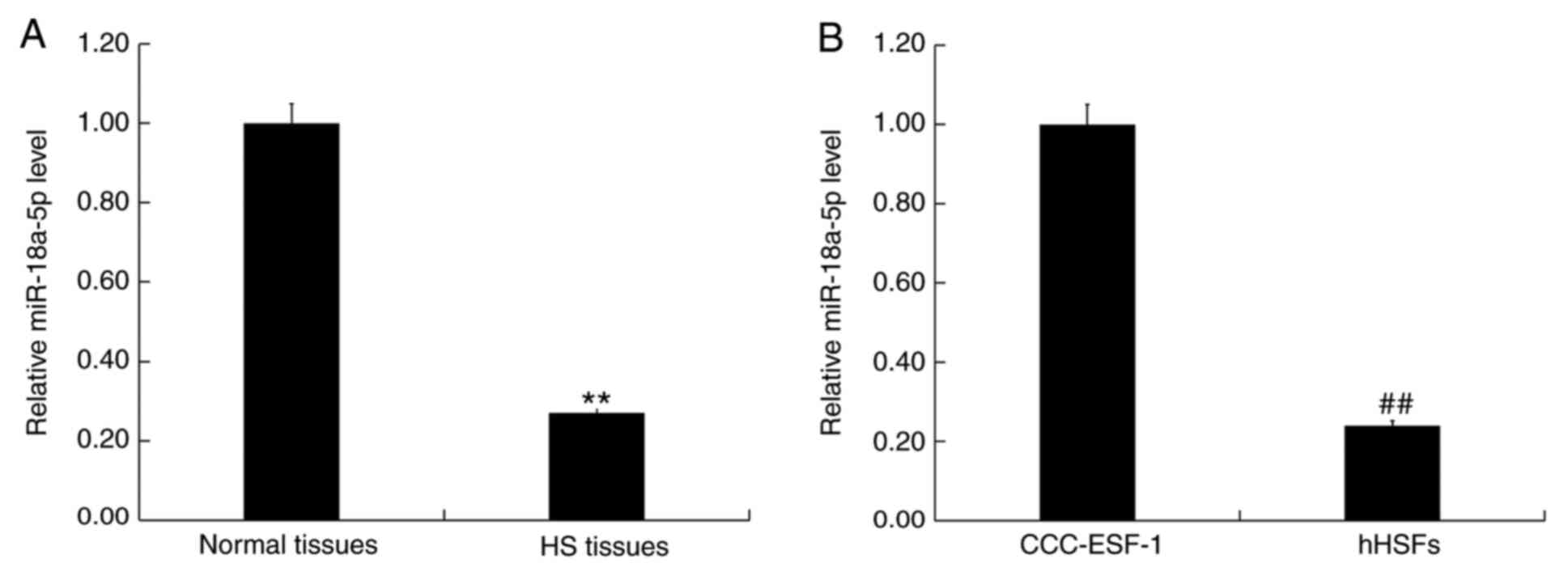
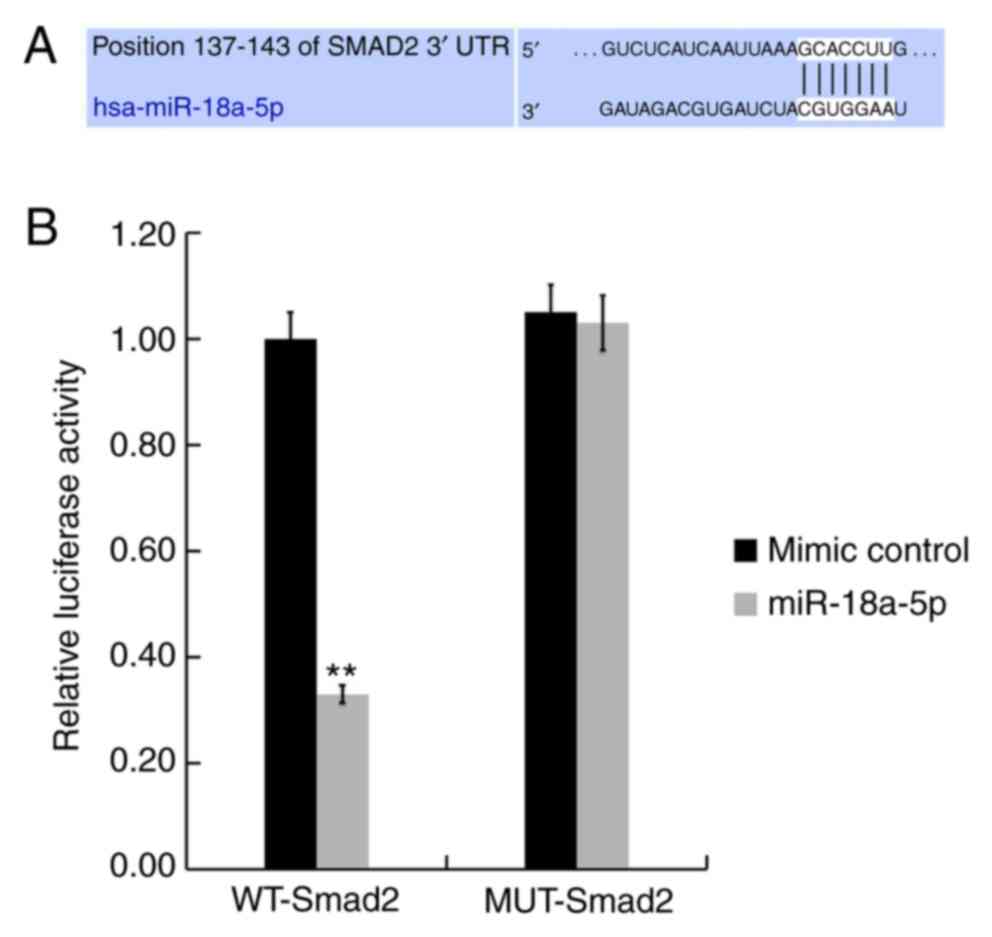
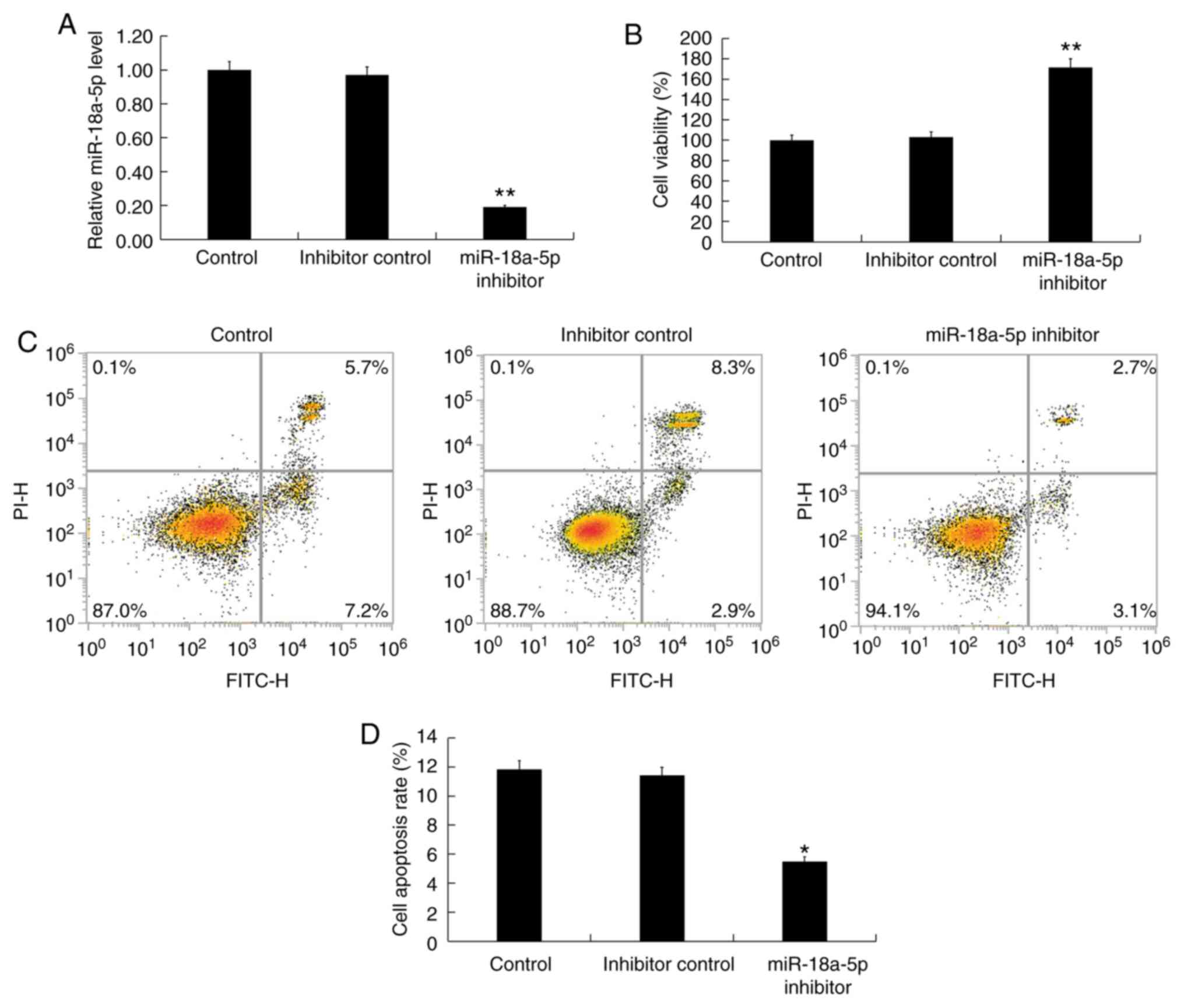
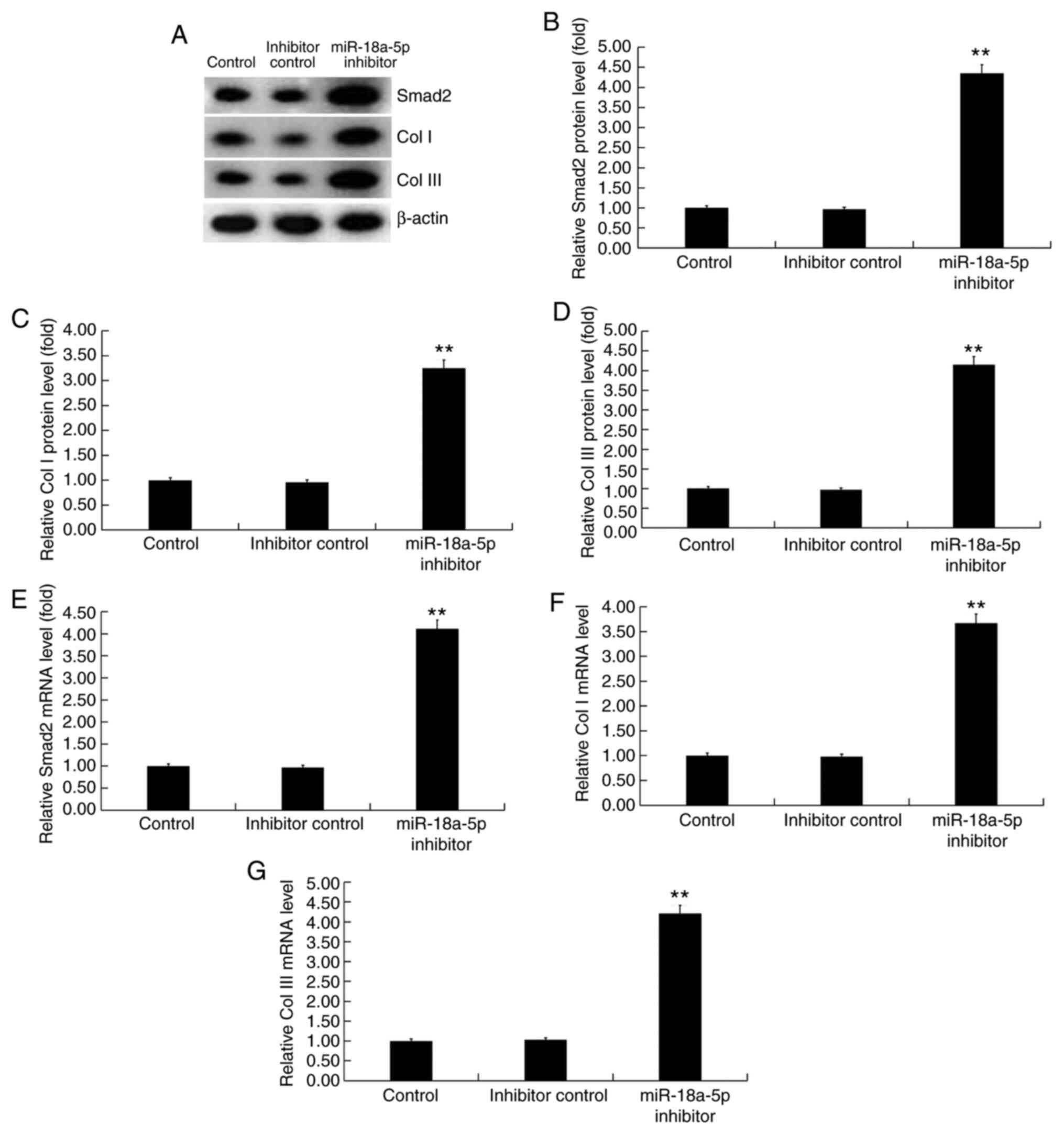
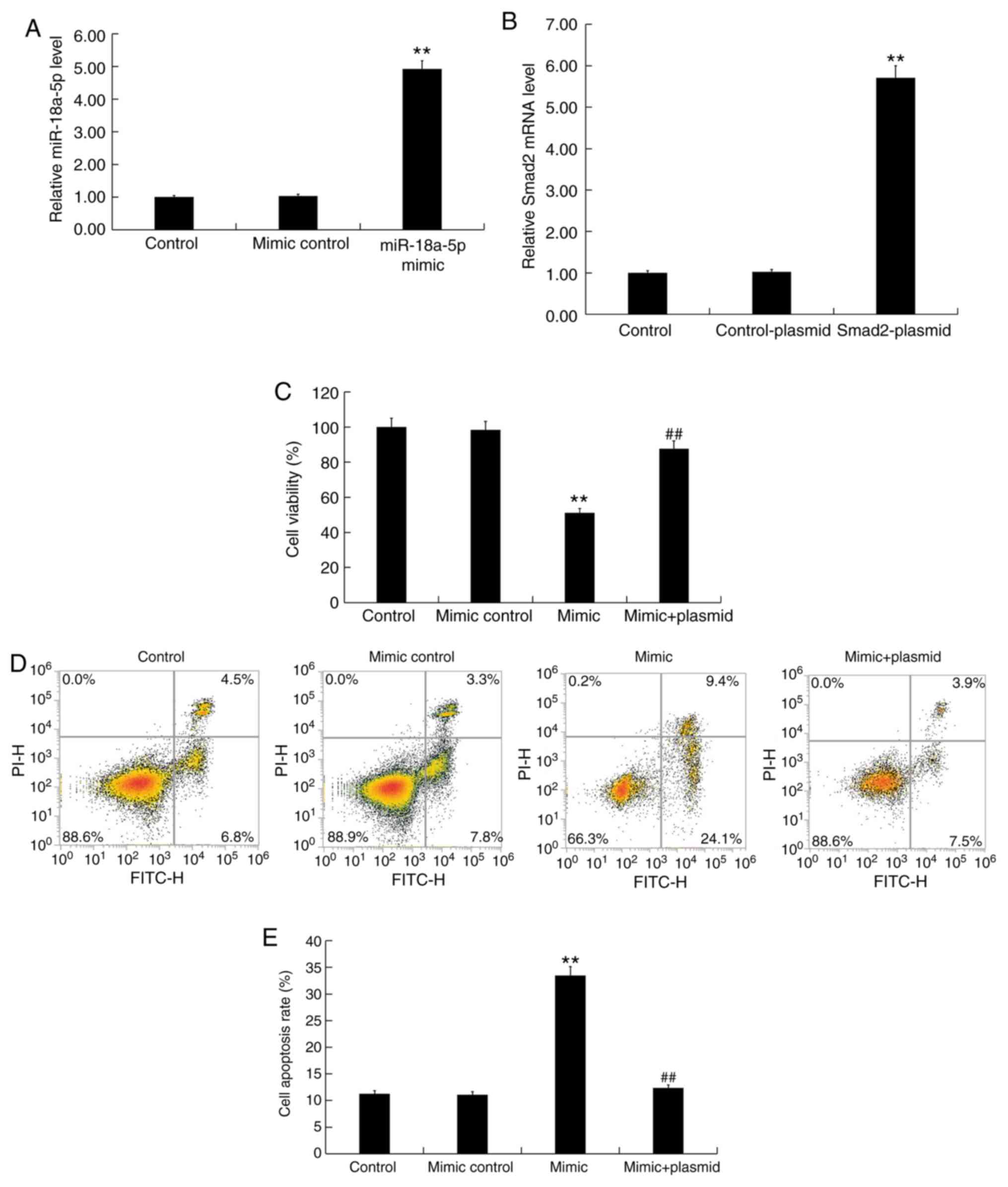
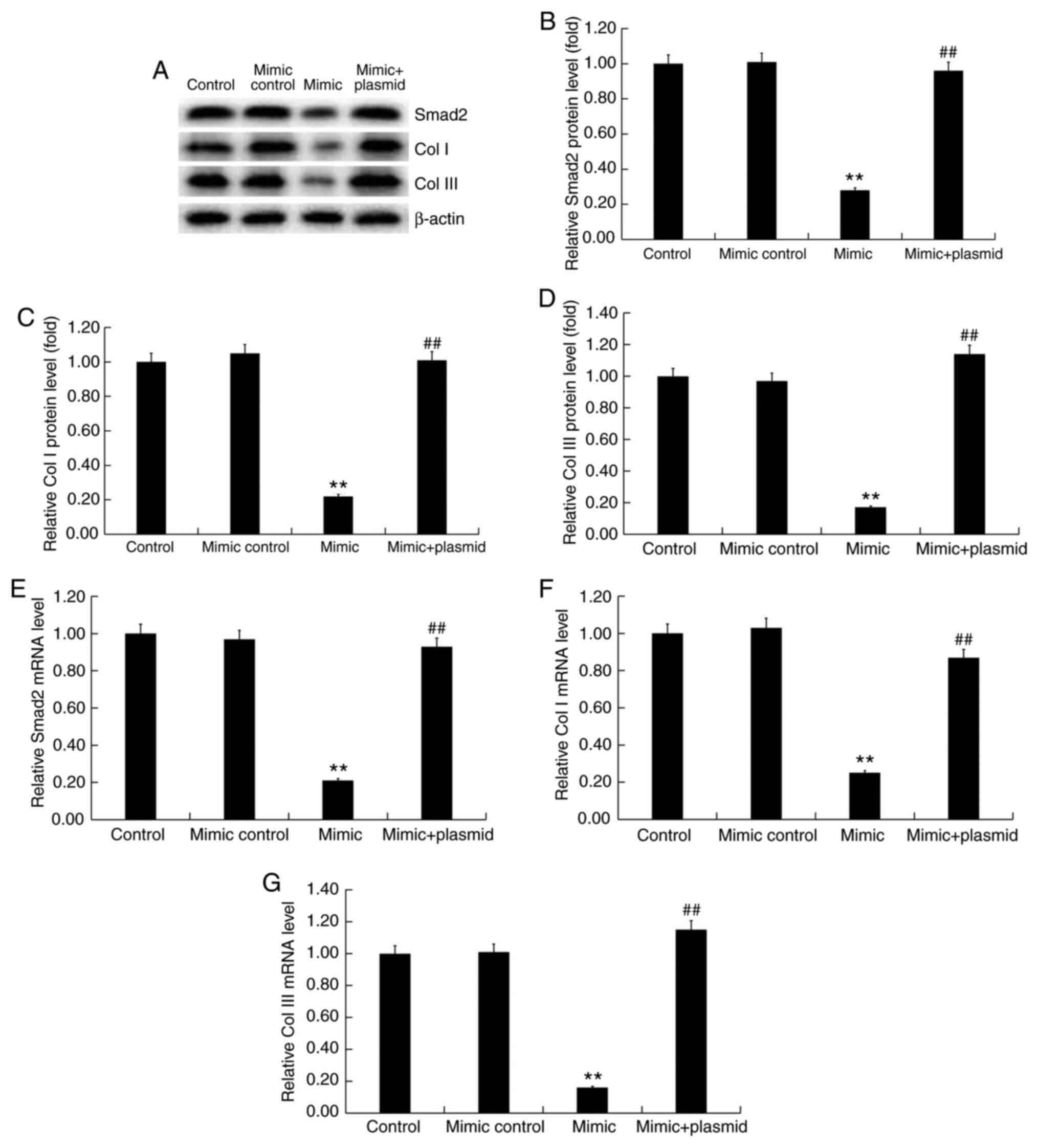
|
1
|
Tyack ZF, Pegg S and Ziviani J: Postburn
dyspigmentation: Its assessment, management, and relationship to
scarring-a review of the literature. J Burn Care Rehabil.
