Introduction
Eyelid cancer represents a specific type of tumor
that involves the skin of the eyelid or glands present at that
level. The eyelid skin is considered the thinnest skin of the human
body and may be easily impaired.
Skin cancer has the highest prevalence among cancers
worldwide (1); head and neck
cancers represent the sixth most common cancer and is defined by
tumors growing in the nose or sinuses, mouth, throat, or larynx, or
around the eyes, in the outer layer of the mucous membranes or the
skin (2). Tumors located at the
eyelid level account for 5 to 10% of all possible skin cancers,
reflecting a rather common site (3).
There are four types of eyelid carcinomas: basal
cell carcinoma (BCC), squamous cell carcinoma (SCC), sebaceous cell
carcinoma, all three considered as non-melanoma skin cancer (NMSC),
and melanoma. Almost 90% of all eyelid tumors are BCC, which is
considered a type of carcinoma with a slow progression rate which
rarely spreads in the surrounding areas. The other types are
considered more aggressive, as they form, grow, or spread to other
sites of the body more quickly.
The main carcinogenic factors for eyelid cancer are
represented by UV exposure mostly during childhood and adolescence,
fair skin, increased age, immunosuppression, and smoking (3,4).
Overall, tobacco is considered the main carcinogenic factor for 16%
of all cancers in developed countries; in less developed regions,
it accounts for around 10%. There is a significant difference
between sexes: for men, smoking is considered responsible for
around 25% of all possible cancers, while for women, it only
accounts for 4% (5,6).
Recent studies performed during the past 5-6 years
have demonstrated that smoking may amplify the aggressiveness of
tumors, especially head and neck carcinomas, favoring a more rapid
progression to higher stages. Thus, tobacco is considered not only
a carcinogenic factor, but it is also associated with an increased
risk of developing aggressive forms of carcinomas. In fact, it was
demonstrated that cigarette smoke can alter cell structures in
indirect and direct ways (at the protein and DNA level) (7,8)
promoting tumoral cell proliferation.
The aim of the present study was to determine
whether smokers present more aggressive forms of eyelid carcinomas,
based on their current stage of tumors identified at first
diagnosis, and the duration of their symptoms.
Patients and methods
We conducted a research study between 2016 and 2019,
on a group of 98 patients admitted to the Ophthalmology Clinic from
the Emergency County Hospital Craiova, Romania.
The inclusion criterion was a diagnosis of eyelid
neoplasm, no matter its form. All patients provided their informed
consent regarding treatment and personal data analysis.
For each patient, we acquired the following data:
sex, age at diagnosis, area of residency, tumor cell type, tumor
stage and extension, status of relapse, treatment, smoking habit,
data regarding the duration of symptoms at the moment of the
initial consultation, as well as several clinical parameters (skin
type, diabetes mellitus, arterial hypertension, cutaneous
infections, actinic keratosis).
Given the available data, a patient was considered
as a smoker if he/she was an active smoker, or he/she was a former
smokers for a significant period of time (at least 1 year);
otherwise, the patient was considered a non-smoker. Based on this
status, we divided our study lot into two groups.
We determined the histopathological type of tumor as
recommended by the World Health Organization (WHO) Classification
of skin tumors (9). Tumor stage was
assessed based on the TNM system (tumor, node, and metastasis)
according to AJCC Cancer Staging 8th edition, which is a
general classification of tumors according to their size and
extent, relative to the original location (10). Following these criteria, tumors were
divided in four ordered stages, named from T1 to T4.
Aggressiveness is a cancer characteristic, and it
expresses the rapidity to which the tumor evolves from lower stages
to higher stages. We assessed the aggressiveness level based on the
tumor stage and the duration of symptoms at diagnosis.
Statistical analysis
We used Statistical Package for Social Sciences
(SPSS), version 20 (IBM Corp.) to regroup patient data, to convert
inputs into categorical parameters, and to perform a statistical
analysis upon the acquired values. Chi-square and Fisher's exact
tests were used to evaluate the studied group distributions and to
compare different results. For each predefined interval of symptom
duration, we used ordinal or binominal logistic regression to
analyze the relation between tumor aggressiveness and smoking
status, adjusting the result with sex and the age at first
diagnosis.
We also determined the relative risk (RR) and 95%
confidence intervals (95% CI) for smokers compared with
non-smokers, in relation with several tumor-related parameters
(P-value <0.05 was considered statistically significant). For
risk calculation, tumor stage was dichotomized in mild (T1-T2) and
severe (T3-T4) and non-smokers represented the reference group.
