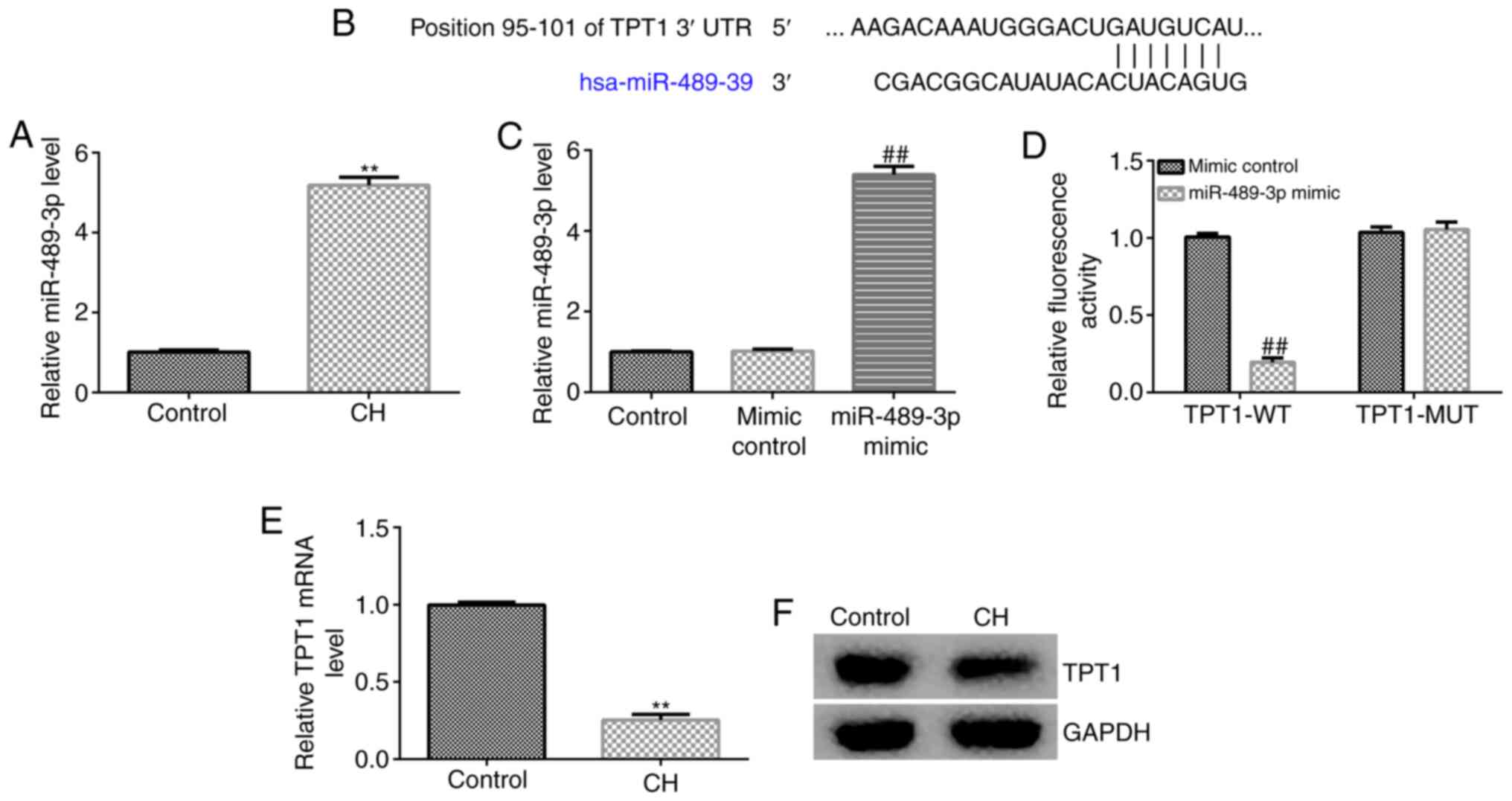
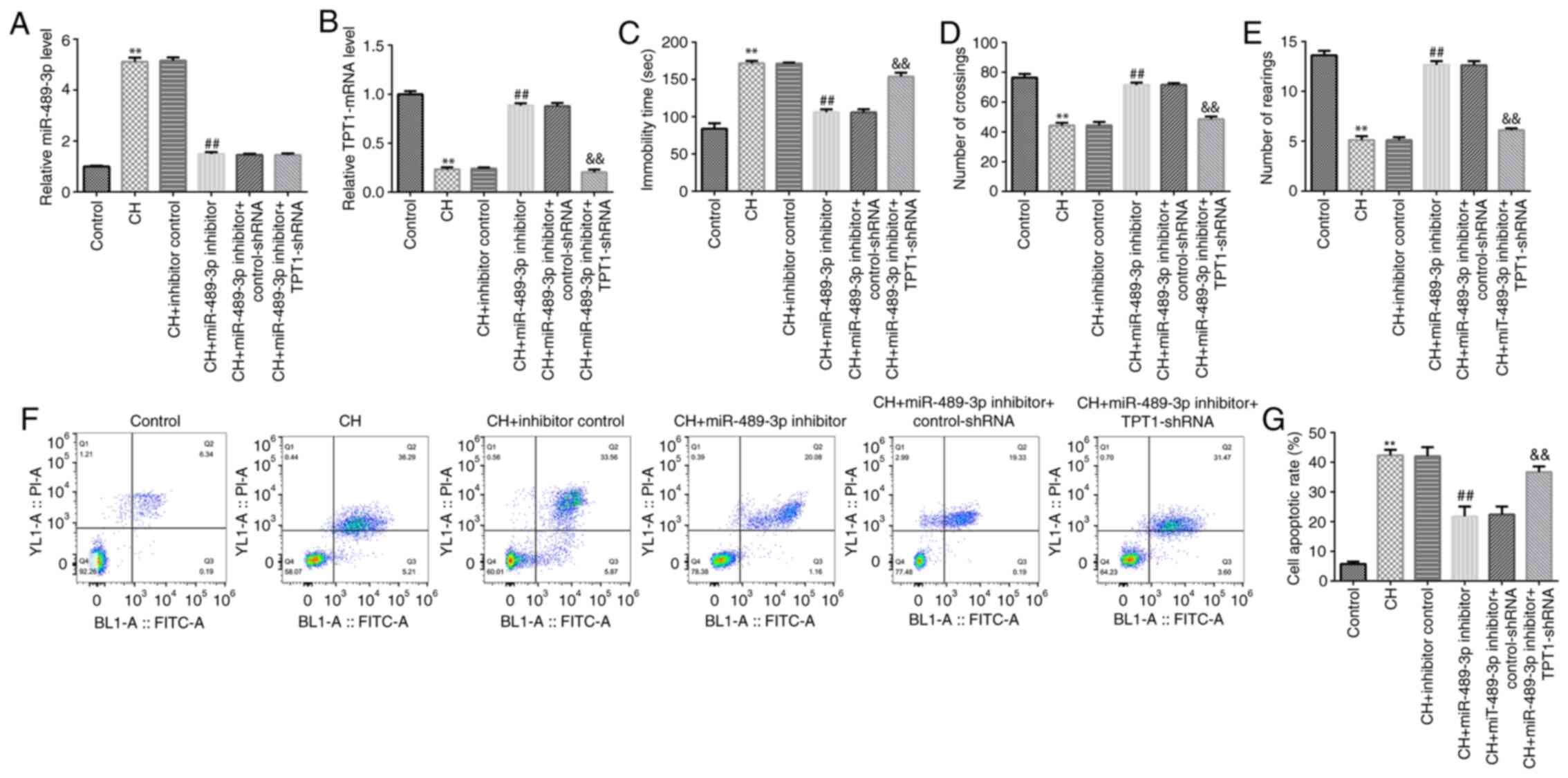
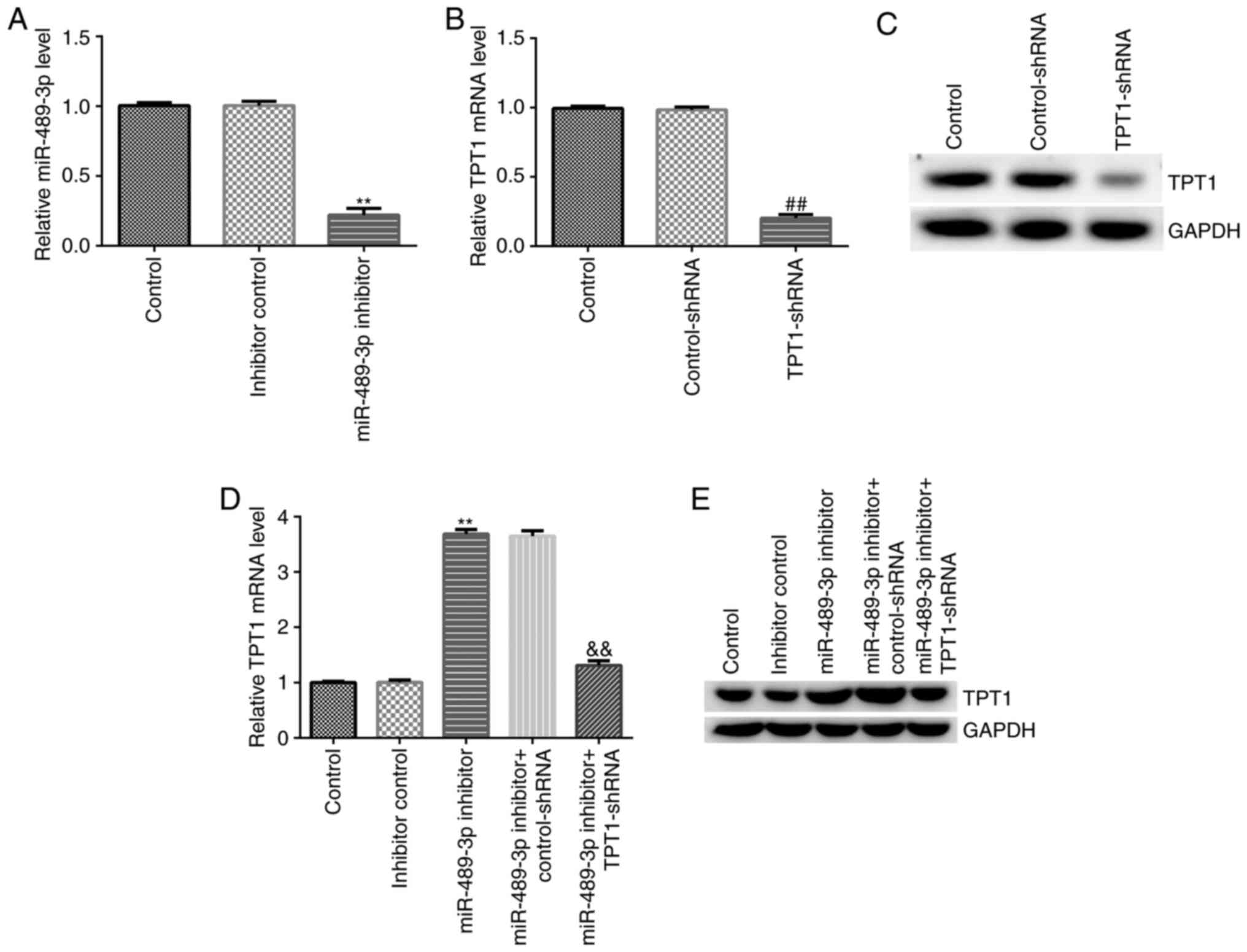
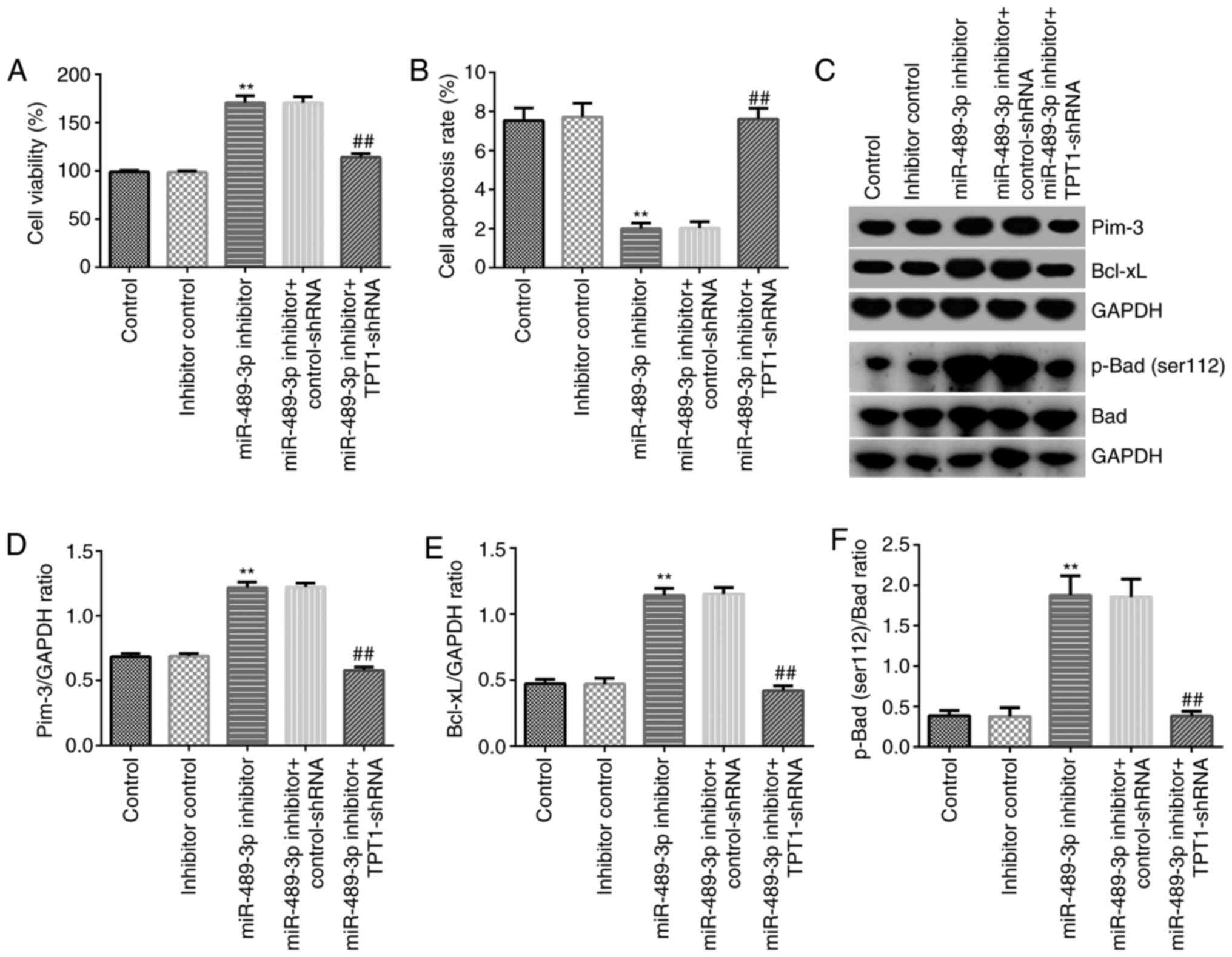
|
1
|
Rousseau JP, Buteau-Poulin A and Kinkead
R: Maternal thyroid hormone deficiency and cardiorespiratory
disorder in rat pups. Exp Neurol. 320(112960)2019.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Tiosano D, Pannain S, Vassart G, Parma J,
Gershoni-Baruch R, Mandel H, Lotan R, Zaharan Y, Pery M, Weiss R,
et al: The hypothyroidism in an inbred kindred with congenital
thyroid hormone and glucocorticoid deficiency is due to a mutation
producing a truncated thyrotropin receptor. Thyroid. 9:887–894.
1999.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Zhang L, Blomgren K, Kuhn HG and
Cooper-Kuhn CM: Effects of postnatal thyroid hormone deficiency on
neurogenesis in the juvenile and adult rat. Neurobiol Dis.
34:366–374. 2009.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Meng T, Shen S, Li C and Liu X:
MicroRNA-1236-3p/translationally controlled tumor protein (TPT1)
axis participates in congenital hypothyroidism progression by
regulating neuronal apoptosis. Exp Ther Med. 19:459–466.
2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Alcigir ME, Dogan HO, Atalay VS and Yilmaz
FM: Neuroprotective activity of cannabinoid receptor-2 against
oxidative stress and apoptosis in rat pups having
experimentally-induced congenital hypothyroidism. Dev Neurobiol.
