Introduction
Strokes are the second leading cause of mortality
worldwide (1). In China, the
age-adjusted incidence of a first-time stroke is not markedly
different compared with developed countries (2). Post-stroke depression (PSD) is a
common occurrence following a stroke (3,4), and
is associated with increased disability, cognitive impairment,
suicidal behavior and mortality (1). PSD has been estimated to affect 33% of
patients worldwide (5-7).
Although the mechanisms underlying the development of PSD remain
largely unknown, a number of hypotheses, including a neurotrophic
effect, have been proposed (8).
Studies have documented that stress decreases the expression of
brain-derived neurotrophic factor (BDNF) mRNA in the hippocampus
(9,10), and direct hippocampal infusions of
BDNF protein can produce antidepressant effects in rodents
(11). In addition, it has been
demonstrated that serum concentrations of BDNF are decreased in
human patients with PSD (12). BDNF
may act through high-affinity interactions with its specific
receptor, tyrosine kinase B (TrkB) (13,14),
as mice lacking TrkB have been demonstrated to exhibit increased
anxiety-like behaviors (15).
Notably, expression of BDNF is partly regulated by cAMP response
element-binding protein (CREB), which is a cellular transcription
factor (16). Dysregulation of CREB
expression or activity has been associated with mood disorders,
including depression (17). In
addition, it has been reported that CREB and the BNDF-TrkB
signaling can form a positive feedback loop (16,18),
CREB can be activated by TrkB signaling through phosphorylation,
and phosphorylated (p)CREB will subsequently activate BDNF to
enhance TrkB signaling (19).
Estrogen is an effective mood regulator that has been demonstrated
to induce antidepressant like effects on a depression model in rats
(20). Estrogen also serves a
neuroprotective role through estrogen-dependent alterations in cell
survival (21), enhancement of
anti-apoptotic gene expression and neurogenesis (22). A previous study demonstrated that
17β-estradiol (E2), which is an antidepressant, can ameliorate PSD.
The similarities between the effects of estrogen and BDNF on
hippocampal physiology and behavior have also been observed
(23-25).
Therefore, the present study hypothesized that CREB/BDNF/TrkB
signaling may serve a role in the E2-mediated improvement of PSD in
rats. In the present study, the CREB/BDNF/TrkB signaling activation
in PSD rats was investigated following moderate doses of E2
treatment. The results of the present study may provide novel
insights into the molecular basis of E2-mediated improvement of
PSD.
Materials and methods
Animals
A total of 120 two-month-old female Sprague-Dawley
(SD) rats, weighing between 220 and 250 g, were obtained from the
Animal Center of Wenzhou Medical University in China. The rats were
housed four per cage and maintained on a 12 h light/dark cycle,
with lights turned on at 7:00 a.m. and temperature at 21±2˚C; and
provided with ad libitum access to food and water. The
groups of ovariectomized (OVX) female SD rats used included the
following: i) Sham-Operated rats treated with vehicle (control +
vehicle; n=16); ii) PSD rats treated with vehicle (PSD + vehicle;
n=16); iii) PSD rats treated with E2 (PSD + E2; n=16); iv) PSD rats
treated with K252a and E2 (PSD + K252a + E2; n=16); v) PSD rats
treated with U0126 and E2 (PSD + U0126 + E2, n=16). PSD rats were
randomly divided into matched subgroups for subsequent K252a and
U0126 with vehicle treatment as follows: PSD rats treated with
K252a and vehicle groups (PSD + K252a + vehicle; n=8) and PSD rats
treated with U0126 vehicle groups (PSD + U0126 + vehicle; n=8). PSD
rats were subcutaneously injected with E2 at 1, 10 or 25 µg (n=8
per group). All experiments were performed in accordance with the
National Institutes of Health Guide for the Care and Use of
Laboratory Animals (26), with the
approval of the Institutional Animal Care and Use Committee at
First Affiliated Hospital, Wenzhou Medical University, Wenzhou. The
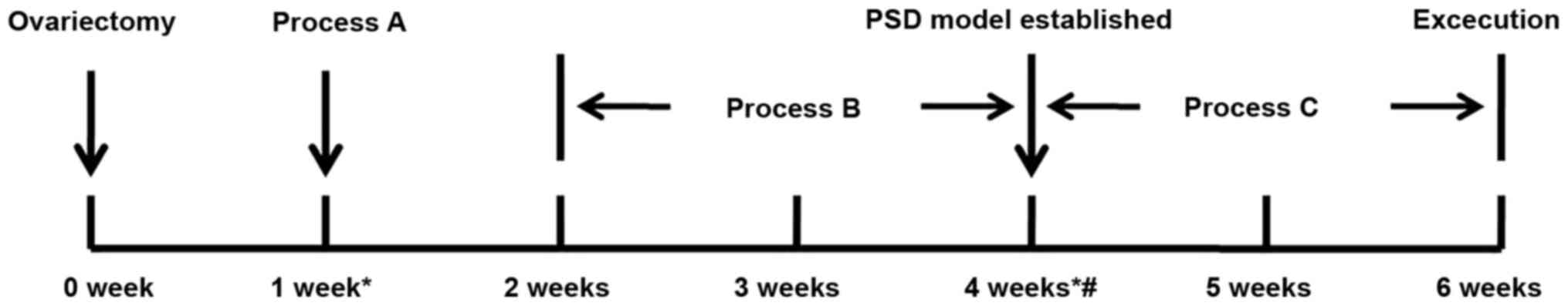
experimental paradigm is described in (Fig. 1). Process A indicates the middle
cerebral artery occlusion. Process B indicates the chronic mild
stress. Process C indicates the estrogen or oil treatment.
OVX surgery
Female SD rats were anesthetized via intraperitoneal
injection with 10% chloral hydrate (320 mg/kg) and showed no
obvious signs of peritonitis. All rats were subjected to an
ovariectomy prior to grouping. Bilateral ovariectomy was performed
according to previously published procedures (27). Briefly, a single midline incision
was made in the lower abdominal area to expose the ovary, and
oviducts were bilaterally ligated and the ovaries removed. After
suturing the muscles and skin, the animals were returned to their
cages to recover for 1 week in the same conditions as
aforementioned.
Transient focal cerebral ischemia
After 1 week had passed, the OVX rats were subjected
to focal ischemia as previously described (24,28).
Briefly, the rats were anesthetized with 10% chloral hydrate (320
mg/kg) and the left common carotid artery was exposed. The external
carotid artery and its branches were subsequently isolated and
coagulated. A 3-0 nylon suture with a blunted tip was inserted into
the internal carotid artery through the external carotid artery
stump, which advanced to the anterior cerebral artery to occlude
the middle cerebral artery (MCA). After occluding the MCA for 90
min, the suture was removed to restore blood flow, and the rats
were placed in a cage under an infrared heating lamp at 35±2˚C
until they had recovered from anesthesia. Rectal temperature was
maintained at 37.0±0.5°C using a thermostat-controlled
heating pad. Sham-operated rats underwent the same surgery and
recovery protocol, except that the MCA was not occluded.
Chronic mild stress
A period of 1 week post-MCAO, the animals were
subjected to chronic mild stress (CMS) for 2 consecutive weeks.
Each week, the stress regime included: i) Food or water deprivation
for 12 h; ii) Cage tilt (45˚); iii) Overnight illumination; iv)
Soiled cage (250 ml water in sawdust bedding); v) Swimming in 4˚C
water; and vi) Placement in a restraining device as previously
described (20,24). Each rat received one form of stress
each day. Control animals were housed in separate rooms and did not
undergo the stress regime.
