Introduction
Obstructive sleep apnea/hypopnea syndrome (OSAHS) is
the most common type of apnea (1).
It is characterized by recurrent episodes of partial or complete
upper airway collapse during sleep (2). OSAHS has been demonstrated to be
associated with several comorbidities, including cardiovascular and
neurodegenerative disorders, such as Alzheimer's disease and
Parkinson's disease (3-5).
Previous findings have shown that the physiological and
pharmacological actions of glucocorticoids are mediated by the
glucocorticoid receptor (GR), which is a member of the nuclear
receptor superfamily of ligand-dependent transcription factors
(6). Following glucocorticoid
binding, GR induces or represses the transcription of target genes,
which comprise up to 10-20% of the human genome (7). For instance, GR has been previously
reported to positively regulate the transcription of FNDC5 in the
liver (8), whilst another study
revealed that Angptl4 is a GR primary target gene in
hepatocytes and adipocytes (9). The
expression of GRα has been demonstrated to be decreased in patients
with OSAHS. For instance, the expression of GRα in the tonsil
tissues of children with OSAHS was significantly lower compared
with the tonsil tissues of children without OSAHS (10). However, the mechanism by which GRα
is dysregulated in patients with OSAHS remains unknown.
Long noncoding RNAs (lncRNAs) are a class of
transcripts >200 nucleotides in length (11). Generally, lncRNAs do not encode
proteins (12). Accumulating data
have indicated that lncRNAs exert crucial functions in gene
regulation, biological processes and several diseases, including
inflammation (13) and immune
responses (14). LncRNA X
inactivate-specific transcript (XIST) is a 17-kb long RNA and is
transcribed from the inactive X chromosome in female mammalians.
Previous studies have indicated that the expression of XIST is
upregulated in various diseases, such as in basal-like human breast
cancer (15) and cystic fibrosis
(16). However, it remains unclear
whether XIST serves a role in OSAHS.
It has been well documented that nuclear factor
κ-light-chain-enhancer of activated B cells (NF-κB) serves a vital
role in inflammation (17). The IκB
kinase enzyme complex is part of the upstream NF-κB signal
transduction cascade and the IκBα protein inactivates transcription
factor of NF-κB by inhibiting the nuclear localization signals of
NF-κB and sequestering it to an inactive state in the cytoplasm
(18). NF-κB is a protein complex
composed of homo- or heterodimers formed by different combinations
of the five monomeric NF-κB subunits: p65/Rel A, Rel B, c-Rel, p50
and p52(19). Activated NF-κB
regulated cell proliferation, apoptosis and the production of
inflammatory cytokines, including tumor necrosis factor (TNF)-α,
interleukin (IL)-1β, IL-6 and IL-8 (20,21).
In conclusion, the present study specially focused
on the role and mechanism of GRα in adenoids of patients with
OSAHS, the results of which may provide a potential novel insight
for treatment of OSAHS.
Materials and methods
Patients and sample collection
The present study was approved by the Ethics
Committee and Animal Care Committee of the Affiliated Hangzhou
First People's Hospital, Zhejiang University School of Medicine,
Hangzhou, China. The legal guardians of all the participants read
and signed informed consent forms. Both inclusion and exclusion
criteria were first assessed through review of the electronic
medical record. Inclusion criteria were: i) Age ≤12 years old; ii)
able to obtain written informed consent from legal guardians; iii)
hospitalized patients having a documented history of congestive
heart failure in agreement with the 2013 ACCF/AHA heart failure
definition (22); iv) anticipated
hospitalization of >24 h. Exclusion criteria were: i)
Established obstructive sleep apnea and/or previous exposure to
positive airway pressure (PAP) therapy; ii) Presence of any active
conditions that the investigators felt would interfere with testing
or potential therapy (hemodynamic instability, respiratory failure,
unconsciousness, pneumothorax, penetrating chest trauma, general
nausea/vomiting, facial anomalies/facial trauma, upper
gastrointestinal bleed or history of recent gastric surgery); iii)
presence of a clinically significant illness or medical condition
that the investigators felt would prohibit the subject from
participating in the study (a number of subjects were ruled due to
advanced COPD, malignancy or having a high baseline home oxygen
requirement).
According to the apnea-hypopnea index, the patients
were diagnosed with OSAHS when apnea-hypopnea occurred ≥ one time
per hour, as previously described (23). Adenoid tissue specimens were
collected from children with OSAHS (n=26) and healthy donors (n=21)
with myocarditis at the Department of Otorhinolaryngology,
Affiliated Hangzhou First People's Hospital, Zhejiang University
School of Medicine, from May 2017 to May 2018. Mean age of the
OSAHS group (19 females and 7 males) was 6.88±3.18 years. Mean age
of the healthy donor group (19 females and 2 males) was 7.2±2.63
years. No local or systemic glucocorticoid treatment was given to
any of the patients within 2 weeks to 2 months prior to the
operations.
Cell cultures
Human nasopharyngeal epithelial cell line NP69
purchased from American Type Culture Collection (ATCC) was cultured
in keratinocyte-SFM medium (Invitrogen; Thermo Fisher Scientific,
Inc.). All cells were grown in a humidified incubator with a 5%
CO2 atmosphere at 37˚C, as previously described
(24). Additionally, EVP4593 (cat.
no. HY-13812; MedChemExpress), an inhibitor of NF-κB signaling, was
used to treat NP69 cells. DMSO was used as the control.
Cell transfections
Total RNA was extracted from adenoid tissue tissues
and NP69 cells using TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) and synthesized into first-strand cDNA
using an M-MLV Reverse Transcriptase kit (cat. no. 28025013;
Invitrogen; Thermo Fisher Scientific, Inc.) as previously described
(25). Full-length XIST was
amplified from female genomic DNA (Roche diagnostics) and then
digested with XbaI to obtain nucleotides 105-10747 of the
XIST cDNA sequence (accession no. M97168) and inserted into
pcDNA3.1 vectors (Invitrogen; Thermo Fisher Scientific, Inc.) to
overexpress XIST. GRα overexpression plasmids were constructed
using the pcDNA3.1 backbone from Sangon Biotechnology Co., Ltd.
XIST short hairpin (sh) RNA sequences were inserted into shRNA
vectors (Shanghai GenePharma Co., Ltd.). The actual DNA sequence of
the shRNA that was ligated into the pGPH1 vector were as follows:
sh-NC, 5'-CACCGCTATGATATCGTCTGTTTCAAGAGAACAGACGATATCATAGCTTTTTTG-3'
and sh-XIST,
5'-CACCGCATCTGACTGTTATGTTTCAAGAGAACATAACAGTCAGATGCTTTTTTG-3'. Cells
(1x105 cells/well) were seeded in 24-well plates and 500
µl of keratinocyte-SFM medium (Invitrogen; Thermo Fisher
Scientific, Inc.) was added to each well. When the cells reached
40-60% confluence, the aforementioned vectors were transfected into
cells at a final concentration of 50 nM using
Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher
Scientific, Inc.) at 37˚C and 5% CO2 for 48 h, according
to the manufacturer's protocol.
