Introduction
Numerous newborn babies are required to receive
anesthesia for diagnostic or surgical purposes (1). The application of anesthetics has been
suggested to be harmful to brain function, leading to impairments
of learning and cognitive functions (2,3).
Isoflurane is one of the most commonly used inhaled anesthetics,
and has been reported to contribute to long-term memory deficits
(4). Anesthesia-associated neuronal
apoptosis is considered to be an important mechanism involved in
the neurological impairment induced by anesthesia (5,6).
Notably, exposure of the developing brain to isoflurane can cause
severe damage to neurological functions, which may result in
persistent learning deficits and cognitive impairment (3,7).
Previous studies performed in developing rodent models have
demonstrated that almost all anesthetics, including isoflurane,
induce widespread neuronal cell death followed by long-term memory
and learning disabilities (8,9). These
findings have raised serious concerns about the safety of
anesthetic use in pregnant women and young children. However, the
underlying mechanisms of anesthesia-induced learning and cognitive
dysfunction remain to be clarified.
MicroRNAs (miRNAs) are a class of short non-coding
RNA molecules, which function as negative regulators of target
genes by binding to their 3'-untranslated regions. miRNAs have been
widely suggested to play important roles in various biological
processes, including cell proliferation, apoptosis and
differentiation, and neuronal inflammation (10,11).
The abnormal expression of miRNAs has been reported to have a
regulatory effect on learning and memory function (12,13).
For example, in one study, miR-132 expression was shown to be
downregulated in the hippocampus of elderly mice compared with
younger mice, and in vivo experiments suggested that
overexpression of miR-132 may reverse the decline in learning and
memory (12). Furthermore, a number
of miRNAs have been identified as being involved in the regulation
of anesthesia-induced cognitive impairment, including miR-24,
miR-124 and miR-214 (3,14,15).
The role of miR-133b, a muscle-specific miRNA, in neuronal
development and dysfunction has also been investigated (16,17).
In a rat model of depression, miR-133b was found to be expressed at
low levels in hippocampal tissues, and the overexpression of
miR-133b inhibited the apoptosis of hippocampal neurons (16). In addition, a study by Takeuchi
et al (18) reported that
miR-133b was downregulated in the plasma of
sevoflurane-anesthetized rats. Therefore, the potential involvement
of miR-133b in the underlying mechanism of isoflurane-induced
learning and memory impairment is a subject of interest.
The present study aimed to investigate changes in
the expression of miR-133b in isoflurane-treated rats.
Additionally, the role and potential mechanism of miR-133b in
isoflurane-induced learning and memory impairment were further
explored.
Materials and methods
Experimental animals and grouping
A total of 80 male or female Sprague-Dawley (SD) rat
pups (age, 7 days; weight, 12-15 g) were purchased from Shanghai
Laboratory Animal Center of the Chinese Academy of Sciences. All
rats were raised in standard conditions, under 50-60% atmospheric
humidity, on a 12-h light/dark cycle, at a room temperature of
24-26˚C and with free access to food and water. All procedures
conducted in this study were approved by the Ethics Committee of
the Experimental Animal Center of Jining No. 1 People's
Hospital.
The rats were randomly divided into four groups,
each comprising 20 rats, as follows: i) Control group, treated with
regular air; ii) isoflurane group, anesthetized with 1.5%
isoflurane for 6 h; iii) isoflurane + agomir NC group, treated with
2 nmol agomir NC (sequence: 5'-UUCUCCGAACGUGUCACGUTT-3'; Guangzhou
RiboBio Co., Ltd.) by lateral cerebroventricular injection and
anesthetized 30 min later with 1.5% isoflurane for 6 h; iv)
isoflurane + miR-agomir group, treated with 2 nmol miR-133b agomir
(sequence: 5'-UUUGGUCCCCUUCAACCAGCUA-3'; Guangzhou RiboBio Co.,
Ltd.) by lateral cerebroventricular injection and anesthetized 30
min later with 1.5% isoflurane for 6 h. Following the air or
isoflurane treatment, 10 rats from each group were euthanized by
cervical dislocation in accordance with the procedure approved by
the Ethics Committee of the Experimental Animal Center of Jining
No. 1 People's Hospital in accordance with AVMA Guidelines, 2020
edition (19) and the hippocampal
tissues were collected for RNA extraction. The remaining 10 rats in
each group were used for subsequent Morris water maze (MWM)
tests.
MWM tests
One week after the various treatments were
administered, rats (postnatal day 14) were trained for a MWM test
to assess spatial learning and memory ability. The MWM test was
performed on 5 consecutive days. The swimming route was observed
using VideoMot software version 2.4.50923 (TSE Systems GmbH). The
time taken for the rats to locate a submerged platform (latency,
using a cut-off time point of 120 sec) and the swimming speed were
recorded on the first 4 days. On day 5, a probe trial was performed
without the platform. The escape latency, meaning the time taken
for the rat to swim to the former location of the platform, was
recorded. The percentage of time that each rat spent in the
quadrant that previously contained the platform in a 120-sec period
was determined.