Results
The study lot had an almost equal distribution of
sexes, with 51 males (52.04%) and 47 females (47.96%). More than
half of the male patients were active smokers (28 patients,
representing 54.90% of males), while only 17 females (representing
36.17% of females) smoked constantly (Table I).
 | Table IDistribution of patients according to
the duration of symptoms, tumor stage and type, sex, and smoking
habit. |
Table I
Distribution of patients according to
the duration of symptoms, tumor stage and type, sex, and smoking
habit.
| Parameter | Total n (%) | Smoker n (%) | Non-smoker n
(%) |
P-valuea
(M, F) |
|---|
| Sex | | | | |
|
Male
(M) | 51 (52.04) | 28 (28.57) | 23 (23.47) | 0.06 |
|
Female
(F) | 47 (47.96) | 17 (17.35) | 30 (30.61) | |
| Residency | | | | 0.49 (M, 0.88; F,
0.59) |
|
Urban | 34 (34.69) | 14 (14.29) | 20 (20.41) | |
|
Rural | 64 (65.31) | 31 (31.63) | 33 (33.67) | |
| Tumor stage | | | | |
|
T1 | 37 (37.76) | 17 (17.35) | 20 (20.41) | 0.99 (M, 0.77; F,
0.85) |
|
T2 | 31 (31.36) | 14 (14.29) | 17 (17.35) | |
|
T3 | 22 (22.45) | 10 (10.20) | 12 (12.24) | |
|
T4 | 8 (8.16) | 4 (4.08) | 4 (4.08) | |
| Type | | | | 0.75 (M, 0.76; F,
0.63) |
|
BCC | 86 (87.76) | 40 (40.82) | 46 (46.94) | |
|
SCC | 12 (12.24) | 5 (5.10) | 7 (7.14) | |
| Duration of
symptoms | | | | |
|
<1
year | 42 (42.86) | 22 (22.45) | 20 (20.41) | 0.11 (M, 0.77; F,
0.03) |
|
1-5
years | 37 (37.76) | 12 (12.24) | 25 (25.51) | |
|
5-10
years | 19 (19.39) | 11 (11.22) | 8 (8.16) | |
Patients included in our study lot had age at
diagnosis of a range between 39 and 91 years, mean value and
standard deviation 67.4±12.53, thus covering a significant age
interval. To ease the subsequent analysis, the lot was divided into
age decades, starting from 30-39 years. We obtained thus 6 decades,
up to 90-99 years of age. To be consistent with the patients' real
age, we named the decades from 3 to 9, instead of 1 to 6. Our study
lot was composed mainly of patients with middle to high ages.
Decades 5 and 6 each had almost a quarter of the entire group,
decade 7 covered approximately 20%, while decades 3, 4, 8 and 9
represented the rest.
Table II contains
the distribution of active smokers among the study lot, divided by
age decade. Thus, decade 8 was the most affected, with 62.5% active
smokers and 37.5% non-smokers. Decades 4 and 7 were equally divided
(50%), while the other decades were dominated by non-smokers.
Greater differences in terms of smoking habit distribution were
present for decades 5 and 8.
 | Table IIDistribution of patients according to
age decade and smoking habit. |
Table II
Distribution of patients according to
age decade and smoking habit.
| Age decade | 30-39 n (%) | 40-49 n (%) | 50-59 n (%) | 60-69 n (%) | 70-79 n (%) | 80-89 n (%) | 90-99 n (%) |
|---|
| Smoking habit | | | | | | | |
|
Smoker | 0 (0) | 3(50) | 9 (34.61) | 11 (45.83) | 10(50) | 10 (62.50) | 2(40) |
|
Non-smoker | 1(100) | 3(50) | 17 (65.39) | 13 (54.17) | 10(50) | 6 (37.50) | 3(60) |
Concerning the area of residence, most patients were
from a rural environment (64 patients, representing 65.31%), 28
females and 36 males, and almost half of all rural residents
(48.44%, mostly males) were smokers (31.63% of the entire lot). The
rest of the 34 patients had an urban residence (34.69%), 19 females
and 15 males, and only 41.18% of the urban residents were active
smokers (14.29% of the entire lot) (Table I).
Two types of neoplasms were identified within our
study lot: 87.76% (86 patients, 43 males and 43 females) had basal
cell carcinoma (BCC), while 12.24% (12 patients, 8 males and 4
females) had squamous cell carcinoma (SCC). Less than half of the
patients with BCC were active smokers (46.51%, 40/86 patients, 60%
of them being males) (40.82% of the entire lot). Among the patients
with SCC, 41.67% were smokers (5/12 patients, only 1 female) (only
5.10% of the total lot). All 4 tumor stages (from T1 to T4) were
identified among the patients (Table
I).
We did not identified correlations between smoking
status and sex, area of residence, tumor stage or neoplasm type
(P>0.05) (Table I).
All patients had unilateral tumors, extended on the
orbit for 8 patients (8.16% from the entire study lot). All 8
patients with orbit extensions had T4 stage tumors (87.5% of them
are from rural areas), and half of them are smokers. From the
entire study lot, only 3 patients had relapse (3.06%) with stages
T1 and T2, all middle-aged males. All 3 were from rural areas, and
two of them (66.67%) were active smokers.
Analysis of symptom duration
During the initial consultation, patients were
requested to declare for how long they presented clinical
manifestations of their eyelid neoplasm; thus, we obtained the
duration of the symptoms. Patients were divided in 3 categories:
symptoms present for less than 1 year, symptoms present for a
period between 1 and 5 years, and symptoms present for a period
between 5 and 10 years (Table
I).