RNA extraction and reverse
transcription-quantitative PCR (RT-qPCR)
Total RNA was extracted from adenoid tissue tissues
and NP69 cells using TRIzol® reagent (Invitrogen; Thermo
Fisher Scientific, Inc.) and synthesized into first-strand cDNA
using an M-MLV Reverse Transcriptase kit (cat. no. 28025013;
Invitrogen; Thermo Fisher Scientific, Inc.) as previously described
(25). qPCR was performed using a
One Step TB Green PrimeScript RT-PCR kit II (cat. no. RR086A;
Takara Bio, Inc.). mRNA expression levels were normalized to U6 and
GAPDH using the 2-ΔΔCq method (26). The following thermocycling
conditions were used: Initial denaturation at 95˚C for 3 min,
followed by 40 cycles at 95˚C for 5 sec, 60˚C for 30 sec and at
72˚C for 45 sec, before final extension at 72˚C for 3 min. Primer
sequences are listed in Table
I.
 | Table IPrimers used for reverse
transcription-quantitative PCR. |
Table I
Primers used for reverse
transcription-quantitative PCR.
| Gene | Forward | Reverse |
|---|
| GRα |
CGAGCTCGTGTCTGTGACG |
AGAAAGTCTCCCATTGCCCA |
| IL-8 |
GAAGAGAGCTCTGTCTGGACC |
ACTGGCATCTTCACTGATTCT |
| TNFα |
CTGGGGCCTACAGCTTTGAT |
GGCCTAAGGTCCACTTGTGT |
| IL-6 |
TATGAACTCCTTCTCCACAAGCG |
AATCTTCTCCTGGGGGTACTGG |
| IL-1β |
GCAGAAGTACCTGAGCTCGC |
CTTGCTGTAGTGGTGGTCGG |
| XIST |
TGCATTTCCTTTCTGCCTCT |
TGCCCACATATGCAAAGAAA |
| GAPDH |
CCGCATCTTCTTTTGCGTCG |
TTCACCTTCCCCATGGTGTC |
Western blotting
Cells were lysed with lysis buffer containing
protease inhibitors (50 mM Tris-HCl pH 8; 50 mM NaCl; 0.5% NP-40).
Total protein was extracted from adenoid tissues and NP69 cells
using RIPA buffer (Thermo Fisher Scientific, Inc.). Protein
concentration was determined using a bicinchoninic acid assay.
Equal amounts of protein (5 µg for each sample) were resolved by
10% SDS-PAGE gels and transferred to PVDF membranes. Following
blocking with 5% non-fat milk at 25˚C for 1 h, the membranes were
immunoblotted with primary antibodies against GRα (cat. no.
ab3580), IL-8 (cat. no. ab18672), tumor necrosis factor α (TNFα;
cat. no. ab1793), IL-6 (cat. no. ab6672), IL-1β (cat. no. ab9722),
Rel-B (cat. no. ab180127), Rel-C (cat. no. ab133251), P52 (cat. no.
ab264236), P50 (cat. no. ab32360) and P65 (cat. no. ab16502) at a
dilution of 1:1,000 overnight at 4˚C. All primary antibodies were
purchased from Abcam. After washed using TBS + 0.1% Tween for 10
min three times, the membranes were exposed to horseradish
peroxidase-conjugated goat anti-rabbit antibodies (1:5,000; cat.
no. sc-2357, Santa Cruz Biotechnology, Inc.) Secondary antibodies
and signals were detected using an ECL detection system (Thermo
Fisher Scientific, Inc.). GAPDH (cat. no. ab8245; 1:1,000; Abcam)
was used as the loading control. Images were analyzed using ImageJ
software (version 1.51; National Institutes of Health).
ELISA
NP69 cell supernatants cultured in 24-well plates
were collected and the production of inflammatory cytokines was
measured using ELISA kits (R&D Systems, Inc.) including
interleukin 6 (IL-6; cat. no. D6050), interleukin 8 (IL-8; cat. no.
D8000C), tumor necrosis factor-α (TNF-α; cat. no. MTA00B) and
interleukin-1β (IL-1β; cat. no. MLB00C), according to the
manufacturer's protocols. Levels of inflammatory cytokines were
quantified by normalizing the protein concentrations (27).
Bioinformatics analysis
The binding site of XIST and GRαwas predicted by
starBase 2.0 website (http://starbase.sysu.edu.cn).
RNA pull-down assay
RNA pull-down assays were used to confirm the
interaction between XIST and GRα. Bio-NC, Bio-XIST and
Bio-XIST-Mutant constructs were synthesized by Shanghai Genepharma
Co., Ltd. The sequences were shown in Supplementary Table I. XIST was labeled with biotin to
generate the constructs. Sequences for the constructs are provided
in Table SI. Bio-NC was used as
the negative control and input, which is the cell lysate, was used
as the positive control. Following this, RIPA lysis buffer (Sangon
Biotech Co., Ltd.) containing RNase inhibitors (Invitrogen; Thermo
Fisher Scientific, Inc.) was utilized to lyse cells. Bio-XIST-WT,
Bio-XIST-MUT or Bio-NC constructs (200 pmol) were added to
supernatants and Dynabeads™ M-270 Streptavidin (1 mg; cat. no.
65305; Invitrogen; Thermo Fisher Scientific, Inc.) and proteinase K
(Sigma-Aldrich; Merck KGaA) were added and incubated with the
supernatants overnight at 4˚C to isolate the RNA. Beads were
isolated from the supernatant after centrifugation (2,500 x g, 5
min, 4˚C) and washed with wash buffer (10 mM Tris-HCl pH 7.5, 1 mM
EDTA, 2 M NaCl and 0.1% Tween-20) followed by another
centrifugation step (2,500 x g, 5 min, 4˚C). The pellet was then
collected and the RNA-RNA complexes were eluted using Tris-EDTA
buffer (Invitrogen; Thermo Fisher Scientific, Inc.) and purified
using ethanol. Subsequently, the degree of XIST and GRα enrichment
was analyzed by RT-qPCR as aforementioned.
