Introduction
Achalasia cardia (AC) is a primary esophageal
motility disorder that presents with esophageal aperistalsis and
impaired lower esophageal sphincter (LES) relaxation (1). Persistent severe AC may lead to
excessive dilation and tortuosity of the esophagus a condition
termed advanced AC (2,3). The esophageal morphology of advanced
AC, including esophageal dilation, sigmoid-shaped esophagus and
sigmoid-shaped megaesophagus, differs among individual patients.
Cases of AC who exhibit subtle differences in their clinical
presentation may exhibit distinctly different responses to current
treatment modalities (4). To date,
there is no definitive information correlating esophageal
morphology and treatment outcome.
Peroral endoscopic myotomy (POEM), a technique based
on submucosal tunneling and myotomy, was introduced into clinical
practice by Inoue et al (5)
in 2008. Now, it is the first line of treatment for AC. Despite
limited experience, POEM has demonstrated superiority in treating
advanced AC due to the capacity to perform long myotomy of the
esophagus and the relative ease in locating the initial site for
myotomy (6,7). To the best of our knowledge, however,
studies addressing advanced AC treatment in cohorts stratified by
their esophageal morphologies, which may impact the therapeutic
results, are currently sparse. In addition, a dilated and tortuous
esophageal lumen may make POEM more challenging and invasive. In
the present study, the technical feasibility, safety and clinical
efficacy of POEM in treating advanced AC were evaluated
retrospectively. The influence of atypical esophageal morphology on
treatment outcomes was determined.
Materials and methods
Patients and data collection
The present retrospective study was approved by the
Institutional Review Board for Human Research of The First
Affiliated Hospital of Zhengzhou University (Zhengzhou, China). All
patients who underwent POEM for the treatment of achalasia (n=209)
between June 2015 and March 2019 were included. The diagnosis of
achalasia was based on the following: Clinical symptoms,
radiological contrast swallow esophagography,
esophagogastroduodenoscopy, esophageal manometry and/or chest
computed tomography (CT) scan (8).
Patients who exhibited coagulopathy or severe systemic disorders,
which may lead to unsuitability for endoscopic therapy and
pseudoachalasia, were excluded.
Advanced achalasia, defined as an esophagus lumen
with a diameter of ≥6 cm and/or sigmoid in shape (9,10), was
diagnosed in 50 patients according to images obtained from a barium
swallow test and/or CT scan. All imaging was performed after
fasting overnight and in the absence of esophageal clearance. A
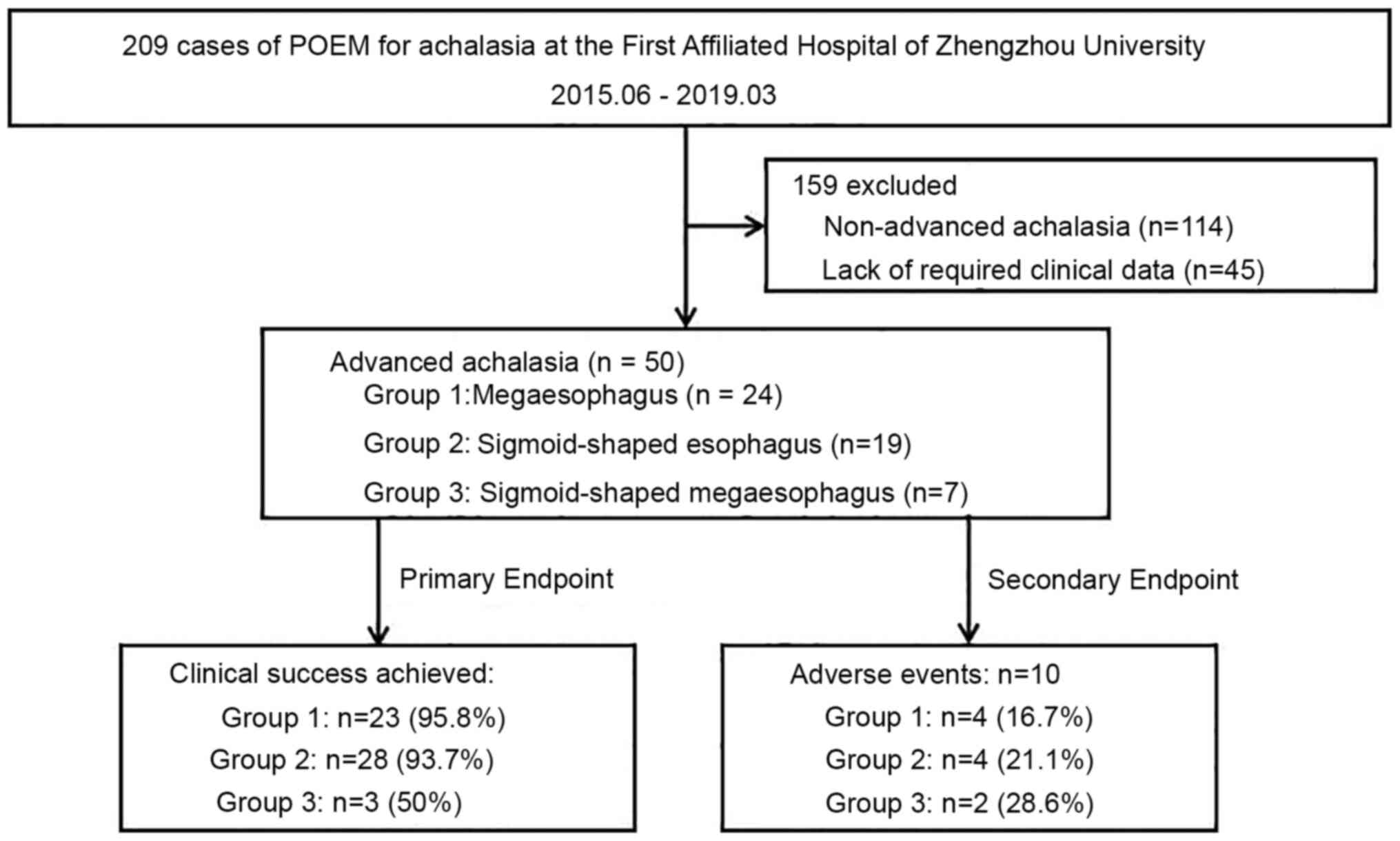
flow chart illustrating the clinical course of the present study is
presented in Fig. 1. Adverse events
(AEs) were defined according to the severity grading system of the
American Society for Gastrointestinal Endoscopy (11).