Behavior tests Open-field test
The open-field test consisted of a wooden box (100
cm in length and 100 cm in width) with the wall painted black (40
cm high); the non-reflective floor was divided into 25 (5x5) equal
squares (20x20 cm). Each room test was dimly illuminated with one
25 W red bulb located 130 cm above the open-field floor. The
animals were placed at the center of the open field arena and
tested in a quiet room. The frequency of rearing and line crossing
activity of the test subject was quantified over a 5-min period by
trained and experienced observers were blinded to the treatment
groups. Rats crossing the line were scored when all four paws were
removed from one square and entered another. Rears were scored when
both front paws were raised from the floor, and climbs were scored
when an animal leaned its front paws against a wall. Open field
activities scores were monitored in all groups at baseline, at 2
weeks after CMS and at 2 weeks following treatment.
Sucrose preference tests
After 1 week of acclimatization to the Animal
Center, rats were trained to consume 1% (w/v) sucrose solution, as
previously described (24). A
period of 3 days after this, and after 23 h of food and water
deprivation, a 1 h baseline test was performed, in which rats could
select between two pre-weighted bottles, one with 1% (w/v) sucrose
solution and the other with tap water. The maximum percentage of
body weight loss following the 23 h food and water deprivation was
as follows: Baseline group 5.9% vs. 2W CMS group 5.5% vs. 2W
Treatment group 5.4%.
The position of the two bottles in the 1 h baseline
test (left/right sides of the cages) varied randomly between
trials. The sucrose preference tests were performed at 2 weeks
following CMS and at 2 weeks following treatment under similar
conditions throughout the experiment. The relative sucrose intake
(g/g) was the absolute sucrose intake per gram of rat body weight,
whereas sucrose preference (SP) was calculated according to the
following ratio: SP=sucrose intake (g)/[sucrose intake (g) + water
intake (g)].
E2 administration
PSD rats were injected subcutaneously with 10 µg E2
(Sigma-Aldrich; Merck KGaA) in 0.1 ml sesame oil at 09:00 and 10:00
am for 14 consecutive days from the establishment of the PSD model.
The PSD group and control group rats received 0.1 ml sesame oil
using the same protocol.
Subventricle zone (SVZ) infusions of
K252a and U0126
PSD rats were anesthetized with 10% chloral hydrate
(320 mg/kg) prior to stereotaxic surgery. A small burr hole was
drilled in the left hemisphere of the brain and a stainless steel
needle (26 gauge) was inserted into the left ventricle (coordinates
0.9 mm posterior from bregma, 1.5 mm from the midline and 4.0 mm
from the brain surface). After fixing the guide cannulas to the
skull, a substitute cannula (length, 8.5 mm; external diameter,
0.35 mm) was inserted into the guide cannula to prevent a block of
tubing. The guide cannula was removed 3 days later, and the wound
was disinfected. K252a, a TrkB specific blocker, was dissolved in
1% sterilized DMSO and prepared into a 40 mmol/l solution. Each 3
µl liquid contained 120 nmol K252a (10). K252a solution (3 µl) was injected
into the lateral ventricle (29).
U0126 is a mitogen activated protein kinase (MAPK) blocker that
inhibits CREB phosphorylation and dissolves U0126 in 1% DMSO (0.3
g/µl), 3 µl of which was injected into the lateral ventricle of the
rats in the present study (30,31).
Each PSD rat was randomly assigned to artificial cerebrospinal
fluid (aCSF), U0126 and K252a groups. Each drug (3 µl in total) was
unilaterally injected into the lateral ventricle via an injection
cannula (external diameter, 0.35 mm) extending 0.5 mm below the tip
of the guide cannula at a rate of 1 µl/min using a 10 ml Hamilton
syringe. Drug administration was performed continuously for 3 days.
After establishing the model, rats were treated with E2 (10 µg) or
vehicle for 2 weeks.
Measurement of serum E2
After 2 weeks of (1, 10 or 25 µg) E2 therapy, rats
were anesthetized with 10% chloral hydrate at a dose of 320 mg/kg,
the abdominal cavity was exposed, the abdominal aorta was isolated,
and 2-3 ml of blood was extracted. Cervical dislocation was used as
the method of euthanasia. No rat exhibited signs of peritonitis
after the administration of 10% chloral hydrate. Rat deaths were
confirmed by observing breathing and cardiac arrest. Glass tubes
containing whole blood from the abdominal aorta were kept at room
temperature for 1-2 h and then centrifuged at 4°C, 1,800
x g for 15 min. Serum was collected into glass vials and stored at
-80°C. Serum levels of estradiol were sent to the
Guangzhou Kingmed Diagnostics Group Co., Ltd. for testing via
Enhanced Estradiol Assay kit (cat. no. CLA-4664; DRG Instruments
GmbH). The ADVIA Centaur Automatic chemiluminescence immunoassay
system (Siemens AG) was used to assay the serum E2 levels,
according to the manufacturer's protocol. The endogenous estradiol
contained in the sample was released from its binding protein using
an estradiol assay kit release agent. The anti-estradiol rabbit
polyclonal antibody (1:100; cat. no. CLA-4664; DRG Instruments
GmbH) labeled with acridine ester was then added to bind the
available estradiol. Finally, estradiol derivatives were added to
the reactants to capture the solid phase and bind to estradiol
competitively and acridin-labeled antibodies. After washing, acids
and bases are added to start the chemiluminescence reaction for 10
min at room temperature. All experiments were performed twice. The
lower level of detectability was 3 pg/ml.
Immunohistochemistry
Animals were sacrificed after 2 weeks of treatment.
Rats were anesthetized with 10% chloral hydrate and decapitated.
The rat brains were fixed in 4% paraformaldehyde in PBS for 24 h at
4°C, and then dehydrated in a graded series of alcohol,
placed in xylene until transparent and embedded in paraffin via
treatment at 60°C oven. Brain tissues were cut to 4
mm-thick sections using a microtome. The sections were blocked in
3% H2O2 for 10 min at room temperature, and
3% normal goat serum (OriGene Technologies, Inc.), and then
incubated with rabbit polyclonal antibodies against phospho cAMP
response element-binding protein (pCREB; 1:800; cat. no. 9198; Cell
Signaling Technology, Inc.), CREB (1:3,000; cat. no. 9197; Cell
Signaling Technology, Inc.), BDNF (1:200; cat. no. ab6201; Abcam)
at 4˚C overnight. HRP-labeled goat anti-rabbit secondary antibody
(1:1,000; cat. no. TA140003; OriGene Technologies, Inc.) for 30 min
at 37˚C and diaminobenzidine from the Streptavidin-Peroxidase kit
(OriGene Technologies, Inc.) were used to visualize the signals.
Hematoxylin was used for counterstaining for 1-2 min at room
temperature. The controls included incubation without any primary
or secondary antibodies at 37°C for 30 min. Finally, the
sections were observed under a NIKON fluorescence microscope
(magnification, x40, x200 and x400; Nikon Corporation).
Western blot analysis
The hippocampus was also dissected from rats
euthanized via cervical dislocation. The tissue samples were
homogenized in ice-cold lysis buffer and phenylmethanesulfonyl
fluoride. The homogenates were centrifuged at 4°C and
21,900 x g for 15 min. Protein concentrations were measured using a
BCA Protein Assay Reagent kit (Beyotime Institute of
Biotechnology). Protein (50 µg per lane) was loaded and separated
via 12% SDS-PAGE and transferred onto PVDF membranes (EMD
Millipore). After blocking for 90 min at room temperature with 5%
non-fat dried milk in TBS, the membranes were incubated overnight
at 4°C with anti-β-actin (1:1,000; cat. no. ab8226;
Abcam), polyclonal anti-CREB (1:5,000; cat. no. 9197; Cell
Signaling Technology, Inc.), anti-phospho-CREB (1:5,000; cat. no.