Cell culture and transfection
Hippocampal cells were prepared as described in a
previous study (20). Three newborn
rats (0-24 h old) were purchased from Shanghai Laboratory Animal
Center of the Chinese Academy of Sciences. Hippocampal tissues were
collected from the newborn rats within 24 h of birth. Hippocampal
neuronal cells were cultured in Neurobasal™ Medium supplemented
with 2% B27, 0.3% glucose, 1 mM glutamine, and 5% FBS (Invitrogen;
Thermo Fisher Scientific, Inc.) in a humidified incubator with 5%
CO2 at 37˚C. The cells were seeded in 96-well plates at
a density of 1x105 cells/well and cultured for 24 h.
miR-133b mimic (50 nM; sequence: 5'-UUUGGUCCCCUUCAACCAGCUA-3') and
negative control (50 nM; miR-NC, sequence:
5'-UUCUCCGAACGUGUCACGUTT-3') were provided by RiboBio Co., Ltd.
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) was used for cell transfection according to the
manufacturer's protocols. In addition to control group, cells in
other groups were cultured in a 37°C incubator with 5%
CO2 and 1.5% isoflurane for 6 h, then transfected with
miR-mimic or miR-NC at 37˚C. At 24 h post transfection, cells in
different groups were collected for futher experiment.
The cells were divided into four groups as follows:
i) Negative control group (control), untreated cells; ii)
isoflurane group, cells treated with 1.5% isoflurane for 6 h; iii)
isoflurane + miR-NC group, cells treated with 1.5% isoflurane for 6
h and transfected with miR-NC; iv) isoflurane + miR-mimic group,
cells treated with 1.5% isoflurane for 6 h and transfected with
miR-133b mimic.
RNA extraction and reverse
transcription quantitative polymerase chain reaction (RT-qPCR)
Total RNA was extracted from hippocampal tissues and
cells using TRIzol® reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) according to the manufacturer's protocol. The
miScript Reverse Transcription Kit (Qiagen GmbH) was used to
transcribe the total RNA into cDNA. The reverse transcription
conditions were as follows: 37˚C for 15 min, followed by 5 sec at
85˚C for RT inactivation. Thereafter, qPCR was conducted using a
SYBR Green Master Mix kit (Invitrogen; Thermo Fisher Scientific,
Inc.) to detect the expression level of miR-133b. The following
thermocycling conditions were used for the PCR: Initial
denaturation at 95˚C for 5 min; 30 cycles of 95˚C for 30 sec, 60˚C
for 30 sec and 72˚C for 20 sec; and a final extension at 72˚C for
10 min. The relative expression of miR-133b was evaluated using the
2-∆∆Cq method with U6 as the reference gene (21). The primer sequences were as follows:
miR-133b forward, 5'-AAAGGACCCCAACAACCAGCAA-3 and reverse,
5'-TTGCTGGTTGTTGGGGTCCTTT-3'; and U6 forward,
5'-CTCGCTTCGGCAGCACATATACT-3 and reverse,
5'-ACGCTTCACGAATTTGCGTGTC-3.
Cell viability
Cell viability was assessed using a Cell Counting
Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc.) assay. Cells
from the different groups were inoculated into 96-well plates at a
density of 5x104 cells per well and cultured
continuously for 3 days. At 0, 24, 48 and 72 h after the initiation
of incubation, 10 ml CCK-8 reagent was added to each well and
incubated for 2 h. The absorbance of each well was then measured at
450 nm.
Flow cytometric assay
A FITC Annexin V Apoptosis Detection kit (BD
Biosciences) was used to measure cell apoptosis. Cells
(5x105) were collected from each group, fixed with 3.7%
formaldehyde for 15 min at room temperature and then permeabilized
with 0.1% Triton X-100 for 5 min at 37˚C. After washing, the cells
were mixed with 5 µl Annexin V-FITC and propidium iodide, and
incubated at room temperature for 10 min. The apoptotic rates of
the cells were detected and recorded using a FACScan™ flow
cytometer in combination with CellQuest Pro v5.1.1 software (BD
Biosciences).
Statistical analysis
GraphPad Prism 5.0 software (GraphPad Software,
Inc.) was used for data analysis. Differences between two groups
were compared using the Student's t-test, while differences among
multiple groups were evaluated using one-way analysis of variance
followed by Tukey's post hoc test. P<0.05 was considered to
indicate a statistically significant difference.
