Introduction
Atherosclerosis is a chronic arterial disease
characterized by the gradual formation of atherosclerotic plaques
in the arterial walls (1). It is
considered to be one of the leading causes (with 2.4 million deaths
and 61% of cardiovascular deaths in 2016) of vascular-associated
mortality worldwide (2), which can
result in ischemic stroke, coronary heart disease and peripheral
arterial disease (3). Despite
advancements in technology and secondary prevention measures (e.g.
percutaneous coronary intervention and oral anticoagulants), the
burden of atherosclerotic cardiovascular disease continues to
increase, posing a significant obstacle to the clinical prevention
and treatment of atherosclerosis (4). Research over the past number of
decades has continued to unravel the pathophysiological molecular
mechanism of atherosclerotic plaque formation (5). Advancements in understanding the
cellular or molecular pathogenesis of atherosclerosis have allowed
the development of novel therapeutic strategies (6). A therapeutic strategy that has been
frequently reported is by protecting vascular endothelial cells
from injury since endothelial cell damage and dysfunction serves a
key role in the formation and progression of atherosclerotic
plaques, leading to subsequent complications (7). Cardiovascular risk factors (e.g.
smoking and alcohol) that induce oxidative stress are the principle
drivers of endothelial dysfunction. Under oxidative stress, excess
reactive oxygen species (ROS) can inhibit the proliferation of
vascular endothelial cells, disrupting repair and induce apoptosis
(8) Therefore, preventing oxidative
stress in the vascular endothelium may protect against or reverse
the development of atherosclerosis (9). Therapeutic approaches designed to
inhibit NAD(P)H oxidase, a major source of ROS, have been
demonstrated to alleviate cardiovascular oxidative stress and
prevent endothelial cell damage (10,11).
Development of novel strategies (e.g. administration of
anti-radical agents and antioxidants) aimed at attenuating
oxidative stress hold significant promise for the prevention and
treatment of atherosclerosis.
Sulforaphane (SFN) is a phytochemical antioxidant
that can be extracted from cruciferous plants, including broccoli,
Brussel sprouts and kale (12). SFN
exerts protective effects by activating the nuclear factor
erythroid-2-related factor 2 (Nrf2)/antioxidant response element
(ARE) pathway, which increases the transcription and activity of
antioxidant enzymes (13).
Cytoprotective properties of SFN against oxidative stress have been
reported both in vivo and in vitro. Yoon et al
(14) previously demonstrated that
SFN pretreatment protects against ischemia and reperfusion-induced
acute renal failure in rats by suppressing oxidative stress in
renal tissues. Furthermore, Chen et al (15) reported that SFN reduces ROS levels
and protects osteoblasts from apoptosis in vitro, whilst Zhu
et al (16) suggested that
SFN prevents rat aortic smooth muscle cells death by inhibiting ROS
production and oxidative cytotoxicity induced by xanthine oxidase.
However, to the best of our knowledge, the potential effects of SFN
on vascular endothelial cells under oxidative stress have not been
previously reported. Further investigation on its underlying
molecular mechanism may facilitate the identification of SFN as a
novel therapeutic agent for atherosclerosis.
The association between microRNAs (miRNAs/miRs) and
oxidative stress has attracted considerable interest over recent
decades. A number of miRNAs (e.g. miR-146a, miR-92 and miR-143)
that are responsive to oxidative stress have been previously
identified constituting a complex regulatory network in
pathological conditions such as atherosclerosis (17). miR-34a is one of the more
extensively studied oxidative stress-responsive miRNAs that has
been previously reported to be upregulated during oxidative stress
and mediate apoptosis in mesenchymal stromal/stem cells, HLE-B3 and
HUVECs (18-20).
There is increasing consensus suggesting that sirtuin-1 (SIRT1),
which regulates cell death/survival balance under oxidative stress
expression, is impaired by ROS (21). Down-regulation SIRT1 under oxidative
stress promoting endothelial dysfunction and vascular disease
progression has also been reported (22). In particular, a previous study has
documented that SIRT1 may also be under the regulation of oxidative
stress-responsive miRNAs, including miR-34a (23).
The present study hypothesized that SFN may
alleviate oxidative stress-induced endothelial cell injury via
regulation of the miR-34a/SIRT1 axis, since a number of previous
studies have also demonstrated that SFN mediates its function by
regulating miRNA expression (24,25).
Materials and methods
Cell culture
Human umbilical vein endothelial cells (HUVECs) were
obtained from the American Type Culture Collection (ATCC) and
cultured in F-12K medium (ATCC) supplemented with 10% FBS (Thermo
Fisher Scientific, Inc.), endothelial cell growth supplements (cat.
no. 354006; BD Biosciences), 0.1 mg/ml heparin and 1%
penicillin/streptomycin (Merck KGaA) at 37˚C in 5%
CO2.
H2O2 treatment
and SFN pretreatment
The concentrations of H2O2 and
SFN used in the present study were determined following a
preliminary study, where treatment with SFN for 4, 8 or 12 h prior
to the addition of H2O2 exhibited similar
protective effects. HUVECs were divided into four groups as
follows: i) Control; ii) H2O2 (cat. no.
H1009; Merck KgaA); iii) H2O2 + SFN; and iv)
SFN (cat. no. S4441; Merck KgaA). The cells cultured at 37˚C and
other culture conditions for each group were as follows: i) Cells
in the control group were treated with PBS; ii) Cells in the
H2O2 group were treated with 200 µmol/l
H2O2 to simulate oxidative stress-induced
cell injury; iii) Cells in the H2O2 + SFN
group were treated with 1.0 µmol/l SFN for 4 h, prior to the
addition of 200 µmol/l H2O2; and iv) Cells in
the SFN group were treated with 1.0 µmol/l SFN.
Cell transfection
pcDNA3.1-SIRT1 and pcDNA3.1-NRF2 (Vi-gene Co., Ltd.)
expression vectors were constructed and transfected (1,000 ng/ml)
into HUVECs to overexpress SIRT1 and NRF2, respectively. Empty
pcDNA3.1 was used as the negative control. A total of 50 nM
siRNA-SIRT1 (Shanghai GenePharma Co., Ltd.) was transfected into
cells to downregulate SIRT1 expression, whilst 50 nM miR-34a mimics
and inhibitors (Shanghai GenePharma Co., Ltd.) were used to
upregulate and downregulate miR-34a expression in cells,
respectively. The Allstar negative control sequence (Qiagen AB)
served as a control for transfection with siRNA, mimics and
inhibitors. Cells were transfected with pcDNA3.1 vectors using
TransPass™ HUVEC transfection reagent (New England Biolabs, Inc.)
according to the manufacturer's protocol. Cells were transfected
with siRNA, mimics and inhibitors using Lipofectamine®
RNAiMAX reagent (Thermo Fisher Scientific, Inc.) according to the
manufacturer's protocol. Subsequent experiments were performed 48 h
after transfection.
