Introduction
With the changes in diet, lifestyle diversification
and the aging of the population, the incidence of acute cerebral
infarction and the associated mortality rate are increasing,
placing heavy burdens on patients and their families (1). Acute cerebral infarction is the most
common cause of stroke and mainly occurs due to narrowing and even
occlusion of blood vessels in the brain. Once the supply of blood
flow suddenly stops, ischemia and hypoxia occur in the brain,
resulting in brain tissue softening and edema, which promotes
increased intracranial pressure and aggravates clinical symptoms,
including impaired neurological function (2,3). Once
the condition arises, effective treatment methods are limited. The
most reliable method for treating acute cerebral infarction is
intravenous thrombolysis in the early stage, which may reduce the
narrow area of the occluded blood vessels, increase the blood
supply in the infarct area, shorten the cerebral ischemia time and
reduce brain tissue damage. Recombinant human tissue plasminogen
activator (rt-PA) has been widely used for intravenous thrombolysis
within 4.5 h after the onset of acute cerebral infarction (4,5).
However, the time window for thrombolytic therapy is short and the
therapeutic effect has not been fully evaluated (6).
In recent years, only a small number of patients
with acute cerebral infarction have been able to receive
thrombolytic therapy in the optimal time window for thrombolysis.
Most patients miss the optimal thrombolysis time, resulting in poor
thrombolytic therapy. Therefore, application of brain protection
approaches during treatment is of great significance for treating
acute cerebral infarction. Associated studies have indicated that
physical methods enable patients to be in a mild hypothermia state,
reduce brain edema and intracranial pressure, slow down brain
tissue metabolism and avoid ischemia-reperfusion injury after
intravenous thrombolysis (7-9),
thus promoting neurological function recovery and improving
prognosis (10). Acute cerebral
infarction is a process of lipid accumulation and chronic
inflammation characterized by the reaction of cytokines and
inflammatory factors and consequently, assessment of inflammatory
factors in the acute cerebral infarction has attracted increasing
interest in recent decades. However, whether the inflammatory
factors interleukin (IL)-1, IL-6, intercellular adhesion molecule
(ICAM)-1 and matrix metalloproteinase (MMP)-2 have a role in
patients with acute cerebral infarction undergoing intravenous
thrombolysis has remained elusive. The present study aimed to
investigate the role of IL-1β, IL-6, C-reactive protein (CRP),
ICAM-1 and MMP-2 in patients with acute cerebral infarction by
measuring their levels prior to and after treatment. The clinical
application value of intravenous thrombolysis combined with mild
hypothermia in patients with acute cerebral infarction was also
explored.
Materials and methods
Research objects and grouping
With the approval of the Medical Ethics Committee of
Wuxi Third People's Hospital (Wuxi, Jiangsu, China), 80 patients
with acute cerebral infarction diagnosed at Wuxi Third People's
Hospital (Wuxi, Jiangsu, China) between January 2016 and December
2017 and were selected as the subjects. The inclusion criteria were
as follows: i) According to the 'China Acute Ischemic Stroke
Diagnosis and Treatment Guide 2014(11), the patients were diagnosed with
acute cerebral infarction; ii) the onset time was within 6 h; iii)
any neurological symptoms were consistent with the diagnosis of
acute cerebral infarction; iv) blood pressure of ≤180/100 mmHg; v)
CT and MRI imaging indications for acute cerebral infarction. The
following exclusion criteria were applied: i) Venous thrombosis in
the limbs; ii) cerebral infarction due to non-angiopathy; iii)
heart, liver or kidney failure; iv) history of cerebral infarction
or myocardial infarction; v) pregnant or lactating females; vi)
subjects participating in other associated medical experimental
research or who are unable/unwilling to cooperate with the
completion of treatment. The cohort included 37 females and 43
males, aged 42-80 years (average age, 44.5±15.7 years). The 80
patients were randomly and equally divided into the conventional
treatment group (group A) and the hypothermia treatment group
(group B). All enrolled patients and their families provided
written informed consent to participate in the present study.