Results
Effects of isoflurane on learning and
memory in rats
MWM tests were conducted to determine whether
isoflurane affects the learning and memory functions of neonatal
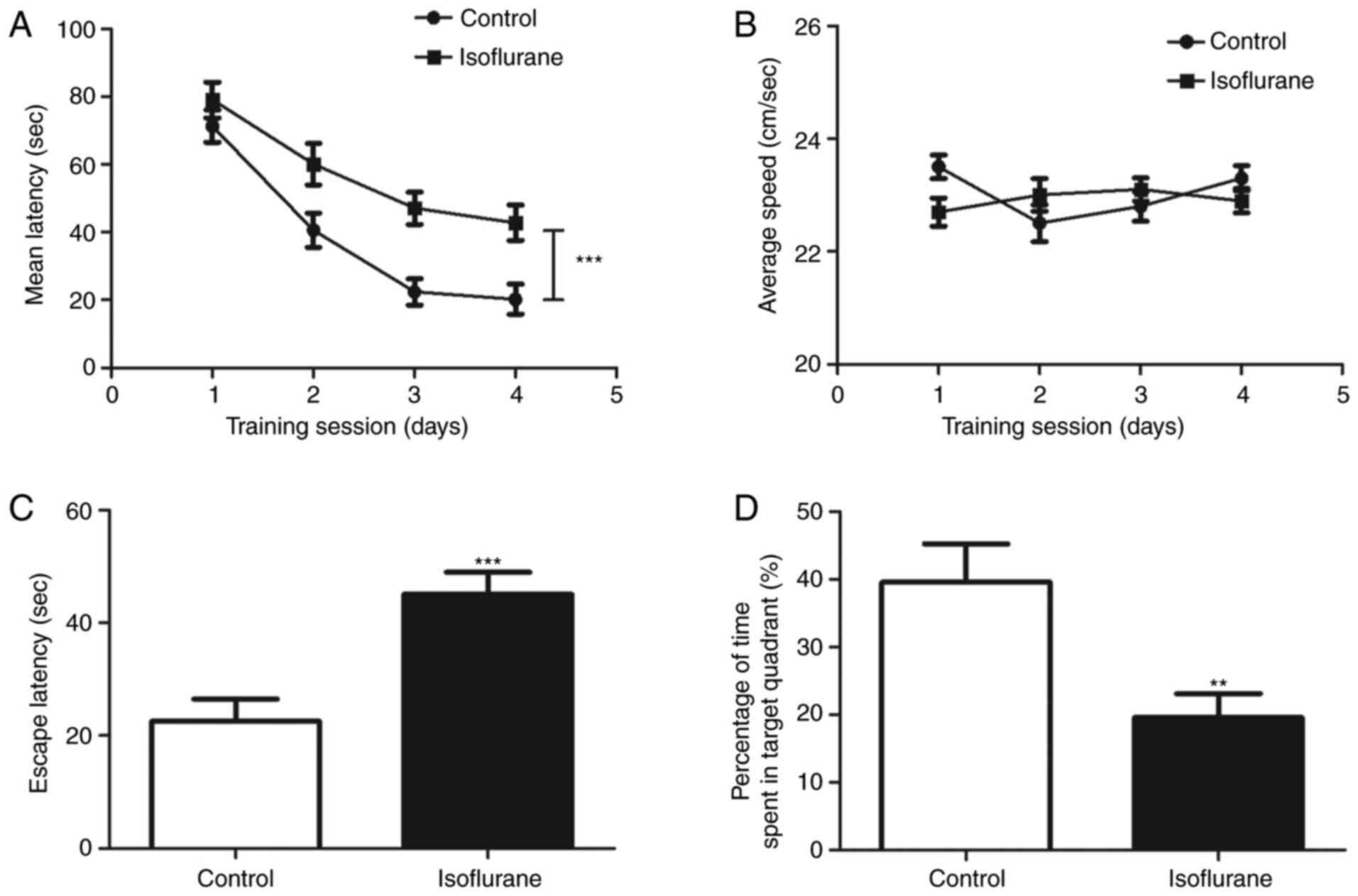
rats. As shown in Fig. 1A, during
the training session, the time taken for the isoflurane-treated
rats to locate the platform was significantly increased compared
with that of the control rats (P<0.001). However, no significant
difference in swimming speed was observed between the
isoflurane-treated and control rats (P>0.05, Fig. 1B). A probe trial was then performed
to assess the effect of isoflurane on memory. It was found that the
rats treated with isoflurane exhibited a significantly higher
escape latency than the control group (P<0.001; Fig. 1C). Additionally, the rats in the
isoflurane group spent significantly less time than the control
rats in the quadrant that previously contained the platform
(P<0.01; Fig. 1D). These results
indicate that isoflurane exposure affected the learning and memory
functions of the rats, and the isoflurane-induced learning and
memory impaired rat model was established successfully.
miR-133b expression is reduced in rats
anesthetized with isoflurane
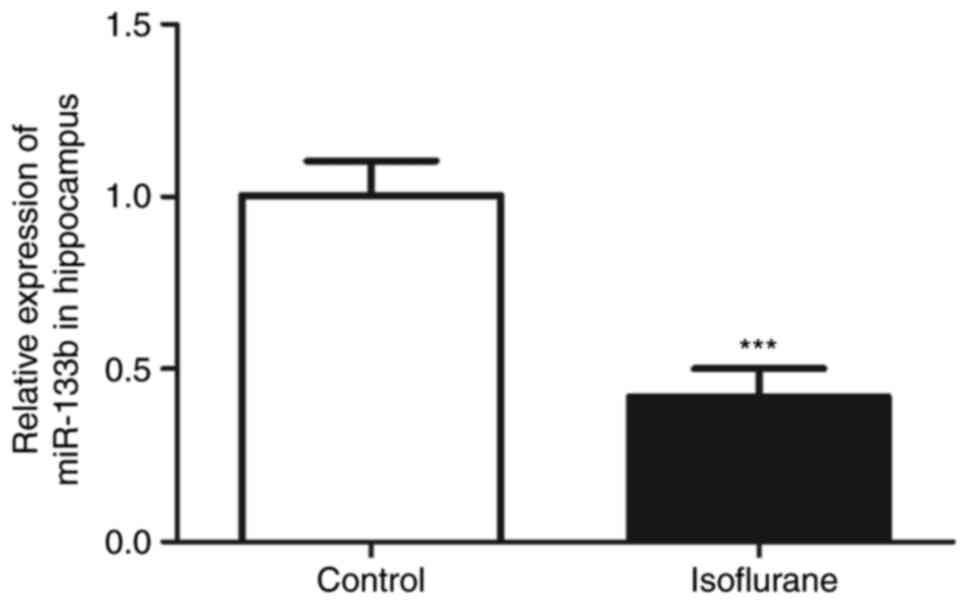
RT-qPCR was used to measure the hippocampal
expression levels of miR-133b in the isoflurane-treated and control
rats. The hippocampal expression of miR-133b in the rats treated
with isoflurane was significantly decreased compared with that of
the control rats (Fig. 2;
P<0.001).