18:435–440. 1997.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Atiyeh BS, El Khatib AM and Dibo SA:
Pressure garment therapy (PGT) of burn scars: Evidence-based
efficacy. Ann Burns Fire Disasters. 26:205–212. 2013.PubMed/NCBI
|
|
3
|
Gauglitz GG, Korting HC, Pavicic T,
Ruzicka T and Jeschke MG: Hypertrophic scarring and keloids:
Pathomechanisms and current and emerging treatment strategies. Mol
Med. 17:113–125. 2011.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Younai S, Nichter LS, Wellisz T, Reinisch
J, Nimni ME and Tuan TL: Modulation of collagen synthesis by
transforming growth factor-beta in keloid and hypertrophic scar
fibroblasts. Ann Plast Surg. 33:148–154. 1994.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Aarabi S, Bhatt KA, Shi Y, Paterno J,
Chang EI, Loh SA, Holmes JW, Longaker MT, Yee H and Gurtner GC:
Mechanical load initiates hypertrophic scar formation through
decreased cellular apoptosis. FASEB J. 21:3250–3261.
2007.PubMed/NCBI View Article : Google Scholar
|
|
6
|
van der Veer WM, Bloemen MC, Ulrich MM,
Molema G, van Zuijlen PP, Middelkoop E and Niessen FB: Potential
cellular and molecular causes of hypertrophic scar formation.
Burns. 35:15–29. 2009.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Atiyeh BS: Nonsurgical management of
hypertrophic scars: Evidence-based therapies, standard practices,
and emerging methods. Aesthetic Plast Surg. 31:468–492, Discussion
493-494. 2007.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zuccaro J, Ziolkowski N and Fish J: A
systematic review of the effectiveness of laser therapy for
hypertrophic burn scars. Clin Plast Surg. 44:767–779.
2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Hammond SM: An overview of microRNAs. Adv
Drug Deliv Rev. 87:3–14. 2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297.
2004.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mo YY: MicroRNA regulatory networks and
human disease. Cell Mol Life Sci. 69(3529)2012.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Laffont B and Rayner KJ: MicroRNAs in the
pathobiology and therapy of atherosclerosis. Can J Cardiol.