Cell viability assay
The CellTiter-Blue® kit (Promega
Corporation) was used to measure the viability of HUVECs in the
control, H2O2, H2O2 +
SFN and SFN groups in accordance with manufacturer's protocol. The
respective cells were seeded into 96-well plates at a density of
6,000 cells/well. After 0, 12, 24 and 48 h, cells were incubated
with 10 µl CellTiter-Blue® reagent at 37˚C, following
which fluorescence was measured using a FLx800™ microplate
fluorescence reader at 560 nm (BioTek Instruments Inc.). Cell
viability was analyzed and normalized to the value at 0 h.
Flow cytometric analysis of
apoptosis
After 24 h treatment, 1x106/ml cells in
the control, H2O2,
H2O2+SFN and SFN groups were collected,
harvested and subsequently resuspended in 500 µl annexin V binding
buffer prior to staining with 5 µl Annexin V-FITC and 10 µl
propidium iodide (Thermo Fisher Scientific, Inc.) at 25˚C for 15
min in the dark. Apoptotic cells were measured using an Epics XL™
flow cytometer (Beckman Coulter, Inc.) and analyzed using FlowJo™
v9.0 (FlowJo LLC).
ROS measurement
After 24 h treatment, intracellular ROS levels in
HUVECs in the control, H2O2,
H2O2 + SFN and SFN groups were measured using
the Reactive Oxygen Species Assay kit (cat. no. S0033; Beyotime
Institute of Biotechnology). Briefly, HUVECs treated with
H2O2 were collected and incubated with
2',7'-dichlorofluorescin diacetate (DCFH-DA) probes (1:1,000) for
30 min at 37˚C. Fluorescence was subsequently measured at the
emission wavelength of 523 nm, using an excitation wavelength of
488 nm in a FLx800 microplate fluorescence reader.
Reverse transcription-quantitative
(RT-q)PCR
Total RNA was extracted from the treated HUVECs 24 h
after treatment with H2O2 and/or SFN or 48 h
post-transfection, using TRIzol® reagent (Thermo Fisher
Scientific, Inc.). To measure SIRT1 mRNA expression, total RNA was
reverse transcribed into cDNA using RevertAid H Minus First Strand
cDNA Synthesis Kit (Thermo Fisher Scientific, Inc.) and amplified
using the SYBR™-Green PCR Master Mix (Thermo Fisher Scientific,
Inc.) in accordance with the manufacturer's protocols. To measure
miR-34a expression, total RNA was reverse transcribed into cDNA
using the miScript II RT kit (Qiagen AB). qPCR was subsequently
performed using the miScript SYBR®-Green PCR kit (Qiagen
AB). The relative expression levels of the target genes were
calculated using the 2-ΔΔCq method (26) and normalized to the internal
reference genes GAPDH for SIRT1 and SNORD-48(27) for miR-34a. The following primer
sequences were used for qPCR: SIRT1 forward,
5'-TAGACACGCTGGAACAGGTTGC-3' and reverse,
5'-CTCCTCGTACAGCTTCACAGTC-3' and GAPDH forward,
5'-GTCTCCTCTGACTTCAACAGCG-3' and reverse,
5'-ACCACCCTGTTGCTGTAGCCAA-3'. miR-34a and SNORD-48 primer sequences
were obtained from the miScript Primer Assays kit (Qiagen AB).
Western blotting
Total protein was extracted from treated HUVECs 24 h
after treatment with H2O2 and/or SFN or 48 h
post-transfection, using a protease inhibitor cocktail added to
RIPA buffer (Beyotime Institute of Biotechnology). Total protein
was quantified using the BSA Protein Assay kit (Beyotime Institute
of Biotechnology) and 30 µg protein/lane was separated by 12%
SDS-PAGE. Following electrophoresis, protein samples were
transferred onto nitrocellulose membranes and blocked with 5%
fat-free milk at room temperature. Membranes were incubated with
primary antibodies against SIRT1 (1:1,000; cat. no. ab189494), Nrf2
(1:1,000; cat. no. ab137550) and GAPDH (1:5,000; cat. no. ab9485;
all from Abcam) for 12 h at 4˚C. The membranes were subsequently
washed and incubated with IRDye®-conjugated secondary
antibody (1:2,000; LI-COR Biosciences, cat. no. 926-32211) at room
temperature for 2 h. Protein bands were visualized using a Li-Cor
Odyssey system v 1.60 (LI-COR Biosciences).
Dual-luciferase reporter assay
Both wild-type and mutant SIRT1 3'-untranslated
region (3'-UTR) sequences were first cloned into firefly luciferase
reporter plasmids pMIR-REPORT™ (Applied Biosystems; Thermo Fisher
Scientific, Inc.). Subsequently, 1,000 ng/ml of luciferase reporter
vectors (SIRT1 3'-UTR-wild-type or SIRT1 3'-UTR-mutant) were
co-transfected with miR-34a mimics (50 nM) or miR-negative controls
into HUVECs s. Transfections were performed using
Lipofectamine® 2000 (Thermo Fisher Scientific, Inc.).
Following incubation for 48 h at 37˚C, firefly and Renilla
luciferase activities were measured using a
Dual-Luciferase® Reporter system (Promega Corporation).
Firefly luciferase activity was normalized to that of
Renilla luciferase activity.
Statistical analysis
Statistical analysis was performed using the SPSS
software (version 19.0; IBM Corp). All experiments were repeated
five times. Data are presented as the mean ± standard deviation.
Unpaired Student's t-test was used to compare differences between
two groups, whilst one-way analysis of variance followed by Tukey's
post hoc test was used to compare differences among multiple
groups. P<0.05 was considered to indicate a statistically
significant difference.
Results
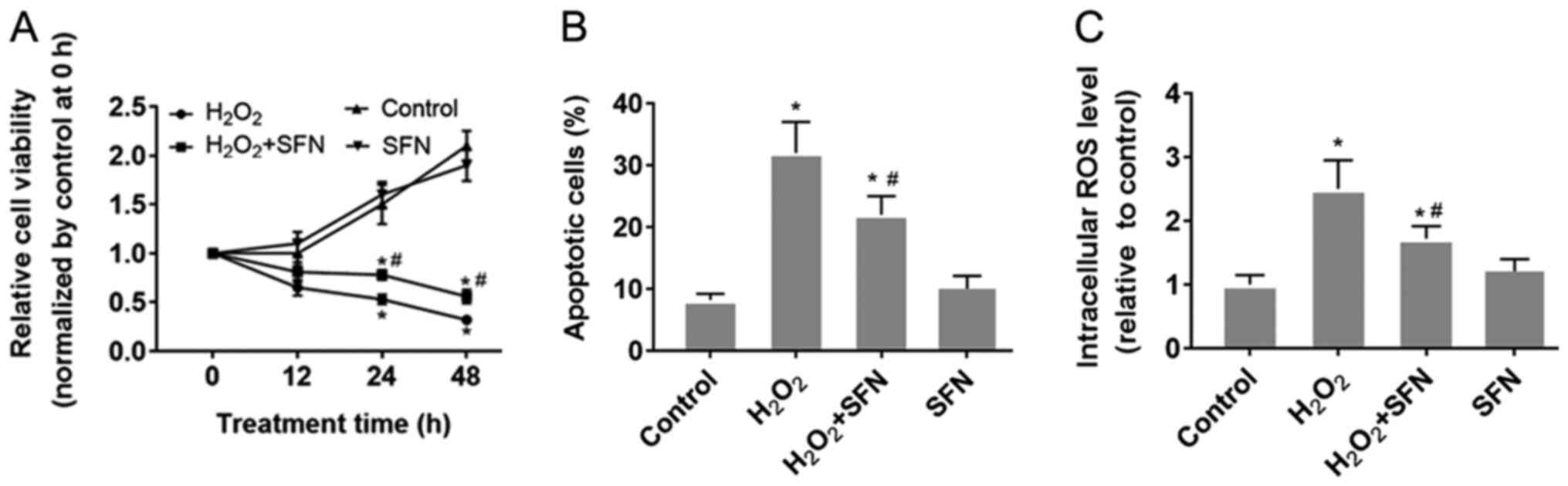
SFN protects HUVECs from oxidative
stress
HUVECs were treated with H2O2
to induce oxidative stress. The results demonstrated that
H2O2 treatment significantly reduced HUVEC
viability whilst significantly increasing apoptosis compared with
those under control conditions (P<0.05; Figs. S1A, 1A and B).