Treatments
The enrolled patients maintained blood glucose and
blood pressure within the normal range after hospitalization with
improved circulation. The two groups were treated with rt-PA
intravenous thrombolysis and intravenously given rt-PA 0.9 mg/kg
(maximum dose, 90 mg). The rt-PA used was all of the same batch,
purchased from Boehringer Ingelheim International. Of the total
amount, 10% was intravenously injected within 1 min and the
remaining amount was intravenously instilled for 1 h. The patients'
condition was closely monitored within 24 h after treatment. The
major monitoring measures during intravenous thrombolytic therapy
included the following: i) The patient was admitted to the
intensive care unit for 24-h dynamic ECG monitoring. ii) After the
first thrombolysis, the blood pressure was monitored every 15 min
for the first 2 h, followed by every 30 min for the next 6 h, and
then once per h until a total of 24 h had passed. If the patient
had a diastolic blood pressure >100 mmHg and/or systolic blood
pressure of 180 mmHg, anti-hypertensive drugs were administrated
and blood pressure was closely monitored. iii) According to the
patient's requirement for neurological assessment, this was
performed 1 time/h. iv) If the blood pressure was severely high
with hiccup or headache, thrombolytic therapy was immediately
stopped and head CT examination was performed. In group B,
intravenous thrombolytic therapy was performed simultaneously with
mild hypothermia. The patient was made to wear a cooling cap, the
head was wrapped and the desuperheater (Kadant Inc.) was started.
The temperature of the cooling cap was maintained at 6-12˚C, the
patient's eardrum temperature was maintained at 33-35˚C and the
treatment time was 6 days. During the hypothermia phase, the
patient's vital signs and electrolytes were closely monitored.
After the treatment with mild hypothermia, the warming cap was
re-warmed on time, i.e., the patient's body temperature was
increased by 1˚C/5 h and the patient's eardrum temperature was
restored to 36.5-37.5˚C after ~20 h. The desuperheater, cooling cap
and infrared thermometer used by the patient were the same.
In the two groups, 5 ml fasting venous blood was
collected prior to treatment and at 7 days after treatment in an
anti-coagulation tube that was placed in a low-temperature
ultracentrifuge and centrifuged at 2,000 x g for 10 min at 4˚C to
collect the supernatant, which was stored at -80˚C for further
analysis.
Observation indicators
Serum IL-1β, IL-6, CRP, ICAM-1 and MMP-2 levels were
determined in the two groups using an ELISA kit. The kits used were
all in the same production batch and purchased from Biyuntian
Biotechnology Co., Ltd. All testing procedures were determined in
strict accordance with the instrument and reagent instructions to
ensure the reliability of the test data.
Evaluation of efficacy
At 7 days after treatment, the clinical treatment
effect was evaluated according to the diagnostic criteria for
cerebrovascular disease and the National Institutes of Health
Stroke Scale (NIHSS) score of cerebral infarction was determined
according to the functional score classification criteria as
determined at the 4th Cerebrovascular Conference (10). The treatment efficacy was rated as
follows: i) Not improved: NIHSS score <17%; ii) improved: NIHSS
score of 18-45%; iii) significantly improved: NIHSS score of
46-90%; iv) almost cured: NIHSS score of 91-100%. The total
efficiency was calculated as follows: Total efficiency=(almost
cured + significantly improved + improved)/total number x100%.
Safety assessment
Pulmonary inflammation, extracranial hemorrhage,
intracranial hemorrhage and mortality were recorded during
treatment and 7 days after treatment. Pulmonary inflammation was
assessed by 18F-fludeoxyglucose positron emission
topography. Intracranial hemorrhage was assessed according to the
score and diagnostic criteria of acute stroke (12). Intracranial hemorrhage may be
symptomatic or asymptomatic. Symptomatic intracranial hemorrhage
refers to an increase in NIHSS score of >4 points or death in
patients with acute cerebral infarction within 36 h of thrombolytic
therapy (13).
Statistical analysis
Data were processed using SPSS 19.0 statistical
software (IBM Corp.). Values are expressed as the mean ± standard
deviation. The unpaired t-test was used for comparison within the
group and an independent-samples t-test was used for comparison
between groups. Count data were presented as a ratio or composition
ratio and a χ2 test was used for comparison between
groups. Pearson correlation analysis was performed to assess
correlations. P<0.05 was considered to indicate a statistically
significant difference.
Results
General information
The basic information of the patients is provided in
Table I. Parameters including age
and body weight were not significantly different between the two
groups (P>0.05).
 | Table IGeneral information of the two groups
of patients (n=40 per group). |
Table I
General information of the two groups
of patients (n=40 per group).
