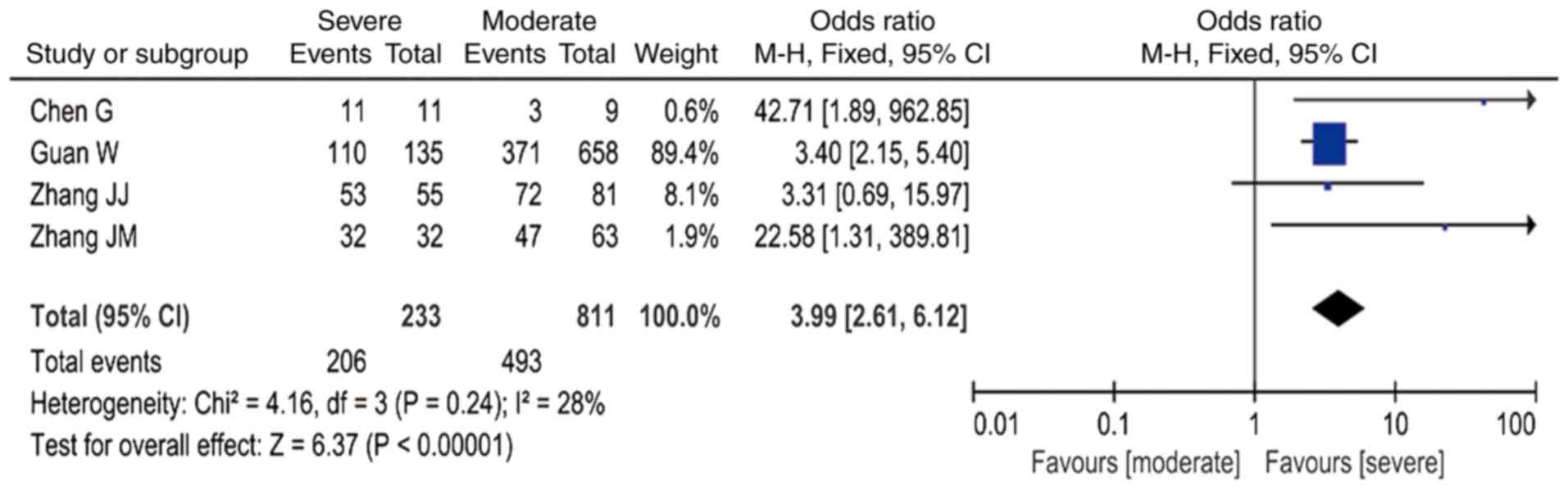
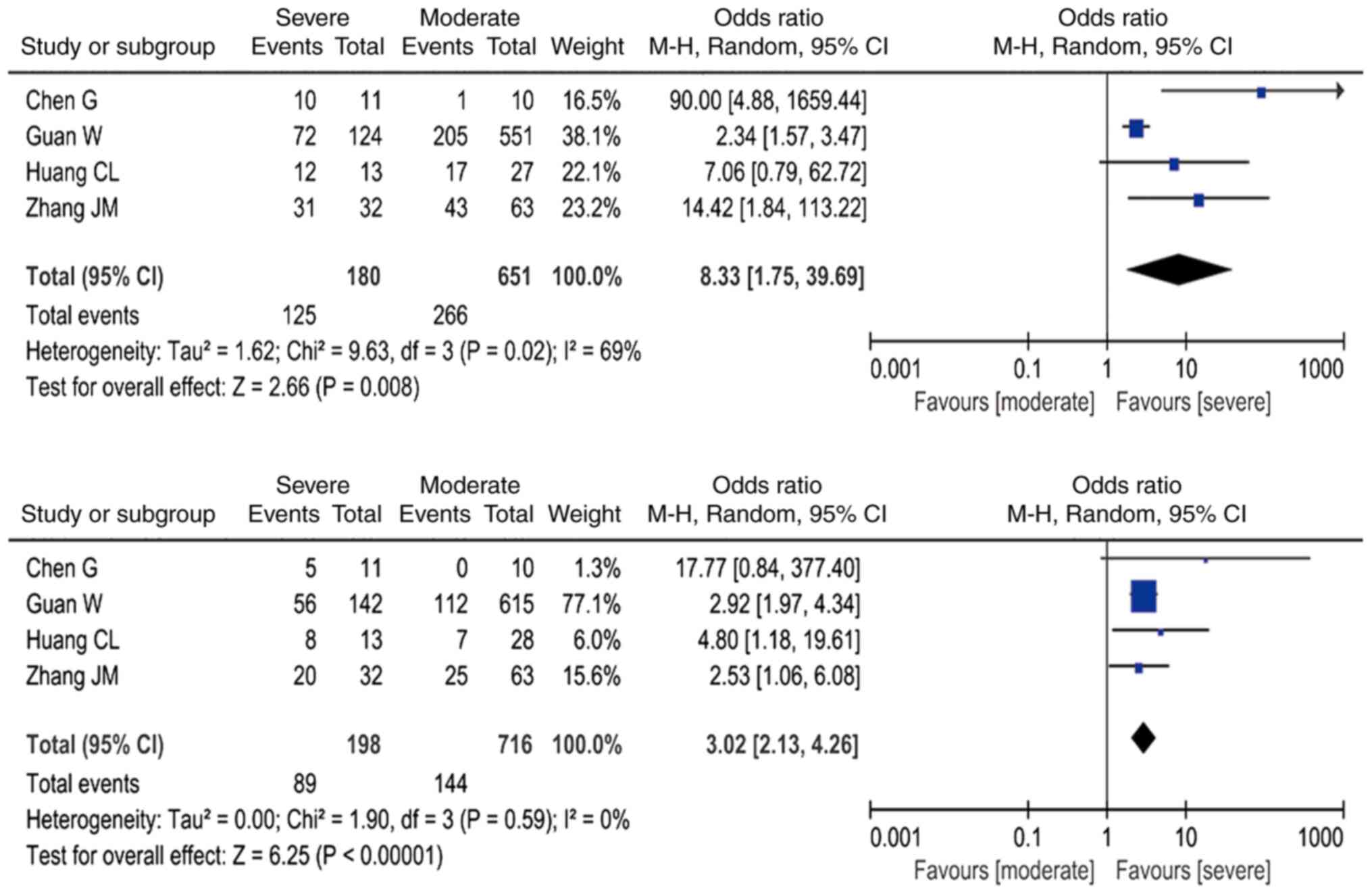
|
1
|
World Health Organization. Coronavirus
disease 2019 (COVID-19): Situation report,73. Available from:
https://www.who.int/emergencies/diseases/novel-coronavirus-2019/situation-reports/.
Accessed, April 2, 2020.
|
|
2
|
Li Q, Guan X, Wu P, Wang X, Zhou L, Tong
Y, Ren R, Leung KSM, Lau EHY, Wong JY, et al: Early transmission
dynamics in Wuhan, China, of novel coronavirus-infected pneumonia.
N Engl J Med. 382:1199–1207. 2020.PubMed/NCBI View Article : Google Scholar
|
|
3
|
The diagnosis and treatment of Coronavirus
disease 2019 (version 7). Available from: http://www.cac.gov.cn/.
|
|
4
|
Sun P, Qie S, Liu Z, Ren J, Li K and Xi J:
Clinical characteristics of hospitalized patients with SARS-CoV-2
infection: A single arm meta-analysis. J Med Virol. 92:612–617.
2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Yang J, Zheng Y, Gou X, Pu K, Chen Z, Guo
Q, Ji R, Wang H, Wang Y and Zhou Y: Prevalence of comorbidities in
the novel Wuhan coronavirus (COVID-19) infection: A systematic
review and meta-analysis. Int J Infect Dis.
4023:S1201–9712(20)30136-3. 2020.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Li LQ, Huang T, Wang YQ, Wang ZP, Liang Y,
Huang TB, Zhang HY, Sun WM and Wang YP: 2019 novel coronavirus
patients' clinical characteristics, discharge rate, and fatality
rate of meta-analysis. J Med Virol. 577–583. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
National Health Commission & State
Administration of Traditional Chinese Medicin. Diagnosis and
Treatment Protocol for COVID-19 (trial version 7). Updated:
2020-03-29. Available from: http://en.nhc.gov.cn/2020-03/29/c_78469.htm.
|
|
8
|
Farrah K, Young K, Tunis MC and Zhao L:
Risk of bias tools in systematic reviews of health interventions:
An analysis of PROSPERO-registered protocols. Syst Rev.
8(280)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Slim K, Nini E, Forestier D, Kwiatkowski
F, Panis Y and Chipponi J: Methodological index for non-randomized
studies (minors): Development and validation of a new instrument.
