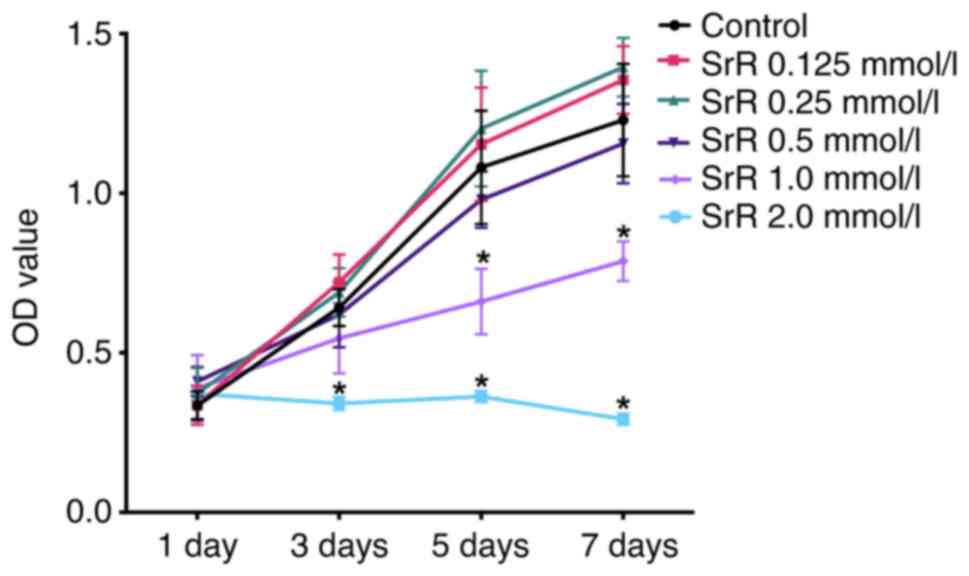
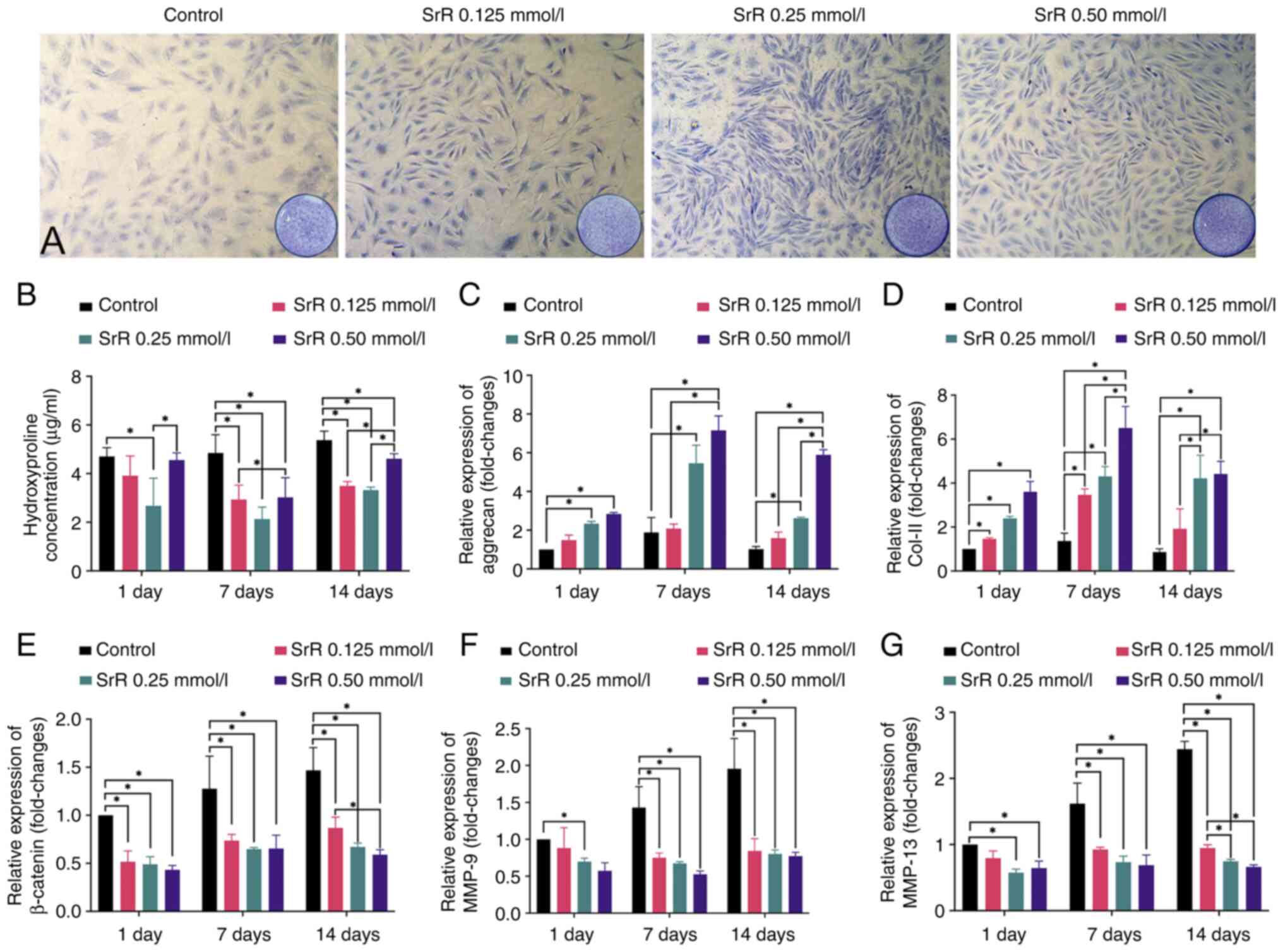
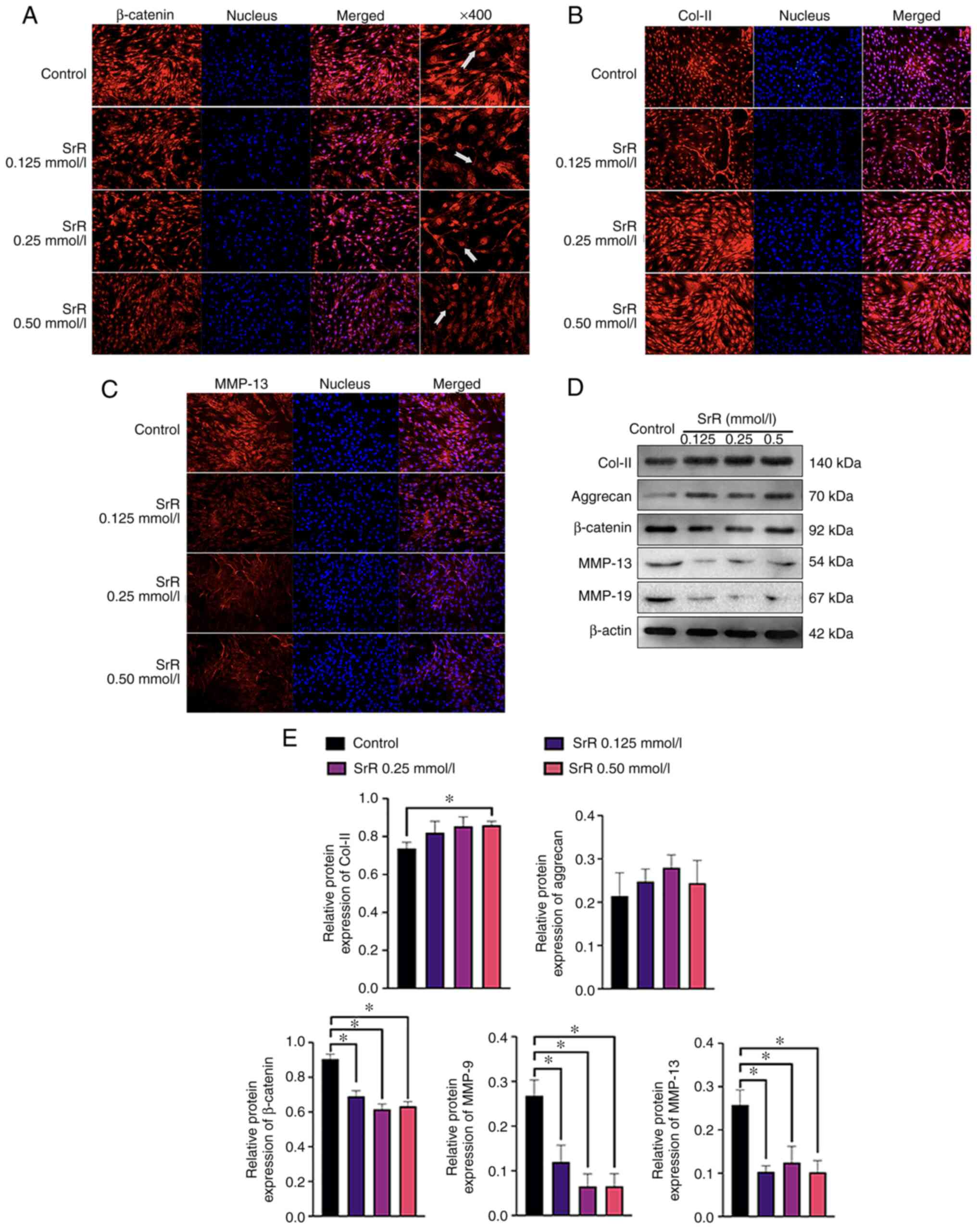
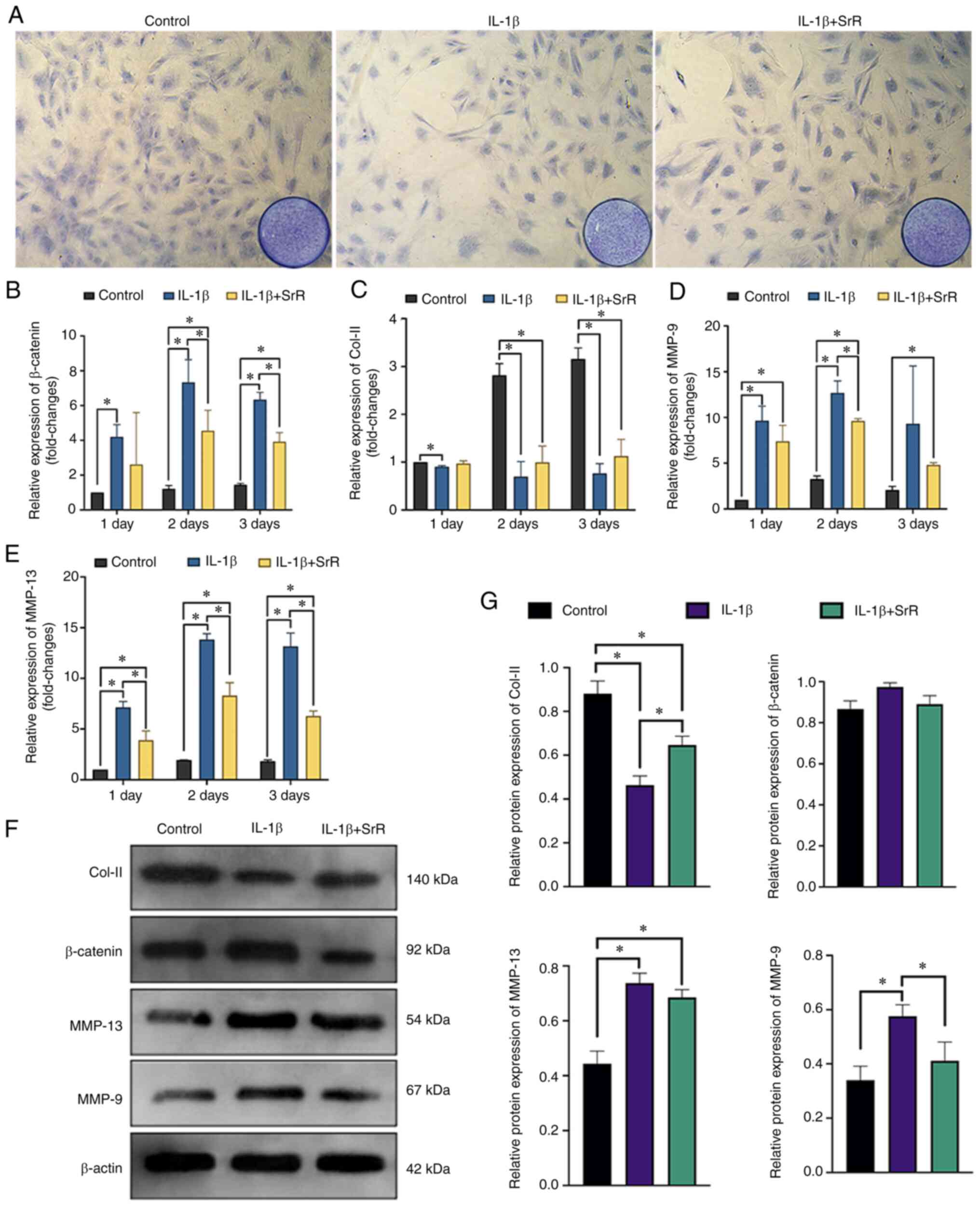
|
1
|
Joseph R, Rahena A, Hassan N, Glen H,
James W and Soichiro I: Epidemiology of temporomandibular disorder
in the general population: A systematic review. Adv Dent Oral
Health. 10(555787)2019.
|
|
2
|
Wang XD, Zhang JN, Gan YH and Zhou YH:
Current understanding of pathogenesis and treatment of TMJ
osteoarthritis. J Dent Res. 94:666–673. 2015.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Charlier E, Deroyer C, Ciregia F, Malaise
O, Neuville S, Plener Z, Malaise M and de Seny D: Chondrocyte