Statistical analysis
Data are presented as mean ± standard deviation. For
comparison between two groups, unpaired Student's t-test was
applied. For comparison between multiple groups, data were analyzed
by one-way ANOVA followed by Tukey's post hoc test. Pearson's
correlation analysis was used to examine the correlation between
the levels of XIST and GRα. Statistical analysis was performed
using SPSS software (version 22.0; IBM Corp.). P<0.05 was
considered to indicate a statistically significant difference.
Results
GRα is decreased in the adenoids of
patients with OSAHS
Previous studies have demonstrated that GRα is an
important factor in the inhibition of inflammation. The current
study verified GRα expression in the adenoids of patients with
OSAHS. GRα mRNA was significantly decreased in the adenoids of
patients with OSAHS compared with healthy controls (Fig. 1A). Additionally, GRα protein levels
were significantly decreased in the adenoids of patients with OSAHS
compared with healthy controls (Fig.
1B and C). In contrast, the
western blot results demonstrated that the protein levels of
inflammatory cytokines, including IL-8, TNFα, IL-6 and IL-1β, were
significantly increased in the adenoids of patients with OSAHS
patients compared with healthy controls (Fig. 1D and E). In summary, GRα expression was
decreased and inflammatory cytokine expression was increased in the
adenoids of patients with OSAHS.
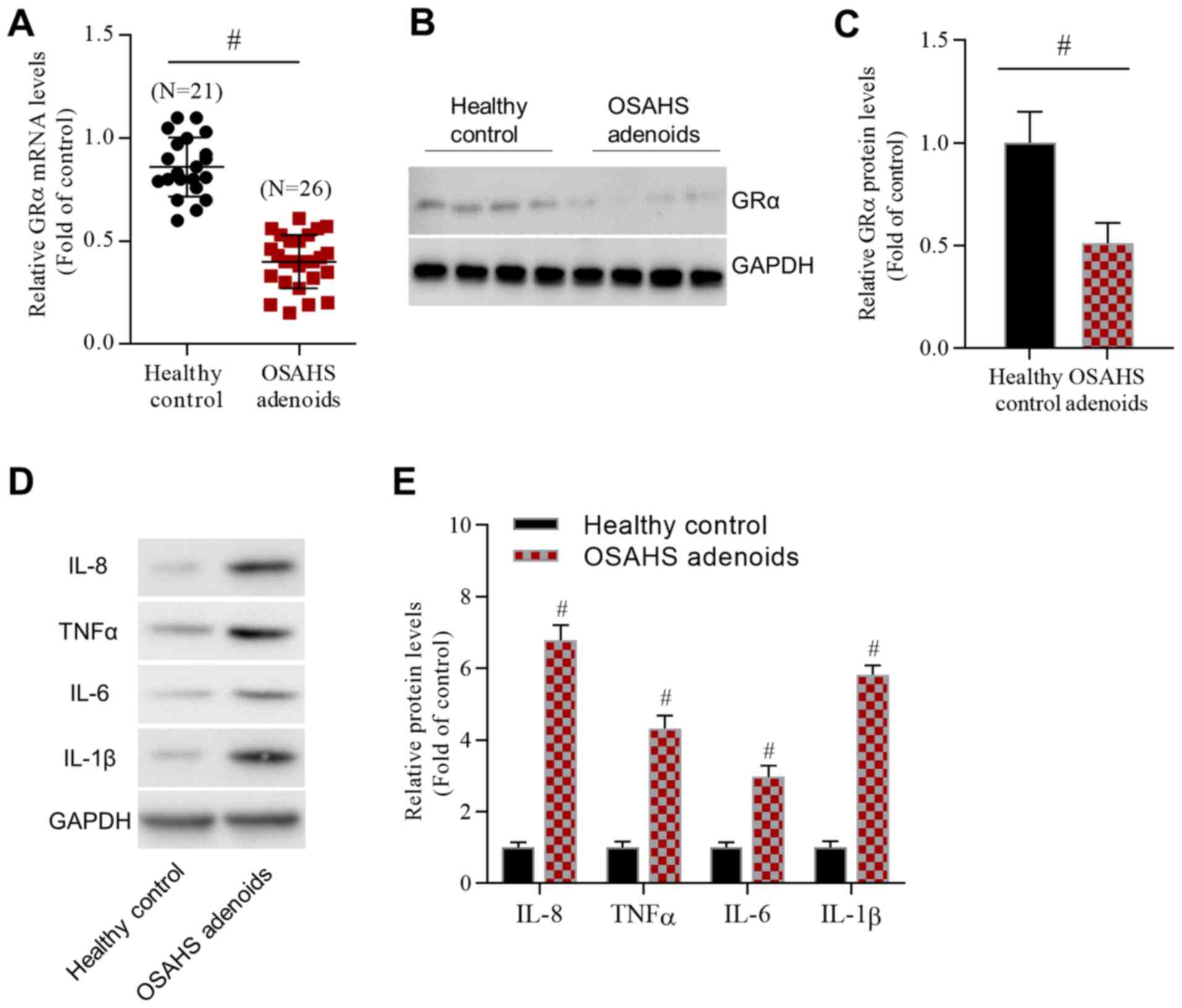 | Figure 1GRα is highly expressed in the
adenoids of patients with OSAHS. (A) Reverse
transcription-quantitative PCR data demonstrated changes in GRα
mRNA levels. (B) Representative western blot images and (C)
analyzed data reported changes in GRα protein levels. (D)
Representative western blot images and (E) analyzed data revealed
changes in the levels of inflammatory cytokines, including IL-8,
TNFα, IL-6 and IL-1β. For IL-8, IL-6 and IL-1β, n=4/group. For
TNFα, n=5/group. #P<0.05 vs. the healthy control
group. GRα, glucocorticoid receptor α; OSAHS, obstructive sleep
apnea/hypopnea syndrome; IL, interleukin; TNFα, tumor necrosis
factor α. |
XIST negatively regulates GRα
expression
A bioinformatics assay using the Starbase database
demonstrated that lncRNA XIST was closely associated with GRα. To
determine whether XIST interacted with GRα, the association between
the expression levels of XIST and GRα was analyzed using Pearson's
correlation analysis. According to the results, the expression of
XIST was negatively correlated with the expression of GRα (Fig. 2A). In contrast to the expression of
GRα, RT-qPCR demonstrated that the expression of XIST was
significantly increased in the adenoids of patients with OSAHS
compared with healthy controls (Fig.
2B). Furthermore, pcDNA-XIST significantly increased XIST
levels and XIST shRNA significantly decreased XIST levels in NP69
cells compared with those in the pcDNA and sh-NC groups,
respectively (Fig. 2C).