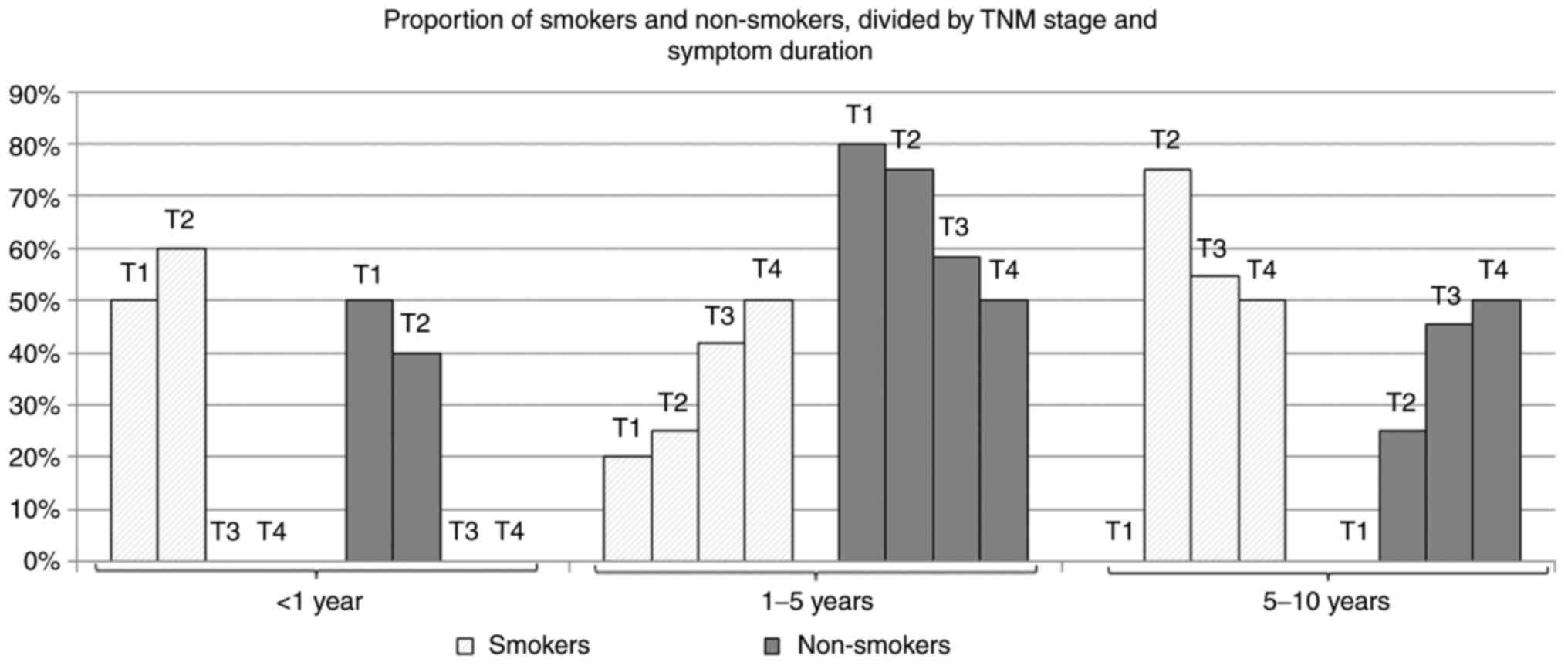
Fig. 1 summarizes
the evolution of smoker and non-smoker distribution among the study
lot, indicating the increased tendency of the smoker proportion by
stage, for patients with symptoms present for less than 5
years.
From the entire study lot, 42 patients (representing
42.86%) came to the doctor after a few months of symptoms (less
than 1 year): 17 females and 25 males (smokers represent 52.38%)
(22.45% of the total lot). Mean age was 66.09±13.19 years. Tumor
distribution was the following: 32 patients (76.19%) (32.65% of the
entire lot) had T1 tumors and 50% were smokers; 10 patients (10.20%
of the entire lot) had T2 tumors, 60% of them were smokers.
The second duration category was 1-5 years and
included 37 patients from the entire study lot (37.76%, 19 females
and 18 males). Mean age was 66.13±11.71 years. Only 32.43% of
patients in this category were smokers (12.24% of the total lot).
Tumor stage distribution was the following: 5 patients had T1
tumors and 20% were smokers; 16 patients had T2 tumors and 25% were
smokers; 12 patients had T3 tumors and 41.67% were smokers; 4
patients had T4 tumors and 50% were smokers.
Within our study lot, we also had 19 patients
(19.39%, 11 females and 8 males) who sought medical treatment after
more than 5 years from their first clinical manifestations. Mean
age was 72.73±11.71 years. More than half of them (11/19 patients,
57.89%) were smokers (11.22% of the total study lot).
Tumor stage distribution was the following: 4
patients had T2 tumors and 75% were smokers; 11 patients had T3
tumors and 54.5% were smokers; 4 patients had T4 tumors and 50%
were smokers.
We identified a significant correlation between
smoking and symptom duration only for females (P<0.05).
For the first two categories, the smoker proportion
increased with the stage tumor; thus, the higher the stage, the
higher percentage of smokers. Thus, for patients with symptoms
present for less than 5 years, smokers had tumors of higher stages,
more aggressive, compared to non-smokers. For category 5-10 years,
the percentage of smokers decreased with the tumor stage. Smokers
with symptoms present for more than 5 years apparently have less
aggressive tumors, compared to non-smokers.