Furthermore, significantly higher intracellular ROS levels were
also observed following H2O2 treatment
compared with those in cells in the control group (P<0.05;
Fig. 1C). SFN pretreatment was
found to relieve the damaging effect of H2O2
in HUVECs compared with that in HUVECs treated with
H2O2 alone (Fig.
1). SFN pretreatment was revealed to partially preserve cell
viability whilst significantly reducing HUVEC apoptosis induced by
H2O2 treatment (P<0.05; Fig. 1A and B). In addition, SFN also alleviated
H2O2-induced ROS generation in HUVECs
(P<0.05; Fig. 1C). However,
HUVECs treated with SFN alone did not exhibit any significant
effects in terms of cell viability, apoptosis or ROS production
compared with the other three groups (Fig. 1).
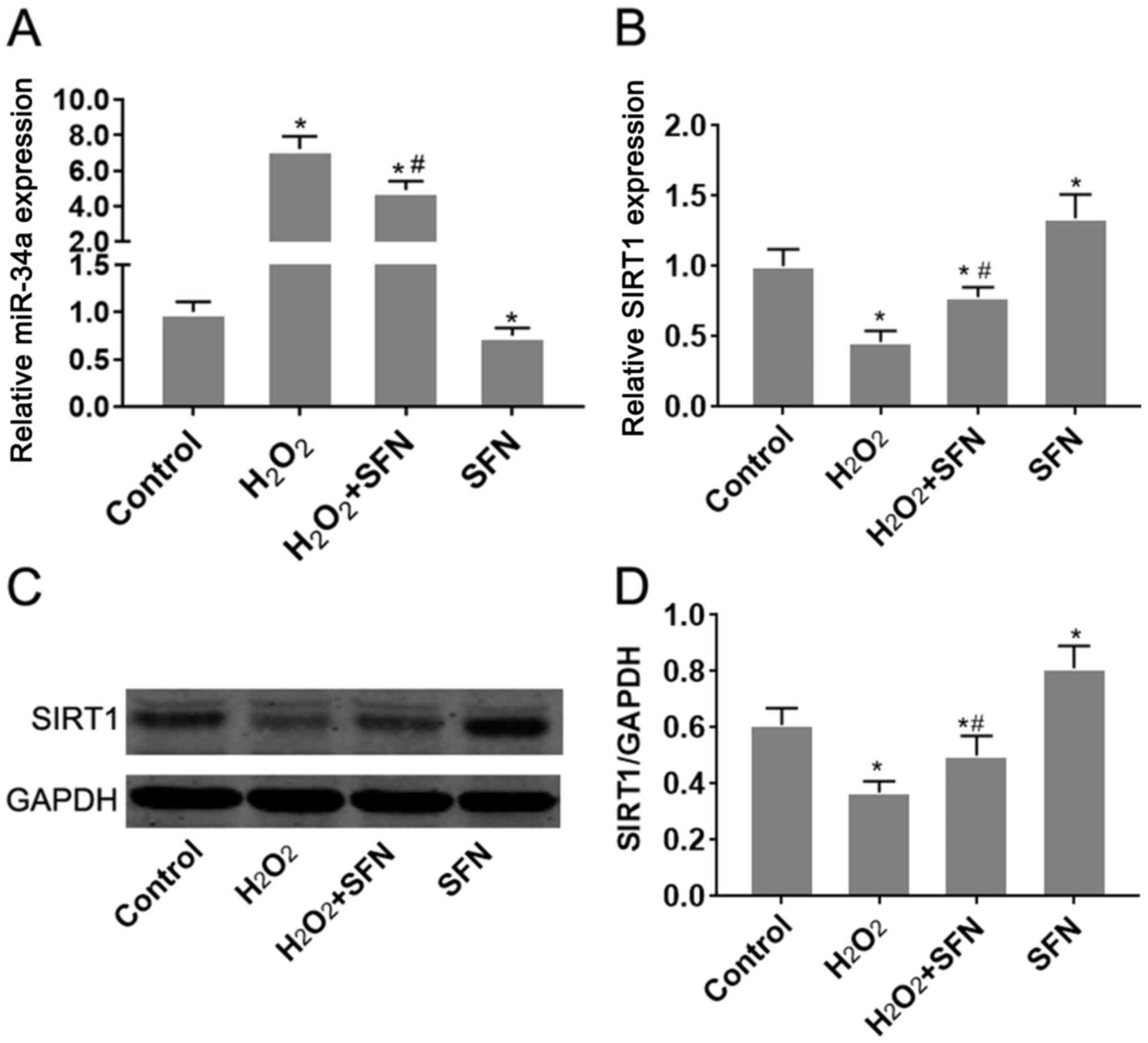
Effect of SFN on miR-34a and SIRT1
expression in HUVECs under oxidative stress
H2O2 treatment was
demonstrated to significantly increase miR-34a expression in HUVECs
(P<0.05; Fig. 2A). Since
TargetScan v7.2 (http://www.targetscan.org/vert_72/) indicated that
SIRT1 is a potential target gene of miR-34a, changes in SIRT1
expression were subsequently measured. SIRT1 mRNA and protein
expression levels were found to be significantly reduced following
treatment with H2O2 (P<0.05; Fig. 2B-D). SFN pretreatment significantly
reversed the effects of H2O2 treatment on
miR-34a and SIRT1 expression (P<0.05; Fig. 2B-D). Additionally, SFN treatment
alone was also found to reduce miR-34a expression whilst increasing
SIRT1 expression in HUVECs, compared with those of control cells
(Fig. 2). Taken together, these
observations suggest that SFN may regulate miR-34a and SIRT1
expression in HUVECs under oxidative stress.