Medical files collected prospectively were used to
record baseline data, medical history, pre-POEM Eckardt score,
intra-procedure parameters, procedure-related AEs and length of
hospital stay. Medical records were also used to collect follow-up
data. Clinical outcomes were evaluated by Eckardt symptom scoring
(8). Delayed AEs were identified
based upon self-declaration and endoscopic examination performed
during follow-up consultation.
Study endpoints
The primary endpoint was the clinical response
assessed by comparing pre- and post-procedural Eckardt scores
collected at baseline and during the final follow-up visit,
respectively. The secondary endpoints included intra-procedural
details, such as operation time, length of myotomy,
procedure-related AEs and hospital stay, as well as delayed AEs,
i.e., gastroesophageal reflux disease (GERD).
Instruments and accessories
Instruments included a single-channel gastroscope
(model type, GIF-H260 or GIF-Q260J), a Hook Knife (model type,
KD-620LR) or triangle-tip knife (model type, KD-640L) and a
CO2 insufflator (model type, UCR; all from Olympus
Medical Systems Corp.). Furthermore, a high-frequency generator
(model type, ICC 200 EA INT; ERBE), injection needle (model type,
ET2522-C4; Endo-Flex), electrosurgical hemostatic forceps (model
type, FD-410LR; Olympus Medical Systems Corp.) and clips (model
type, ROCC-D-26-195; Micro-Tech Co., Ltd.; or no. HX-610-135;
Olympus Medical Systems Corp.) were used.
Endoscopic myotomy techniques
Patients were fasted 24-48 h pre-operatively; food
remnants were cleared from the esophagus prior to general
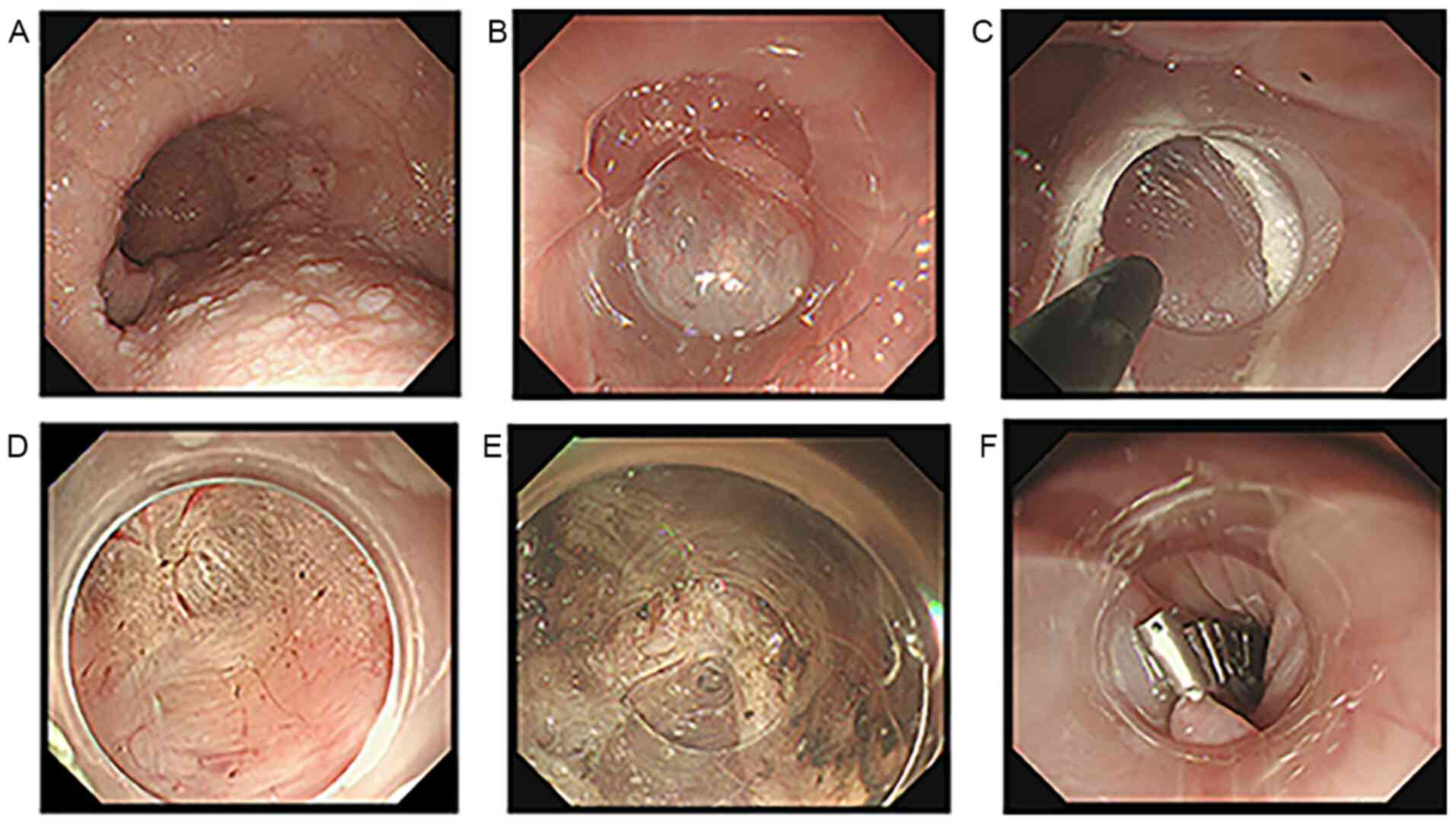
anesthesia. Conventional POEM includes the following four steps
described previously: i) Submucosal injection and mucosal incision;