dedifferentiation and osteoarthritis (OA). Biochem Pharmacol.
165:49–65. 2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wojdasiewicz P, Poniatowski ŁA and
Szukiewicz D: The role of inflammatory and anti-inflammatory
cytokines in the pathogenesis of osteoarthritis. Mediators Inflamm.
2014(561459)2014.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Loeser RF: Molecular mechanisms of
cartilage destruction: Mechanics, inflammatory mediators, and aging
collide. Arthritis Rheum. 54:1357–1360. 2006.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Johnson CI, Argyle DJ and Clements DN: In
vitro models for the study of osteoarthritis. Vet J. 209:40–49.
2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Samavedi S, Diaz-Rodriguez P, Erndt-Marino
JD and Hahn MS: A three-dimensional chondrocyte-macrophage
coculture system to probe inflammation in experimental
osteoarthritis. Tissue Eng Part A. 23:101–114. 2017.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Gu YT, Chen J, Meng ZL, Ge WY, Bian YY,
Cheng SW, Xing CK, Yao JL, Fu J and Peng L: Research progress on
osteoarthritis treatment mechanisms. Biomed Pharmacother.
93:1246–1252. 2017.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Xia B, Chen D, Zhang J, Hu S, Jin H and
Tong P: Osteoarthritis pathogenesis: A review of molecular
mechanisms. Calcif Tissue Int. 95:495–505. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Grässel S and Muschter D: Recent advances
in the treatment of osteoarthritis. F1000Res. 9(325)2020.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Pelletier JP, Roubille C, Raynauld JP,
Abram F, Dorais M, Delorme P and Martel-Pelletier J:
Disease-modifying effect of strontium ranelate in a subset of
patients from the phase III knee osteoarthritis study SEKOIA using
quantitative MRI: Reduction in bone marrow lesions protects against
cartilage loss. Ann Rheum Dis. 74:422–429. 2015.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tenti S, Cheleschi S, Guidelli GM,
Galeazzi M and Fioravanti A: What about strontium ranelate in
osteoarthritis? Doubts and securities. Mod Rheumatol. 24:881–884.