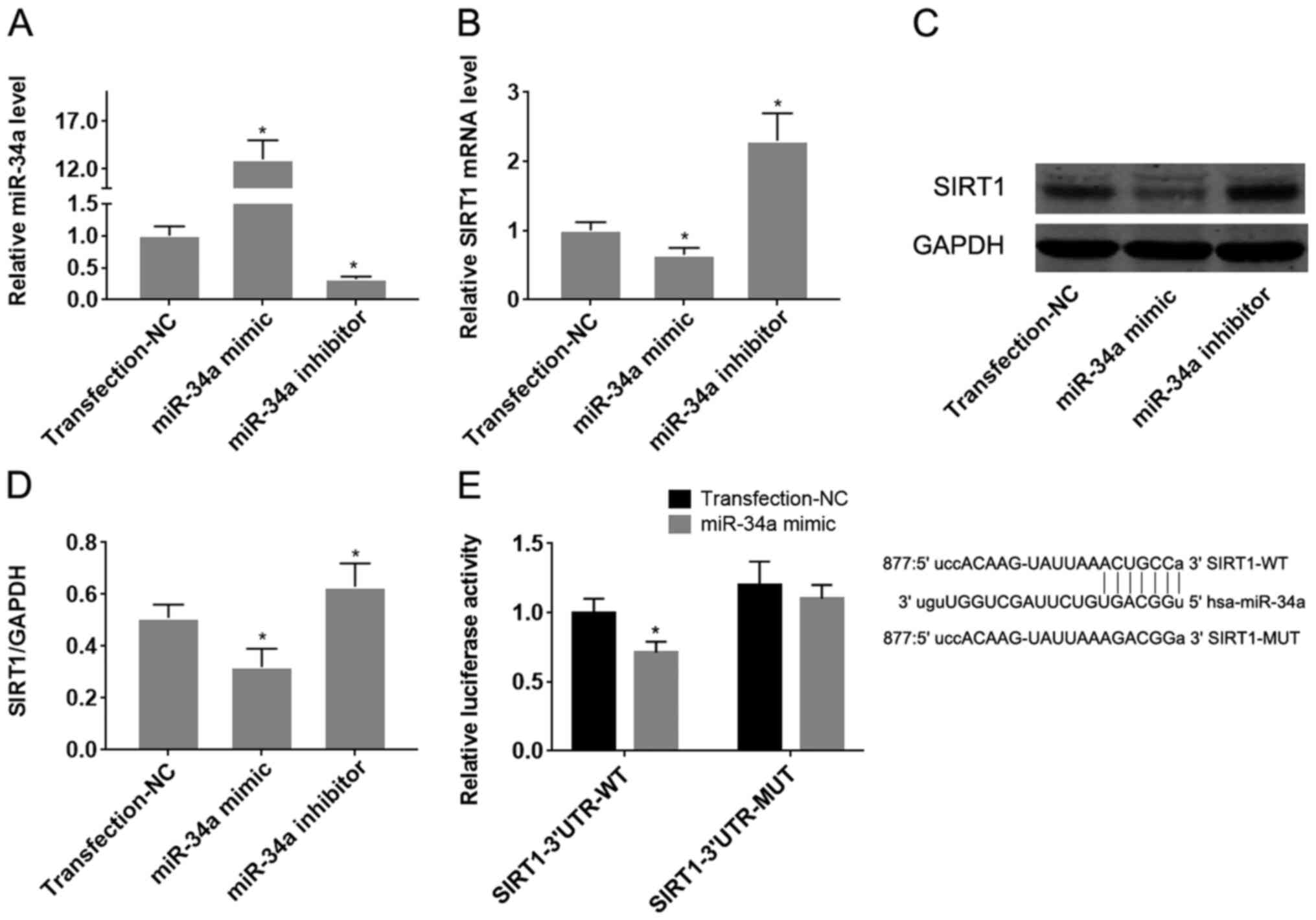
miR-34a/SIRT1 axis regulates oxidative
stress-induced HUVEC injury and underlies the protective effect of
SFN
To confirm the target effect of miR-34a on SIRT1 in
HUVECs, they were transfected with miR-34a mimics or inhibitors.
Transfection with miR-34a mimics and inhibitors significantly was
found to significantly increase and reduce miR expression in
HUVECs, respectively (Fig. 3A;
P<0.05). Overexpression of miR-34a significantly reduced the
expression levels of both SIRT1 mRNA and protein (Fig. 3B-D), whilst downregulation of
miR-34a mediated the opposite effect on SIRT1 expression, compared
with that observed following the upregulation of miR-34a
(P<0.05; Fig. 3B-D).
Dual-luciferase reporter assay subsequently demonstrated that
co-transfection of pMIR-REPORT-SIRT1-3'-UTR-wild-type with the
miR-34a mimic significantly reduced luciferase activity, compared
with pMIR-REPORT-SIRT1-3'-UTR-mutant-type (P<0.05; Fig. 3E). These results suggest that
miR-34a directly targets SIRT1 mRNA by binding to its 3'-UTR.
The role of the miR-34a/SIRT1 axis on oxidative
stress-induced injury in HUVECs was next analyzed. Transfection of
HIUVECs with pcDNA3.1-SIRT1 and siRNA-SIRT1 was demonstrated to
significantly upregulate and downregulate SIRT1 expression in
HUVECs compared with the respective negative controls (Fig. 4A-C). Overexpression of miR-34a was
revealed to significantly aggravate
H2O2-induced cell viability inhibition and
apoptosis in HUVECs (P<0.05; Fig.
4D and E). Conversely,
inhibition of miR-34a antagonized
H2O2-induced viability inhibition and
apoptosis in HUVECs (P<0.05; Figs.
S1B, 4D and E). Following H2O2
treatment, HUVECs transfected the pcDNA3.1-SIRT1 plasmid exhibited
significantly increased cell viability and lower apoptotic rates
compared with those in cells transfected with negative controls
(P<0.05; Figs. S1B, 4D and E).
By contrast, opposite effects were observed in HUVECs transfected
with siRNA-SIRT1 (P<0.05; Figs.
S1B, 4D and E). Taken together, these results
collectively suggest that the miR-34a/SIRT1 axis partly underlies
the mechanism of oxidative stress-induced injury in HUVECs, where
SFN may serve a protective role in HUVECs under oxidative stress by
regulating this miR-34a/SIRT1 axis.