33:313–324. 2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Chen L and Li J, Li Q, Yan H, Zhou B, Gao
Y and Li J: Non-coding RNAs: The new insight on hypertrophic scar.
J Cell Biochem. 118:1965–1968. 2017.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li P, He QY and Luo CQ: Overexpression of
miR-200b inhibits the cell proliferation and promotes apoptosis of
human hypertrophic scar fibroblasts in vitro. J Dermatol.
41:903–911. 2014.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang X, Zhang Y, Jiang BH, Zhang Q, Zhou
RP, Zhang L and Wang C: Study on the role of Hsa-miR-31-5p in
hypertrophic scar formation and the mechanism. Exp Cell Res.
361:201–209. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Xiao YY, Fan PJ, Lei SR, Qi M and Yang XH:
MiR-138/peroxisome proliferator-activated receptor β signaling
regulates human hypertrophic scar fbroblast proliferation and
movement in vitro. J Dermatol. 42:485–495. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Lu C, Peng K, Guo H, Ren X, Hu S, Cai Y,
Han Y, Ma L and Xu P: miR-18a-5p promotes cell invasion and
migration of osteosarcoma by directly targeting IRF2. Oncol Lett.
16:3150–3156. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Fei D, Zhang X, Liu J, Tan L, Xing J, Zhao
D and Zhang Y: Long noncoding RNA FER1L4 suppresses tumorigenesis
by regulating the expression of PTEN targeting miR-18a-5p in
osteosarcoma. Cell Physiol Biochem. 51:1364–1375. 2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Liu Q, Yu W, Zhu S, Cheng K, Xu H, Lv Y,
Long X, Ma L, Huang J, Sun S and Wang K: Long noncoding RNA GAS5
regulates the proliferation, migration, and invasion of glioma
cells by negatively regulating miR-18a-5p. J Cell Physiol.
234:757–768. 2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang N, Zhang H, Liu Y, Su P, Zhang J,
Wang X, Sun M, Chen B, Zhao W, Wang L, et al: SREBP1, targeted by
miR-18a-5p, modulates epithelial-mesenchymal transition in breast
cancer via forming a co-repressor complex with Snail and HDAC1/2.
Cell Death Differ. 26:843–859. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhang G, Han G, Zhang X, Yu Q, Li Z, Li Z
and Li J: Long non-coding RNA FENDRR reduces prostate cancer
malignancy by competitively binding miR-18a-5p with RUNX1.
Biomarkers. 23:435–445. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhang Y, Qian W, Feng F, Cao Q, Li Y, Hou
Y, Zhang L and Fan J: Upregulated lncRNA CASC2 may inhibit
malignant melanoma development through regulating miR-18a-5p/RUNX1.
Oncol Res. 27:371–377. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Zhang W, He W, Gao J, Wang Y, Zang W, Dong
Z and Zhao G: RETRACTED: The long noncoding RNA CASC2 inhibits
tumorigenesis through modulating the expression of PTEN by
targeting miR-18a-5p in esophageal carcinoma. Exp Cell Res.
361:30–38. 2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhou L, Li Z, Pan X, Lai Y, Quan J, Zhao
L, Xu J, Xu W, Guan X, Li H, et al: Identification of miR-18a-5p as
an oncogene and prognostic biomarker in RCC. Am J Transl Res.
10:1874–1886. 2018.PubMed/NCBI
|
|
26
|
Geng H and Guan J: MiR-18a-5p inhibits
endothelial-mesenchymal transition and cardiac fibrosis through the
Notch2 pathway. Biochem Biophys Res Commun. 491:329–336.
2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Zhang Q, Ye H, Xiang F, Song LJ, Zhou LL,
Cai PC, Zhang JC, Yu F, Shi HZ, Su Y, et al: miR-18a-5p inhibits
sub-pleural pulmonary fibrosis by targeting TGF-β receptor II. Mol
Ther. 25:728–738. 2017.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Tredget EE, Nedelec B, Scott PG and
Ghahary A: Hypertrophic scars, keloids, and contractures. The
cellular and molecular basis for therapy. Surg Clin North Am.