2014.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Tat SK, Pelletier JP, Mineau F, Caron J
and Martel-Pelletier J: Strontium ranelate inhibits key factors
affecting bone remodeling in human osteoarthritic subchondral bone
osteoblasts. Bone. 49:559–567. 2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Yu DG, Ding HF, Mao YQ, Liu M, Yu B, Zhao
X, Wang XQ, Li Y, Liu GW, Nie SB, et al: Strontium ranelate reduces
cartilage degeneration and subchondral bone remodeling in rat
osteoarthritis model. Acta Pharmacol Sin. 34:393–402.
2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yu H, Liu Y, Yang X, He J, Zhang F, Zhong
Q and Guo X: Strontium ranelate promotes chondrogenesis through
inhibition of the Wnt/β-catenin pathway. Stem Cell Res Ther.
12(296)2021.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Sun Y, Zhou L, Lv D, Liu H, He T and Wang
X: Poly(ADP-ribose) polymerase 1 inhibition prevents
interleukin-1β-induced inflammation in human osteoarthritic
chondrocytes. Acta Biochim Biophys Sin (Shanghai). 47:422–430.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Alves SM, Abreu SC, Lemos JC, Gomes FI,
Alves SM, do Val DR, Freitas RS, Pereira KM, de Paulo Teixeira
Pinto V, de Castro Brito GA, et al: Anti-inflammatory and
anti-nociceptive effects of strontium ranelate on the
zymosan-induced temporomandibular joint inflammatory
hypernociception in rats depend on TNF-α inhibition. Pharmacol Rep.
69:764–772. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Henrotin Y, Labasse A, Zheng SX, Galais P,
Tsouderos Y, Crielaard JM and Reginster JY: Strontium ranelate
increases cartilage matrix formation. Bone Miner Res. 16:299–308.
2001.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang L, Lazebnik M and Detamore MS:
Hyaline cartilage cells outperform mandibular condylar cartilage
cells in a TMJ fibrocartilage tissue engineering application.
Osteoarthritis Cartilage. 17:346–353. 2009.PubMed/NCBI View Article : Google Scholar
|
|
20
|
National Research Council. Guide for the
care and use of laboratory animals: Eighth edition. Washington, DC:
The National Academies Press, 2011. https://doi.org/10.17226/12910.
|
|
21
|
Guo X, Wei S, Lu M, Shao Z, Lu J, Xia L,
Lin K and Zou D: Dose-dependent effects of strontium ranelate on
ovariectomy rat bone marrow mesenchymal stem cells and human
umbilical vein endothelial cells. Int J Biol Sci. 12:1511–1522.
2016.PubMed/NCBI View Article : Google Scholar
|
|
22
|
da Silva CM, Spinelli E and Rodrigues SV:
Fast and sensitive collagen quantification by alkaline
hydrolysis/hydroxyproline assay. Food Chem. 173:619–623.
2015.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Aimaiti A, Maimaitiyiming A, Boyong X, Aji
K, Li C and Cui L: Low-dose strontium stimulates osteogenesis but
high-dose doses cause apoptosis in human adipose-derived stem cells
via regulation of the ERK1/2 signaling pathway. Stem Cell Res Ther.
8(282)2017.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mao Z, Fang Z, Yang Y, Chen X, Wang Y,
Kang J, Qu X, Yuan W and Dai K: Strontium ranelate-loaded PLGA
porous microspheres enhancing the osteogenesis of MC3T3-E1 cells.
RSC Adv. 7:24607–24615. 2017.
|
|
26
|
Pilmane M, Salma-Ancane K, Loca D, Locs J
and Berzina-Cimdina L: Strontium and strontium ranelate: Historical
review of some of their functions. Mater Sci Eng C Mater Biol Appl.
78:1222–1230. 2017.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Deepthi S, Abdul Gafoor AA, Sivashanmugam
A, Nair SV and Jayakumar R: Nanostrontium ranelate incorporated
injectable hydrogel enhanced matrix production supporting
chondrogenesis in vitro. J Mater Chem B. 4:4092–4103.