| Group | Sex
(male/female) | Age (years) | Body weight (kg) |
|---|
| A | 22/18 | 44.2±15.6 | 51.6±12.7 |
| B | 21/19 | 41.2±16.4 | 56.3±18.2 |
| P-value | 0.084 | 0.135 | 0.116 |
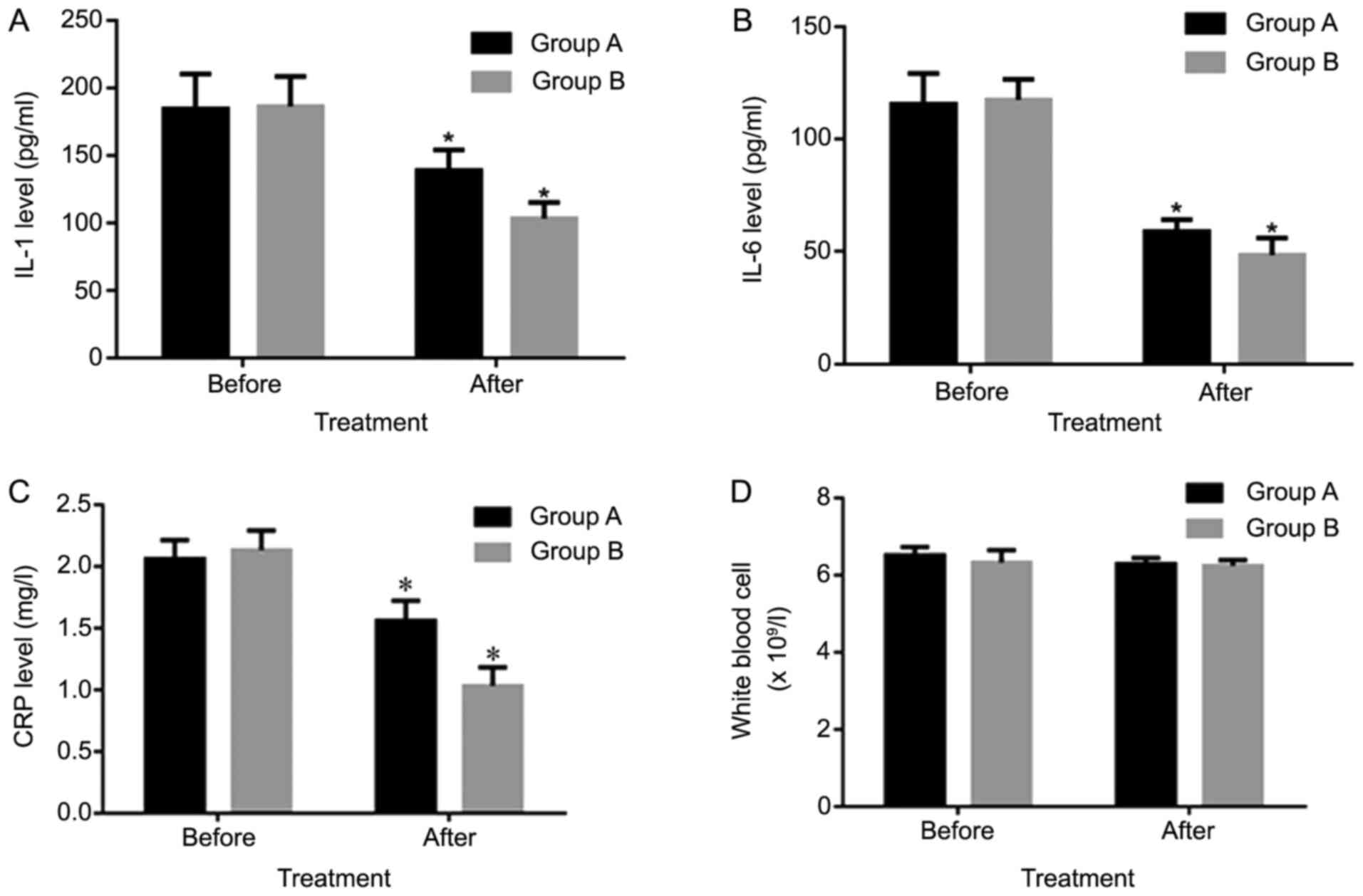
Serum levels of IL-1β, IL-6, CRP,
ICAM-1 and MMP-2 in the two groups of patients
Compared with those in group A, the levels of IL-1β,
IL-6 and CRP in group B were decreased at 7 days after treatment
and the differences were statistically significant (P<0.05;
Table II, Fig. 1A-C). However, there were no
differences in white blood cells prior to and after treatment in
the two groups (Fig. 1D).
 | Table IIChanges in serum IL-1β, IL-6, ICAM-1
and MMP-2 levels in the two groups (n=40 per group). |
Table II
Changes in serum IL-1β, IL-6, ICAM-1
and MMP-2 levels in the two groups (n=40 per group).