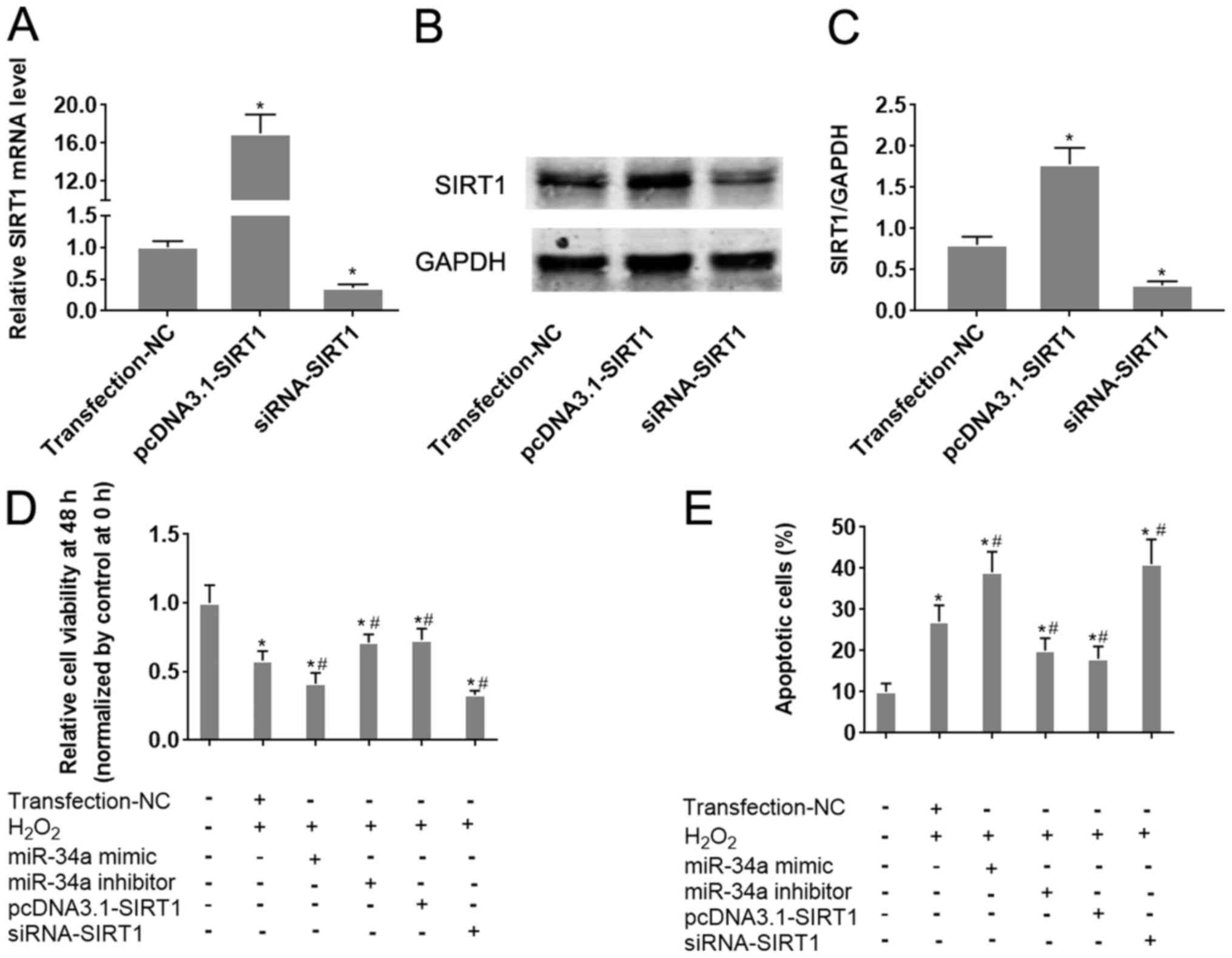 | Figure 4Alteration of the miR-34a/SIRT1 axis
influences HUVEC cell viability and apoptosis following
H2O2 treatment. miR-34a mimics and inhibitors
were transfected into HUVECs 24 h before H2O2
treatment. To change SIRT1 expression in cells, SIRT1-siRNA or
pcDNA3.1-SIRT1 were transfected into HUVECs. HUVECs were treated
with H2O2. (A) The effect of siRNA-SIRT1 and
pcDNA3.1-SIRT1 transfection on SIRT1 mRNA expression was verified
by reverse transcription-quantitative PCR analysis. (B) The effect
of siRNA-SIRT1 and pcDNA3.1-SIRT1 transfection on SIRT1 protein
expression was verified by western blot analysis and (C)
quantified. (D and E) HUVEC cells were transfected with the
indicated combination of miR-34a mimic, miR-34a inhibitor,
pcDNA3.1-SIRT1 or siRNA-SIRT1, following which they were treated
with H2O2. (D) HUVEC cell viability was
measured after 48 h using CellTiter-Blue® assay. (E)
HUVEC apoptosis was measured by flow cytometry.
*P<0.05 vs. Transfection-NC; #P<0.05
vs. H2O2 + Transfection-NC. Data are
presented as the mean ± standard deviation, n=5. miR, microRNA;
SIRT1, sirtuin-1; HUVECs, human umbilical vein endothelial cells;
siRNA, small interfering RNA; NC, negative control;
Transfection-NC, co-transfection with the empty pcDNA3.1 vector and
Allstar. |
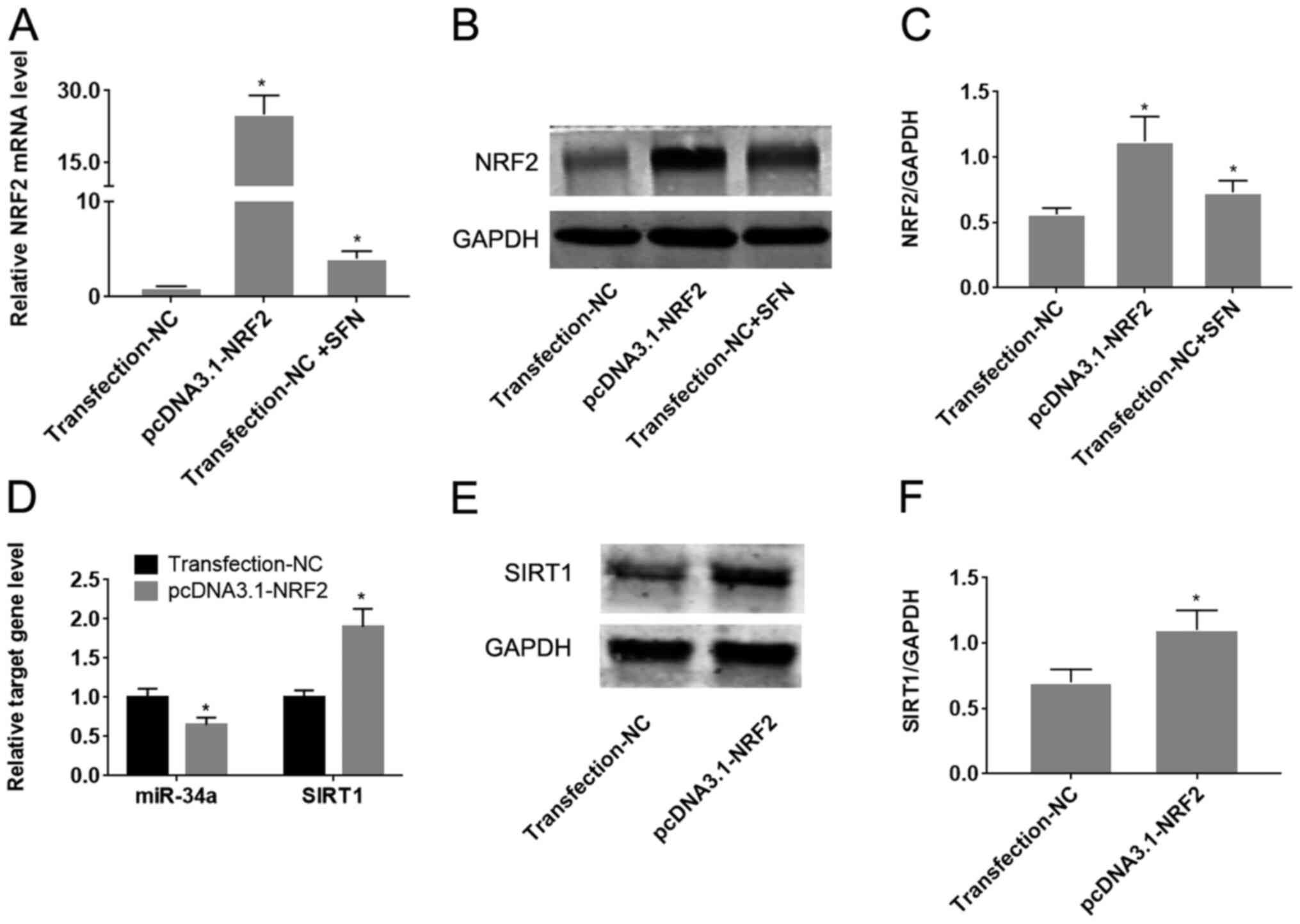
SFN regulates the miR-34a/SIRT1 axis
by inducing Nrf2 expression
To determine whether Nrf2 regulates the
miR-34a/SIRT1 axis in HUVECs, Nrf2 was upregulated by transfection
with the pcDNA3.1-Nrf2 plasmid. Transfection with pcDNA3.1-Nrf2 was
found to significantly increase Nrf2 expression compared with cells
transfected with the empty plasmid (P<0.05; Fig. 5A-C). Treatment of HUVECs with SFN
significantly increased the expression of Nrf2 on both mRNA and
protein levels (P<0.05; Fig.
5B-D). Overexpression of Nrf2 was also demonstrated to
significantly reduce miR-34a expression, whilst increasing SIRT1
expression compared with cells transfected with the empty plasmid
(P<0.05; Fig. 5D-F).
Discussion
Vascular endothelial cells are particularly
sensitive to oxidative stress, where ROS production results in the
induction of inflammatory genes such as vascular cell adhesion
protein 1 and monocyte chemoattractant protein-1, oxidation of low
density lipoprotein, endothelial cell adhesion and infiltration
(28). These alterations disrupt
the structure and function of the vascular endothelium, which
contributes to the initiation and development of atherosclerosis
(29). SFN is a natural antioxidant
that can be extracted from cruciferous vegetables (30). The abundant availability of raw
materials means that SFN confers certain advantages in clinical
applications over other potential naturally occurring agents
(31). Previous studies have
demonstrated that SFN protects cells against oxidative stress both
in vivo and in vitro. Saleh et al (32) reported that SFN has hepatic
anti-aging potential through the amelioration of oxidative stress
in rats. Another previous in vitro analysis demonstrated
that pretreatment of H9c2 rat myoblasts with SFN reduces
doxorubicin-induced apoptosis by preventing oxidative stress
(33). Although the role of SFN in
the protection of endothelial cells through an antioxidant
mechanism has been previously reported, information regarding this
subject remains insufficient. Campbell et al (34) previously demonstrated that SFN
treatment improves the expression of selenoenzymes in the human