9198; Cell Signaling Technology, Inc.) at Ser133, anti-BDNF
(1:1,000; cat. no. ab6201; Abcam), anti-pTrkB (1:3,000; cat. no.
4168; Cell Signaling Technology, Inc.) or TrkB (1:3,000; cat. no.
4603; Cell Signaling Technology, Inc.) and shaken on a rotator at
4˚C for overnight. The membranes were washed three times with TBS
containing 0.1% Tween-20 (TBST) for 7 min each, and subsequently
incubated in HRP-labeled goat anti-rabbit secondary antibody
(1:5,000; cat. no. A0208; Beyotime Institute of Biotechnology) with
TBST for 1 h at room temperature. Membranes were then washed three
times with TBST for 10 min each. The resulting
antigen-antibody-peroxidase complexes were detected by an ECL
detection system (Beyotime Institute of Biotechnology).
Densitometric analysis of the western blot images was performed
using a Bio-Rad GS-710 Calibrated Imaging Densitometer and
quantified using Quantity One software (Bio-Rad Laboratories, Inc;
version 4.5.2).
Statistical analysis
Significant differences among groups were determined
using one-way ANOVA using SPSS software (version 23.0; IBM, Corp).
Post-hoc comparisons were performed using Tukey's post hoc test.
All values are reported as the mean ± standard error of the mean.
P<0.05 was considered to indicate a statistically significant
difference.
Results
Successful PSD model
To establish a PSD model, rats that had undergone
surgical removal of the ovaries were subjected to MCAO and 2 weeks
of CMS (Fig. 1). OVX rats without
MCAO and CMS were used as controls. As presented in Table I, the rats undergoing CMS displayed
a significant decrease in crossing activity and rearing activity
compared with control rats (P<0.05), and they drank sucrose
significantly less compared with the control rats (P<0.05;
Table II), suggesting that these
rats had developed PSD.
 | Table IOpen field activities scores in all
groups at baseline, at 2 weeks after CMS and at 2 weeks following
treatment. |
Table I
Open field activities scores in all
groups at baseline, at 2 weeks after CMS and at 2 weeks following
treatment.
| | Crossing
activity | Rearing
activity |
|---|
| Group (n) | Baseline | 2W CMS | 2W Treatment | Baseline | 2W CMS | 2W Treatment |
|---|
| CON (16) | 71.4±5.74 | 64.5±4.47 | 65.3±4.75 | 10.4±1.66 | 9.6±1.64 | 10.1±1.24 |
| PSD (16) | 70.8±5.35 |
36.8±3.42a | 32.7±6.15 | 11.8±1.24 |
7.6±1.21a | 6.3±1.02 |
| E2(16) | 68.2±6.48 |
36.5±3.47a |
58.6±4.45b | 10.2±2.22 |
5.4±0.77a |
9.6±0.80b |
| E2+U0126(16) | 72.1±7.32 |
35.6±4.17a |
41.1±5.92c | 13.2±1.86 |
4.2±0.53a |
7.3±0.61c |
| E2+K252a (16) | 73.5±8.22 |
40.4±5.24a |
40.2±4.37c | 11.1±1.84 |
5.7±0.76a |
6.9±0.52c |
 | Table IISucrose preference intake (%) in all
groups at baseline, at 2 weeks after CMS and at 2 weeks following
treatment. |
Table II
Sucrose preference intake (%) in all
groups at baseline, at 2 weeks after CMS and at 2 weeks following
treatment.
| Group (n) | Baseline | 2W CMS | 2W Treatment |
|---|
| CON (16) | 0.74±0.142 | 0.65±0.073 | 0.64±0.135 |
| PSD (16) | 0.78±0.111 |
0.35±0.062a | 0.36±0.074 |
| E2(16) | 0.69±0.124 |
0.39±0.076a |
0.65±0.082b |
| E2+U0126(16) | 0.73±0.076 |
0.41±0.072a | 0.48±0.083 |
| E2+K252a (16) | 0.72±0.117 |
0.33±0.035a |
0.41±0.106c |
Middle dose E2 improves PSD
symptoms
To test if PSD symptoms could be relieved by E2, the
present study first determined the dosages of injected E2 to
restore the physiological level of E2 in the OVX rats. Females with
ovaries exhibit circulating estradiol levels ranging from
undetectable to 50 pg/ml. The present study revealed that
estradiol, which was injected at 1, 10 and 25 µg every day,
resulted in the low (12.1-14.3 pg/ml), middle (40.3-50.8 pg/ml) and
high (100.2-127.5 pg/ml) levels in serum (Table III), suggesting that 10 µg E2
administration for 1 or 2 weeks restored physiological E2 levels,
which was thus used for PSD treatment. As listed in Tables I and II, PSD symptoms were significantly
improved following E2 treatment, based on crossing activity
(P<0.05), rearing frequency (P<0.05; Table I), and SP (P<0.05; Table II), in comparison with
vehicle-treated group.
 | Table IIISerum levels of 17β-estradiol
detected after injecting 1, 10 or 25 µg for 1 or 2 weeks. |
Table III
Serum levels of 17β-estradiol
detected after injecting 1, 10 or 25 µg for 1 or 2 weeks.
| E2 dose (n) | 0 week | 1 week | 2 weeks |
|---|
| 0 µg (8) | 7.6±2.1 | 8.3±3.0 | 6.3±1.9 |
| 1 µg (8) | 12.1±2.3 | 14.3±3.1 | 12.1±2.9 |
| 10 µg (8) | 11.2±3.8 | 40.3±7.2 | 50.8±6.9 |
| 25 µg (8) | 12.5±2.5 | 100.2±15.6 | 143.5±20.8 |
Effects of U0126 and K252a on the
behavior tests of PSD rats
To determine the degree at which E2 effect is
mediated by TrkB and MEK-mediated signaling, PSD rats were treated
with E2 alone, or E2 combined with SVZ infusions of K252a or U0126,
the inhibitors of TrkB and MAPK (MEK), respectively. The present
study revealed that PSD rats treated with E2 and K252a or E2 and
U0126 exhibited a decrease in frequency of crossing activity and
rearing activity compared with the group treated with E2 only
(P<0.05; Table I). PSD rats
treated with E2 and K252a also showed a significant reduction in SP
compared with E2 treatment alone (P<0.05; Table II). The E2 and U0126 treated group
also exhibited a decrease in SP compared with the E2 treated group,
even though the decrease was not statistically significant
(P>0.05; Table II).