ii) submucosal tunneling; iii) myotomy to the LES and cardia; and
iv) closure of the mucosal entry with endoscopic clips (Fig. 2) (12). Modified POEM incorporated tunneling
and myotomy into a single step as described by Liu et al
(13). All procedures were
performed by 3 experienced endoscopists with at least 5 years of
experience of performing conventional and modified POEM. A
full-thickness myotomy at the LES and cardia, including the
internal circular and longitudinal muscular layers, was performed
on all patients.
Post-operative management and
follow-up
Patients remained nil per os until the morning of
the post-operative day. A clear liquid diet was then provided if no
contraindications were identified. A CT scan and/or endoscopic
re-check were scheduled to evaluate the possibility and severity of
AE if any bleeding, chest pain, dyspnea, abdominal pain or
distention, or emphysema occurred post-operatively. Further
management of each patient was individualized. Asymptomatic
patients were usually discharged on day 3. Upon discharge, all
patients were prescribed a 4-week liquid diet and an oral proton
pump inhibitor for 2 weeks.
Reflux esophagitis was evaluated and the current
Eckardt score was determined at 1, 3, 6 and 12 months of follow-up.
Esophagogastroduodenoscopy was recommended if serious symptoms
occurred or refractory reflux was present.
Statistical analysis
All statistical analyses were performed using SPSS
software, version 17.0 for Windows (SPSS, Inc.). Variables were
expressed as the median (range). Count data were compared by
Chi-square tests. Groups were compared using the Wilcoxon
matched-pairs signed rank-sum test, Kruskal-Wallis test and
Chi-square test. P<0.05 was considered to indicate statistical
significance.