Effect of miR-133b on
isoflurane-induced learning and memory impairment in rats
To further investigate the role of miR-133b in
isoflurane-induced learning and memory impairment in vivo,
the expression level of miR-133b was regulated by the injection of
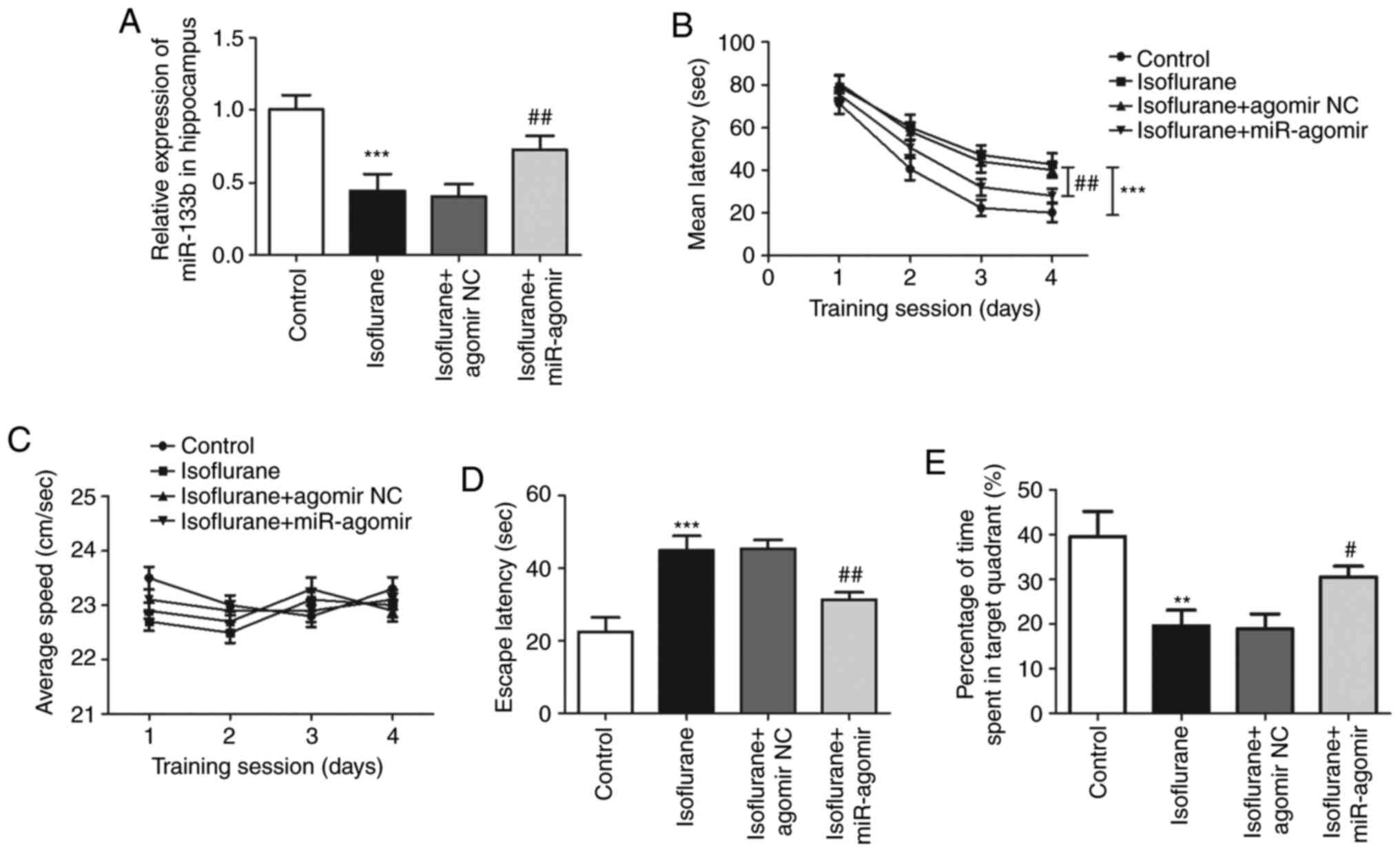
miR-133b agomir. As shown in Fig.