Effects of estrogen therapy on BDNF
expression in PSD rats
The present study performed western blot analysis
and immunocytochemistry to determine the BDNF expression in the
hippocampus of PSD rats following E2 treatment. As presented in
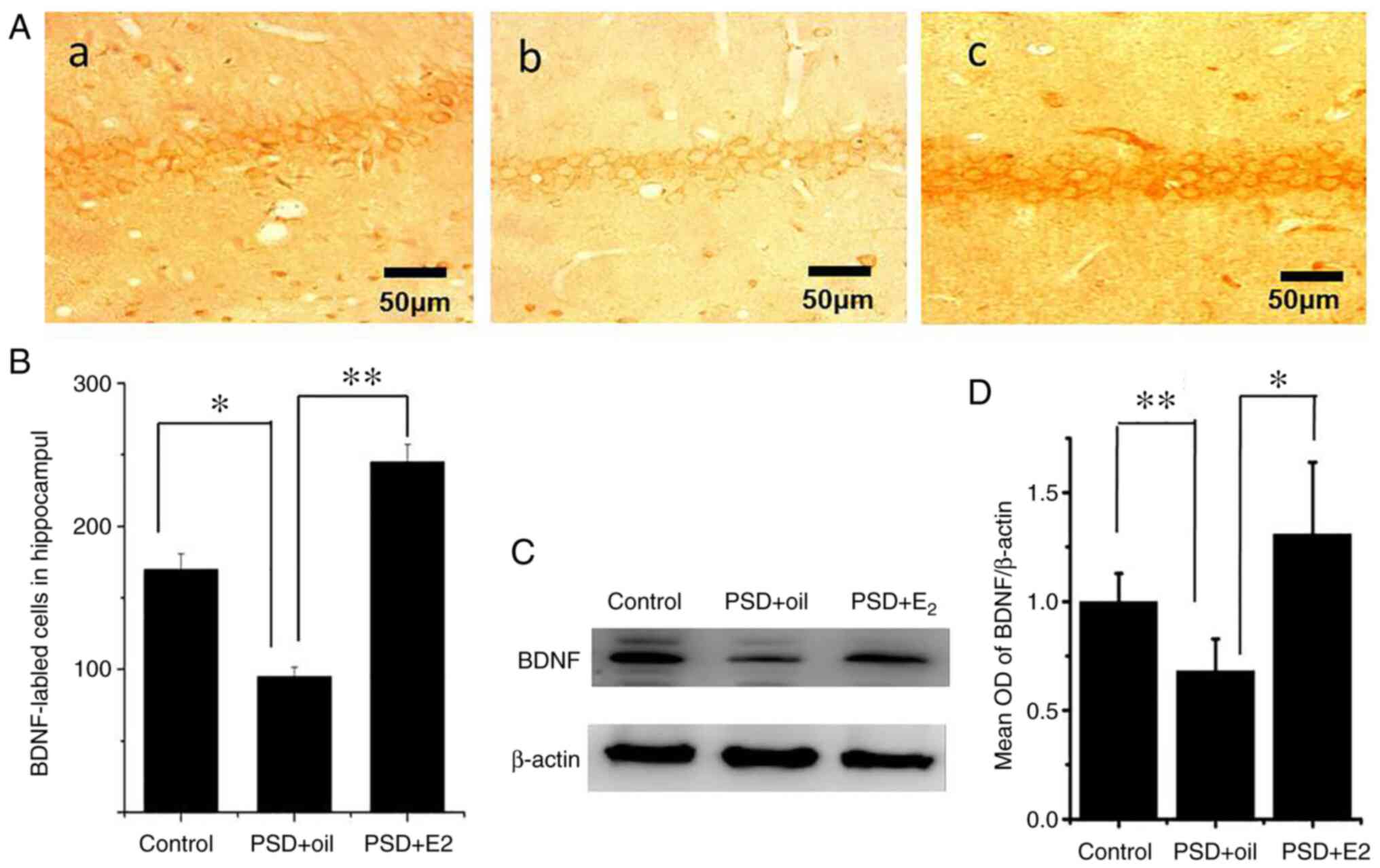
Fig. 2, BDNF was detected in the
cytoplasm and nuclei of hippocampal cells (Fig. 2Aa-Ac). Immunocytochemistry indicated
that the number of BDNF positive cells was significantly lower in
the hippocampus CA1 region of PSD rats (P<0.05 Fig. 2B), which significantly increased
following E2 administration (P<0.01; Fig. 2B). Western blot analysis revealed a
significant decrease in BDNF protein expression levels in the
hippocampus of PSD rats (P<0.01; Fig. 2C and D), which was ameliorated following E2
administration (P<0.05; Fig. 2C
and D).
Effects of estrogen therapy on the
ratio pCREB/CREB and pTrkB/TrkB in PSD rats
The present study assessed whether CREB and TrkB
activity were affected in PSD rats and by subsequent E2 treatment.
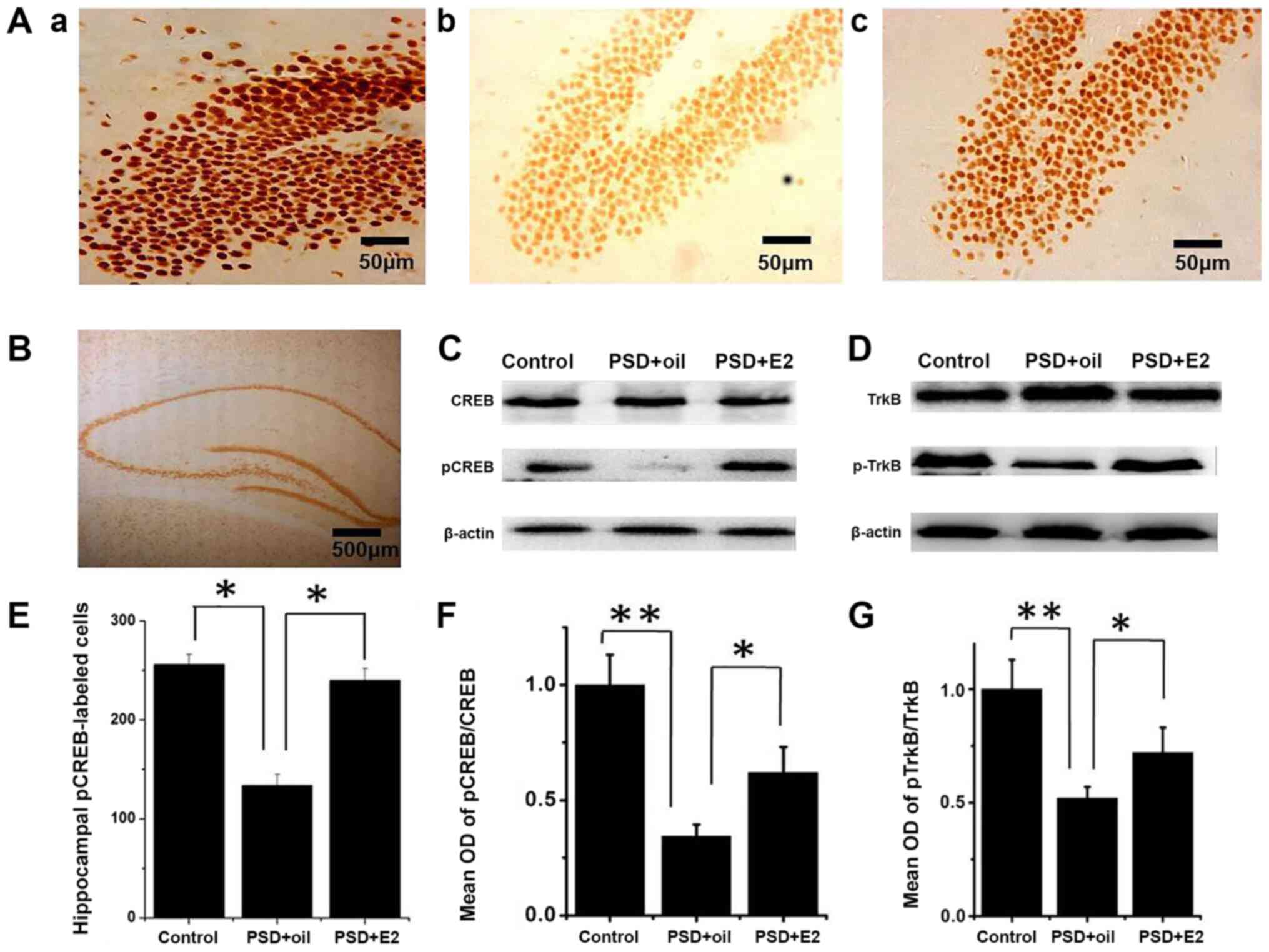
Immunolabeling indicated that pCREB and CREB proteins were
expressed in the nuclei of hippocampal dentategyrus region cells
(Fig. 3; Aa, control group; Ab, PSD
group; Ac, E2 group; and 3B). Immunocytochemistry indicated that
the number of pCREB positive cells was significantly decreased in
the hippocampus dentategyru region of PSD rats (P<0.05; Fig. 3E), which significantly improved
following E2 administration (P<0.05; Fig. 3E). Western blot analysis revealed
that the pCREB/CREB ratio was significantly decreased in the
hippocampus of PSD rats compared with control rats (P<0.01;
Fig. 3C and F). E2 treatment restored the pCREB/CREB
ratio compared with the untreated PSD group (P<0.05; Fig. 3C and F). Consistently, the ratio of pTrkB/TrkB
was decreased in the PSD hippocampus compared with the control
group (P<0.01; Fig. 3D and
G), and E2 treatment increased the
ratio of pTrkB/TrkB (P<0.05; Fig.