Additionally, the potential effect of XIST on GRα expression was
investigated. XIST overexpression significantly decreased GRα mRNA
levels and XIST shRNA significantly increased GRα mRNA levels
compared with those in the pcDNA and sh-NC groups, respectively
(Fig. 2D). Similarly, pcDNA-XIST
significantly decreased GRα protein levels and XIST shRNA
significantly increased the GRα protein levels compared with those
in the pcDNA and sh-NC groups, respectively (Fig. 2E and F). Following this, the interaction between
XIST and GRα was examined. The predicted binding sites for XIST and
GRα were obtained from the Starbase database (Fig. 2G). Bio-XIST-WT and Bio-XIST-MUT
constructs were generated for RNA pull-down assays. The results
demonstrated that XIST and GRα were enriched in the Bio-XIST-WT
group compared with the Bio-XIST-MUT group (Fig. 2H). These results indicated that XIST
negatively regulated GRα.
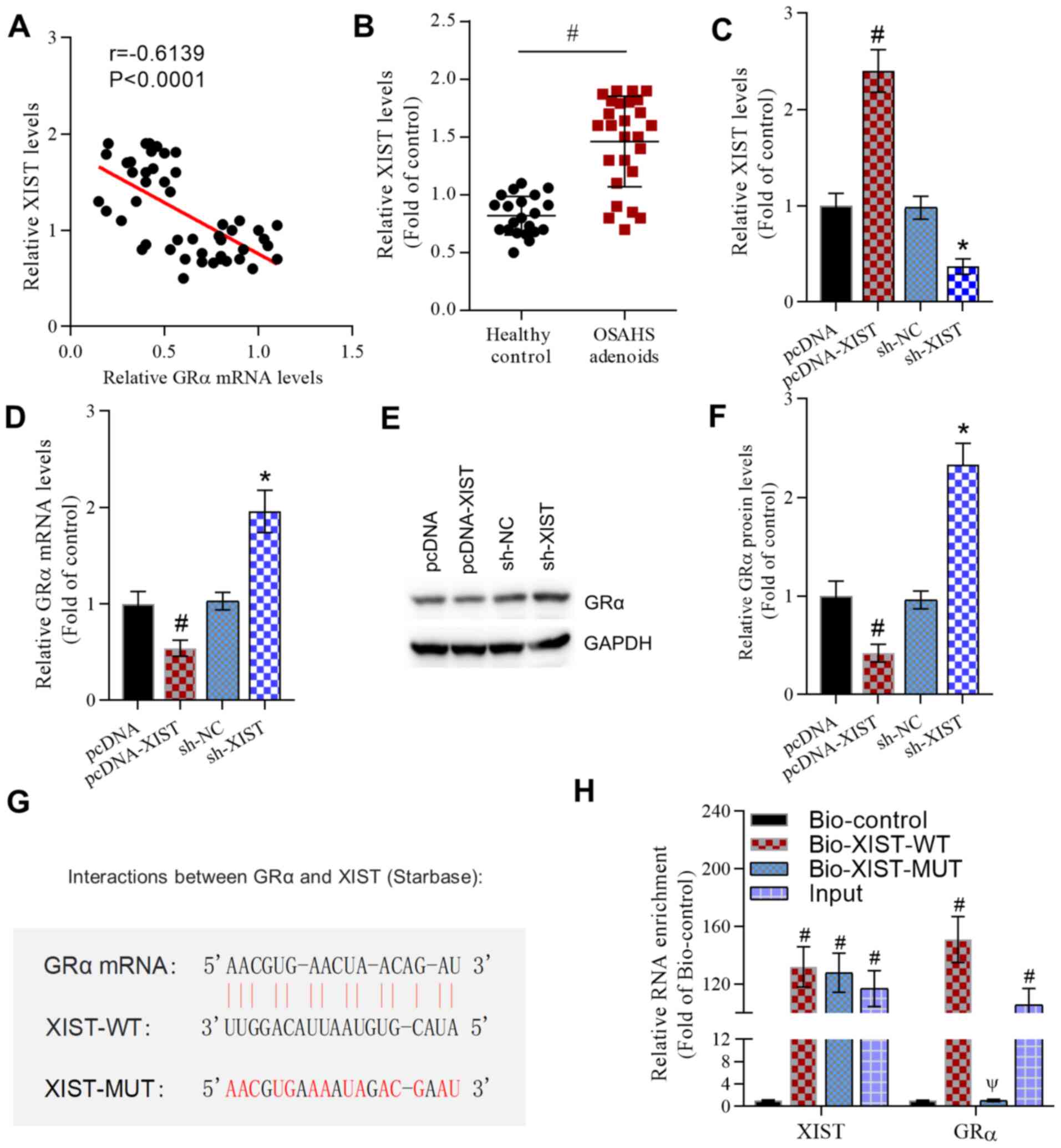 | Figure 2XIST negatively regulates GRα
expression. (A) Pearson's correlation analysis indicated a negative
correlation between the XIST and GRα mRNA levels. r=-0.6139.
P<0.0001. (B) RT-qPCR revealed increased XIST expression levels
in the adenoids of patients with OSAHS compared with healthy
controls. #P<0.05 as indicated. (C) RT-qPCR data
demonstrated the effect of the overexpression or knockdown of XIST
on XIST levels in NP69 cells. #P<0.05 vs. the pcDNA
group and *P<0.05 vs. the sh-NC group. (D) RT-qPCR
data revealed the effect of the overexpression or knockdown of XIST
on GRα mRNA levels in NP69 cells. #P<0.05 vs. the
pcDNA group and *P<0.05 vs. the sh-NC group. (E)
Representative western blot images and (F) analyzed data
demonstrated the effect of the overexpression or knockdown of XIST
on GRα protein levels in NP69 cells. #P<0.05 vs. the
pcDNA group and *P<0.05 vs. the sh-NC group. (G) The
binding sites and mutated sites of XIST and GRα. (H) RNA pull-down
assays confirmed the interaction between XIST and GRα.