2016.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Pan FY, Li ZM, Liu XW, Luo Y, Ma Z, Feng
SX and Xu N: Effect of strontium ranelate on rabbits with
steroid-induced osteonecrosis of femoral head through TGF-β1/BMP2
pathway. Eur Rev Med Pharmacol Sci. 24:1000–1006. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Jackson A and Gu W: Transport properties
of cartilaginous tissues. Curr Rheumatol Rev. 5(40)2009.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Troeberg L and Nagase H: Proteases
involved in cartilage matrix degradation in osteoarthritis. Biochim
Biophys Acta. 1824:133–145. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Liu-Bryan R and Terkeltaub R: Emerging
regulators of the inflammatory process in osteoarthritis. Nat Rev
Rheumatol. 11:35–44. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Pelletier JP, Kapoor M, Fahmi H,
Lajeunesse D, Blesius A, Maillet J and Martel-Pelletier J:
Strontium ranelate reduces the progression of experimental dog
osteoarthritis by inhibiting the expression of key proteases in
cartilage and of IL-1β in the synovium. Ann Rheum Dis. 72:250–257.
2013.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Lefebvre V, Behringer RR and de
Crombrugghe B: L-Sox5, Sox6 and Sox9 control essential steps of the
chondrocyte differentiation pathway. Osteoarthritis Cartilage. 9
(Suppl A):S69–S75. 2001.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Oh CD, Lu Y, Liang S, Mori-Akiyama Y, Chen
D, de Crombrugghe B and Yasuda H: SOX9 regulates multiple genes in
chondrocytes, including genes encoding ECM proteins, ECM
modification enzymes, receptors and transporters. PLoS One.
9(e107577)2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Kim HJ and Im GI: Electroporation-mediated
transfer of SOX trio genes (SOX-5, SOX-6, and SOX-9) to enhance the
chondrogenesis of mesenchymal stem cells. Stem Cells Dev.
20:2103–2114. 2011.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Rodrigues TA, Freire AO, Bonfim BF,
Cartágenes MS and Garcia JB: Strontium ranelate as a possible
disease-modifying osteoarthritis drug: A systematic review. Braz J
Med Biol Res. 51(e7440)2018.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Sassi N, Laadhar L, Allouche M, Achek A,
Kallel-Sellami M, Makni S and Sellami S: WNT signaling and
chondrocytes: From cell fate determination to osteoarthritis
physiopathology. J Recept Signal Transduct Res. 34:73–80.
2014.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Xia H, Cao D and Yang F, Yang W, Li W, Liu
P, Wang S and Yang F: Jiawei Yanghe decoction ameliorates cartilage
degradation in vitro and vivo via Wnt/β-catenin signaling pathway.
Biomed Pharmacother. 122(109708)2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Ma B, van Blitterswijk CA and Karperien M:
A Wnt/β-catenin negative feedback loop inhibits
interleukin-1-induced matrix metalloproteinase expression in human
articular chondrocytes. Arthritis Rheum. 64:2589–2600.
2012.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Yoshida Y, Yamasaki S, Oi K, Kuranobu T,
Nojima T, Miyaki S, Ida H and Sugiyama E: IL-1β enhances Wnt signal
by inhibiting DKK1. Inflammation. 41:1945–1954. 2018.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Yun K and Im SH: Transcriptional
regulation of MMP13 by Lef1 in chondrocytes. Biochem Biophys Res
Commun. 364:1009–1014. 2007.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Zhu M, Tang D, Wu Q, Hao S, Chen M, Xie C,
Rosier RN, O'Keefe RJ, Zuscik M and Chen D: Activation of
beta-catenin signaling in articular chondrocytes leads to
osteoarthritis-like phenotype in adult beta-catenin conditional
activation mice. J Bone Miner Res. 24:12–21. 2009.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Akiyama H, Lyons JP, Mori-Akiyama Y, Yang
X, Zhang R, Zhang Z, Deng JM, Taketo MM, Nakamura T, Behringer RR,
et al: Interactions between Sox9 and beta-catenin control
chondrocyte differentiation. Genes Dev. 18:1072–1087.
2004.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Han W, Fan S, Bai X and Ding C: Strontium
ranelate, a promising disease modifying osteoarthritis drug. Expert
Opin Investig Drugs. 26:375–380. 2017.PubMed/NCBI View Article : Google Scholar
|