3D and G). These data
demonstrated that the CREB activity and TrkB signaling are
decreased in PSD rats, which can be reversed by E2
administration.
Effects of k252a therapy on BDNF and
pTrkB expression in PSD rats
Since TrkB signaling is able to activate CREB, which
in turn activates BDNF, the present study assessed whether the
elevation of TrkB signaling by E2 administration could explain the
elevation of BDNF expression levels. To test this, the present
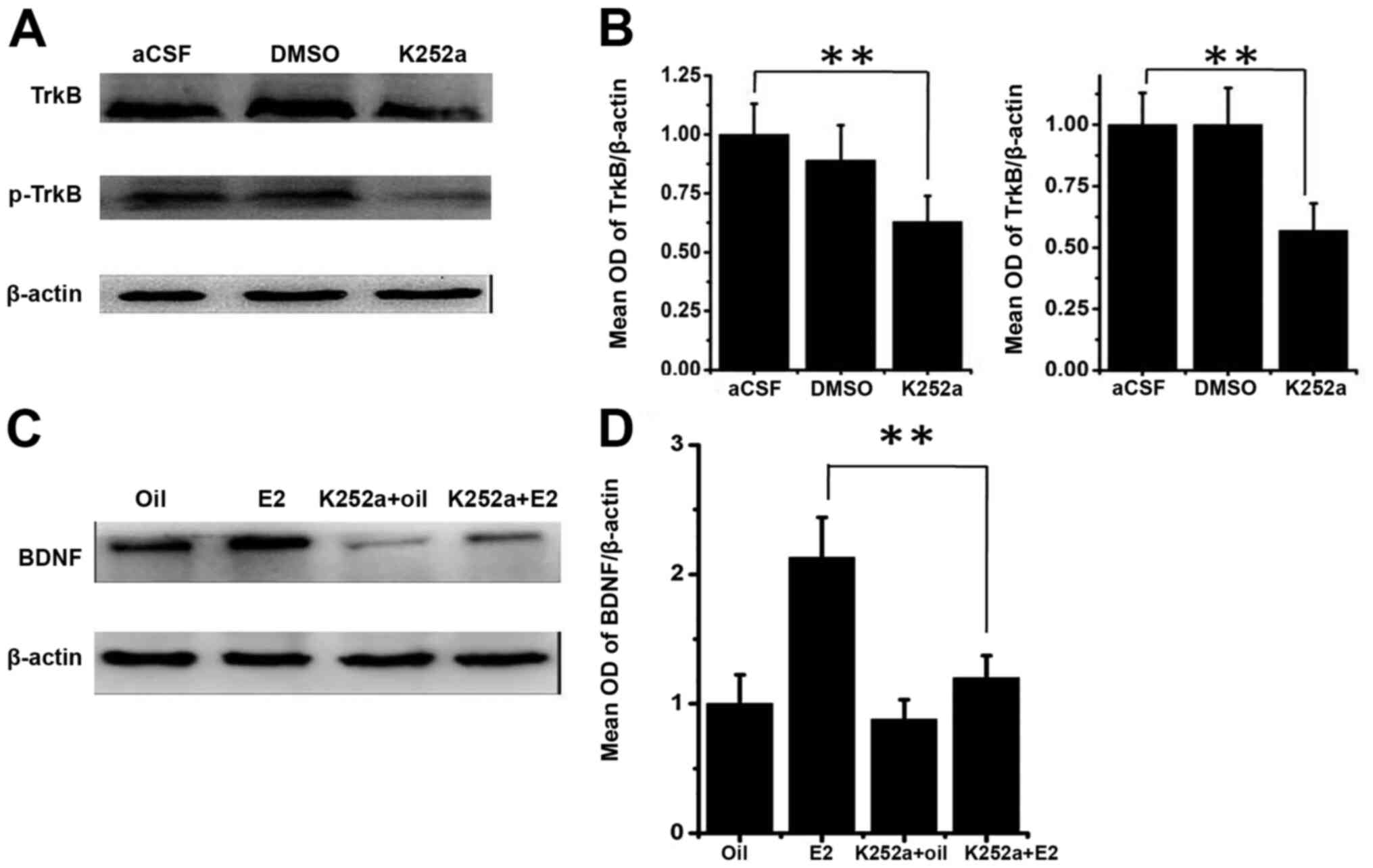
study injected k252a, the TrkB inhibitor, into SVZ to inhibit TrkB
signaling. The results revealed that K252a infusion causes a
decrease in total TrkB and pTrkB in comparison with the infusion of
aCSF (P<0.01; Fig. 4A and
B). Notably, the increase of BDNF
expression following E2 administration in PSD rats was nearly
completely blocked by the presence of K252a (P<0.01; Fig. 4C and D), suggesting that E2 indeed acts through
enhancing TrkB signaling to promote BDNF expression.