#P<0.05 vs. the Bio-control group and
ΨP<0.05 vs. the Bio-XIST-WT group. No significance
between GRα enrichment in Bio-XIST-MUT and Bio-control group. For
XIST, n=4/group. For GRα, n=5/group. XIST, X inactivate-specific
transcript; GRα, glucocorticoid receptor α; RT-qPCR, reverse
transcription-quantitative PCR; OSAHS, obstructive sleep
apnea/hypopnea syndrome; sh, short hairpin; NC, negative control;
WT, wild-type; MUT, mutant. |
The XIST-GRα signaling pathway
regulates the production of inflammatory cytokines
Whether the XIST-GRα axis regulated inflammation was
investigated. GRα expression was significantly overexpressed by
pcDNA-GRα transfection compared with that following pcDNA
transfection (Fig. 3A). XIST
significantly increased the mRNA levels (Fig. 3B), protein levels (Fig. 3C and D and E)
concentrations of inflammatory cytokines, including IL-8, TNFα,
IL-6 and IL-1β, compared with those in the pcDNA group. However,
these stimulatory effects were reversed by XIST-MUT. Therefore, GRα
overexpression significantly decreased the mRNA levels, protein
levels and concentrations of IL-8, TNFα, IL-6 and IL-1β compared
with those in the pcDNA group. Furthermore, the stimulatory effect
of XIST on the production of inflammatory cytokines was
significantly reversed by GRα compared with that in the pcDNA and
pcDNA-XIST + pcDNA-GRα groups (Fig.
3F). These data indicated that the XIST-GRα signaling pathway
contributed to the regulation of inflammatory cytokine
production.
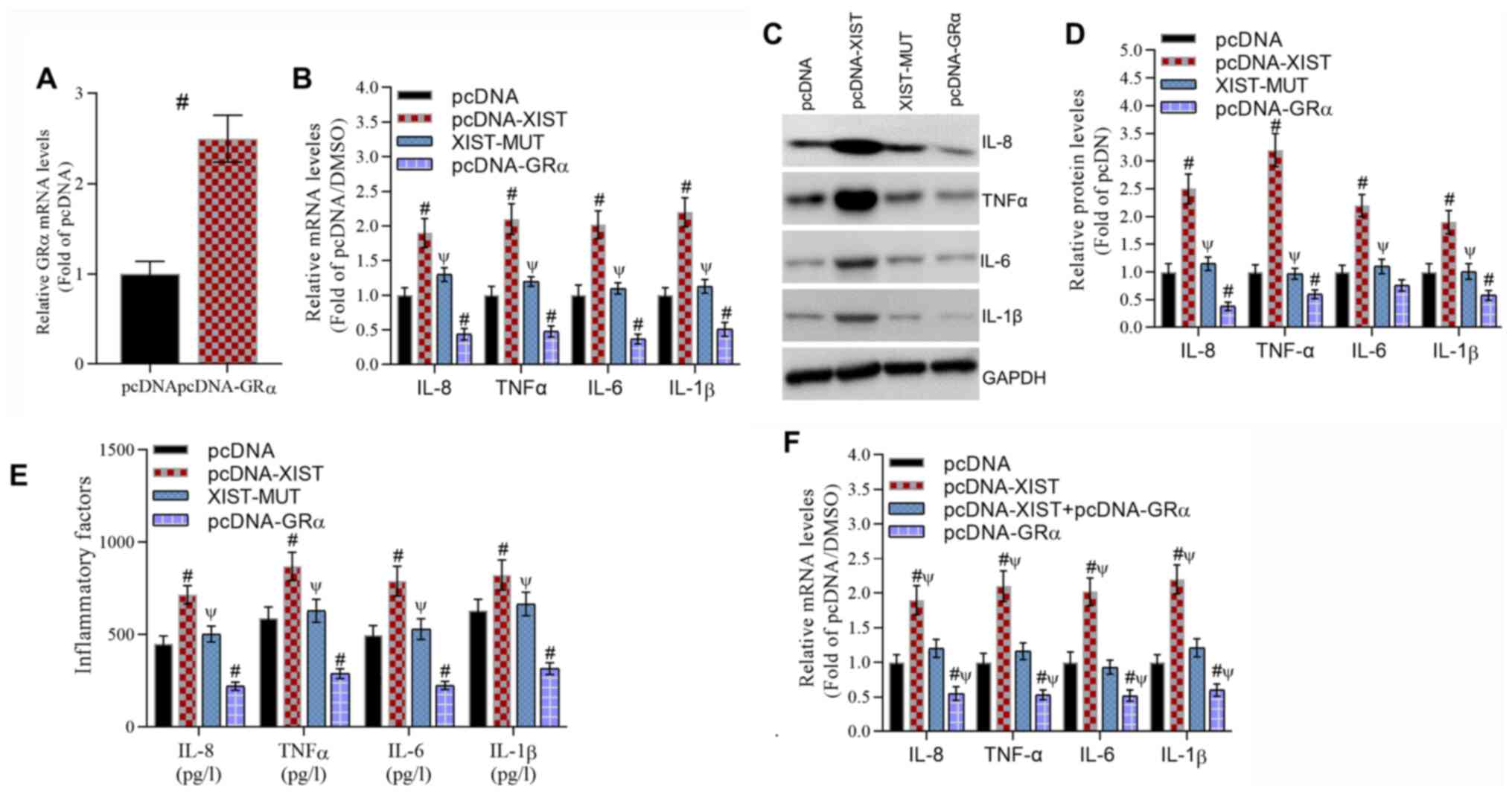 | Figure 3XIST promotes the production of
inflammatory cytokines, including IL-8, TNFα, IL-6 and IL-1β, that
depend on the binding of XIST and GRα. (A) RT-qPCR data
demonstrated the effect of GRα overexpression on GRα mRNA levels.
#P<0.05 vs. the pcDNA group. (B) RT-qPCR data
revealed the effect of XIST-WT, XIST-MUT and GRα overexpression on
the mRNA levels of inflammatory cytokines. #P<0.05
vs. the pcDNA group and ΨP<0.05 vs. the XIST group.
(C) Representative western blotting images and (D) analyzed data
demonstrated the effect of XIST-WT, XIST-MUT and GRα overexpression
on the protein levels of inflammatory cytokines.
#P<0.05 vs. the pcDNA group and ΨP<0.05
vs. the XIST group. (E) Analyzed data revealed the effect of
XIST-WT, XIST-MUT and GRα overexpression on the protein levels of
inflammatory cytokines as determined by ELISA.
#P<0.05 vs. the pcDNA group and ΨP<0.05
vs. the XIST group. (F) Analyzed data demonstrated that the
inhibitory effect of GRα overexpression reversed the stimulatory
effect of XIST on the protein levels of inflammatory cytokines as
determined by ELISA. #P<0.05 vs. the pcDNA group and
ΨP<0.05 vs. the XIST+GRα group. For IL-8, IL-6 and
IL-1β, n=5/group. For TNFα, n=6/group. XIST, X inactivate-specific
transcript; GRα, glucocorticoid receptor α; RT-qPCR, reverse
transcription-quantitative PCR; WT, wild-type; MUT, mutant; IL,
interleukin; TNFα, tumor necrosis factor α. |
XIST regulates adenoidal inflammation
through the GRα-mediated NF-κB pathway
Finally, we evaluated whether NF-κB was involved in
XIST-GRα-induced inflammation. XIST significantly increased the
protein levels of Rel-B, c-Rel, P52, P50 and P65 compared with that
in the pcDNA/DMSO group (Fig. 4A
and B). These effects were
significantly inhibited by the NF-κB inhibitor EVP4593.