Results
Baseline characteristics
A total of 50 patients (22 females and 28 males)
with a median age of 49 (21-86) years were enrolled between June
2015 and March 2019 (Table I). Each
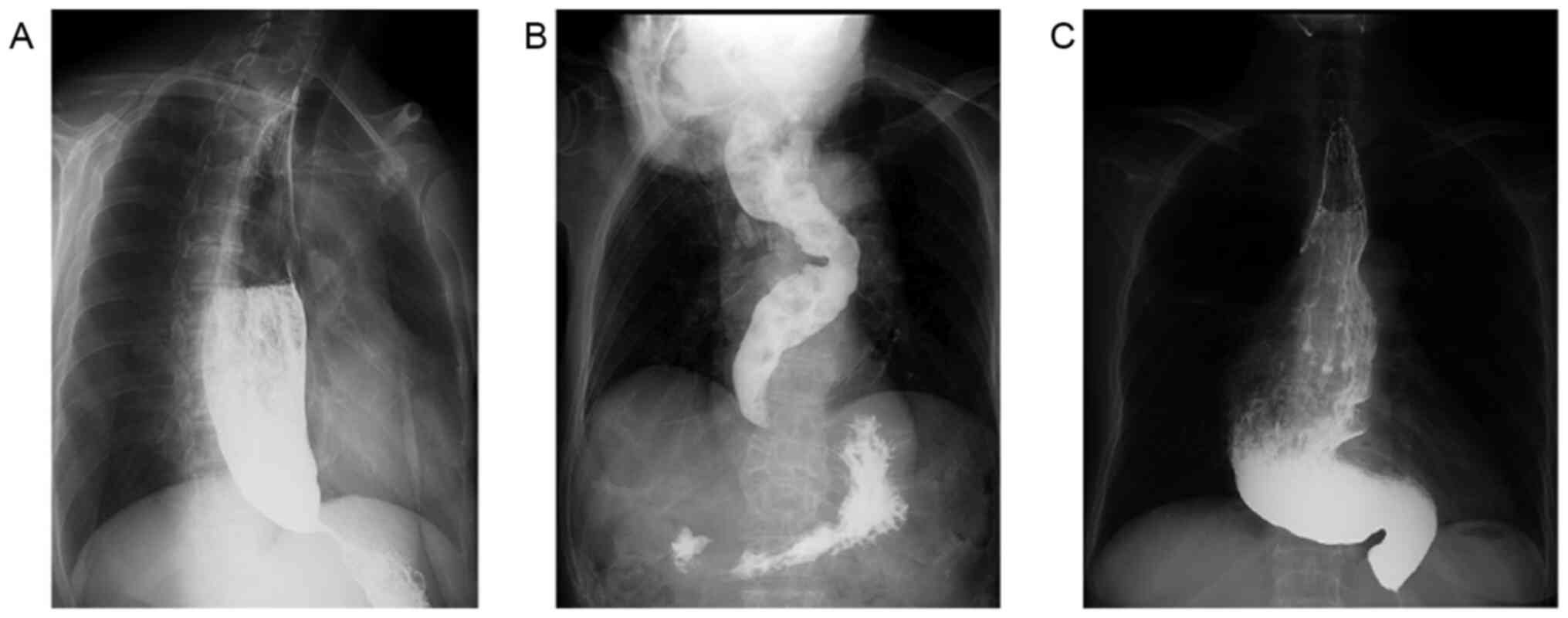
patient was categorized into one of three subgroups based upon
esophageal morphology according to the Chicago Classification:
Group 1, megaesophagus; group 2, sigmoid-shaped esophagus; and
group 3, sigmoid-shaped megaesophagus (Fig. 3). The median history of symptoms was
91 (6-600) months; 40% of patients had a history of >10 years.
The patients in group 3 usually presented a longer history than the
patients in the other two groups (i.e., 240 months compared with
108 and 60 months for group 1 and 2, respectively; Table II), but the differences were not
statistically significant. A total of 12 patients received
treatment prior to POEM [endoscopic dilation (n=8), surgical
myotomy (n=5) or both procedures (n=1)]. Treatment prior to POEM
was performed in four patients in group 3 (57.1%); this was a
significantly higher percentage of patients than in the other two
subgroups (P=0.047; Table II).
Over the course of a median of 25.3 (6-50) months, 46 patients
completed the final follow-up visit. However, three patients in
group 2 and one patient in group 3 did not complete the follow-up,
one patient died because of acute obstructive suppurative
cholangitis.
 | Table IClinical characteristics and
procedure-related parameters (n=50). |
Table I
Clinical characteristics and
procedure-related parameters (n=50).
| Variable | Value |
|---|
| Patient
characteristics | |
|
Age
(years) | 49 (21-86) |
|
Sex
(female/male) | 22/28 |
| Subtype | |
|
Megaesophagus | 24(48) |
|
Sigmoid-shaped
esophagus | 19(38) |
|
Sigmoid-shaped
megaesophagus | 7(14) |
| Symptom duration
(months) | 91 (6-600) |
| Previous
treatments | |
|
Dilatation | 8(16) |
|
Laparoscopic
Heller myotomy | 4(8) |
|
Open surgery
myotomy | 1(2) |
| Operation | |
|
POEM | 20(40) |
|
Modified
POEM | 30(60) |
| Operation time
(min) | |
|
POEM | 43 (16-163) |
|
Modified
POEM | 29 (11-65) |
| Length of myotomy
(cm) | 8 (5-14) |
| Depth of myotomy | |
|
Full-thickness
myotomy | 50(100) |
|
Circular
myotomy | 0 (0) |
| Length of hospital
stay (days) | 5.5 (3-11) |
| Adverse events | |
|
Mucosal
injury | 2(4) |
|
Bleeding | 3(6) |
|
Subcutaneous
emphysema | 3(6) |
|
Chest
pain | 5(10) |
|
Perforation | 1(2) |
 | Table IIClinical characteristics and
procedural parameters comparing three groups classified on the
basis of esophageal morphology. |
Table II
Clinical characteristics and
procedural parameters comparing three groups classified on the
basis of esophageal morphology.