77:1334–1347. 2017.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Akin MA, Aydogan S, Gunes T, Artis AS,
Karakukcu M and Kurtoglu S: Changes of red blood cell rheology in
newborns with congenital hypothyroidism during treatment. J Matern
Fetal Neonatal Med. 26:1532–1536. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Vetrovoy O, Sarieva K, Lomert E,
Nimiritsky P, Eschenko N, Galkina O, Lyanguzov A, Tyulkova E and
Rybnikova E: Pharmacological HIF1 inhibition eliminates
down-regulation of the pentose phosphate pathway and prevents
neuronal apoptosis in rat hippocampus caused by severe hypoxia. J
Mol Neurosci. 70:635–646. 2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Shao Q, Jiang W and Jin Y: MiR-124 effect
in neurons apoptosis in newborn rat with thyroid hypofunction. Int
J Clin Exp Pathol. 8:14465–14471. 2015.PubMed/NCBI
|
|
9
|
Li W, Song D, Sun Y, Lv Y and Lv J:
microRNA-124-3p inhibits the progression of congenital
hypothyroidism via targeting programmed cell death protein 6. Exp
Ther Med. 15:5001–5006. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Huang XW, Yin HM, Ji C, Qin YF, Yang RW
and Zhao ZY: Effects of perinatal hypothyroidism on rat behavior
and its relation with apoptosis of hippocampus neurons. J
Endocrinol Invest. 31:8–15. 2008.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Schira-Heinen J, Czapla A, Hendricks M,
Kloetgen A, Wruck W, Adjaye J, Kögler G, Werner Müller H, Stühler K
and Trompeter HI: Functional omics analyses reveal only minor
effects of microRNAs on human somatic stem cell differentiation.
Sci Rep. 10(3284)2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Omidkhoda N, Wallace HA, Reiter RJ and
Karimi G: The role of microRNAs on endoplasmic reticulum stress in
myocardial ischemia and cardiac hypertrophy. Pharmacol Res.
150(104516)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wu S, Wang J, Li J and Li F: microRNA-21
aggravates lipopolysaccharide-induced inflammation in MH7A cells
through targeting SNF5. Inflammation. 43:441–454. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Sohrabifar N, Ghaderian S, Vakili H,
Ghaedi H, Rouhani B, Jafari H and Heidari L: MicroRNA-copy number
variations in coronary artery disease patients with or without type
2 diabetes mellitus. Arch Physiol Biochem. 1–7. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
You D, Wang D, Liu P, Chu Y, Zhang X, Ding
X, Li X, Mao T, Jing X, Tian Z and Pan Y: MicroRNA-498 inhibits the
proliferation, migration and invasion of gastric cancer through
targeting BMI-1 and suppressing AKT pathway. Hum Cell. 33:366–376.
2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zhou X, Lu H, Li F, Hao X, Han L, Dong Q
and Chen X: MicroRNA-429 inhibits neuroblastoma cell proliferation,
migration and invasion via the NF-κB pathway. Cell Mol Biol Lett.
25(5)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Chen L, Chen L, Qin Z, Lei J, Ye S, Zeng
K, Wang H, Ying M, Gao J, Zeng S and Yu L: Up-regulation of
miR-489-3p and miR-630 inhibits oxaliplatin uptake in renal cell
carcinoma by targeting OCT2. Acta Pharm Sin B. 9:1008–1020.
2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Kuppa SS, Jia W, Liu S, Nguyen H, Smyth
SS, Mills GB, Dobbin KK, Hardman WJ and Murph MM: Autotaxin
exacerbates tumor progression by enhancing MEK1 and overriding the
function of miR-489-3p. Cancer Lett. 432:84–92. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Sun X, Su S, Zhang G, Zhang H and Yu X:
MiR-204 suppresses cell proliferation and promotes apoptosis in
ovarian granulosa cells via targeting TPT1 in polycystic ovary
syndrome. Biochem Cell Biol. 97:554–562. 2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Neuhäuser K, Küper L, Christiansen H and
Bogdanova N: Assessment of the role of translationally controlled
tumor protein 1 (TPT1/TCTP) in breast cancer susceptibility and ATM
signaling. Clin Transl Radiat Oncol. 15:99–107. 2019.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Li R, Zhu H, Yang D, Xia J and Zheng Z:
Long noncoding RNA lncBRM promotes proliferation and invasion of
colorectal cancer by sponging miR-204-3p and upregulating TPT1.