3A, RT-qPCR analysis demonstrated that the injection of
miR-133b agomir significantly increased the expression of miR-133b,
counteracting the reduction of miR-133b expression induced by
isoflurane treatment (P<0.01; Fig.
3A). In the MWM test, during the training session, the latency
time for location of the submerged platform was significantly
increased by isoflurane treatment (P<0.001), and this increase
was significantly attenuated by the overexpression of miR-133b
(P<0.01, Fig. 3B). The swimming
speeds of the isoflurane-treated rats during the training session
were not observed to differ significantly according to whether or
not miR-133b agomir was administered (P>0.0.05, Fig. 3C). Additionally, the probe trial
test results indicated that the overexpression of miR-133b reversed
the effect of isoflurane on the escape latency (P<0.01) and time
spent in the target quadrant (P<0.05) (Fig. 3D and E). These data suggest that the
overexpression of miR-133b attenuated the isoflurane-induced
learning and memory impairment of the rats.
Effect of miR-133b on hippocampal
neuron viability and apoptosis
Rat hippocampal neurons were used in vitro to
further explore the effect of miR-133b on neuron viability and
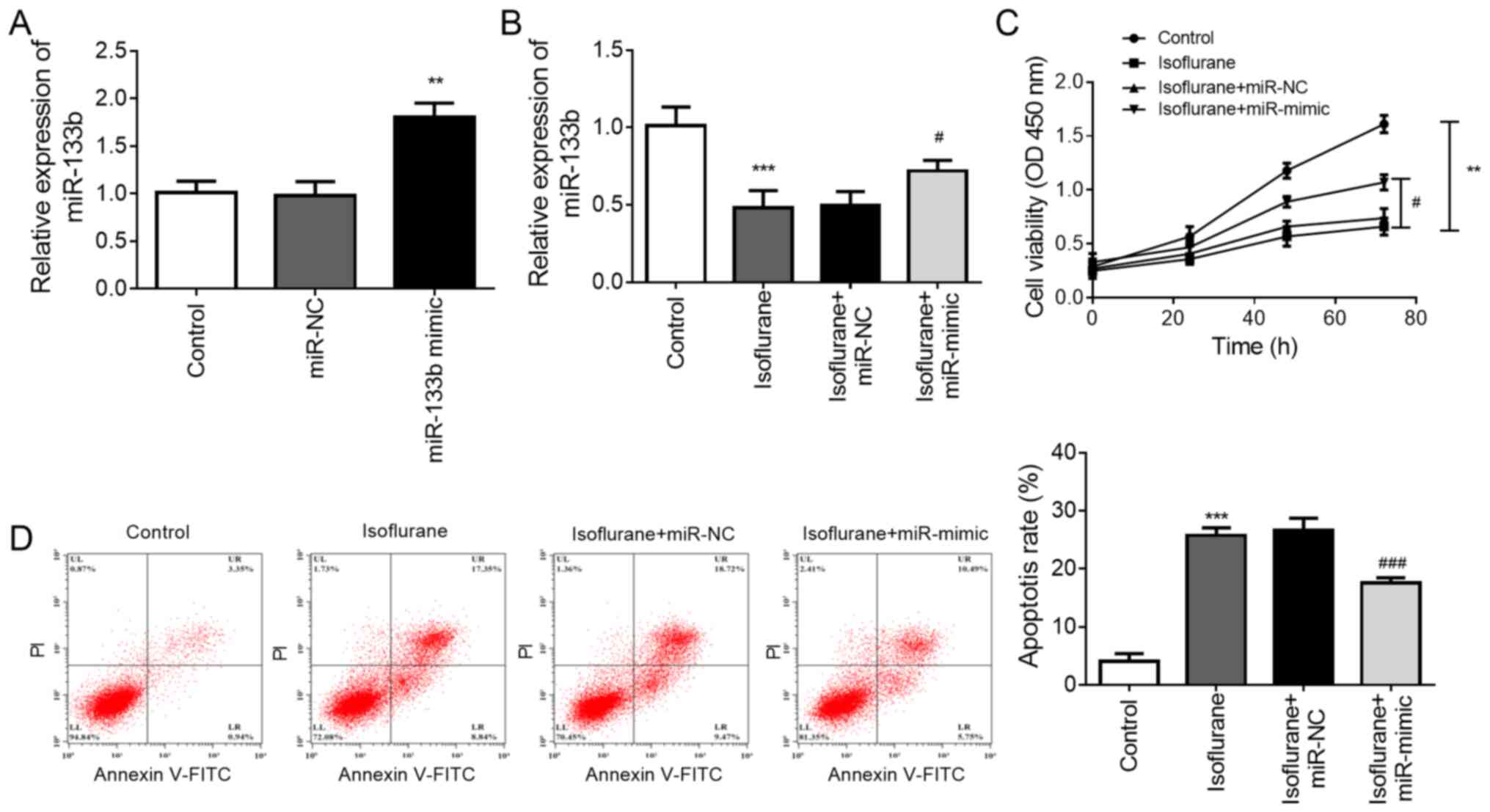
apoptosis. The expression of miR-133b was regulated by
transfection, and it was found that transfection of the neurons
with miR-133b mimic significantly increased the level of miR-133b
compared with that in the control group (P<0.01, Fig. 4A). RT-qPCR results also revealed
that treatment with isoflurane significantly reduced the expression
level of miR-133b in neurons in vitro (P<0.001), and this
reduction was attenuated by transfection with miR-133b mimic
(P<0.05, Fig. 4B). The results
of the CCK-8 assay indicated that isoflurane treatment reduced the
viability of the neurons (P<0.01), and this reduced viability
was significantly attenuated by the upregulation of miR-133b
(P<0.05, Fig. 4C). Additionally,
flow cytometric analysis demonstrated that isoflurane induced
neuronal apoptosis (P<0.001), and the overexpression of miR-133b
significantly attenuated the isoflurane-induced apoptosis
(P<0.001, Fig. 4D).