| Item | Group 1:
Megaesophagus (n=24) | Group 2:
Sigmoid-shaped esophagus (n=19) | Group 3:
Sigmoid-shaped megaesophagus (n=7) | P-value |
|---|
| Sex
(female/male) | 8/16 | 9/10 | 5/2 | 0.819 |
| Age (years) | 48.5 (21-82) | 48.5 (21-82) | 57 (25-74) | 0.952 |
| Symptom duration
(years) | 108 | 60 | 240 | 0.202 |
| Interventions prior
to POEM | 6 | 2 | 4 | 0.047 |
| Procedure time
(min) | 41 (13-163) | 25 (11-62) | 41 (25-66) | 0.071 |
| Myotomy length
(cm) | 8 (6-14) | 7 (5-12) | 8 (5-12) | 0.201 |
| Hospital stay
(days) | 6 (3-9) | 5 (3-11) | 7 (4-9) | 0.524 |
| Adverse events | | | | |
|
Subcutaneous
emphysema | 2 | 1 | 0 | 0.256 |
|
Bleeding | 0 | 3 | 0 | 0.074 |
|
Mucosal
injuries | 1 | 0 | 1 | 0.749 |
|
Perforation | 0 | 1 | 0 | 0.813 |
|
Chest
pain | 2 | 2 | 1 | 0.435 |
| Eckardt
scorea | | | | |
|
Pre-treatment | 7 (3-11) | 8 (3-10) | 9 (4-10) |
6.71x10-9 |
|
Post-treatment | 1 (0-9) | 1 (0-4) | 3 (0-11) | |
| GERD score | 0 (0-3) | 0 (0-2) | 1 (0-3) | 0.202 |
| Clinical
successa | 23 (95.8%) | 15 (93.7%) | 3 (50%) | 0.004 |
| Last follow-up
visit (months) | 22.5 | 26.5 | 22.2 | 0.691 |
POEM procedure and related AEs
Endoscopic myotomy was performed successfully on all
50 patients (conventional POEM on 20 patients and modified POEM on
30 patients). The median operating time of POEM was 43 (16-163)
min, and the time of modified POEM was 29 (11-65) min. The length
of myotomy was determined endoscopically by each operator taking
the proximal condition of the esophageal lumen into account. The
median length of the esophageal myotomy was 8 (5-14) cm, albeit
longer than 10 cm in 10 patients (21.7%; Table I). The median length of
post-operative hospital stay was 5.5 (3-11) days.
Procedure-related AEs were reported in 10 patients
and were as follows: Air-related emphysema, intra-operative and
post-operative bleeding, mucosal injury, perforation and
post-operative chest pain (Table
I). Occasionally, two or more AEs occurred simultaneously in
the same patient. All AEs were mild (n=8) or moderate (n=6) and did
not require surgical conversion or admission to the intensive care
unit. None of the patients with cutaneous emphysema had dyspnea and
they were diagnosed by self-reporting and physical examination,
without drainage or decompression. The mucosal failures, which
occurred in the 3 patients who suffered mucosal injury or
perforation, were detected prior to withdrawing the endoscope and
managed similarly: Clipping, esophageal or gastric decompression
and post-operative antibiotics. A total of three cases of bleeding
occurred among the patients in group 2 (Table II). Hemostasis was achieved
intraoperatively using endoscopic vessel coagulation.
Sengstaken-Blakemore tube compression was applied after hemostasis
in one patient in order to avoid re-bleeding of the vascular stump.
The Sengstaken-Blakemore tube was removed on day 3 and the patient
was discharged on day 9 without any signs of bleeding. In a second
patient, post-operative bleeding manifested as melena and a drop in
hemoglobin on the third day after POEM. Upon endoscopic
re-examination, a submucosal hematoma was detected as a sign of
recent bleeding along the submucosal tunnel. A Sengstaken-Blakemore
tube was placed and compressed for 3 days. This patient reported no
further symptoms of bleeding and was discharged without any further
intervention.