| | IL-1β (pg/ml) | IL-6 (pg/ml) | ICAM-1 (µg/ml) | MMP-2 (µg/l) |
|---|
| Group | Before | 7d | Before | 7d | Before | 7d | Before | 7d |
|---|
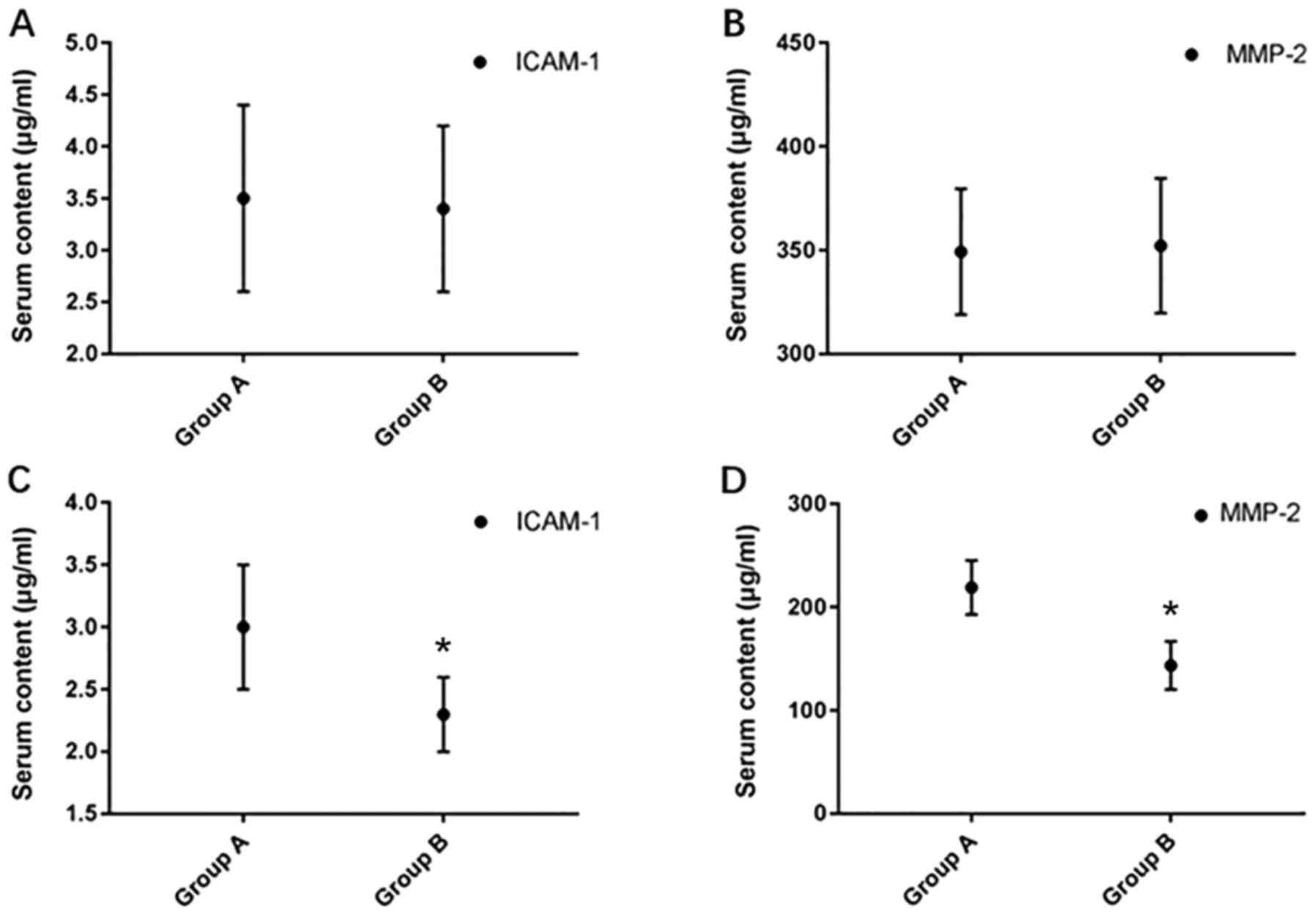
| A | 184.18±26.18 | 138.57±15.48 | 115.43±13.77 | 58.77±5.46 | 3.5±0.9 | 3.0±0.5 | 349.27±30.39 | 219.12±26.33 |
| B | 186.20±22.23 | 103.33±11.71 | 117.44±9.13 | 48.49±7.51 | 3.4±0.8 | 2.3±0.3 | 352.19±32.51 | 143.73±23.25 |
| P-value | 0.135 | 0.014 | 0.163 | 0.017 | 0.161 | 0.023 | 0.172 | 0.015 |
Compared with those in conventional treatment group
(group A) and the hypothermia treatment group (group B), the levels
of ICAM-1 and MMP-2 in group B were significantly decreased at 7
days after treatment (P<0.05), while there was no significant
difference between group A and B before treatment (P>0.05;
Table II, Fig. 2A-D).