endothelial cell line EAhy926, protecting the cells from oxidative
stress. In brain vascular endothelium, SFN has been found to
upregulate antioxidative stress responses, redox signaling and
phase 2 drug metabolism/detoxification, thereby exerting
neurovascular protective effects (35). Consistent with the aforementioned
previous findings, results from the present study demonstrated that
SFN inhibited oxidative stress to protect vascular endothelial
cells. Pretreatment with SFN was observed to significantly reduce
the production of ROS in H2O2-treated HUVECs
and suppress apoptosis. SFN has been frequently documented to
protects cells against oxidative stress at low-to-moderate doses in
cardiomyocytes and nerve cells (36). However, high concentrations of SFN
are cytotoxic and confer an acute pro-oxidant effect through the
depletion of intracellular glutathione by the formation and export
of SFN-glutathione complexes (37).
Therefore, the treatment dose of SFN needs to be considered
prudently to investigate the potential protective effects of SFN on
normal cells.
Prior to its clinical application, it is important
to determine the molecular mechanism by which SFN exerts
endothelial cell protection. A canonical pathway by which SFN
performs a protective role is the Nrf2-ARE pathway (38). SFN can induce the expression or
activation of Nrf2 to enhance its binding to ARE (39). Nrf2 is a helix-loop-helix basic
leucine zipper transcription factor that serves as a key regulator
in the cellular defense system against oxidative stress (40). Through binding to the promoter
region of ARE, Nrf2 activation induces the transcription of a
series of anti-oxidative stress/detoxifying enzymes, including heme
oxygenase-1, NAD(P)H: Quinone oxidoreductase-1,
UDP-glucuronosyltransferases and glutathione-S-transferases
(41). Accumulating evidence
suggests that in addition to inducing the level of antioxidative
stress enzymes via the NRF2-ARE pathway, SFN may also alter of
expression profile of miRNAs under oxidative stress. Eren et
al (42) previously reported
that SFN alleviates lipopolysaccharide-induced cell injury and
suppresses oxidative stress by inhibiting miR-155. The function of
miRNA regulatory networks during oxidative stress in
atherosclerosis and arterial remodeling have been discussed in
previous studies (43,44). It is therefore possible that SFN may
be involved in the regulation of miRNA expression during the
protection of endothelial cells from oxidative stress damage.
The present study selected miR-34a as the candidate
target gene of SFN in HUVECs under oxidative stress conditions.
miR-34a has been previously confirmed as an miRNA that is
upregulated in response to oxidative stress. For example, Tong
et al (45) demonstrated
that miR-34a induction in response to ROS reduced the tolerance of
spontaneously arising retinal pigment epithelia cells to oxidative
stress. Furthermore, increased miR-34a expression has been
previously reported to mediate visfatin-induced apoptosis and
oxidative stress in human osteoarthritic chondrocytes, which was
reversed by the inhibition of miR-34a (46). However, role of SFN on miR-34a
expression remains to be investigated.
SIRT1 is an NAD-dependent deacetylase that regulates
apoptosis in response to oxidative and genotoxic stress that has
also been found to be a potential target gene of miR-34a (47). The role of the miR-34a/SIRT1 axis in
oxidative stress-induced cell injury has been confirmed in
endothelial progenitor cells (48).