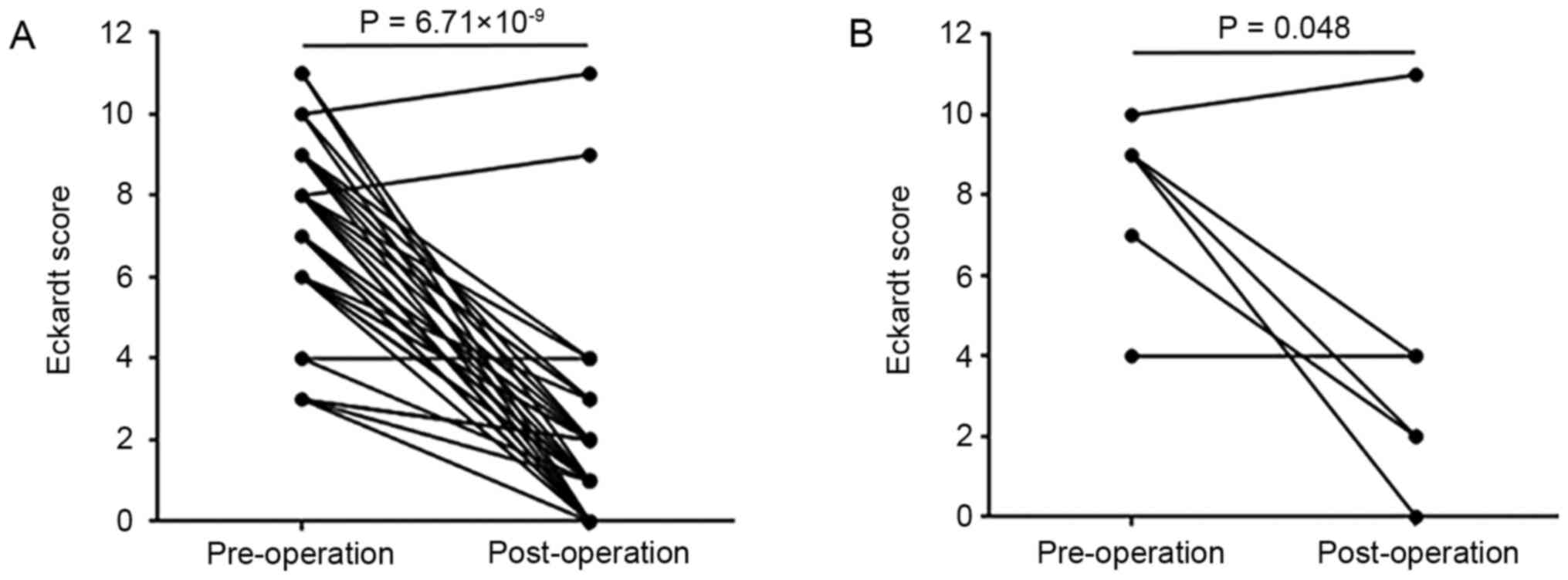
Clinical outcomes of endoscopic
myotomy on advanced AC
Clinical success, defined as a final Eckardt score
≤3 achieved during the 6- to 50-month follow-up period, occurred in
41 patients in whom the mean Eckardt score decreased from 7 (3-11)
to 1 (0-11) (Fig. 4A). A
post-procedure Eckardt score of 0 was achieved and sustained (a
complete response) in 13 patients during a 14.6 (7.2-33.8) month
follow-up period. A total of 5 patients, who did not respond
successfully, relapsed in a median time of 19.8 (15.6-38.0) months.
One of these patients never experienced any symptomatic relief and
underwent thoracic surgical myotomy 6 months after modified POEM;
the remaining 4 patients did not pursue any further intervention
after relapsing. Of note, although 50% of the patients in group 3
achieved clinical success (Fig.
4B), this rate was significantly lower than that achieved for
the other two groups (95.8% for group 1 and 93.7% for group 2;
P=0.004; Table II).
Post-operative reflux
Symptomatic reflux occurred in 13 of 46 patients who
completed their follow-up visit. Among these patients, 2
experienced episodes of reflux every day and reflux occurred in the
other patients occasionally. A total of 5 patients took a proton
pump inhibitor to relieve the symptoms of reflux; all symptoms were
readily controlled by intermittent oral medication. Upper endoscopy
was performed on 8 patients chosen at random to evaluate the
severity of reflux esophagitis. Furthermore, seven patients had
mild esophagitis (Los Angeles classification A) and 1 patient
suffered severe esophagitis (Los Angeles classification C)
(14). There was no significant
difference in the occurrence and severity of GERD among the
morphological subgroups (P=0.202; Table II).
Discussion
A limited number of studies have reported on the
clinical outcomes of POEM for advanced AC; most of these studies
focused on the sigmoid-shaped esophageal morphology (9,15-17).
In a clinical setting, however, morphologic changes in the
esophagus vary dependent upon multiple factors, such as symptomatic
history, esophageal body contraction and LES pressure. A proportion
of patients who exhibit a significantly dilated esophagus for a
relatively long period with no signs of angulation (also termed
advanced AC) experience dysfunction and poor clinical outcomes
(18). Consequently, a
retrospective analysis was performed in the present study to
determine the safety and efficacy of POEM on advanced AC
categorized based upon esophageal morphology, i.e., megaesophagus,
sigmoid-shaped esophagus and sigmoid-shaped megaesophagus.
Although laparoscopic Heller myotomy is recommended
as the initial treatment for patients with advanced achalasia,
myotomy does not provide relief of dysphagia for most of these
individuals. Rather, patients with end-stage achalasia esophagus
may ultimately require esophagectomy, where symptom resolution
reportedly occurs in 75-100% of patients (10,19-21).
Due to high risk associated with the surgery and its unclear impact
on the long-term quality of life, however, surgeons are reluctant
to perform esophagectomy for benign conditions such as achalasia.
Initial results indicated that POEM was less invasive and an
effective technique for treating advanced AC. Endoscopists reported
clinical success rates of 95.6 and 100% for sigmoid-type advanced
AC after a minimum follow-up period of 12 months (15,16).
Although the overall success rate was 89.1% in the present study,
patients with a sigmoid-shaped megaesophagus (group 3) did not
respond as well as patients in the other two subgroups. Indeed, the
clinical success rate for patients with prior treatment in group 3
was only 33.3%. Given the low incidence of advanced AC, only 7
patients with sigmoid-shaped megaesophagus were enrolled in the
present study. Therefore, these results require further
confirmation.