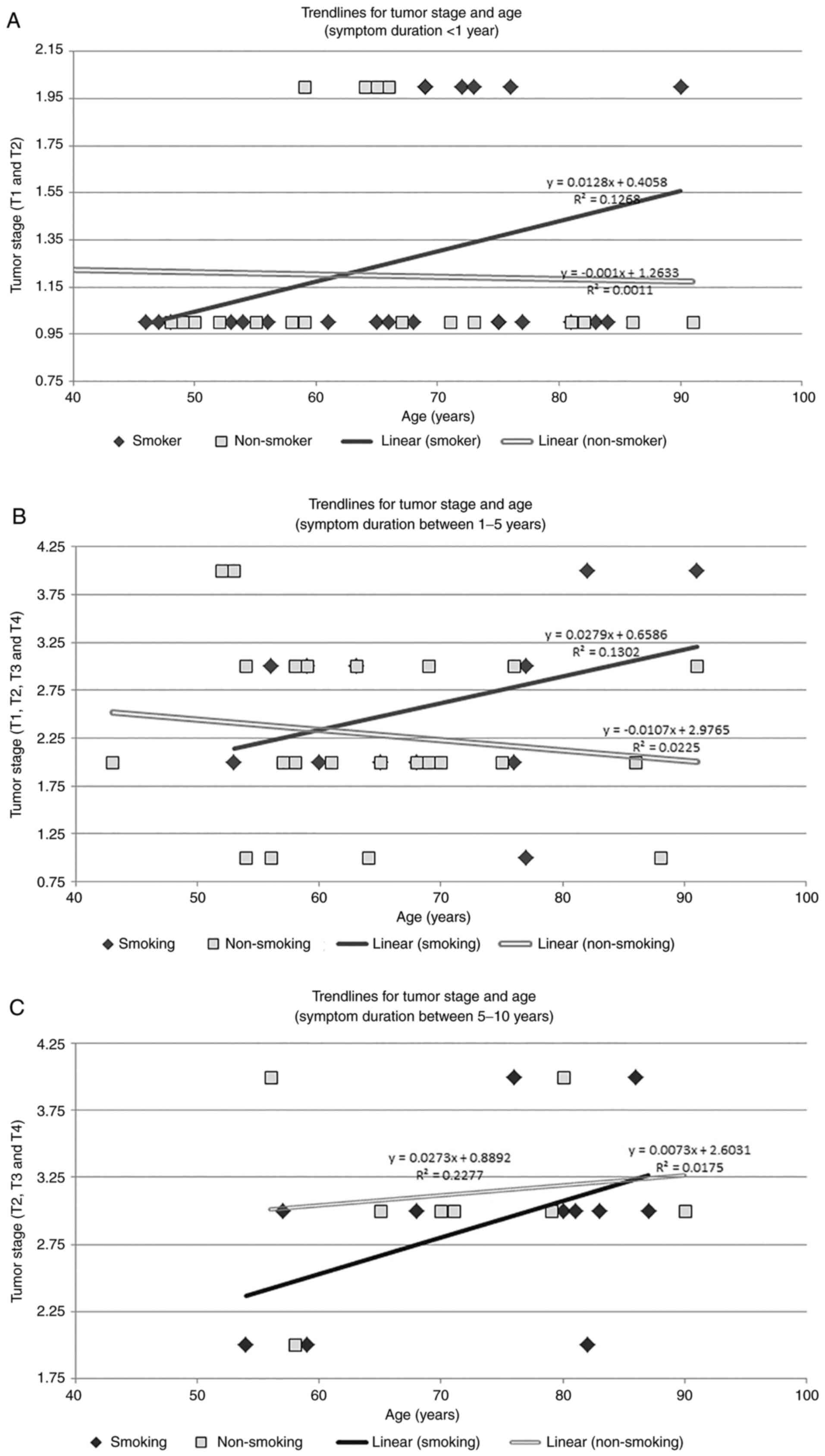
Age plays a role in tumor progression as well. For
smoker patients whose symptoms were present for less than 1 year,
tumor stage increased with age, compared with non-smokers, where
the trend line indicated a decrease in stage with age (Fig. 2A). Only T1 and T2 stages are present
in this category, thus we used a binomial logistic regression model
to determine the potential effect of smoking status, sex, and age,
upon the current tumor stage at first diagnosis. We initially used
the Box-Tidwell procedure to test the linearity of age (the only
continuous variable), and the results confirmed that it was
linearly related to the logit of the tumor stage (P=0.972, which
was greater than the standard value 0.05, and also value 0.0125,
computed using the Bonferroni correction). Our model was
statistically significant, with χ2(4)=27.402, P<0.0005. Only 2 of the 3
variables used as predictors were significant: age and smoking
status. Smokers presented 6.044 times higher odds to have more
advanced tumor stages, compared to non-smokers (95% CI,
1.240-54.141), χ2(1)=4.768, P=0.029). Similarly, older
patients had 1.141 times higher odds, compared to younger patients
(95% CI, 1.203-14.171), χ2(1)=4.167, P=0.041).