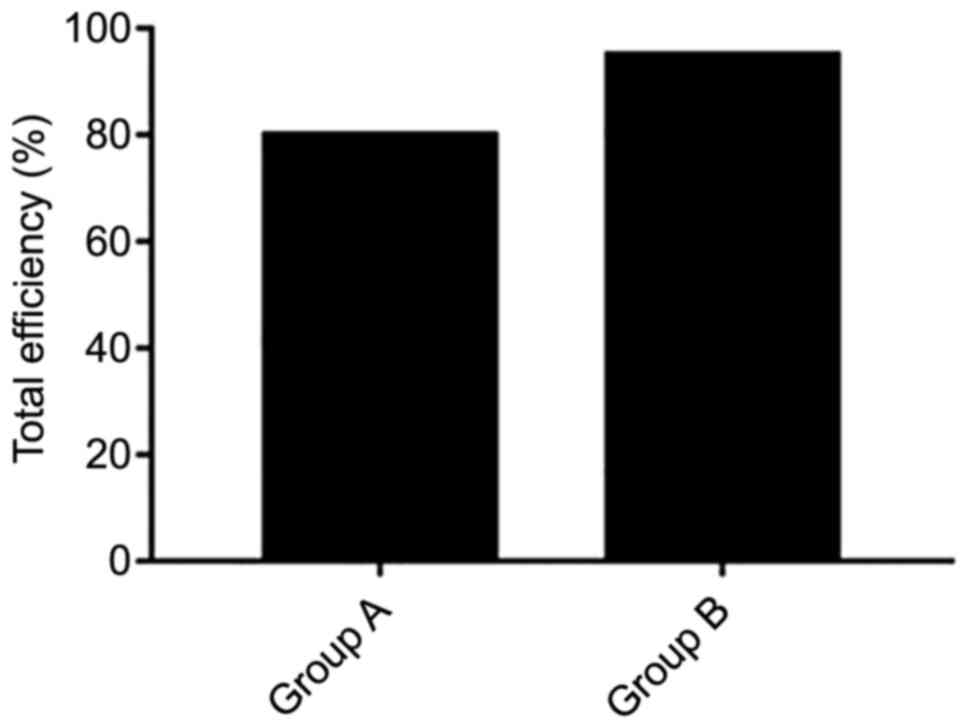
Evaluation of effectiveness
At 7 days after treatment, the total effective rate
was 80% in group A and 95% in group B. There was a significant
difference between the two groups (P<0.05; Fig. 3).
Safety evaluation
The incidence of adverse reactions in group A and
group B was 35 and 20%, respectively, and the mortality rates were
10 and 5%, respectively. There were no significant differences in
adverse events and mortality between the two groups (P>0.05,
Table III).
 | Table IIIComparison of incidence of adverse
reactions between the two groups (n=40 per group). |
Table III
Comparison of incidence of adverse
reactions between the two groups (n=40 per group).
| Group | Symptomatic
intracranial hemorrhage | Asymptomatic
intracranial hemorrhage | Extracranial
hemorrhage | Pulmonary
infection | Adverse reaction rate
(%) | Mortalities |
|---|
| A | 2 | 2 | 6 | 4 | 35 | 4(10) |
| B | 0 | 0 | 4 | 4 | 20 | 2(5) |
| P-value | | | | | 0.128 | 0.152 |
Correlation analysis of IL-1β, IL-6,
CRP, ICAM-1 and MMP-2 with neurological function
To evaluate whether there is an association of
IL-1β, IL-6, CRP, ICAM-1 or MMP-2 with neurological function, a
correlation analysis was performed in all patients prior to
treatment, revealing a positive correlation of the level of IL-1β,
IL-6, CRP, ICAM-1 and MMP-2 with the NIHSS score (Table IV).
 | Table IVCorrelation of inflammatory
factors/cytokines with the National Institutes of Health Stroke
Scale score. |
Table IV
Correlation of inflammatory
factors/cytokines with the National Institutes of Health Stroke
Scale score.
| Parameter | IL-1β | IL-6 | CRP | ICAM-1 | MMP-2 |
|---|
| r | 0.461 | 0.547 | 0.612 | 0.367 | 0.436 |
| P-value | 0.021 | 0.019 | 0.006 | 0.031 | 0.029 |
Discussion
Acute cerebral infarction is one of the most common
cerebrovascular diseases in neurology and is common among
middle-aged and elderly individuals. The annual incidence rate in
China is as high as 120/100,000-180/100,000, with continuously
increasing mortality rate (14).