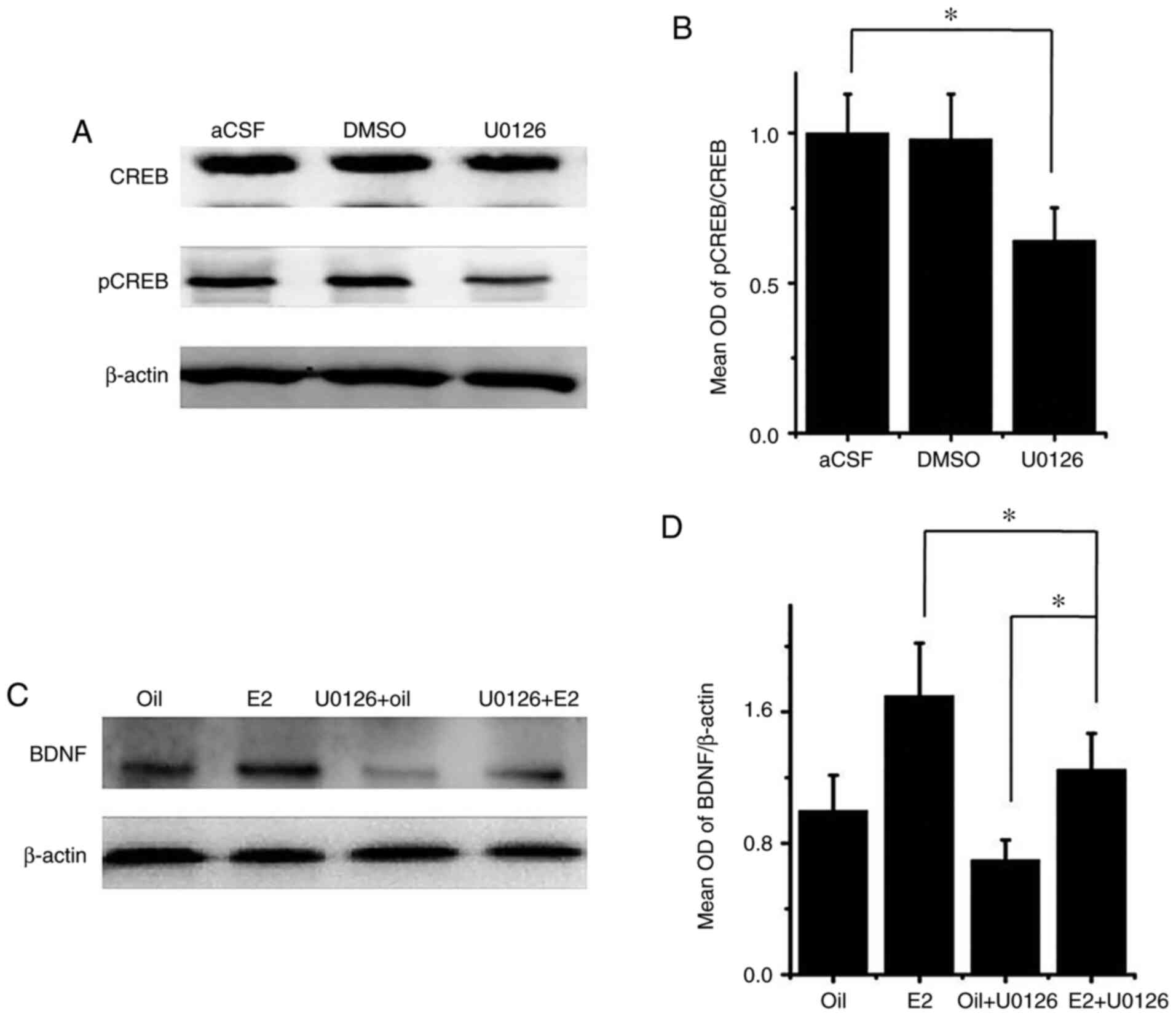
Effects of U0126 therapy on BDNF and
pCREB expression in PSD rats
To assess the mechanism of E2, the present study
examined the role of MEK by injecting the MEK inhibitor U0126 into
the SVZ. Western blot analyses showed that the ratio of pCREB/CREB
decreased by 34% following U0126 administration, compared with the
aCSF group (P<0.05; Fig. 5A and
B), suggesting that CREB activation
in the hippocampus is partially regulated by MEK. Finally, it was
revealed that in rats with PSD, the increase in BDNF expression
following E2 treatment, indicated by the BDNF/β-actin ratio, was
attenuated, but not fully blocked by cotreatment of E2 and U0126.
The E2 + U0126 group significantly increased the expression of BDNF
compared with the oil + U0126 group (P<0.05; Fig. 5C and D), suggesting that the E2 effect on BDNF
expression is partially dependent on MEK signaling.
Discussion
At present, there are few studies on the correlation
between estrogen and PSD. Previous studies have found that estrogen
can significantly improve the depressive behaviors of PSD rats
(24), but the mechanism is still
unclear. The study established a PSD model in rats by left MCAO
followed by exposure to CMS for 2 weeks. It was then revealed that
PSD symptoms can be relieved through E2 administration through
CREB/BDNF/TrkB signaling.
Depression is a heterogeneous, multifaceted disorder
with psychological and behavioral components (8). Translating the complexities of human
affective disease (for example, guilt/suicidal symptoms) or of
behavioral interventions, such as psychotherapy, into animal models
poses a tremendous challenge to the experimental researcher. The
present study revealed that a putative PSD model can be developed
in rats using MCAO followed by CMS. Rats were subjected to MCAO,
which resulted in cerebral infarction that is similar to the
pathophysiological mechanisms observed in patients that have
experienced a stroke. CMS has been reported to provide an effective
equivalent to the precipitation of depression by chronic, low-grade
stressors in humans, inducing an anhedonic-like state in rats
(32). Both variability and
unpredictability during the stress regime are pivotal triggers in
the induction of depressive-like behaviors (33). The present study used various
behavioral tests to induce depression-like symptoms in rats. SP is
regarded to be a key indicator of depression symptoms such as
anhedonia, which indicates loss of interest or pleasure (34,35).
The frequency of line crossing and rearing activity in the
open-field test subjects demonstrated the rats' locomotor activity
and desire to explore. The experimental data in the present study
demonstrated that MCAO/CMS rats demonstrated marked performance
deficits in both SP index and the open-field test, suggesting the
development of PSD symptoms.
The present study also revealed that the depression
symptoms in MCAO/CMS rats could largely be reversed following E2
administration. Estrogen has a wide range of effects in the body
and brain, and its therapeutic potential for mood, among other
physiological and psychological processes, has been recognized for
some time (36). E2 administration
to young women with low E2 levels may alter mood (37). In women with postpartum depression,
administration of sublingual or transdermal E2 sufficiently
improves depressive symptoms to meet the definition of clinical
recovery (38). E2 also can affect
anxiety and depression behavior in animal models. Ovariectomy,
which equates to the removal of the primary source of E2, is
utilized as a model of E2 deprivation to assess the behavioral
effects of E2(39). OVX rats
exhibit increased anxiety and depression behavior, and subcutaneous
administration of E2 can reverse these effects (40,41).
The experimental data in the present study also indicated that E2
has a robust effect on improving anxiety and depression behavior in
rat PSD models.
Mechanistically, estrogen acts through two ERs (ER-α
and ER-β) to regulate multiple functions in the central nervous
system (42). A previous study on
rodent brains have revealed that ER-α is the predominant ER in the
hypothalamus, and controls reproduction (43). ER-β influences non-reproductive
processes and seems to be the main ER subtype expressed in the
cerebral cortex, hippocampus, cerebellum and dorsal raphe (44). Estrogen binds to estrogen receptors,
acting on MAPK, or directly activating tyrosine kinase, which leads
to transcription factors after CREB phosphorylation (19). This further strengthens the cAMP
response element-mediated downstream target genes of BDNF
transcription, and BDNF then serves an anti-depression role by
activating specific receptors, such as TrkB (19,45,46).
Phosphorylated TrkB then activates a variety of proteins and
enzymes through the cytoplasmic pathway, allowing signals to pass
from the cytoplasm into the nucleus, which finally leads to changes
in gene expression patterns, affecting the proliferation of neural
stem cells, and thus serving a role in regulating depression
(47). The present study revealed
that in rats with PSD, the activated levels of pTrkB and pCREB were
all decreased, which may lead to decreased levels of BDNF. E2 was
indicated to enhance TrkB signaling, and was accordingly able to
elevate activated CREB and BDNF expression levels. When TrkB
signaling was concurrently blocked by k252a, E2 was not able to
elevate BDNF expression or to relieve depression symptoms in
MCAO/CMS rats. The results of the present study further suggest
that this E2 effect is partially dependent on MEK signaling.
The present study consolidated the neurotrophic
hypothesis in the development of psychological disorders. BDNF is
the key factor of neuronal plasticity, and is closely associated
with major depression (48).
Antidepressants and the mood stabilizer lithium have both been
demonstrated to increase BDNF levels in the brain (49). A previous study reported that a
region-specific knockdown of BDNF in the dentate gyrus induces
depression-like behavior (50).
Conversely, increased TrkB signaling produced by overexpression of
TrkB decreases anxiety and depressive-like behavior (51,52).
In patients with PSD, serum concentrations of BDNF decrease
(12), and the decreased BDNF-TrkB
signaling may therefore serve an important role in the pathogenesis
of the illness (53). This
coincides with the results observed in the PSD rat model developed
in the present study.