A similar evolution was identified for patients with
symptoms present between 1 and 5 years, where tumors were more
aggressive with age for smokers, compared to non-smokers (Fig. 2B). Given the fact that patients from
this category exhibited all 4 types of tumors, we used ordinal
logistic regression to identify the potential effect of smoking
status, sex, and age, upon the current tumor stage at first
diagnosis. We assessed the proportional odds through a full
likelihood ratio test (our fitted model was compared to a model
characterized by varying location parameters), obtaining
χ2(6)=7.221, P=0.301.
The final model was able to assess the tumor stage,
χ2(4)=12.891, P=0.005.
The computed odds of smokers having advanced tumor stages were
4.501 times greater than for non-smokers (95% CI, 1.076-18.820),
χ2(1)=4.246, P=0.039.
Sex had no statistically significant influence over more advanced
tumor stages, χ2(1)=1.410, P=0.235. We identified an
association between a higher age and an increase of odds for
patients presenting higher tumor stages, with a computed odds ratio
of 1.077 (95% CI, 1-1.119), χ2(1)=3.697, and a borderline value of
P=0.05.
For patients in the last category of symptom
duration, there was an increased tendency to higher stages with
age, for both smokers and non-smokers (Fig. 2C). With 3 tumor stages identified
for this category, from T2 to T4, we ran a similar ordinal logistic
regression model. Odds were proportional for our parameters:
χ2(3)=3.055, P=0.383.
The final model was able to assess the tumor stage,
χ2(3)=8.915, P=0.030.
Smoking status or sex had no significant effects over advanced
tumor stages, with χ2(1)=1.464, P=0.226, respectively
χ2(1)=0.154, P=0.695.
However, we also identified an association between a higher age and
an increase of odds for patients presenting higher tumor stages,
with an odds ratio of 1.150 (95% CI, 1.028-1.287),
χ2(1)=5.942,
P=0.015.
We also performed various analyses upon clinical and
demographical data, by sex and age decades, but we did not find any
specific tumor progression correlations (P>0.05). Table III contains the relative risk
analysis for smokers with non-smokers as reference, determined for
sex, residency, cell type, tumor stage and symptom duration.
 | Table IIIRisk estimation for smokers compared
to non-smokers. |
Table III
Risk estimation for smokers compared
to non-smokers.
| Parameter | Smokers RR (95%
CI) |
|---|
| Sex | |
|
Female | 0.667
(0.429-1.039) |
|
Male | 1.434
(0.978-2.102) |
| Residency | |
|
Urban | 0.824
(0.473-1.437) |
|
Rural | 1.106
(0.830-1.474) |
| Type | |
|
SCC | 0.841
(0.287-2.469) |
|
BCC | 1.024
(0.884-1.187) |
| Tumor stage | |
|
T1-T2 | 0.987
(0.758-1.285) |
|
T3-T4 | 1.031
(0.567-1.873) |
| Symptoms <1
year | |
|
T1 | 0.909
(0.649-1.273) |
|
T2 | 1.364
(0.449-4.141) |
| Symptoms 1-5
years | |
|
T1-T2 | 0.781
(0.413-1.478) |
|
T3-T4 | 1.389
(0.643-3.000) |
| Symptoms 5-10
years | |
|
T1-T2 | 2.182
(0.275-17.322) |
|
T3-T4 | 0.831
(0.532-1.299) |
Discussion
Tobacco smoking has been around for more than 2000
years ago, either part of religious ceremonies, or simply for
entertainment in more recent times. Before 1800, there were just a
few attempts to link smoking with diseases. But this habit (later
defined as a vice) started to be considered dangerous only around
the 1920's; studies continued in a more organized manner until
1950-1960, when a series of major results were clearly reported and
confirmed that tobacco smoking led to lung cancer (11).
Almost half of the patients included in our study
lot were smokers. The simple notion of a smoker is however complex
since several types of smoking are defined: active (or first-hand)
and passive (or second hand). A series of studies have demonstrated
that passive smoking is frequent. Whether it is smoke from a
burning cigarette, or the smoke exhaled by a smoker nearby, we may
consider that most people are exposed to tobacco smoke. Data from
the literature indicate that more than a quarter of non-smokers
(27.5%) are exposed to secondhand smoke; women being more exposed
than men (12-14).
Similar results were obtained by Oberg et al (15) in Easter Mediterranean and South-East
Asia, with other authors also reporting that women are at least 50%
more susceptible of passive smoking than men (16,17).
Several years ago, a series of studies analyzed the
remainder of particles from first-hand tobacco smoke. These
particles get attached to dust and various surfaces and they remain
there for a long period after the original smoke is no longer
present. In this case, individuals present in this area are exposed
to third hand smoke (also known as residual tobacco smoke)
(18).
