinflammation and
cell apoptosis. Biomed Pharmacother. 127(110125)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Yin X, Feng L, Ma D, Yin P, Wang X, Hou S,
Hao Y, Zhang J, Xin M and Feng J: Roles of astrocytic connexin-43,
hemichannels, and gap junctions in oxygen-glucose
deprivation/reperfusion injury induced neuroinflammation and the
possible regulatory mechanisms of salvianolic acid B and
carbenoxolone. J Neuroinflammation. 15(97)2018.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Vignal N, Boulay AC, San C, Cohen-Salmon
M, Rizzo-Padoin N, Sarda-Mantel L, Declèves X, Cisternino S and
Hosten B: Astroglial Connexin 43 deficiency protects against
LPS-induced neuroinflammation: A TSPO Brain µPET Study with
[18F]FEPPA. Cells. 9(389)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Zhou F, Cao W, Xu R, Zhang J, Yu T, Xu X,
Zhi T, Yin J, Cao S, Liu N, et al: MicroRNA-206 attenuates glioma
cell proliferation, migration, and invasion by blocking the
WNT/β-catenin pathway via direct targeting of Frizzled 7 mRNA. Am J
Transl Res. 11:4584–4601. 2019.PubMed/NCBI
|
|
20
|
Jiao D, Chen J, Li Y, Tang X, Wang J, Xu
W, Song J, Li Y, Tao H and Chen Q: miR-1-3p and miR-206 sensitizes
HGF-induced gefitinib-resistant human lung cancer cells through
inhibition of c-Met signalling and EMT. J Cell Mol Med.
22:3526–3536. 2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Liu C, Li J, Wang W, Zhong X, Xu F and Lu
J: miR-206 inhibits liver cancer stem cell expansion by regulating
EGFR expression. Cell Cycle. 19:1077–1088. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Duan X, Zohaib A, Li Y, Zhu B, Ye J, Wan
S, Xu Q, Song Y, Chen H and Cao S: miR-206 modulates
lipopolysaccharide-mediated inflammatory cytokine production in
human astrocytes. Cell Signal. 27:61–68. 2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Liu TJ, Wang B, Li QX, Dong XL, Han XL and
Zhang SB: Effects of microRNA-206 and its target gene IGF-1 on
sevoflurane-induced activation of hippocampal astrocytes in aged
rats through the PI3K/AKT/CREB signaling pathway. J Cell Physiol.
233:4294–4306. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li H, Xiang Y, Fan LJ, Zhang XY, Li JP, Yu
CX, Bao LY, Cao DS, Xing WB, Liao XH, et al: Myocardin inhibited
the gap protein connexin 43 via promoted miR-206 to regulate
vascular smooth muscle cell phenotypic switch. Gene. 616:22–30.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Chen W, Tong W, Guo Y, He B, Chen L, Yang
W, Wu C, Ren D, Zheng P and Feng J: Up-regulation of Connexin-43 is
critical for irradiation-induced neuroinflammation. CNS Neurol
Disord Drug Targets. 17:539–546. 2018.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-ΔΔC(T)) method. Methods. 25:402–408. 2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Nutma E, van Gent D, Amor S and Peferoen
LAN: Astrocyte and oligodendrocyte cross-talk in the central
nervous system. Cells. 9(600)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Sofroniew MV and Vinters HV: Astrocytes:
Biology and pathology. Acta Neuropathol. 119:7–35. 2010.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Pawlowski J and Jones WE III: Radiation
Therapy For Anal Cancer. StatPearls Publishing, Treasure Island,
FL, 2021.
|
|
30
|
Wormhoudt TL, Boss MK, Lunn K, Griffin L,
Leary D, Dowers K, Rao S and LaRue SM: Stereotactic radiation
therapy for the treatment of functional pituitary adenomas
associated with feline acromegaly. J Vet Intern Med. 32:1383–1391.
2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chang Y: The effect of far infrared
radiation therapy on inflammation regulation in
lipopolysaccharide-induced peritonitis in mice. SAGE Open Med: Sep
10, 2018 (Epub ahead of print). doi: 10.1177/2050312118798941.
|
|
32
|
Li S, Zhou C, Zhu Y, Chao Z, Sheng Z,
Zhang Y and Zhao Y: Ferrostatin-1 alleviates angiotensin II (Ang
II)- induced inflammation and ferroptosis in astrocytes. Int
Immunopharmacol. 90(107179)2021.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Sun JB, Li Y, Cai YF, Huang Y, Liu S,
Yeung PK, Deng MZ, Sun GS, Zilundu PL, Hu QS, et al: Scutellarin
protects oxygen/glucose-deprived astrocytes and reduces focal
cerebral ischemic injury. Neural Regen Res. 13:1396–1407.
2018.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Ishihara Y, Itoh K, Oguro A, Chiba Y, Ueno
M, Tsuji M, Vogel CF and Yamazaki T: Neuroprotective activation of
astrocytes by methylmercury exposure in the inferior colliculus.
Sci Rep. 9(13899)2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Cabezas R, Baez-Jurado E, Hidalgo-Lanussa
O, Echeverria V, Ashrad GM, Sahebkar A and Barreto GE: Growth
factors and neuroglobin in astrocyte protection against
neurodegeneration and oxidative stress. Mol Neurobiol.
56:2339–2351. 2019.PubMed/NCBI View Article : Google Scholar
|
|
36
|
McBean GJ: Astrocyte antioxidant systems.
Antioxidants (Basel). 7(112)2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Karki P, Smith K, Johnson J Jr and Lee E:
Astrocyte-derived growth factors and estrogen neuroprotection: Role
of transforming growth factor-α in estrogen-induced upregulation of
glutamate transporters in astrocytes. Mol Cell Endocrinol.
389:58–64. 2014.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Nalewajska M, Marchelek-Myśliwiec M,
Opara-Bajerowicz M, Dziedziejko V and Pawlik A:
Connexins-therapeutic targets in cancers. Int J Mol Sci.
21(E9119)2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Li T, Niu J, Yu G, Ezan P, Yi C, Wang X,
Koulakoff A, Gao X, Chen X, Sáez JC, et al: Connexin 43 deletion in
astrocytes promotes CNS remyelination by modulating local
inflammation. Glia. 68:1201–1212. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Sanchez MC, Benitez A, Ortloff L and Green
LM: Alterations in glutamate uptake in NT2-derived neurons and
astrocytes after exposure to gamma radiation. Radiat Res.
171:41–52. 2009.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Yang H, Yan H, Li X, Liu J, Cao S, Huang
B, Huang D and Wu L: Inhibition of Connexin 43 and phosphorylated
NR2B in spinal astrocytes attenuates bone cancer pain in mice.
Front Cell Neurosci. 12(129)2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Bylicky MA, Mueller GP and Day RM:
Radiation resistance of normal human astrocytes: The role of
non-homologous end joining DNA repair activity. J Radiat Res
(Tokyo). 60:37–50. 2019.PubMed/NCBI View Article : Google Scholar
|