Discussion
Isoflurane is a widely used anesthetic in pediatric
and obstetric patients; millions of infants and children are
exposed to general anesthetic agents during surgical or treatment
procedures, which may influence their neuronal functions and brain
development (22,23). Therefore, interest in the biological
mechanisms underlying anesthesia-induced learning and cognitive
dysfunction has been stimulated. A number of in vivo studies
have suggested that isoflurane leads to spatial learning and memory
impairment, and causes cognitive dysfunction (24-26).
In order to investigate this, the present study constructed animal
models of isoflurane exposure using neonatal SD rats. The results
of MWM tests indicated that isoflurane exposure influenced the
learning ability of the rats, which was reflected by an increase in
the time required to locate a submerged platform during the
training session. Additionally, probe trial test results indicated
that isoflurane exposure increased the escape latency of the
neonatal rats, and reduced their time spent in the area where the
platform was previously located. These data suggest that exposure
to isoflurane influenced the learning and memory function of the
rats, which is consistent with previous studies (24,25,26)
and indicates that the rat model of isoflurane-induced learning and
memory impairment was established successfully.
miR-133b is a muscle-specific miRNA that exhibits an
anesthesia-regulated expression pattern and participates in various
pathological processes (18,27).
In a study of the sevoflurane-associated dynamics of circulating
miRNAs, the plasma levels of miR-133b were found to be
downregulated in sevoflurane-anesthetized rats, which is consistent
with the findings of the present study (18). In the present study, the results
indicated that in the rat model of isoflurane exposure, miR-133b
was expressed at lower levels in the hippocampal tissue of rats
exposed to isoflurane than in those of control rats, which is
supported by previous studies (18,27).
These results suggest that miR-133b potentially serves a role in
isoflurane-induced learning and memory impairment.
To further elucidate the role of miR-133b in
isoflurane-induced learning and memory impairment in vivo,
the expression level of miR-133b was modified by the injection of
miR-133b agomir. The MWM test results indicated that the
overexpression of miR-133b attenuated the isoflurane-induced
learning and memory impairment of the rats. The present results are
supported by previous studies that have focused on the role of
miR-133b in neuronal development and neurological disease (17,28).
In a study of spinal cord regeneration, the overexpression of
miR-133b was demonstrated to promote neurite outgrowth by targeting
RhoA (29). Another study suggested
that miR-133b promoted neural plasticity and functional recovery in
a mouse model of stroke (30). All
this evidence supports the present results indicating that miR-133b
may be involved in the regulation of isoflurane-induced learning
and memory impairment.
Isoflurane is considered to influence neuronal cell
viability and apoptosis, which are involved in the regulation of
learning and cognitive functions (26,31).
To gain further insight into the role of miR-133b in
isoflurane-induced neuronal injury, a cell model of isoflurane
exposure was established using primary hippocampal neurons and the
expression of miR-133b was regulated by transfection. It was
observed that the overexpression of miR-133b attenuated the effect
of isoflurane on neuronal cell viability and apoptosis. Consistent
with these findings, the neuroprotective effect of miR-133b has
been demonstrated in a number of previous studies. For example, in
a rat model of depression, miR-133b expression was identified to be
downregulated, and the elevation of miR-133b expression inhibited
the apoptosis of hippocampal neurons, thereby providing a
protective effect against neural injury in depressed rats (16). Another study, concerning the
pathogenesis of Parkinson's disease (PD), indicated that the
expression of miR-133b was decreased in the brain tissues of a
mouse model of PD, and was involved in the mechanism by which long
non-coding RNA SNHG14 contributes to dopaminergic neuron injury
(17). However, the pathological
changes and viability of cells in rat brain tissues were not
investigated in the present study, which is a limitation of the
study. In future studies, such indicators should be analyzed using
in vivo experiments to better reflect the effect of miR-133b
on neuronal cell viability.