From our study lot, 45.92% were active smokers, thus
exposed to first-hand tobacco smoke. People who smoke actively, or
simply stay in an environment where there is cigarette smoke, are
exposed to an ensemble of more than 7000 chemicals; among them,
there are at least 250 toxins with carcinogenic potential. Some of
them have an upregulatory effect on several oncogenes and
transcriptional constituents that may favor carcinogenesis
(19). Others are involved in
carcinogenesis through various mechanisms that interact with cancer
genes or may produce changes at the molecular level and alter the
normal cell cycle, deregulate apoptosis or autophagy processes, or
increase the ability to invade the surrounding areas (20-23).
Studies have found that residues from smoke may combine with gases
from the surrounding air, thus forming cancer-inducing components
that remain on hands or on surfaces (24-26).
These substances have the potential to damage human DNA or impact
blood clotting (27-29).
Tumor aggressiveness
Recent studies have reported increased tumor
progression rates for smokers, compared to non-smokers. This
aggressiveness is due to catabolic transporters and oxidative
stress, since tobacco smoke may favor tumor stroma shifting toward
glycolysis (2). Stroma cells have a
supporting role within the tumor itself and represent more than
half of all tumoral cells. Fibroblasts are the most common stroma
cells and promote tumor progression by generating metabolic
products that act like a fuel for cancer cells. Domingo-Vidal et
al reported that fibroblasts exposed to cigarette smoke favor
increased glycolysis, thus generating more metabolites for tumor
cells, which accelerated their proliferation (2). Moreover, tumor cells become more
resistant to apoptosis and acquire increased mobility (30). Based on tumor cell types, smokers
from our study had similar rates of prevalence for basal cell
carcinoma (BCC) and squamous cell carcinoma (SCC); however
Leonardi-Bee et al reported that tobacco consumption
increases the risk of developing SCC with 52% (1). Still, the number of our SCC patients
was rather small, thus we will continue research in this direction.
Other authors have reported that there also are other factors
activated by nicotine that favor tumoral cell proliferation
(31-34).
Overall, our findings are similar, as active smokers present higher
stage tumors, compared to non-smokers. Periodic screening and early
risk assessment may shift this balance in the future years, as the
use of artificial intelligence, especially machine learning,
increases the efficiency of these processes and diminishes the
burden of physicians in this direction (35-38).
According to our data, eyelid carcinoma and smoking
status are correlated with females, and similar results have been
reported by MercuŢ et al (39) and Wojno (40). In addition, smokers present a higher
risk to develop severe forms of tumors in the first months/years of
symptomatology. From all patients with symptoms present for less
than a year, for all tumor stages in this category (only mild), the
number of smokers was at least equal or higher compared to
non-smokers. T1 was predominant in this group, since patients
sought medical treatment from their very first symptoms. T2 group
was dominated by smokers. A similar status was valid for patients
with a symptom duration between 1 and 5 years. As the tumor stage
increased, so was the number of smoker patients within that
category. Therefore, comparing the distribution of smokers vs.
non-smokers, tobacco consumers had, once again, more advanced tumor
stages, supporting the fact that smoking accelerates tumor
progression rates, which indicates more aggressive behaviors. The
same analysis yielded different results for group with symptoms
between 5 and 10 years. This category was the smallest, with only
19 patients, from whom 13 had a rural residency (61.54% of them
were smokers). After at least 5 years, it was obvious that there
were no patients with T1 tumors, but most smokers had T2 tumors,
followed by T3 and T4 tumors. Compared to the other two groups, the
number of smokers was decreased when the stage was increased. We
can only say that, at this point, smokers are either no longer
smoking (they quit smoking before the diagnosis), or this is
possible evidence that smoking, in association with carcinomas,
does not sustain a long-life expectancy.
Our study presents several limitations. The smoker
status was assessed only based on active smoking or former smoking
for a significant period of time, as we encountered difficulties in
gathering data regarding the smoking period, expressed in number of
years, estimated number of cigars/day, or type of cigarettes/pipe.
Also, the duration of symptoms was the one reported by each patient
before inclusion in our study, without having a consistent method
of definition, and it is based on their personal perception of
symptoms, which may be a subjective estimation as it lacks a common
reference.
In conclusion, tobacco smoke contains many
components that are involved in carcinogenesis and tumor
progression and aggressiveness. Smoking accelerates the progression
rate, thus reaching a higher stage if the patient is an active
smoker, compared to non-smokers for whom the tumor stage evolution
is less rapid.
Acknowledgements
Not applicable.
Funding
Funding: This work was supported by a research grant entitled
“Development of a prediction system regarding the evolution of
non-melanoma skin cancer, for individualized treatment”, financed
by the University of Medicine and Pharmacy of Craiova, Romania,
2021 (Internal competition).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
RM, IMM and MI conceptualized the study, prepared
the initial draft, and share first authorship. ADG, AT and ASD
performed the literature data collection. MEC provided critical
revision in light of the collected data. All authors have read and
approved the final version of the manuscript for publication.
Ethics approval and consent to
participate
Informed consent was obtained from all patients. For
our study, we obtained prior approval from the Ethics Committee of
the University of Medicine and Pharmacy of Craiova, Romania.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Leonardi-Bee J, Ellison T and Bath-Hextall
F: Smoking and the risk of nonmelanoma skin cancer: Systematic
review and meta-analysis. Arch Dermatol. 148:939–946.