Biochem Biophys Res Commun. 508:1259–1263. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zhang L, Wang Q, Wang F, Zhang X, Zhang L,
Tang Y and Wang S: LncRNA LINC01446 promotes glioblastoma
progression by modulating miR-489-3p/TPT1 axis. Biochem Biophys Res
Commun. 503:1484–1490. 2018.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Fabian ID, Rosner M, Fabian I,
Vishnevskia-Dai V, Zloto O, Shinderman Maman E, Cohen K, Ellis M,
Lin HY, Hercbergs A, et al: Low thyroid hormone levels improve
survival in murine model for ocular melanoma. Oncotarget.
6:11038–11046. 2015.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Sidibé el H: Reflections on mental
retardation and congenital hypothyroidism: Effects of trace mineral
deficiencies. Sante. 17:41–50. 2007.PubMed/NCBI(In French).
|
|
26
|
Boen J, Gevaert AB, De Keulenaer GW, Van
Craenenbroeck EM and Segers V: The role of endothelial miRNAs in
myocardial biology and disease. J Mol Cell Cardiol. 138:75–87.
2020.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Segaran RC, Chan LY, Wang H, Sethi G and
Tang FR: Neuronal development-related miRNAs as biomarkers for
Alzheimer's disease, Depression, Schizophrenia and Ionizing
Radiation Exposure. Curr Med Chem: Jan 21, 2020 (Epub ahead of
print) doi: 10.2174/092986732766620012112291.
|
|
28
|
Sadlon A, Takousis P, Alexopoulos P,
Evangelou E, Prokopenko I and Perneczky R: miRNAs identify shared
pathways in Alzheimer's and Parkinson's diseases. Trends Mol Med.
25:662–672. 2019.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Condrat CE, Thompson DC, Barbu MG, Bugnar
OL, Boboc A, Cretoiu D, Suciu N, Cretoiu SM and Voinea SC: miRNAs
as biomarkers in disease: Latest findings regarding their role in
diagnosis and prognosis. Cells. 9(276)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Jian M, Du Q, Zhu D, Mao Z, Wang X, Feng
Y, Xiao Z, Wang H and Zhu Y: Tumor suppressor miR-145-5p sensitizes
prolactinoma to bromocriptine by downregulating TPT1. J Endocrinol
Invest. 42:639–652. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Dong J, Liu Y, Zhan Z and Wang X:
MicroRNA-132 is associated with the cognition improvement following
voluntary exercise in SAMP8 mice. Brain Res Bull. 140:80–87.
2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Li LM, Luo FJ and Song X: MicroRNA-370-3p
inhibits cell proliferation and induces chronic myelogenous
leukaemia cell apoptosis by suppressing PDLIM1/Wnt/β-catenin
signalling. Neoplasma. 67:509–518. 2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Wu H, Wang X, Wu T and Yang S: miR-489
suppresses multiple myeloma cells growth through inhibition of
LDHA-mediated aerobic glycolysis. Genes Genomics. 42:291–297.
2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Gao S, Liu H, Hou S, Wu L, Yang Z, Shen J,
Zhou L, Zheng SS and Jiang B: MiR-489 suppresses tumor growth and
invasion by targeting HDAC7 in colorectal cancer. Clin Transl
Oncol. 20:703–712. 2018.PubMed/NCBI View Article : Google Scholar
|
|
35
|
You Y, Tan J, Gong Y, Dai H, Chen H, Xu X,
Yang A, Zhang Y and Bie P: MicroRNA-216b-5p functions as a
tumor-suppressive RNA by targeting TPT1 in pancreatic cancer cells.
J Cancer. 8:2854–2865. 2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zan T, Piao L, Yang X, Gu Y and Liu B:
Down-regulation of microRNA-124 prevents the development of acute
liver failure through the upregulation of PIM-3. Exp Physiol.
105:108–119. 2020.PubMed/NCBI View
Article : Google Scholar
|
|
37
|
Fan X, Xie Y, Zhang L, Gao X, Han J, Chen
Y, Yang J and Li S: Effect of Pim-3 down-regulation on
proliferation and apoptosis in lung adenocarcinoma A549 cells. Ann
Clin Lab Sci. 49:770–776. 2019.PubMed/NCBI
|
|
38
|
Liu J, Qu X, Shao L, Hu Y, Yu X, Lan P,
Guo Q, Han Q, Zhang J and Zhang C: Pim-3 enhances melanoma cell
migration and invasion by promoting STAT3 phosphorylation. Cancer
Biol Ther. 19:160–168. 2018.PubMed/NCBI View Article : Google Scholar
|