POEM may be technically challenging and
time-consuming in cases of advanced AC that involve morphological
changes and submucosal fibrosis caused by prior treatment (22). Mucosal damage, small perforations
and gas-related AEs were the most frequent procedure-related AEs,
which occurred in 37.5 and 34.8% of patients, respectively, in
previous studies (15,16). In the present study, the overall
incidence of AEs in the cohort was 20%. Bleeding and mucosal damage
were the most severe AEs encountered and usually required
intervention. The frequency of AEs was much lower than that
previously reported, possibly due to the fact that CT scans were
only recommended for patients who exhibited alarm symptoms
post-operatively, while previous studies routinely used CT scans
after POEM (23). Since the results
of a majority of CT scans are minor and clinically irrelevant,
routine scans are probably not warranted.
In the present study, patients with a sigmoid-shaped
esophagus were more prone to AE compared to the other two
morphological subgroups. Major intra-procedural bleeding and
post-procedural bleeding were noticed intra-operatively. Bleeding
spots came from large vessels penetrating the muscularis, which may
be complicated by angulation along the cutting direction during
myotomy. Our experience suggests that it is helpful to compress the
active bleeding site with a cap attached to the tip of the
endoscope in an effort to maintain clear vision and identify the
bleeding vessels. The addition of a water jet allows for accurate
localization when active bleeding occurs. Hemostasis with
coagulation forceps was absolutely essential when major active
bleeding occurred. Finally, Sengstaken-Blakemore tube compression
worked for both intra- and post-operative bleeding under certain
circumstances, as described previously (24). It should be emphasized that in the
present study, a Sengstaken-Blakemore tube was only used during the
early period of the POEM application, i.e., when precise submucosal
dissection and hemostasis techniques were fairly new (24). Otherwise, Sengstaken-Blakemore tubes
should only be used in carefully selected cases based upon the
bleeding site and whether hemostasis was readily achieved (24).
Despite the promising results, the present study had
several limitations. Firstly, it was a retrospective study.
Furthermore, the POEM and modified POEM procedures were performed
by different experienced endoscopists, and the operative skills may
be different among endoscopists. In addition, only 34% of cases
underwent high-resolution manometry before operation, due to
difficult placement in cases of severely dilated and tortuous
esophagi amongst patients with advanced AC.
In conclusion, the present study provided broad
support for the efficacy of the POEM procedure in treating patients
with advanced AC. POEM is also a technically feasible and safe
option in treating patients with a sigmoid-shaped megaesophagus,
although the rate of clinical success was less than that achieved
for the other treatment groups. Additional prospective, multicenter
randomized controlled studies are required.
Acknowledgements
The authors would like to thank Dr Stephen H.
Gregory (Department of Gastroenterology, Rhode Island Hospital,
Brown University, Providence, RI, USA) for his help with the
writing and editing of this manuscript.
Funding
The present study was supported by the Science and
Technology Foundation of Henan Province (grant no. 2018020115) and
the Overseas Research and Training Project of Henan Province (grant
no. 2018143).
Availability of data and materials
All data generated and analyzed in the present study
are included in this article.
Authors' contributions
DL: Conception, design and critical revision of the
manuscript. YYL and JXC: Data acquisition, preparation of the
figures and writing the first draft of the manuscript. LS, SU, YYZ,
LXZ and BH: Acquisition of the clinical data and images. QFZ, LXZ
and BH: Patient follow-up. DYL and DZH: POEM. BRL: POEM and
critical revision of the manuscript. DL and YYL confirm the
authenticity of all the raw data. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
This study was approved by the Institutional Review
Board for Human Research of The First Affiliated Hospital of
Zhengzhou University (Zhengzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Boeckxstaens GE, Zaninotto G and Richter
JE: Achalasia. Lancet. 383:83–93. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Patti MG, Gorodner MV, Galvani C, Tedesco
P, Fisichella PM, Ostroff JW, Bagatelos KC and Way LW: Spectrum of
esophageal motility disorders: Implications for diagnosis and
treatment. Arch Surg. 140:442–448; discussion 448-449.
2005.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Shiino Y, Houghton SG, Filipi CJ, Awad ZT,
Tomonaga T and Marsh RE: Manometric and radiographic verification
of esophageal body decompensation for patients with achalasia. J Am
Coll Surg. 189:158–163. 1999.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Andolfi C and Fisichella PM: Meta-analysis
of clinical outcome after treatment for achalasia based on
manometric subtypes. Br J Surg. 106:332–341. 2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Inoue H, Minami H, Kobayashi Y, Sato Y,
Kaga M, Suzuki M, Satodate H, Odaka N, Itoh H and Kudo S: Peroral
endoscopic myotomy (POEM) for esophageal achalasia. Endoscopy.
42:265–271. 2010.PubMed/NCBI View Article : Google Scholar
|
|
6
|
von Rahden BH, Filser J, Reimer S, Inoue H
and Germer CT: Peroral endoscopic myotomy for treatment of
achalasia. Literature review and own initial experience. Chirurg.