In a previous study on the role of isoflurane in
dopaminergic neurons, isoflurane was demonstrated to inhibit
synaptic transmission in rat midbrain dopaminergic (mDA) neurons
(32). mDA neurons have been
reported to be involved in the regulation of learning and memory
function (33,34). Sanchez-Simon et al (35) used zebrafish embryos as a model to
investigate the role of morphine in neuronal development, and found
that miR-133b expression was downregulated by morphine treatment,
demonstrating that miR-133b has a regulatory role in neuronal
development, mediated via the regulation of dopaminergic neuron
differentiation. In the present study, low expression levels of
miR-133b were detected in the hippocampal tissues of rats treated
with isoflurane, and the in vivo experiments suggested that
the overexpression of miR-133b attenuated isoflurane-induced
learning and memory impairment in rats. Considering the crucial
role of mDA neurons in learning and memory function, we hypothesize
that miR-133b may participate in isoflurane-induced neurological
impairment via the regulation of mDA neurons. However, further
studies are required to explore the exact mechanism by which
miR-133b participates in isoflurane-induced learning and memory
impairment. Additionally, the present results in developing rats
raise questions about the role of miR-133b in isoflurane-induced
cognitive impairment in aged rats, which is an important topic for
further exploration.
In conclusion, the results of the present study
indicate that the overexpression of miR-133b attenuated
isoflurane-induced learning and memory impairment in rats. During
exposure to isoflurane in vitro, the viability and apoptosis
resistance of hippocampal neurons were promoted by the
overexpression of miR-133b. These data provide a theoretical basis
for the prevention and treatment of the neurological deficits
induced by isoflurane exposure.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
YZ, JL, CX and PW participated in the design and
interpretation of the study, analysis of the data and review of the
manuscript. YZ and JL conducted the experiments. PW wrote the
manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to
participate
All procedures of this study were approved by the
Ethics Committee of the Experimental Animal Center of Jining No. 1
People's Hospital.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
McCann ME and Soriano SG: Progress in
anesthesia and management of the newborn surgical patient. Semin
Pediatr Surg. 23:244–248. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Tojo A, Uchimoto K, Inagawa G and Goto T:
Desflurane impairs hippocampal learning on day 1 of exposure: a
prospective laboratory study in rats. BMC Anesthesiol.
19(119)2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Li N, Yue L, Wang J, Wan Z and Bu W:
MicroRNA-24 alleviates isoflurane-induced neurotoxicity in rat
hippocampus via attenuation of oxidative stress. Biochem Cell Biol.
98:208–218. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Belrose JC and Noppens RR: Anesthesiology
and cognitive impairment: A narrative review of current clinical
literature. BMC Anesthesiol. 19(241)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Wang L, Zheng M, Wu S and Niu Z:
MicroRNA-188-3p is involved in sevoflurane anesthesia-induced
neuroapoptosis by targeting MDM2. Mol Med Rep. 17:4229–4236.
2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Yong J, Yan L, Wang J, Xiao H and Zeng Q:
Effects of compound 21, a nonpeptide angiotensin II type 2 receptor
agonist, on general anesthesiainduced cerebral injury in neonatal
rats. Mol Med Rep. 18:5337–5344. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Lu J, Zhou N, Yang P, Deng L and Liu G:
MicroRNA-27a-3p downregulation inhibits inflammatory response and
hippocampal neuronal cell apoptosis by upregulating
Mitogen-Activated Protein Kinase 4 (MAP2K4) expression in epilepsy:
In vivo and in vitro studies. Med Sci Monit. 25:8499–8508.
2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Twaroski D, Bosnjak ZJ and Bai X:
MicroRNAs: New players in anesthetic-induced developmental
neurotoxicity. Pharm Anal Acta. 6(357)2015.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kang E, Jiang D, Ryu YK, Lim S, Kwak M,
Gray CD, Xu M, Choi JH, Junn S, Kim J, et al: Early postnatal
exposure to isoflurane causes cognitive deficits and disrupts
development of newborn hippocampal neurons via activation of the
mTOR pathway. PLoS Biol. 15(e2001246)2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bao F, Kang X, Xie Q and Wu J: HIF-α/PKM2
and PI3K-AKT pathways involved in the protection by dexmedetomidine
against isoflurane or bupivacaine-induced apoptosis in hippocampal
neuronal HT22 cells. Exp Ther Med. 17:63–70. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Duan Q, Sun W, Yuan H and Mu X:
MicroRNA-135b-5p prevents oxygen-glucose deprivation and
reoxygenation-induced neuronal injury through regulation of the
GSK-3beta/Nrf2/ARE signaling pathway. Arch Med Sci. 14:735–744.
2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Liu G, Yin F, Zhang C, Zhang Y, Li X and
Ling Y: Effects of regulating miR-132 mediated GSK-3beta on
learning and memory function in mice. Exp Ther Med. 20:1191–1197.
2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Sun H, Hu H, Xu X, Tao T and Liang Z: Key
miRNAs associated with memory and learning disorder upon exposure
to sevoflurane determined by RNA sequencing. Mol Med Rep.