Additionally, GRα inactivated the NF-κB pathway and significantly
reversed XIST-induced expression of NF-κB pathway-associated
proteins compared with that in the pcDNA-XIST/DMSO group.
Similarly, XIST induced a significant increase in the mRNA levels
(Fig. 4C) and concentrations
(Fig. 4D) of IL-8, TNFα, IL-6 and
IL-1β compared with those in the pcDNA/DMSO group. Furthermore,
compared with those in the pcDNA-XIST/DMSO group, these levels were
significantly inhibited by EVP4593 or GRα, counteracting the
effects of XIST in facilitating the inflammatory response. These
results indicated that XIST induced an inflammatory response
through the GRα-mediated NF-κB pathway.
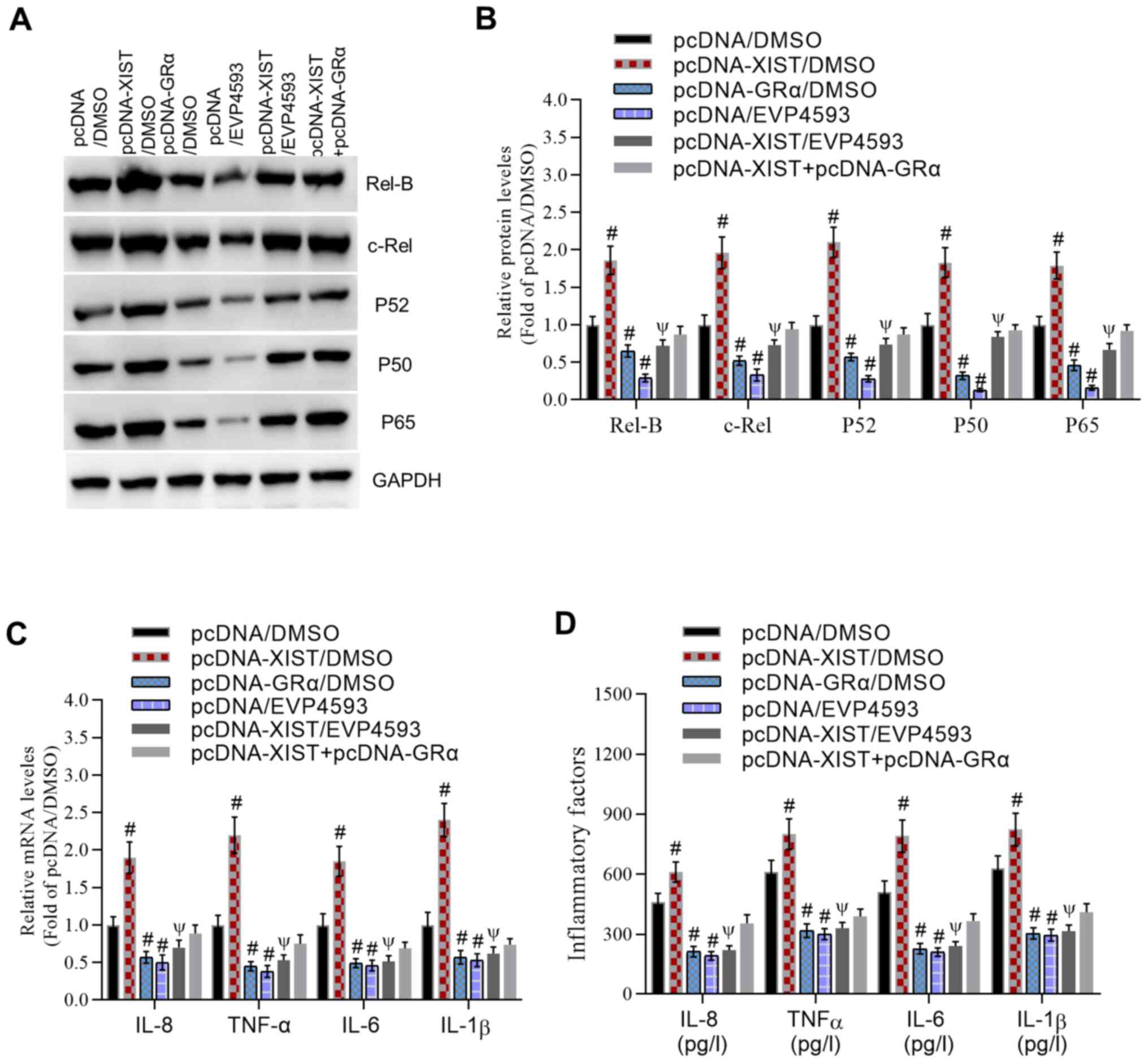 | Figure 4XIST regulates adenoidal inflammation
through the GRα-mediated NF-κB pathway. (A) Representative western
blot images and (B) analyzed data demonstrated the inhibitory
effect of the NFκB inhibitor EVP4593 and GRα on the XIST-induced
increase in protein levels of Rel-B, c-Rel, P52, P50 and P65. (C)
Reverse transcription-quantitative PCR data revealed the inhibitory
effect of EVP4593 and GRα on the XIST-induced increase in the mRNA
levels of inflammatory cytokines. (D) Analyzed data reported the
inhibitory effect of EVP4593 and GRα on the XIST-induced increase
in the protein levels of inflammatory cytokines as determined by
ELISA. For IL-8, IL-6 and IL-1β, n=5/group. For TNFα, n=6/group.
#P<0.05 vs. the pcDNA/DMSO group and
ΨP<0.05 vs. the XIST/DMSO group. XIST, X
inactivate-specific transcript; GRα, glucocorticoid receptor α; NF-
κB, nuclear factor κ-light-chain-enhancer of activated B cells; IL,
interleukin; TNFα, tumor necrosis factor α. |
Discussion
OSAHS is a risk factor for hypertension, arrhythmia,
coronary heart disease and other diseases (28). Therefore, investigating the specific
molecular regulatory mechanisms of OSAHS is crucial. The current
study evaluated whether GRα was involved in the inflammatory
response in OSAHS. The results demonstrated that GRα expression was
significantly decreased and XIST expression was significantly
increased in the adenoids of patients with OSAHS compared with
healthy controls. Further research revealed that XIST interacted
with GRα and decreased GRα expression. Functionally, XIST increased
the production of inflammatory cytokines, including IL-8, TNF-α,
IL-6 and IL-1β, while GRα decreased their production. Furthermore,
the results demonstrated that XIST-GRα axis-mediated inflammation
was significantly inhibited by the NF-κB inhibitor EVP4593. These
results indicated that the XIST-GRα-NF-κB signaling pathway
contributed to inflammation in the adenoids of patients with
OSAHS.