2012.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Domingo-Vidal M, Whitaker-Menezes D,
Martos-Rus C, Tassone P, Snyder CM, Tuluc M, Philp N, Curry J and
Martinez-Outschoorn U: Cigarette smoke induces metabolic
reprogramming of the tumor stroma in head and neck squamous cell
carcinoma. Mol Cancer Res. 17:1893–1909. 2019.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Silverman N and Shinder R: What's new in
eyelid tumors. Asia Pac J Ophthalmol (Phila). 6:143–152.
2017.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Balasubramanian A and Kannan N: Eyelid
malignancies-always quite challenging. J Clin Diagn Res.
11:XR01–XR04. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Sasco AJ, Secretan MB and Straif K:
Tobacco smoking and cancer: A brief review of recent
epidemiological evidence. Lung Cancer. 45 (Suppl 2):S3–S9.
2004.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Khani Y, Pourgholam-Amiji N, Afshar M,
Otroshi O, Sharifi-Esfahani O, Sadeghi-Gandomani H, Vejdani M and
Salehiniya H: Tobacco smoking and cancer types: A review. Biomed
Res Ther. 5:2142–2159. 2018.
|
|
7
|
Tonini G, D'Onofrio L, Dell'Aquila L and
Pezzuto A: New molecular insights in tobacco-induced lung cancer.
Future Oncol. 9:649–655. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Pezzuto A, Citarella F, Croghan I and
Tonini G: The effects of cigarette smoking extracts on cell cycle
and tumor spread: novel evidence. Future Sci OA.
5(FSO394)2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Elder DE, Massi D, Scolyer RA and Willemze
R (eds): World Health Organization (WHO) Classification of Skin
Tumours. 4th edition. International Agency for Research on Cancer
(IARC) Press, Lyon, pp7-14, 2018.
|
|
10
|
Amin MB, Edge S, Greene F, Byrd DR,
Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR,
Sullivan DC (eds), et al: AJCC cancer staging manual (8th edition).
Springer International Publishing, American Joint Commission on
Cancer, 2017.
|
|
11
|
Brandt AM: Cigarette Century: the Rise,
Fall and Deadly Persistence of the Product that Defined America.
Basic Books, New York, NY, 2007.
|
|
12
|
Salimzadeh H, Najafipour H, Mirzaiepour F,
Navadeh S, Shadkam-Farrokhi M and Mirzazadeh A: Prevalence of
active and passive smoking among adult population: Findings of a
population-based survey in Kerman (KERCADRS), Iran. Addict Health.
8:16–24. 2016.PubMed/NCBI
|
|
13
|
Parkin D, Boyd L and Walker L: 16. The
fraction of cancer attributable to lifestyle and environmental
factors in the UK in 2010. Br J Cancer. 105 (Suppl 2):S77–S81.
2011.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Lim SS, Vos T, Flaxman AD, Danaei G,
Shibuya K, Adair-Rohani H, Amann M, Anderson HR, Andrews KG, Aryee
M, et al: A comparative risk assessment of burden of disease and
injury attributable to 67 risk factors and risk factor clusters in
21 regions, 1990-2010: A systematic analysis for the global burden
of disease study 2010. Lancet. 380:2224–2260. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Oberg M, Jaakkola MS, Woodward A, Peruga A
and Prüss-Ustün A: Worldwide burden of disease from exposure to
second-hand smoke: A retrospective analysis of data from 192
countries. Lance. 377:139–146. 2011.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ferrante G, Simoni M, Cibella F, Ferrara
F, Liotta G, Malizia V, Corsello G, Viegi F and La Grutta S:
Third-hand smoke exposure and health hazards in children. Monaldi
Arch Chest Dis. 79:38–43. 2013.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Martins-Green M, Adhami N, Frankos M,
Valdez M, Goodwin B, Lyubovitsky J, Dhall S, Garcia M, Egiebor I,
Martinez B, et al: Cigarette smoke toxins deposited on surfaces:
Implications for human health. PLoS One. 9(e86391)2014.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Jacob P III, Benowitz NL, Destaillats H,
Gundel L, Hang B, Martins-Green M, Matt GE, Quintana PJE, Samet JM,
Schick SF, et al: Thirdhand smoke: New evidence, challenges, and
future directions. Chem Res Toxicol. 30:270–294. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Hussain A, Dulay P, Rivera MN, Aramuni C
and Saxena V: Neoplastic pathogenesis associated with cigarette
carcinogens. Cureus. 11(e3955)2019.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Kim CW, Lee HM, Lee K, Kim B, Lee MY and
Choi KC: Effects of cigarette smoke extracts on cell cycle, cell
migration and endocrine activity in human placental cells. Reprod
Toxicol. 73:8–19. 2017.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Xu L, Mao XY, Fan CF and Zheng HC: MTA1
expression correlates significantly with cigarette smoke in
non-small cell lung cancer. Virchows Arch. 459:415–422.