ANZ J Surg. 73:712–716. 2003.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chen G, Wu D, Guo W, Cao Y, Huang D, Wang
H, Wang T, Zhang X, Chen H, Yu H, et al: Clinical and immunological
features of severe and moderate coronavirus disease 2019. J Clin
Invest. 130:2620–2629. 2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He
JX, Liu L, Shan H, Lei CL, Hui DSC, et al: Clinical characteristics
of coronavirus disease 2019 in China. N Engl J Med. 382:1708–1720.
2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu
Y, Zhang L, Fan G, Xu J, Gu X, et al: Clinical features of patients
infected with 2019 novel coronavirus in Wuhan, China. Lancet.
395:497–506. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J,
Wang B, Xiang H, Cheng Z, Xiong Y, et al: Clinical characteristics
of 138 hospitalized patients with 2019 novel coronavirus-infected
pneumonia in Wuhan, China. JAMA. 323:1061–1069. 2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Zhang JJ, Dong X, Cao YY, Yuan YD, Yang
YB, Yan YQ, Akdis CA and Gao YD: Clinical characteristics of 140
patients infected with SARS-CoV-2 in Wuhan, China. Allergy.
75:1730–1741. 2020.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Zhang G, Zhang J, Wang B, Zhu X, Wang Q
and Qiu S: Analysis of clinical characteristics and laboratory
findings of 95 cases of 2019 novel coronavirus pneumonia in Wuhan,
China: A retrospective analysis. Respir Res. 21(74)2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Lu R, Zhao X, Li J, Niu P, Yang B, Wu H,
Wang W, Song H, Huang B, Zhu N, et al: Genomic characterisation and
epidemiology of 2019 novel coronavirus: Implications for virus
origins and receptor binding. Lancet. 395:565–574. 2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Su S, Wong G, Shi W, Liu J, Lai ACK, Zhou
J, Liu W, Bi Y and Gao GF: Epidemiology, genetic recombination, and
pathogenesis of coronaviruses. Trends Microbiol. 24:490–502.
2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Lippi G and Plebani M: A modern and
pragmatic defnition of laboratory medicine. Clin Chem Lab Med.
58(1171)2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lippi G and Plebani M: Laboratory
abnormalities in patients with COVID-2019 infection. Clin Chem Lab
Med. 58:1131–1134. 2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Rodriguez-Morales AJ, Cardona-Ospina JA,
Gutiérrez-Ocampo E, Villamizar-Peña R, Holguin-Rivera Y,
Escalera-Antezana JP, Alvarado-Arnez LE, Bonilla-Aldana DK,
Franco-Paredes C, Henao-Martinez AF, et al: Clinical, laboratory
and imaging features of COVID-19: A systematic review and
meta-analysis. Travel Med Infect Dis. 34(101623)2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Lin L, Lu L, Cao W and Li T: Hypothesis
for potential pathogenesis of SARS-CoV-2 infection-a review of
immune changes in patients with viral pneumonia. Emerg Microbes
Infect. 9:727–732. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Rokni M, Ghasemi V and Tavakoli Z: Immune
responses and pathogenesis of SARS-CoV-2 during an outbreak in
Iran: Comparison with SARS and MERS. Rev Med Virol.
30(e2107)2020.PubMed/NCBI View
Article : Google Scholar
|
|
23
|
Azkur AK, Akdis M, Azkur D, Sokolowska M,
van de Veen W, Brüggen MC, O'Mahony L, Gao Y, Nadeau K and Akdis
CA: Immune response to SARS-CoV-2 and mechanisms of
immunopathological changes in COVID-19. Allergy. 75:1564–1581.
2020.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Qin C, Zhou L, Hu Z, Zhang S, Yang S, Tao
Y, Xie C, Ma K, Shang K, Wang W and Tian DS: Dysregulation of
immune response in patients with coronavirus 2019 (COVID-19) in
Wuhan, China. Clin Infect Dis. 71:762–768. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Zhang Y, Zheng L, Liu L, Zhao M, Xiao J
and Zhao Q: Liver impairment in COVID-19 patients: A retrospective
analysis of 115 cases from a single centre in Wuhan city, China.
Liver Int. 40:2095–2103. 2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Possamai LA, Thursz MR, Wendon JA and
Antoniades CG: Modulation of monocyte/macrophage function: A
therapeutic strategy in the treatment of acute liver failure. J
Hepatol. 61:439–445. 2014.PubMed/NCBI View Article : Google Scholar
|