Acute cerebral infarction is a process resulting from lipid
accumulation and chronic inflammation characterized by the reaction
of cytokines and inflammatory factors. One of the major causes of
cerebral ischemia-reperfusion injury is brain inflammation caused
by cerebral infarction (15,16).
The core of the treatment approach for acute cerebral infarction is
to restore the blood circulation as soon as possible, correct brain
ischemia and hypoxia, reduce inflammation and cerebral edema,
reduce intracranial pressure, and thus ameliorate the degree of
brain damage. However, due to intravenous thrombolytic therapy, the
incidence of intracranial hemorrhage may be as high as 10.9-32.0%
and the mortality rate after bleeding is high. The comorbidities
are unacceptably high, resulting in an unsatisfactory curative
effect and poor prognosis. The treatment of acute cerebral
infarction remains challenging and has become a hot topic in
clinical research. An appropriate therapeutic strategy should not
only resolve cerebral ischemia and hypoxia, but also protect brain
tissue function and improve the prognosis.
MMP-2 is one of the key members of the MMPs and acts
as an inflammatory molecule in atherosclerotic plaques of brain
tissues, which contributes to the damage of the blood-brain barrier
after brain tissue ischemia and increases the permeability of the
blood-brain barrier (17), causing
brain edema. Relevant studies suggested that IL-1β, IL-6 and MMP-2
may be used as indicators for the detection of the condition and
prognosis of patients with acute cerebral infarction, and may
accurately reflect the condition and treatment effect of patients
with acute cerebral infarction (18,19).
IL-1β is a cytokine that has multiple biological effects. Studies
have indicated that when brain tissue is subjected to ischemia, the
IL-1β content increases rapidly, promotes brain cell apoptosis,
accelerates free radical release, induces endothelial cells to
secrete pro=coagulant factors, inhibits anti-coagulant activity and
reduces cerebral blood flow (20,21).
IL-6 is an immunological and inflammatory cytokine that is used as
a major risk indicator for detecting ischemic brain injury
(22) and is associated with an
inflammatory response to acute cerebral infarction. ICAM-1 is one
of the largest adhesion molecules that mediate adhesion reactions
and belongs to the immunoglobulin superfamily, which enhances the
adhesion of the inflammatory factors.
In recent years, sub-hypothermia in the range of
33-35˚C has been reported to reduce the oxygen consumption of brain
tissue, slow down the metabolism of brain cells, increase the blood
supply to the brain, relieve the degree of cerebral edema and
reduce intracranial pressure (23,24).
It has also been suggested that 33±1˚C is the most significant
temperature range for maintaining brain tissue function (25). In mice subjected to experimental
cerebral ischemia with brain inflammation and mild hypothermia,
Deng et al (26) determined
that the content of ICAM-1 was significantly lower than that in the
control group, which indicated a certain inhibitory effect of mild
hypothermia on brain inflammation. At the same time, associated
animal experiments indicated that local hypothermia treatment may
reduce the area of cerebral infarction (27,28).
With the continuous improvement of scientific research, an
increasing number of studies have confirmed through experimental
and clinical research that hypothermia treatment has a protective
effect on neurological function, with good prognosis in the later
stage and fewer side effects (29-31).
Therefore, in the present clinical study, a treatment temperature
of 33-35˚C was adopted, which was compared with conventional
treatment to determine the efficacy of intravenous thrombolysis
combined with mild hypothermia treatment for acute cerebral
infarction.
The results of the present study suggested that,
compared with those in group A, the levels of IL-1β, IL-6, CRP and
ICAM-1 in group B were significantly decreased At 7 days after
treatment (P<0.05). Compared with those prior to treatment, the
levels of MMP-2 were significantly decreased At 7 days after
treatment, and the levels of MMP-2 in group B was significantly
lower than those in group A (P<0.05). At 7 days after treatment,
the total effective rate was 80% in group A and 95% in group B and
there was a significant difference between the two groups
(P<0.05). The incidence of adverse reactions in group A and
group B was 35 and 20%, respectively, and the mortality rates were
10 and 5%, respectively. However, there were no significant
differences in adverse events and mortalities between the two
groups (P>0.05). In addition, a positive correlation of the
level of IL-1β, IL-6, CRP, ICAM-1 and MMP-2 with the NIHSS score
was identified in the total cohort of patients prior to treatment.