The current study reported that GRα was decreased in
patients with OSAHS, which was consistent with a previous study
that revealed that GRα was decreased in the tonsils of children
with OSAHS (10). Furthermore, the
present study demonstrated that decreased GRα contributed to
increased inflammation in patients with OSAHS. Furthermore, the
present study reported that XIST was upregulated and decreased the
expression of GRα in patients with OSAHS. Consistent with the
present study, accumulating data have demonstrated that XIST is
upregulated and involved in various diseases, including basal-like
human breast cancer (15) and
cystic fibrosis (16). Shenoda
et al (29) reported that
XIST promotes inflammation through the pro-inflammatory
transcription factor yin yang 1 in patients with complex regional
pain syndrome. Those studies indicated that XIST served a critical
role in inflammation. Additionally, XIST promotes the progression
of non-small cell lung cancer through the microRNA
(miR)-335/superoxide dismutase 2/reactive oxygen species pathway
(30). Inhibition of XIST
suppresses cell proliferation via modulation of the miR-744/ring
finger protein 1 axis in non-small cell lung cancer (31). Suppression of XIST increases the
secretion of exosomal miR-503, which triggers M1-M2 polarization of
microglia and promotes brain metastasis (32). The results of the current study
indicated that XIST interacted with GRα and inhibited GRα
expression. GRα has been previously reported to serve an
anti-inflammatory role in cardiomyocytes (33). The present study revealed that GRα
alleviated the inflammatory response and inactivated the NF-κB
pathway in NP69 cells.
Numerous previous studies have demonstrated that
NF-κB serves a vital role in inflammation. For instance, it has
been reported that NF-κB upregulated the synthesis and secretion of
various pro-inflammatory cytokines, including TNF-α, IL-1β and
IL-6. Fu et al (34)
reported that pharmacologically inhibiting neddylation with MLN4924
suppressed pro-inflammatory cytokine generation through the NF-κB
pathway in lipopolysaccharide (LPS)-stimulated HK2 cells and
attenuates renal inflammation in LPS-induced acute kidney damage.
Zhao et al (35) revealed
that XIST upregulated TNF-α induced protein 1 expression to
increase NF-kB activity and, therefore, exacerbating neuropathic
pain in a rat model. XIST alleviates LPS-induced cell injury by
modulating the Janus kinase/signal transducer and activator of
transcription and NF-κB pathways (36). However, Ma et al (17) demonstrated that the expression of
XIST was promoted by the activation of the NF-κB pathway and that
XIST generated a negative feedback loop to regulate the NF-κB/NLR
family CARD domain containing 6, leucine-rich repeat and PYD
domain-containing protein 3 inflammasome pathways to mediate
inflammatory processes. The aforementioned studies indicated that
the association between XIST and NF-κB is complex. The present
study identified that GRα suppressed activation of the NF-κB
pathway and that XIST activated the NF-κB pathway through the
inhibition of GRα. However, the underlying mechanism by which GRα
inactivates the NF-κB pathway remains unclear. Future studies will
investigate the mechanism by which GRα suppresses the activation of
the NF-κB pathway in the progression of OSAHS.
In summary, the present study demonstrated that XIST
decreased the expression of GRα, thereby contributing to the
initiation and development of OSAHS through an NF-κB-dependent
signaling pathway. This may provide a potential target for the
treatment of OSAHS.
Supplementary Material
Sequences for the Bio-control,
Bio-XIST and Bio-XIST-Mutant.
Acknowledgements
Not applicable.
Funding
The current study was supported by the Hangzhou Science and
Technology Development Plan Project (grant nos. 20170533B94 and
20170533B27).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Author's contributions
ZZ conceived and designed the experiments. ZZ, HN,
YL and BJ performed the experiments. ZZ and HN analyzed the data.
ZZ and HN can authenticate the raw data in this study. All authors
agreed to be accountable for all aspects of the work. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
The current study was approved by the Ethics
Committee and Animal Care Committee of the Affiliated Hangzhou
First People's Hospital, Zhejiang University School of Medicine
(Hangzhou, China). Informed consent was obtained from all
participants included in the current study. All procedures
performed in studies involving human participants were in
accordance with the ethical standards of the institutional and/or
national research committee and with the 1964 Declaration of
Helsinki and its later amendments or comparable ethical
standards.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zhang L, Ou X, Zhu T and Lv X: Beneficial
effects of estrogens in obstructive sleep apnea hypopnea syndrome.
Sleep Breath. 24:7–13. 2020.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gonzaga C, Bertolami A, Bertolami M,
Amodeo C and Calhoun D: Obstructive sleep apnea, hypertension and
cardiovascular diseases. J Hum Hypertens. 29:705–12.
2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Jordan AS and McEvoy RD: Gender
differences in sleep apnea: Epidemiology, clinical presentation and
pathogenic mechanisms. Sleep Med Rev. 7:377–389. 2003.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Morong S, Hermsen B and de Vries N:
Sleep-disordered breathing in pregnancy: A review of the physiology
and potential role for positional therapy. Sleep Breath. 18:31–37.
2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Snyder B and Cunningham RL: Sex
differences in sleep apnea and comorbid neurodegenerative diseases.
Steroids. 133:28–33. 2018.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Evans RM: The steroid and thyroid hormone
receptor superfamily. Science. 240:889–895. 1988.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ren R, Oakley RH, Cruz-Topete D and
Cidlowski JA: Dual role for glucocorticoids in cardiomyocyte
hypertrophy and apoptosis. Endocrinology. 153:5346–5360.
2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Kim HK, Jeong YJ, Song IS, Noh YH, Seo KW,
Kim M and Han J: Glucocorticoid receptor positively regulates
transcription of FNDC5 in the liver. Sci Rep.
7(43296)2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Kuo T, Chen TC, Yan S, Foo F, Ching C,
McQueen A and Wang JC: Repression of glucocorticoid-stimulated
angiopoietin-like 4 gene transcription by insulin. J Lipid Res.
55:919–28. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Chen X and Li JR: Glucocorticoid receptor
expression in the tonsils of children with obstructive sleep apnea
hypopnea syndrome. Genet Mol Res. 15:2016.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Choi SW, Kim HW and Nam JW: The small
peptide world in long noncoding RNAs. Brief Bioinform.