77:701–730. 1997.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Syed F, Ahmadi E, Iqbal SA, Singh S,
McGrouther DA and Bayat A: Fibroblasts from the growing margin of
keloid scars produce higher levels of collagen I and III compared
with intralesional and extralesional sites: Clinical implications
for lesional site-directed therapy. Br J Dermatol. 164:83–96.
2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lu Q, Guo Z and Qian H: Role of
microRNA-150-5p/SRCIN1 axis in the progression of breast cancer.
Exp Ther Med. 17:2221–2229. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Aarabi S, Longaker MT and Gunner GC:
Hypertrophic scar formation following burns and trauma: New
approaches to treatment. PLoS Med. 4(e234)2007.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Gangemi EN, Gregori D, Berchialla P,
Zingarelli E, Cairo M, Bollero D, Ganem J, Capocelli R, Cuccuru F,
Cassano P, et al: Epidemiology and risk factors for pathologic
scarring after burn wounds. Arch Facial Plast Surg. 10:93–102.
2008.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Schneider JC, Holavanahalli R, Helm P,
Goldstein R and Kowalske K: Contractures in burn injury: Defining
the problem. J Burn Care Res. 27:508–514. 2006.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Aberdam D, Candi E, Knight RA and Melino
G: miRNAs, ‘sternness’ and skin. Trends Biochem Sci. 33:583–591.
2008.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Botchkareva NV: MicroRNA/mRNA regulatory
networks in the control of skin development and regeneration. Cell
Cycle. 11:468–474. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Butzelaar L, Ulrich MM, Mink van der Molen
AB, Niessen FB and Beelen RH: Currently known risk factors for
hypertrophic skin scarring: A review. J Plast Reconstr Aesthet
Surg. 69:163–169. 2016.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xiao Y, Sun Y, Zhu B, Wang K, Liang P, Liu
W, Fu J, Zheng S, Xiao S and Xia Z: Risk factors for hypertrophic
burn scar pain, pruritus, and paresthesia development. Wound Repair
Regen. 26:172–181. 2018.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Qi J, Liu Y, Hu K, Zhang Y, Wu Y and Zhang
X: MicroRNA-26a inhibits hyperplastic scar formation by targeting
Smad2. Exp Ther Med. 15:4332–4338. 2018.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Yin L, Zhao X, Ji S, He C, Wang G, Tang C,
Gu S and Yin C: The use of gene activated matrix to mediate
effective Smad2 gene silencing against hypertrophic scar.
Biomaterials. 35:2488–2498. 2014.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Cao S, Xiao L, Rao JN, Zou T, Liu L, Zhang
D, Turner DJ, Gorospe M and Wang JY: Inhibition of Smurf2
translation by miR-322/503 modulates TGF-β/Smad2 signaling and
intestinal epithelial homeostasis. Mol Biol Cell. 25:1234–1243.
2014.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yin K, Yin W, Wang Y, Zhou L, Liu Y, Yang
G, Wang J and Lu J: MiR-206 suppresses epithelial mesenchymal
transition by targeting TGF-β signaling in estrogen receptor
positive breast cancer cells. Oncotarget. 7:24537–24548.
2016.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Shi Y, Wang L, Yu P, Liu Y and Chen W:
MicroRNA-486-5p inhibits the growth of human hypertrophic scar
fibroblasts by regulating Smad2 expression. Mol Med Rep.
19:5203–5210. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Ichihara A, Wang Z, Jinnin M, Izuno Y,
Shimozono N, Yamane K, Fujisawa A, Moriya C, Fukushima S, Inoue Y
and Ihn H: Upregulation of miR-18a-5p contributes to epidermal
necrolysis in severe drug eruptions. J Allergy Clin Immunol.
133:1065–1074. 2014.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Zhou R, Zhang Q, Zhang Y, Fu S and Wang C:
Aberrant miR-21and miR-200b expression and its pro-fbrotic
potential in hypertrophic scars. Exp Cell Res. 339:360–366.
2015.PubMed/NCBI View Article : Google Scholar
|