22:1567–1575. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yang W, Guo Q, Li J, Wang X, Pan B, Wang
Y, Wu L, Yan J and Cheng Z: microRNA-124 attenuates
isoflurane-induced neurological deficits in neonatal rats via
binding to EGR1. J Cell Physiol. 234:23017–23032. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yan H, Xu T, Zhao H, Lee KC, Wang HY and
Zhang Y: Isoflurane increases neuronal cell death vulnerability by
downregulating miR-214. PLoS One. 8(e55276)2013.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Pei G, Xu L, Huang W and Yin J: The
protective role of microRNA-133b in restricting hippocampal neurons
apoptosis and inflammatory injury in rats with depression by
suppressing CTGF. Int Immunopharmacol. 78(106076)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Zhang LM, Wang MH, Yang HC, Tian T, Sun
GF, Ji YF, Hu WT, Liu X, Wang JP and Lu H: Dopaminergic neuron
injury in Parkinson's disease is mitigated by interfering lncRNA
SNHG14 expression to regulate the miR-133b/ alpha-synuclein
pathway. Aging (Albany NY). 11:9264–9279. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Takeuchi J, Sakamoto A and Takizawa T:
Sevoflurane anesthesia persistently downregulates muscle-specific
microRNAs in rat plasma. Int J Mol Med. 34:291–298. 2014.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Bailey RI, Cheng HH, Chase-Topping M, Mays
JK, Anacleto O, Dunn JR and Doeschl-Wilson A: Pathogen transmission
from vaccinated hosts can cause dose-dependent reduction in
virulence. PLoS Biol. 18(e3000619)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Nunez J: Primary culture of hippocampal
neurons from P0 newborn rats. J Vis Exp. 29(895)2008.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Xu L, Shen J, Yu L, Sun J, McQuillan PM,
Hu Z and Yan M: Role of autophagy in sevoflurane-induced
neurotoxicity in neonatal rat hippocampal cells. Brain Res Bull.
140:291–298. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Sun LS, Li G, Dimaggio C, Byrne M, Rauh V,
Brooks-Gunn J, Kakavouli A and Wood A: Coinvestigators of the
Pediatric Anesthesia Neurodevelopment Assessment (PANDA) Research
Network. Anesthesia and neurodevelopment in children: Time for an
answer? Anesthesiology. 109:757–761. 2008.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li C, Wang Q, Li L, Liu Y and Diao H:
Arachidonic acid attenuates learning and memory dysfunction induced
by repeated isoflurane anesthesia in rats. Int J Clin Exp Med.
8:12365–12373. 2015.PubMed/NCBI
|
|
25
|
Wang H, Xu Z, Feng C, Wang Y, Jia X, Wu A
and Yue Y: Changes of learning and memory in aged rats after
isoflurane inhalational anaesthesia correlated with hippocampal
acetylcholine level. Ann Fr Anesth Reanim. 31:e61–66.
2012.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Pang X, Zhang P, Zhou Y, Zhao J and Liu H:
Dexmedetomidine pretreatment attenuates isoflurane-induced
neurotoxicity via inhibiting the TLR2/NF-κB signaling pathway in
neonatal rats. Exp Mol Pathol. 112(104328)2019.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Werner W, Sallmon H, Leder A, Lippert S,
Reutzel-Selke A, Morgul MH, Jonas S, Dame C, Neuhaus P, Iacomini J,
et al: Independent effects of sham laparotomy and anesthesia on
hepatic microRNA expression in rats. BMC Res Notes.
7(702)2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Zhou Y, Zhu J, Lv Y, Song C, Ding J, Xiao
M, Lu M and Hu G: Kir6.2 deficiency promotes mesencephalic neural
precursor cell differentiation via regulating miR-133b/GDNF in a
parkinson's disease mouse model. Mol Neurobiol. 55:8550–8562.
2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Lu XC, Zheng JY, Tang LJ, Huang BS, Li K,
Tao Y, Yu W, Zhu RL, Li S and Li LX: miR-133b Promotes neurite
outgrowth by targeting RhoA expression. Cell Physiol Biochem.
35:246–258. 2015.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Xin H, Li Y, Liu Z, Wang X, Shang X, Cui
Y, Zhang ZG and Chopp M: miR-133b promotes neural plasticity and
functional recovery after treatment of stroke with multipotent
mesenchymal stromal cells in rats via transfer of exosome-enriched
extracellular particles. Stem Cells. 31:2737–2746. 2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Guo M, Zhu X, Xu H, Li J, Yang S, Zuo Z
and Lin D: Ulinastatin attenuates isoflurane-induced cognitive
dysfunction in aged rats by inhibiting neuroinflammation and
β-amyloid peptide expression in the brain. Neurol Res. 41:923–929.
2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Torturo CL, Zhou ZY, Ryan TA and Hemmings
HC: Isoflurane inhibits dopaminergic synaptic vesicle exocytosis
coupled to CaV2.1 and CaV2.2 in rat midbrain neurons. eNeuro.
6(ENEURO.0278-18.2018)2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Coddington LT and Dudman JT: Learning from
action: Reconsidering movement signaling in midbrain dopamine
neuron activity. Neuron. 104:63–77. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Heyer MP, Pani AK, Smeyne RJ, Kenny PJ and
Feng G: Normal midbrain dopaminergic neuron development and
function in miR-133b mutant mice. J Neurosci. 32:10887–10894.
2012.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Sanchez-Simon FM, Zhang XX, Loh HH, Law PY
and Rodriguez RE: Morphine regulates dopaminergic neuron
differentiation via miR-133b. Mol Pharmacol. 78:935–942.
2010.PubMed/NCBI View Article : Google Scholar
|