20:1853–1864. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Barth DA, Slaby O, Klec C, Juracek J,
Drula R, Calin GA and Pichler M: Current concepts of non-coding
RNAs in the pathogenesis of non-clear cell renal cell carcinoma.
Cancers (Basel). 11(1580)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Rapicavoli NA, Qu K, Zhang J, Mikhail M,
Laberge RM and Chang HY: A mammalian pseudogene lncRNA at the
interface of inflammation and anti-inflammatory therapeutics.
Elife. 2(e00762)2013.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Carpenter S, Aiello D, Atianand MK, Ricci
EP, Gandhi P, Hall LL, Byron M, Monks B, Henry-Bezy M, Lawrence JB,
et al: A long noncoding RNA mediates both activation and repression
of immune response genes. Science. 341:789–792. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Richardson AL, Wang ZC, De Nicolo A, Lu X,
Brown M, Miron A, Liao X, Iglehart JD, Livingston DM and Ganesan S:
X chromosomal abnormalities in basal-like human breast cancer.
Cancer Cell. 9:121–132. 2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
McKiernan PJ, Molloy K, Cryan SA,
McElvaney NG and Greene CM: Long noncoding RNA are aberrantly
expressed in vivo in the cystic fibrosis bronchial epithelium. Int
J Biochem Cell Biol. 52:184–191. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ma M, Pei Y, Wang X, Feng J, Zhang Y and
Gao MQ: LncRNA XIST mediates bovine mammary epithelial cell
inflammatory response via NF-kappaB/NLRP3 inflammasome pathway.
Cell Prolif. 52(e12525)2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Sadeghi A, Rostamirad A, Seyyedebrahimi S
and Meshkani R: Curcumin ameliorates palmitate-induced inflammation
in skeletal muscle cells by regulating JNK/NF-kB pathway and ROS
production. Inflammopharmacology. 26:1265–1272. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Nedjai B, Hitman GA, Church LD, Minden K,
Whiteford ML, McKee S, Stjernberg S, Pettersson T, Ranki A, Hawkins
PN, et al: Differential cytokine secretion results from p65 and
c-Rel NF-κB subunit signaling in peripheral blood mononuclear cells
of TNF receptor-associated periodic syndrome patients. Cell
Immunol. 268:55–59. 2011.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Ruland J: Return to homeostasis:
Downregulation of NF-κB responses. Nature Immunol. 12:709–714.
2011.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Lai JL, Liu YH, Liu C, Qi MP, Liu RN, Zhu
XF, Zhou QG, Chen YY, Guo AZ and Hu CM: Indirubin inhibits
LPS-induced inflammation via TLR4 abrogation mediated by the NF-κB
and MAPK signaling pathways. Inflammation. 40:1–12. 2017.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yancy CW, Jessup M, Bozkurt B, Butler J,
Casey DE Jr, Drazner MH, Fonarow GC, Geraci SA, Horwich T, Januzzi
JL, et al: 2013 ACCF/AHA guideline for the management of heart
failure: A report of the American College of Cardiology
Foundation/American Heart Association Task Force on Practice
Guidelines. J Am Coll Cardiol. 62:147–239. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Ye XH, Chen H, Yu Q and Zhu QL: Liver X
receptor gene expression is enhanced in children with obstructive
sleep apnea-hyperpnoea syndrome and cyclooxygenase-2 (COX-2) is
correlated with severity of obstructive sleep apnea-hypopnea
syndrome (OSAHS). Med Sci Monit. 23:3261–3268. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Guo Z, Wang Y, Yang J, Zhong J, Liu X and
Xu M: KAI1 overexpression promotes apoptosis and inhibits
proliferation, cell cycle, migration, and invasion in
nasopharyngeal carcinoma cells. Am J Otolaryngol. 38:511–517.
2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Pathare AD and Deshpande AS: HGV-HCV/HBV
co-infection in India: A pilot study. Asian J Transfus Sci.
7:48–50. 2013.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–8. 2001.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Yu X, Wang D, Wang X, Sun S, Zhang Y, Wang
S, Miao R, Xu X and Qu X: CXCL12/CXCR4 promotes inflammation-driven
colorectal cancer progression through activation of RhoA signaling
by sponging miR-133a-3p. J Exp Clin Cancer Res.
38(32)2019.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Burman D: Sleep disorders: Sleep-related
breathing disorders. FP Essent. 460:11–21. 2017.PubMed/NCBI
|
|
29
|
Shenoda BB, Tian Y, Alexander GM,
Aradillas-Lopez E, Schwartzman RJ and Ajit SK: miR-34a-mediated
regulation of XIST in female cells under inflammation. J Pain Res.
11:935–945. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Liu J, Yao L, Zhang M, Jiang J, Yang M and
Wang Y: Downregulation of LncRNA-XIST inhibited development of
non-small cell lung cancer by activating miR-335/SOD2/ROS signal
pathway mediated pyroptotic cell death. Aging. 11:7830–7846.
2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Wang J, Cai H, Dai Z and Wang G:
Down-regulation of lncRNA XIST inhibits cell proliferation via
regulating miR-744/RING1 axis in non-small cell lung cancer. Clin
Sci (Lond). 133:1567–1579. 2019.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Xing F, Liu Y, Wu SY, Wu K, Sharma S, Mo
YY, Feng J, Sanders S, Jin G, Singh R, et al: Loss of XIST in
breast cancer activates MSN-c-Met and reprograms microglia via
exosomal miRNA to promote brain metastasis. Cancer Res.
78:4316–4330. 2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Song YX, Ou YM and Zhou JY: Gracillin
inhibits apoptosis and inflammation induced by lipopolysaccharide
(LPS) to alleviate cardiac injury in mice via improving miR-29a.
Biochem Biophys Res Commun. 523:580–587. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Fu Z, Liao W, Ma H, Wang Z, Jiang M, Feng
X and Zhang W: Inhibition of neddylation plays protective role in
lipopolysaccharide-induced kidney damage through CRL-mediated
NF-kappaB pathways. Am J Transl Res. 11:2830–2842. 2019.PubMed/NCBI
|
|
35
|
Zhao Y, Li S, Xia N, Shi Y and Zhao CM:
Effects of XIST/miR-137 axis on neuropathic pain by targeting
TNFAIP1 in a rat model. J Cell Physiol. 233:4307–4316.
2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhang Y, Zhu Y, Gao G and Zhou Z:
Knockdown XIST alleviates LPS-induced WI-38 cell apoptosis and
inflammation injury via targeting miR-370-3p/TLR4 in acute
pneumonia. Cell Biochem Funct. 37:348–358. 2019.PubMed/NCBI View
Article : Google Scholar
|


