Therefore, the present study hypothesized that SFN may protect
endothelial cells partly through regulation of the miR-34a/SIRT1
axis. The results of the present study demonstrated that
H2O2 treatment upregulated miR-34a
expression, whilst inhibiting SIRT1 expression. Down-regulation of
miR-34a and up-regulation of SIRT1 was found to protect cells from
oxidative stress. These results are consistent with previous
findings, suggesting a role for the miR-34a/SIRT1 axis in HUVECs
(48). SFN treatment was
demonstrated to reduce the expression levels of miR-34a and induce
SIRT1 in HUVECs, indicating that miR-34a/SIRT1 axis counteracted
the protective effect of SFN. Taken together, these results suggest
that SFN may modulate the miR-34a/SIRT1 axis to protect against
oxidative stress-induced HUVEC injury. Furthermore, overexpression
of Nrf2 exerted a regulatory effect on the miR-34a/SIRT1 axis,
similar to that exhibited by SFN. These results are consistent with
previous findings (38,39), which have demonstrated that SFN
regulates Nrf2, suggesting that SFN may also regulate the
miR-34a/SIRT1 axis via Nrf2 in HUVECs. Nrf2 is considered to be a
regulator of miRNA expression during antioxidant responses
(35). Nrf2 can directly regulate
the expression of miRNAs that contain the functional
Nrf2-responsive elements (49).
Nrf2 can also indirectly regulate miRNA expression by controlling
transcription. For example, miR-125B1 and miR-29B1 were also
identified as direct transcriptional targets of NRF2 in acute
myeloid leukemia cells (50).
However, the present study failed to investigate if Nrf2 directly
or indirectly regulates miR-34a. Therefore, prospective studies
will aim to perform chromatin immunoprecipitation or luciferase
assays to address this.
In conclusion, results from the present study
suggest that SFN protects HUVECs against oxidative stress by
regulating the miR-34a/SIRT1 axis via Nrf2. Furthermore, SFN
exhibited cytoprotective effects on vascular endothelial cells
under oxidative stress, putting it forward as a potential candidate
for the therapeutic intervention of atherosclerosis.
Supplementary Material
Representative flow cytometry dot
plots. Representative flow cytometry dot diagrams for (A) Fig. 1B and (B) Fig. 4E. SFN, sulforaphane; miR, microRNA;
SIRT1, sirtuin-1; NC, negative control.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analysed during the present
study are available from the corresponding author upon reasonable
request.
Authors' contributions
TL and ZZ designed the study; TL and QP analyzed and
interpreted the data; TL, YL and QP drafted the manuscript; MB and
YP performed statistical analysis; TL and YP performed data
visualization; YL also contributed to the acquisition and analysis
part of data. ZZ supervised the study. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Kobiyama K and Ley K: Atherosclerosis.
Circ Res. 123:1118–1120. 2018.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Zhao D, Liu J, Wang M, Zhang X and Zhou M:
Epidemiology of cardiovascular disease in China: Current features
and implications. Nat Rev Cardiol. 16:203–212. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Herrington W, Lacey B, Sherliker P,
Armitage J and Lewington S: Epidemiology of atherosclerosis and the
potential to reduce the global burden of atherothrombotic disease.
Circ Res. 118:535–546. 2016.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Torres N, Guevara-Cruz M,
Velázquez-Villegas LA and Tovar AR: Nutrition and atherosclerosis.
Arch Med Res. 46:408–426. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Criqui MH and Aboyans V: Epidemiology of
peripheral artery disease. Circ Res. 116:1509–1526. 2015.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Fredman G and Tabas I: Boosting
inflammation resolution in atherosclerosis: The next frontier for
therapy. Am J Pathol. 187:1211–1221. 2017.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ma H, Su L, He X and Miao J: Loss of
HMBOX1 promotes LPS-induced apoptosis and inhibits LPS-induced
autophagy of vascular endothelial cells in mouse. Apoptosis.
24:946–957. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Marchio P, Guerra-Ojeda S, Vila JM,
Aldasoro M, Victor VM and Mauricio MD: Targeting early
atherosclerosis: A focus on oxidative stress and inflammation. Oxid
Med Cell Longev. 2019(8563845)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kattoor AJ, Pothineni NV, Palagiri D and
Mehta JL: Oxidative stress in atherosclerosis. Curr Atheroscler
Rep. 19(42)2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Libby P, Bornfeldt KE and Tall AR:
Atherosclerosis: Successes, surprises, and future challenges. Circ
Res. 118:531–534. 2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Li Z, Hyseni X, Carter JD, Soukup JM,
Dailey LA and Huang YC: Pollutant particles enhanced
H2O2 production from NAD(P)H oxidase and
mitochondria in human pulmonary artery endothelial cells. Am J
Physiol Cell Physiol. 291:C357–C365. 2006.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Sita G, Hrelia P, Graziosi A and Morroni
F: Sulforaphane from cruciferous vegetables: Recent advances to
improve glioblastoma treatment. Nutrients. 10(1755)2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Huang C, Wu J, Chen D, Jin J, Wu Y and
Chen Z: Effects of sulforaphane in the central nervous system. Eur
J Pharmacol. 853:153–168. 2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yoon HY, Kang NI, Lee HK, Jang KY, Park JW
and Park BH: Sulforaphane protects kidneys against
ischemia-reperfusion injury through induction of the nrf2-dependent
phase 2 enzyme. Biochem Pharmacol. 75:2214–2223. 2008.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Chen Q, Wu S, Lu T and Chen J, Xu Z and
Chen J: The effect of sulforaphane on the activity and
mineralization of osteoblasts under oxidative stress. Pharmacology.
104:147–156. 2019.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zhu H, Jia Z, Strobl JS, Ehrich M, Misra
HP and Li Y: Potent induction of total cellular and mitochondrial
antioxidants and phase 2 enzymes by cruciferous sulforaphane in rat
aortic smooth muscle cells: Cytoprotection against oxidative and
electrophilic stress. Cardiovasc Toxicol. 8:115–125.
2008.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Feinberg MW and Moore KJ: MicroRNA
regulation of atherosclerosis. Circ Res. 118:703–720.
2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Liu Y, Zhang X, Chen J and Li T:
Inhibition of mircoRNA-34a enhances survival of human bone marrow
mesenchymal stromal/stem cells under oxidative stress. Med Sci
Monit. 24:264–271. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang S, Guo C, Yu M, Ning X, Yan B, Zhao
J, Yang A and Yan H: Identification of H2O2 induced oxidative
stress associated microRNAs in HLE-B3 cells and their clinical
relevance to the progression of age-related nuclear cataract. BMC
Ophthalmol. 18(93)2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Zhong X, Li P, Li J, He R, Cheng G and Li
Y: Downregulation of microRNA34a inhibits oxidized lowdensity
lipoproteininduced apoptosis and oxidative stress in human
umbilical vein endothelial cells. Int J Mol Med. 42:1134–1144.
2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Singh P, Hanson PS and Morris CM: SIRT1
ameliorates oxidative stress induced neural cell death and is
down-regulated in parkinson's disease. BMC Neurosci.