The above results suggest that, compared with intravenous
thrombolytic therapy alone, intravenous thrombolysis combined with
mild hypothermia treatment has the most obvious effects on the
levels of IL-1β, IL-6, CRP, ICAM-1 and MMP-2, and may significantly
improve brain ischemia and hypoxia, reduce inflammation and
ameliorate brain tissue damage.
In conclusion, intravenous thrombolysis combined
with mild hypothermia treatment may significantly improve the
clinical symptoms of patients with acute cerebral infarction and
control the inflammatory response after cerebral infarction,
indicating that this treatment is effective and may be used in the
clinic. However, the exact mechanism of how the treatment affects
the cytokines or markers in patients with acute cerebral infarction
remains elusive. In the future, in vitro experiments are
required to investigate the mechanisms of how this treatment alters
the inflammatory cytokines.
Acknowledgements
Not applicable.
Funding
Funding: This study was supported by the Youth Medical Key
Talent Project of Wuxi (grant no. QNRC084) and Health and Family
Planning Commission of Wuxi (grant no. Q201610).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
CL and LHu contributed to conception and design and
performed the experiments. JZ, MD, CF, XZ and LHan contributed to
acquisition, analysis and interpretation of data. XZ, CL, JZ and
LHan drafted the manuscript and revised it critically for important
intellectual content and gave final approval of the version to be
published. All authors approved revisions and the final paper, and
agreed to be accountable for all aspects of the work in ensuring
that questions related to the accuracy or integrity of any part of
the work are appropriately investigated and resolved. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
All enrolled patients and their families provided
written informed consent to participate in the present study and
the study was approved by the Medical Ethics Committee of Wuxi
Third People's Hospital (Wuxi, Jiangsu, China).
Patient consent for publication
All enrolled patients provided consent for the
publication of this article.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Zhang M: Clinical research on traumatic
cerebral infarction treated with mild-hypothermia and acupuncture.
Zhongguo Zhen Jiu. 32:697–700. 2012.PubMed/NCBI(In Chinese).
|
|
2
|
Zhang X, Li Y, Liang Y, Sun P, Wu X, Song
J, Sun X, Hong M, Gao P and Deng D: Distinguishing Intracerebral
Hemorrhage from Acute Cerebral Infarction through Metabolomics. Rev
Invest Clin. 69:319–328. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Muramatsu K, Fujino Y, Kubo T, Otani M,
Fushimi K and Matsuda S: Efficacy of antimicrobial catheters for
prevention of catheter-associated urinary tract infection in acute
cerebral infarction. J Epidemiol. 28:54–58. 2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Juttler E, Kohrmann M and Schellinger PD:
Therapy for early reperfusion after stroke. Nat Clin Pract
Cardiovasc Med. 3:656–663. 2006.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Varga V, Waje-Andreassen U, Naess H, Naess
H, Lundstadsveen MT and Thomassen L: Thrombolytic therapy in
cerebral infarction. Tidsskr Nor Laegeforen. 129:2214–2117.
2009.PubMed/NCBI View Article : Google Scholar : (In Norwegian).
|
|
6
|
Peng X, Yue W, Liu W, Dan B, Lin L and
Tang Z: Protective roles of intra-arterial mild hypothermia and
arterial thrombolysis in acute cerebral infarction. Springerplus.
5(1988)2016.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Cechmanek BK, Tuor UI, Rushforth D and
Barber PA: Very Mild hypothermia (35°C) postischemia reduces
infarct volume and blood/brain barrier breakdown following tPA
Treatment in the Mouse. Ther Hypothermia Temp Manag. 5:203–208.
2015.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Moon Y, Lee JH, Cho HJ, Roh HG, Choi JW,
Chun YI and Kim HY: Intravenous thrombolysis in a patient with
acute ischemic stroke attributable to intracranial dissection.
Neurologist. 18:136–138. 2012.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Miyauchi T, Wei EP and Povlishock JT:
Evidence for the therapeutic efficacy of either mild hypothermia or
oxygen radical scavengers after repetitive mild traumatic brain
injury. J Neurotrauma. 31:773–781. 2014.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Guan Y, Zhao GD, Jiang L, et al:
Meta-analysis of mild hypothermia in the treatment of acute
ischemic stroke. J Practical Med. 29:1518–1521. 2013.
|
|
11
|
Chinese Medical Association Neurology
Branch. Chinese Medical Association Neurology Branch
Cerebrovascular Disease Group: Chinese guidelines for the diagnosis
and treatment of acute ischemic stroke, 2014. Chin J Neurol.