Finally, the results of the present study provide
novel evidence for a functional interaction between CREB/BDNF/TrkB
signaling and estrogen receptor signaling in the hippocampus. In
neurons, CREB can be activated by TrkB signaling through
phosphorylation, and pCREB will then activate BDNF to enhance TrkB
signaling (45). It has been
revealed that there is complementary expression of estrogen
receptors and BDNF in the hippocampus, and there are similar
effects of estrogen and BDNF on hippocampal physiology and behavior
(54-56).
Previous studies have demonstrated that E2 administration in OVX
rats increases BDNF mRNA and protein expression levels in the
hippocampus (57,58), and the mRNA and protein expression
of BDNF fluctuate across the estrous cycle in female rats, with the
highest levels of BDNF mRNA detected during late diestrus when
estrogen reaches the highest levels (27,59).
The present study demonstrated that in rats with PSD, E2 treatment
is able to elevate CREB/BDNF/TrkB signaling, which in turn leads to
a decrease in depression-like symptoms.
In conclusion, the present study developed a PSD
model in rats, whose depression-like symptoms can be relieved by
estrogen treatment that acts to promote CREB/BDNF/TrkB signaling.
Considering that serum BDNF levels were reported to be decreased in
human patients with PSD, future studies should focus on whether
estrogen treatment could be a therapeutic agent that can be used to
treat these patients.
Acknowledgements
Not applicable.
Funding
The current study was supported by grants from Wenzhou
Technology Foundation (grant no. H20090067) and the National
Institute of Health (nos. AG21980 and NS057186).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
HJ, LX, KJ and BS designed the study and performed
the experiments. HJ and LX established the animal models, collected
the data and analyzed the data. HJ wrote the manuscript. BS revised
the manuscript. All authors read and approved the final version of
the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Animal Ethics
Committee of First Affiliated Hospital, Wenzhou Medical University
(Wenzhou, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing
interests.
References
|
1
|
Kim AS and Johnston SC: Temporal and
geographic trends in the global stroke epidemic. Stroke. 44 (Suppl
1):S123–S125. 2013.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Liu M, Wu B, Wang WZ, Lee LM, Zhang SH and
Kong LZ: Stroke in China: Epidemiology, prevention, and management
strategies. Lancet Neurol. 6:456–464. 2007.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Boden-Albala B, Litwak E, Elkind MS,
Rundek T and Sacco RL: Social isolation and outcomes post stroke.
Neurology. 64:1888–1892. 2005.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ellis C, Zhao Y and Egede LE: Depression
and increased risk of death in adults with stroke. J Psychosomatic
Res. 68:545–551. 2010.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Hackett ML, Yapa C, Parag V and Anderson
CS: Frequency of depression after stroke: A systematic review of
observational studies. Stroke. 36:1330–1340. 2005.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Poynter B, Shuman M, Diaz-Granados N,
Kapral M, Grace SL and Stewart DE: Sex differences in the
prevalence of post-stroke depression: A systematic review.
Psychosomatics. 50:563–569. 2009.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Hackett ML and Pickles K: Part I:
Frequency of depression after stroke: An updated systematic review
and meta-analysis of observational studies. Int J Stroke.
9:1017–1025. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Loubinoux I, Kronenberg G, Endres M,
Schumann-Bard P, Freret T, Filipkowski RK, Kaczmarek L and
Popa-Wagner A: Post-stroke depression: Mechanisms, translation and
therapy. J Cell Mol Med. 16:1961–1969. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Duman RS and Monteggia LM: A neurotrophic
model for stress-related mood disorders. Biol Psychiatry.
59:1116–1127. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
McEwen BS: Protective and damaging effects
of stress mediators: Central role of the brain. Dialogues Clin
Neurosci. 8:367–381. 2006.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Krishnan V and Nestler EJ: The molecular
neurobiology of depression. Nature. 455:894–902. 2008.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yang L, Zhang Z, Sun D, Xu Z, Yuan Y,
Zhang X and Li L: Low serum BDNF may indicate the development of
PSD in patients with acute ischemic stroke. Int J Geriatric
Psychiatry. 26:495–502. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
13
|
Pillai A: Brain-derived neurotropic
factor/TrkB signaling in the pathogenesis and novel pharmacotherapy
of schizophrenia. Neurosignals. 16:183–193. 2008.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Horch HW and Katz LC: BDNF release from
single cells elicits local dendritic growth in nearby neurons. Nat
Neurosci. 5:1177–1184. 2002.PubMed/NCBI View
Article : Google Scholar
|
|
15
|
Bergami M, Rimondini R, Santi S, Blum R,
Götz M and Canossa M: Deletion of TrkB in adult progenitors alters
newborn neuron integration into hippocampal circuits and increases
anxiety-like behavior. Proc Natil Acad Sci. 105:15570–15575.
2008.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sanchez-Huertas C and Rico B:
CREB-Dependent regulation of GAD65 transcription by BDNF/TrkB in
cortical interneurons. Cereb Cortex. 21:777–788. 2011.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Blendy JA: The role of CREB in depression
and antidepressant treatment. Biol Psychiatry. 59:1144–1150.
2006.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Nibuya M, Nestler EJ and Duman RS: Chronic
antidepressant administration increases the expression of cAMP
response element binding protein (CREB) in rat hippocampus. J
Neurosci. 16:2365–2372. 1996.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Carlezon WA Jr, Duman RS and Nestler EJ:
The many faces of CREB. Trends Neurosci. 28:436–445.
2005.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Romano-Torres M and Fernández-Guasti A:
Estradiol valerate elicits antidepressant-like effects in
middle-aged female rats under chronic mild stress. Behav Pharmacol.
21:104–111. 2010.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lebesgue D, Traub M, De Butte-Smith M,
Chen C, Zukin RZ, Kelly MJ and Etgen AM: Acute administration of
non-classical estrogen receptor agonists attenuates
ischemia-induced hippocampal neuron loss in middle-aged female
rats. PLoS One. 5(e8642)2010.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Ormerod BK, Lee TT and Galea LA: Estradiol
enhances neurogenesis in the dentate gyri of adult male meadow
voles by increasing the survival of young granule neurons.
Neuroscience. 128:645–654. 2004.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Scharfman HE and MacLusky NJ: Similarities
between actions of estrogen and BDNF in the hippocampus:
Coincidence or clue? Trends Neurosci. 28:79–85. 2005.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Su Q, Cheng Y, Jin K, Cheng J, Lin Y, Lin
Z, Wang L and Shao B: Estrogen therapy increases BDNF expression
and improves post-stroke depression in ovariectomy-treated rats.
Exp Ther Med. 12:1843–1848. 2016.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Kight KE and McCarthy MM: Sex differences
and estrogen regulation of BDNF gene expression, but not propeptide
content, in the developing hippocampus. J Neurosci Res. 95:345–354.
2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Yang NB, Pan XJ, Cheng JJ, Lin JQ and Zhu
JY: Ethical inspection about laboratory animals. Zhongguo Ying Yong
Sheng Li Xue Za Zhi. 31:504–507. 2015.PubMed/NCBI
|
|
27
|
Allen AL and McCarson KE: Estrogen
increases nociception-evoked brain-derived neurotrophic factor gene
expression in the female rat. Neuroendocrinology. 81:193–199.
2005.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Gao BY, Sun CC, Xia GH, Zhou ST, Zhang Y,
Mao YR, Liu PL, Zheng Y, Zhao D, Li XT, et al: Paired associated
magnetic stimulation promotes neural repair in the rat middle
cerebral artery occlusion model of stroke. Neural Regen Res.