2011.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Zaharie M, Carstea D, Streba CT, Mitrut P,
Glodeanu AD, Carstea AP, Zaharie SI, Dascalu IT, Tuculina MJ,
Bunget A, et al: Renal dysfunction-a possible marker of severity of
heart failure. Rev Chim. 69:1435–1440. 2018.
|
|
23
|
Tian D, Zhu M, Li J, Ma Y and Wu R:
Cigarette smoke extract induces activation of beta-catenin/TCF
signaling through inhibiting GSK3beta in human alveolar epithelial
cell line. Toxicol Lett. 187:58–62. 2009.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Sleiman M, Gundel LA, Pankow JF, Jacob P
III, Singer BC and Destaillats H: Formation of carcinogens indoors
by surface-mediated reactions of nicotine with nitrous acid,
leading to potential thirdhand smoke hazards. Proc Natl Acad Sci
USA. 107:6576–6581. 2010.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Thomas JL, Hecht SS, Luo X, Ming X,
Ahluwalia JS and Carmella SG: Thirdhand tobacco smoke: A
tobacco-specific lung carcinogen on surfaces in smokers' homes.
Nicotine Tob Res. 16:26–32. 2014.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Hoh E, Hunt RN, Quintana PJ, Zakarian JM,
Chatfield DA, Wittry BC, Rodriguez E and Matt GE: Environmental
tobacco smoke as a source of polycyclic aromatic hydrocarbons in
settled household dust. Environ Sci Technol. 46:4174–4183.
2012.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Hang B, Sarker AH, Havel C, Saha S, Hazra
TK, Schick S, Jacob P III, Rehan VK, Chenna A, Sharan D, et al:
Thirdhand smoke causes DNA damage in human cells. Mutagenesis.
28:381–391. 2013.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Cârstea D, Streba LA, Glodeanu AD, Cârstea
AP, Vancu M and Ninulescu AM: The accuracy of combined physical
examination and ultrasonography for the detection of abdominal
aorta aneurysm. Rom J Morphol Embryol. 49:569–572. 2008.PubMed/NCBI
|
|
29
|
Constantinescu AF, Ionescu M, Iovanescu
VF, Ciurea ME, Ionescu AG, Streba CT, Bunescu MG, Rogoveanu I and
Vere CC: A computer-aided diagnostic system for intestinal polyps
identified by wireless capsule endoscopy. Rom J Morphol Embryol.
57:979–984. 2016.PubMed/NCBI
|
|
30
|
Schaal C and Chellappan SP:
Nicotine-mediated cell proliferation and tumor progression in
smoking-related cancers. Mol Cancer Res. 12:14–23. 2014.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Paleari L, Catassi A, Ciarlo M, Cavalieri
Z, Bruzzo C, Servent D, Cesario A, Chessa L, Cilli M, Piccardi F,
et al: Role of alpha7-nicotinic acetylcholine receptor in human
non-small cell lung cancer proliferation. Cell Prolif. 41:936–959.
2008.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Oh JJ, West AR, Fishbein MC and Slamon DJ:
A candidate tumor suppressor gene, H37, from the human lung cancer
tumor suppressor locus 3p21.3. Cancer Res. 62:3207–3213.
2002.PubMed/NCBI
|
|
33
|
Bechara E, Sebestyén E, Bernardis I, Eyras
E and Valcárcel J: RBM5, 6, and 10 differentially regulate NUMB
alternative splicing to control cancer cell proliferation. Mol
Cell. 52:720–733. 2013.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Sutherland LC, Wang K and Robinson AG:
RBM5 as a putative tumor suppressor gene for lung cancer. J Thorac
Oncol. 5:294–298. 2010.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Ahmad Z, Rahim S, Zubair M and
Abdul-Ghafar J: Artificial intelligence (AI) in medicine, current
applications and future role with special emphasis on its potential
and promise in pathology: Present and future impact, obstacles
including costs and acceptance among pathologists, practical and
philosophical considerations. A comprehensive review. Diagn Pathol.
16(24)2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Streba CT, Ionescu M, Vere CC and
Rogoveanu I: Artificial intelligence and automatic image
interpretation in modern medicine. In: Translational Bioinformatics
and Its Application. Translational Medicine Research. Wei DQ, Ma Y,
Cho W, Xu Q and Zhou F (eds). Springer, Dordrecht, pp371-407,
2017.
|
|
37
|
Venkadesh KV, Setio AAA, Schreuder A,
Scholten ET, Chung K, W Wille MM, Saghir Z, van Ginneken B, Prokop
M and Jacobs C: Deep learning for malignancy risk estimation of
pulmonary nodules detected at low-dose screening CT. Radiology.
300:438–447. 2021.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Joy Mathew C, David AM and Joy Mathew CM:
Artificial intelligence and its future potential in lung cancer
screening. EXCLI J. 19:1552–1562. 2020.PubMed/NCBI View Article : Google Scholar
|
|
39
|
MercuŢ IM, Ilia LC, TĂnasie CA, Ionescu M,
MercuŢ R, PÎrvĂnescu V and Ciurea ME: Analysis of tumour related
data and clinical features of eyelid carcinomas. Curr Health Sci J.
46:222–229. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Wojno TH: The association between
cigarette smoking and basal cell carcinoma of the eyelids in women.
Ophthalmic Plast Reconstr Surg. 15:390–392. 1999.PubMed/NCBI View Article : Google Scholar
|