48:246–257. 2015.
|
|
12
|
Hacke W, Kaste M, Fieschi C, Toni D,
Lesaffre E, von Kummer R, Boysen G, Bluhmki E, Höxter G, Mahagne
MH, et al: Intravenous thrombolysis with recombinant tissue
plasminogen activator for acute hemispheric stroke. The European
Cooperative Acute Stroke Study (ECASS). JAMA. 274:1017–1025.
1995.PubMed/NCBI
|
|
13
|
Kruyt ND and Roos YWBM: Hyperglycemia in
Acute Stroke: Humana Press, 2010.
|
|
14
|
Wu Y and Zhang X: Cost-effectiveness
comparison of four drug regimens in the treatment of acute cerebral
infarction. Chin J Biochem Pharmaceutics. 31:57–58. 2010.(In
Chinese).
|
|
15
|
Wang X, Shen B, Sun D and Cui X: Aspirin
ameliorates cerebral infarction through regulation of
TLR4/NF-κB-mediated endoplasmic reticulum stress in mouse model.
Mol Med Rep. 17:479–487. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Nakatani Y, Suto Y, Fukuma K, Yamawaki M,
Sakata R, Takahashi S, Nakayasu H and Nakashima K: Intrathecal
isoniazid for refractory tuberculous meningitis with cerebral
infarction. Int Med. 56:953–957. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Fang ZQ: Experimental Methodology of
Dialectical Treatment of Rats/Mice, Science Press, 2009.
|
|
18
|
Jiang Y and Lian YJ: Effects of Danhong
injection on hemodynamics and the inflammation-related NF-κB
signaling pathway in patients with acute cerebral infarction. Genet
Mol Res. 14:16929–16937. 2015.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Yang J and Yang Z: Changes of serum TNF-α
sICAM-1 in patients with cerebral infarction and its clinical
significance. J Brain Nervous Dis. 10:39–41. 2002.(In Chinese).
|
|
20
|
Kochanek PM and Hallenbeck JM:
Polymorphonuclear leukocytes and monocytes/macrophages in the
pathogenesis of cerebral ischemia and stroke. 23:1367–1379.
1992.PubMed/NCBI View Article : Google Scholar
|
|
21
|
del Zoppo GJ, Schmidschönbein GW, Mori E,
Copeland BR and Chang CM: Polymorphonuclear leukocytes occlude
capillaries following middle cerebral artery occlusion and
reperfusion in baboons. Stroke. 22:1276–1283. 1991.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Yang Y, Wu J and Sui Z: Tumor necrosis
factor and cerebral ischemia. Zhejiang J Integrated Trad Chin
Western Med. 19:60–61. 2009.(In Chinese).
|
|
23
|
Hu Xiao, Qi Hao, Li Shirong, et al: The
efficacy and possible mechanism of intravenous thrombolysis
combined with mild hypothermia in patients with acute cerebral
infarction. Chin J Neuropsychiatric Dis. 42:15–21. 2016.(In
Chinese).
|
|
24
|
Gao HN, Lu HZ, Cao B, Du B, Shang H, Gan
JH, Lu SH, Yang YD, Fang Q, Shen YZ, et al: Clinical findings in
111 cases of influenza A (H7N9) virus infection. N Engl J Med.
368:2277–2285. 2013.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Husain M: Avian influenza A (H7N9) virus
infection in humans: epidemiology, evolution, and pathogenesis.
Infect Genet Evol. 28:304–312. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Deng H, Han HS, Cheng D, Sun GH and Yenari
MA: Mild hypothermia inhibits inflammation after experimental
stroke and brain inflammation. Stroke. 34:2495–2501.
2003.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Chang YP, Liao PT, Shen EY and Liu IH:
Protective effect against focal cerebral ischemia injury in acute
phase of a novel invasive device for regional hypothermia. J Chin
Med Assoc. 78:67–75. 2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Liang J: Effects of body temperature
control on serum S-100B protein, LDH expression and prognosis in
patients with severe head injury. Chin Laboratory Diagnosis.
20(755)2016.(In Chinese).
|
|
29
|
Hu W, Dai Y, Chen S, et al: Clinical study
of brain hypothermia on brain protection in patients with cerebral
infarction. Chongqing Med J. 45:4817–4819. 2016.(In Chinese).
|
|
30
|
Andresen M, Gazmuri JT, Marín A, Regueira
T and Rovegno M: Therapeutic hypothermia for acute brain injuries.
Scand J Trauma Resusc Emerg Med. 23(42)2015.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Guo XY, Li AQ, Zhan J, et al: Effects of
local mild hypothermia on serum NSE in patients with severe
cerebral infarction. Stroke and Neurological Diseases. 19:214–216.
2012.
|