15:2047–2056. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Li X, Chen C, Yang X, Wang J, Zhao ML, Sun
H, Zhang S and Tu Y: Acupuncture improved neurological recovery
after traumatic brain injury by activating BDNF/TrkB pathway. Evid
Based Complement Alternat Med. 2017(8460145)2017.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wang ZQ, Chen XC, Yang GY and Zhou LF:
U0126 prevents erk pathway phosphorylation and interleukin-1β mRNA
prodution after cerebral ischemia. Chin Med Sci J. 19:270–275.
2004.PubMed/NCBI
|
|
31
|
Yi LT, Li J, Liu BB, Luo L, Liu Q and Geng
D: BDNF-ERK-CREB signalling mediates the role of miR-132 in the
regulation of the effects of oleanolic acid in male mice. J
Psychiatry Neurosci. 39:348–359. 2014.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Willner P: Chronic mild stress (CMS)
revisited: Consistency and behavioural-neurobiological concordance
in the effects of CMS. Neuropsychobiology. 52:90–110.
2005.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Chang CH and Grace AA: Amygdala-Ventral
pallidum pathway decreases dopamine activity after chronic mild
stress in rats. Biol Psychiatry. 76:223–230. 2014.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Kalueff AV, Gallagher PS and Murphy DL:
Are serotonin transporter knockout mice'depressed'?: Hypoactivity
but no anhedonia. Neuroreport. 17:1347–1351. 2006.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Strekalova T, Gorenkova N, Schunk E,
Dolgov O and Bartsch D: Selective effects of citalopram in a mouse
model of stress-induced anhedonia with a control for chronic
stress. Behav Pharmacol. 17:271–287. 2006.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Newhouse P and Albert K: Strogen, stress,
and depression: A neurocognitive model. JAMA Psychiatry.
72:727–729. 2015.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Schmidt PJ: Depression, the perimenopause,
and estrogen therapy. Ann NY Acad Sci. 1052:27–40. 2005.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Ahokas A, Kaukoranta J, Wahlbeck K and
Aito M: Estrogen deficiency in severe postpartum depression:
Successful treatment with sublingual physiologic 17beta-estradiol:
A preliminary study. J Clin Psychiatry. 62(332)2001.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Cheng Y, Su Q, Shao B, Cheng J, Wang H,
Wang L, Lin Z, Ruan L, ZhuGe Q and Jin K: 17β-Estradiol attenuates
poststroke depression and increases neurogenesis in female
ovariectomized rats. BioMed Res Int. 2013:1–10. 2013.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Bowman R, Ferguson D and Luine VN: Effects
of chronic restraint stress and estradiol on open field activity,
spatial memory, and monoaminergic neurotransmitters in
ovariectomized rats. Neuroscience. 113:401–410. 2002.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Walf AA and Frye CA: Estradiol's effects
to reduce anxiety and depressive behavior may be mediated by
estradiol dose and restraint stress. Neuropsychopharmacology.
30:1288–1301. 2005.
|
|
42
|
Mitra SW, Hoskin E, Yudkovitz J, Pear L,
Wilkinson HA, Hayashi S, Pfaff DW, Ogawa S, Rohrer SP, Schaeffer
JM, et al: Immunolocalization of estrogen receptor beta in the
mouse brain: Comparison with estrogen receptor alpha.
Endocrinology. 144:2055–2067. 2003.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Lorsch ZS, Loh YE, Purushothaman I, Walker
DM, Parise EM, Salery M, Cahill ME, Hodes GE, Pfau ML, Kronman H,
et al: Estrogen receptor alpha drives pro-resilient transcription
in mouse models of depression. Nat Commun. 9(1116)2018.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Suzuki S, Gerhold LM, Böttner M, Rau SW,
Cruz CD, Yang E, Zhu H, Yu J, Cashion AB, Kindy MS, et al:
Estradiol enhances neurogenesis following ischemic stroke through
estrogen receptors alpha and beta. J Comp Neurol. 500:1064–1075.
2007.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Nair A, Vadodaria KC, Banerjee SB,
Benekareddy M, Dias BG, Duman RS and Vaidya VA: Stressor-Specific
regulation of distinct brain-derived neurotrophic factor
transcripts and cyclic AMP response element-binding protein
expression in the postnatal and adult rat hippocampus.
Neuropsychopharmacology. 32:1504–1519. 2007.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Wu Q, Chambliss K, Umetani M, Mineo C and
Shaul PW: Non-nuclear estrogen receptor signaling in the
endothelium. J Biol Chem. 286:14737–14743. 2011.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Rantamäki T and Castrén E: Targeting TrkB
neurotrophin receptor to treat depression. Expert Opin Ther
Targets. 12:705–715. 2008.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Pezawas L, Meyer-Lindenberg A, Goldman AL,
Verchinski BA, Chen G, Kolachana BS, Egan MF, Mattay VS, Hariri AR
and Weinberger DR: Evidence of biologic epistasis between BDNF and
SLC6A4 and implications for depression. Mol Psychiatry. 13:709–716.
2008.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Yasuda S, Liang M, Marinova Z, Yahyavi A
and Chuang D: The mood stabilizers lithium and valproate
selectively activate the promoter IV of brain-derived neurotrophic
factor in neurons. Mol Psychiatry. 14:51–59. 2007.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Taliaz D, Stall N, Dar D and Zangen A:
Knockdown of brain-derived neurotrophic factor in specific brain
sites precipitates behaviors associated with depression and reduces
neurogenesis. Mol Psychiatry. 15:80–92. 2009.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Koponen E, Rantamäki T, Voikar V,
Saarelainen T, MacDonald E and Castrén E: Enhanced BDNF signaling
is associated with an antidepressant-like behavioral response and
changes in brain monoamines. Cell Mol Neurobiol. 25:973–980.
2005.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Koponen E, Võikar V, Riekki R, Saarelainen
T, Rauramaa T, Rauvala H, Taira T and Castrén E: Transgenic mice
overexpressing the full-length neurotrophin receptor trkB exhibit
increased activation of the trkB-PLCγ pathway, reduced anxiety, and
facilitated learning. Mol Cell Neurosci. 26:166–181.
2004.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Zhang E and Liao P: Brain-Derived
neurotrophic factor and post-stroke depression. J Neurosci Res.
89:537–548. 2020.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Scharfman HE and MacLusky NJ: Estrogen and
brain-derived neurotrophic factor (BDNF) in hippocampus: Complexity
of steroid hormone-growth factor interactions in the adult CNS.
Front Neuroendocrinology. 27:415–435. 2006.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Scharfman HE, Mercurio TC, Goodman JH,
Wilson MA and MacLusky NJ: Hippocampal excitability increases
during the estrous cycle in the rat: A potential role for
brain-derived neurotrophic factor. J Neurosci. 23:11641–11652.
2003.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Harte-Hargrove L, MacLusky NJ and
Scharfman HE: Brain-derived neurotrophics factor-estrogen
interactions in hippocampal mossy fiber pathway: Implications for
normal brain function and disease. Neuroscience. 3:46–66.
2012.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Jezierski M and Sohrabji F: Neurotrophin
expression in the reproductively senescent forebrain is refractory
to estrogen stimulation. Neurobiol Aging. 22:309–319.
2001.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Zhou J, Zhang H, Cohen RS and Pandey SC:
Effects of estrogen treatment on expression of brain-derived
neurotrophic factor and cAMP response element-binding protein
expression and phosphorylation in rat amygdaloid and hippocampal
structures. Neuroendocrinology. 81:294–310. 2005.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Gibbs RB: Levels of trkA and BDNF mRNA,
but not NGF mRNA, fluctuate across the estrous cycle and increase
in response to acute hormone replacement. Brain Res. 787:259–268.
1998.PubMed/NCBI View Article : Google Scholar
|