85:420–432. 2014.PubMed/NCBI View Article : Google Scholar : (In German).
|
|
7
|
Awaiz A, Yunus RM, Khan S, Memon B and
Memon MA: Systematic review and meta-analysis of perioperative
outcomes of peroral endoscopic myotomy (POEM) and laparoscopic
Heller myotomy (LHM) for achalasia. Surg Laparosc Endosc Percutan
Tech. 27:123–131. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Schlottmann F and Patti MG: Esophageal
achalasia: Current diagnosis and treatment. Expert Rev
Gastroenterol Hepatol. 12:711–721. 2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Bazerbachi F, Blackmon SH, Ravi K and Wong
Kee Song LM: Endoscopic esophagoplasty for megaesophagus with sump
stasis in end-stage achalasia. VideoGIE. 2:274–275. 2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Aiolfi A, Asti E, Bonitta G, Siboni S and
Bonavina L: Esophageal Resection for End-Stage Achalasia. Am Surg.
84:506–511. 2018.PubMed/NCBI
|
|
11
|
Cotton PB, Eisen GM, Aabakken L, Baron TH,
Hutter MM, Jacobson BC, Mergener K, Nemcek A Jr, Petersen BT,
Petrini JL, et al: A lexicon for endoscopic adverse events: Report
of an ASGE workshop. Gastrointest Endosc. 71:446–454.
2010.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Liu ZQ, Li QL, Chen WF, Zhang XC, Wu QN,
Cai MY, Qin WZ, Hu JW, Zhang YQ, Xu MD, et al: The effect of prior
treatment on clinical outcomes in patients with achalasia
undergoing peroral endoscopic myotomy. Endoscopy. 51:307–316.
2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Liu BR, Song JT and Omar Jan M: Video of
the month. Modified peroral endoscopic myotomy. Am J Gastroenterol.
110(499)2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Armstrong D, Bennett JR, Blum AL, Dent J,
De Dombal FT, Galmiche JP, Lundell L, Margulies M, Richter JE,
Spechler SJ, et al: The endoscopic assessment of esophagitis: A
progress report on observer agreement. Gastroenterology. 111:85–92.
1996.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Hu JW, Li QL, Zhou PH, Yao LQ, Xu MD,
Zhang YQ, Zhong YS, Chen WF, Ma LL, Qin WZ, et al: Peroral
endoscopic myotomy for advanced achalasia with sigmoid-shaped
esophagus: Long-term outcomes from a prospective, single-center
study. Surg Endosc. 29:2841–2850. 2015.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Lv L, Liu J, Tan Y and Liu D: Peroral
endoscopic full-thickness myotomy for the treatment of sigmoid-type
achalasia: Outcomes with a minimum follow-up of 12 months. Eur J
Gastroenterol Hepatol. 28:30–36. 2016.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kumbhari V, Tieu AH, Onimaru M, El Zein
MH, Teitelbaum EN, Ujiki MB, Gitelis ME, Modayil RJ, Hungness ES,
Stavropoulos SN, et al: Peroral endoscopic myotomy (POEM) vs
laparoscopic Heller myotomy (LHM) for the treatment of Type III
achalasia in 75 patients: A multicenter comparative study. Endosc
Int Open. 3:E195–E201. 2015.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Rhee K, Jeon H, Kim JH, Yoon YH, Park H
and Lee SI: An evidence of esophageal decompensation in patients
with achalasia in the view of its subtype: A retrospective study. J
Neurogastroenterol Motil. 19:319–323. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Salvador R, Costantini M, Zaninotto G,
Morbin T, Rizzetto C, Zanatta L, Ceolin M, Finotti E, Nicoletti L,
Da Dalt G, et al: The preoperative manometric pattern predicts the
outcome of surgical treatment for esophageal achalasia. J
Gastrointest Surg. 14:1635–1645. 2010.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Aiolfi A, Asti E, Bonitta G and Bonavina
L: Esophagectomy for end-stage achalasia: Systematic review and
meta-analysis. World J Surg. 42:1469–1476. 2018.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Devaney EJ, Lannettoni MD, Orringer MB and
Marshall B: Esophagectomy for achalasia: Patient selection and
clinical experience. Ann Thorac Surg. 72:854–858. 2001.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Wu QN, Xu XY, Zhang XC, Xu MD, Zhang YQ,
Chen WF, Cai MY, Qin WZ, Hu JW, Yao LQ, et al: Submucosal fibrosis
in achalasia patients is a rare cause of aborted peroral endoscopic
myotomy procedures. Endoscopy. 49:736–744. 2017.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Cai M-Y, Zhou P-H, Yao L-Q, Zhu B-Q, Liang
L and Li Q-L: Thoracic CT after peroral endoscopic myotomy for the
treatment of achalasia. Gastrointest Endosc. 80:1046–1055.
2014.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Li QL, Zhou PH, Yao LQ, Xu MD, Chen WF, Hu
JW, Cai MY, Zhang YQ, Zhong YS, Qin WZ, et al: Early diagnosis and
management of delayed bleeding in the submucosal tunnel after
peroral endoscopic myotomy for achalasia (with video). Gastrointest
Endosc. 78:370–374. 2013.PubMed/NCBI View Article : Google Scholar
|