18(46)2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhang W, Huang Q, Zeng Z, Wu J, Zhang Y
and Chen Z: Sirt1 inhibits oxidative stress in vascular endothelial
cells. Oxid Med Cell Longev. 2017(7543973)2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Chen Z, Shentu TP, Wen L, Johnson DA and
Shyy JY: Regulation of SIRT1 by oxidative stress-responsive miRNAs
and a systematic approach to identify its role in the endothelium.
Antioxid Redox Signal. 19:1522–1538. 2013.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Dacosta C and Bao Y: The role of microRNAs
in the chemopreventive activity of sulforaphane from cruciferous
vegetables. Nutrients. 9(902)2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Rafiei H, Ashrafizadeh M and Ahmadi Z:
MicroRNAs as novel targets of sulforaphane in cancer therapy: The
beginning of a new tale? Phytother Res. 34:721–728. 2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Bignotti E, Calza S, Tassi RA, Zanotti L,
Bandiera E, Sartori E, Odicino FE, Ravaggi A, Todeschini P and
Romani C: Identification of stably expressed reference small
non-coding RNAs for microRNA quantification in high-grade serous
ovarian carcinoma tissues. J Cell Mol Med. 20:2341–2348.
2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Perrotta I and Aquila S: The role of
oxidative stress and autophagy in atherosclerosis. Oxid Med Cell
Longev. 2015(130315)2015.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Förstermann U, Xia N and Li H: Roles of
vascular oxidative stress and nitric oxide in the pathogenesis of
atherosclerosis. Circ Res. 120:713–735. 2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Vanduchova A, Anzenbacher P and
Anzenbacherova E: Isothiocyanate from broccoli, sulforaphane, and
its properties. J Med Food. 22:121–126. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Yang L, Palliyaguru DL and Kensler TW:
Frugal chemoprevention: Targeting nrf2 with foods rich in
sulforaphane. Semin Oncol. 43:146–153. 2016.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Saleh DO, Mansour DF, Hashad IM and Bakeer
RM: Effects of sulforaphane on D-galactose-induced liver aging in
rats: Role of keap-1/nrf-2 pathway. Eur J Pharmacol. 855:40–49.
2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Li B, Kim DS, Yadav RK, Kim HR and Chae
HJ: Sulforaphane prevents doxorubicin-induced oxidative stress and
cell death in rat h9c2 cells. Int J Mol Med. 36:53–64.
2015.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Campbell L, Howie F, Arthur JR, Nicol F
and Beckett G: Selenium and sulforaphane modify the expression of
selenoenzymes in the human endothelial cell line EAhy926 and
protect cells from oxidative damage. Nutrition. 23:138–144.
2007.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Sajja RK, Kaisar MA, Vijay V, Desai VG,
Prasad S and Cucullo L: In vitro modulation of redox and metabolism
interplay at the brain vascular endothelium: Genomic and proteomic
profiles of sulforaphane activity. Sci Rep. 8(12708)2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Guerrero-Beltrán CE, Calderón-Oliver M,
Pedraza-Chaverri J and Chirino YI: Protective effect of
sulforaphane against oxidative stress: Recent advances. Exp Toxicol
Pathol. 64:503–508. 2012.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Yanaka A, Zhang S, Tauchi M, Suzuki H,
Shibahara T, Matsui H, Nakahara A, Tanaka N and Yamamoto M: Role of
the nrf-2 gene in protection and repair of gastric mucosa against
oxidative stress. Inflammopharmacology. 13:83–90. 2005.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Bai Y, Wang X, Zhao S, Ma C, Cui J and
Zheng Y: Sulforaphane protects against cardiovascular disease via
nrf2 activation. Oxid Med Cell Longev. 2015(407580)2015.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Houghton CA, Fassett RG and Coombes JS:
Sulforaphane and other nutrigenomic nrf2 activators: Can the
clinician's expectation be matched by the reality? Oxid Med Cell
Longev. 2016(7857186)2016.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Su X, Jiang X, Meng L, Dong X, Shen Y and
Xin Y: Anticancer activity of sulforaphane: The epigenetic
mechanisms and the nrf2 signaling pathway. Oxid Med Cell Longev.
2018(5438179)2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Dinkova-Kostova AT, Fahey JW, Kostov RV
and Kensler TW: KEAP1 and done? Targeting the NRF2 pathway with
sulforaphane. Trends Food Sci Technol. 69:257–269. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Eren E, Tufekci KU, Isci KB, Tastan B,
Genc K and Genc S: Sulforaphane inhibits lipopolysaccharide-induced
inflammation, cytotoxicity, oxidative stress, and miR-155
expression and switches to mox phenotype through activating
extracellular signal-regulated kinase 1/2-nuclear factor erythroid
2-related factor 2/antioxidant response element pathway in murine
microglial cells. Front Immunol. 9(36)2018.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Zampetaki A, Dudek K and Mayr M: Oxidative
stress in atherosclerosis: The role of microRNAs in arterial
remodeling. Free Radic Biol Med. 64:69–77. 2013.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Chen T, Huang Z, Wang L, Wang Y, Wu F,
Meng S and Wang C: MicroRNA-125a-5p partly regulates the
inflammatory response, lipid uptake, and ORP9 expression in
oxLDL-stimulated monocyte/macrophages. Cardiovasc Res. 83:131–139.
2009.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Tong N, Jin R, Zhou Z and Wu X:
Involvement of microRNA-34a in age-related susceptibility to
oxidative stress in ARPE-19 cells by targeting the silent mating
type information regulation 2 homolog 1/p66shc pathway:
Implications for age-related macular degeneration. Front Aging
Neurosci. 11(137)2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Cheleschi S, Tenti S, Mondanelli N,
Corallo C, Barbarino M, Giannotti S, Gallo I, Giordano A and
Fioravanti A: MicroRNA-34a and microRNA-181a mediate
visfatin-induced apoptosis and oxidative stress via NF-kappaB
pathway in human osteoarthritic chondrocytes. Cells.
8(874)2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Yamakuchi M, Ferlito M and Lowenstein CJ:
MiR-34a repression of SIRT1 regulates apoptosis. Proc Natl Acad Sci
USA. 105:13421–13426. 2008.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Guo Y, Li P, Gao L, Zhang J, Yang Z,
Bledsoe G, Chang E, Chao L and Chao J: Kallistatin reduces vascular
senescence and aging by regulating microRNA-34a-SIRT1 pathway.
Aging Cell. 16:837–846. 2017.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Kurinna S and Werner S: NRF2 and
microRNAs: New but awaited relations. Biochem Soc Trans.
43:595–601. 2015.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Shah NM, Zaitseva L, Bowles KM, MacEwan DJ
and Rushworth SA: NRF2-Driven miR-125B1 and miR-29B1
transcriptional regulation controls a novel anti-apoptotic miRNA
regulatory network for AML survival. Cell Death Differ. 22:654–664.
2015.PubMed/NCBI View Article : Google Scholar
|