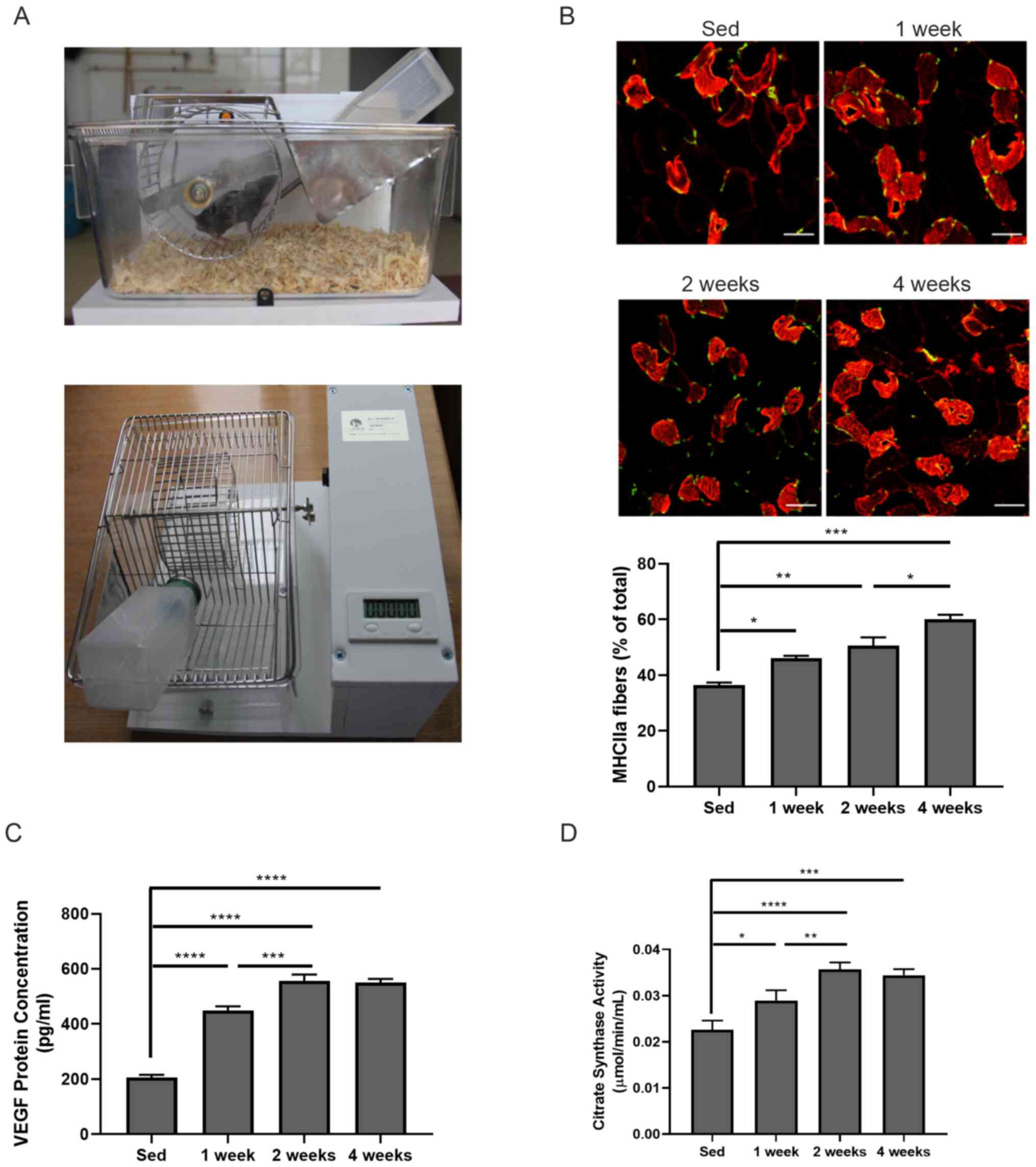
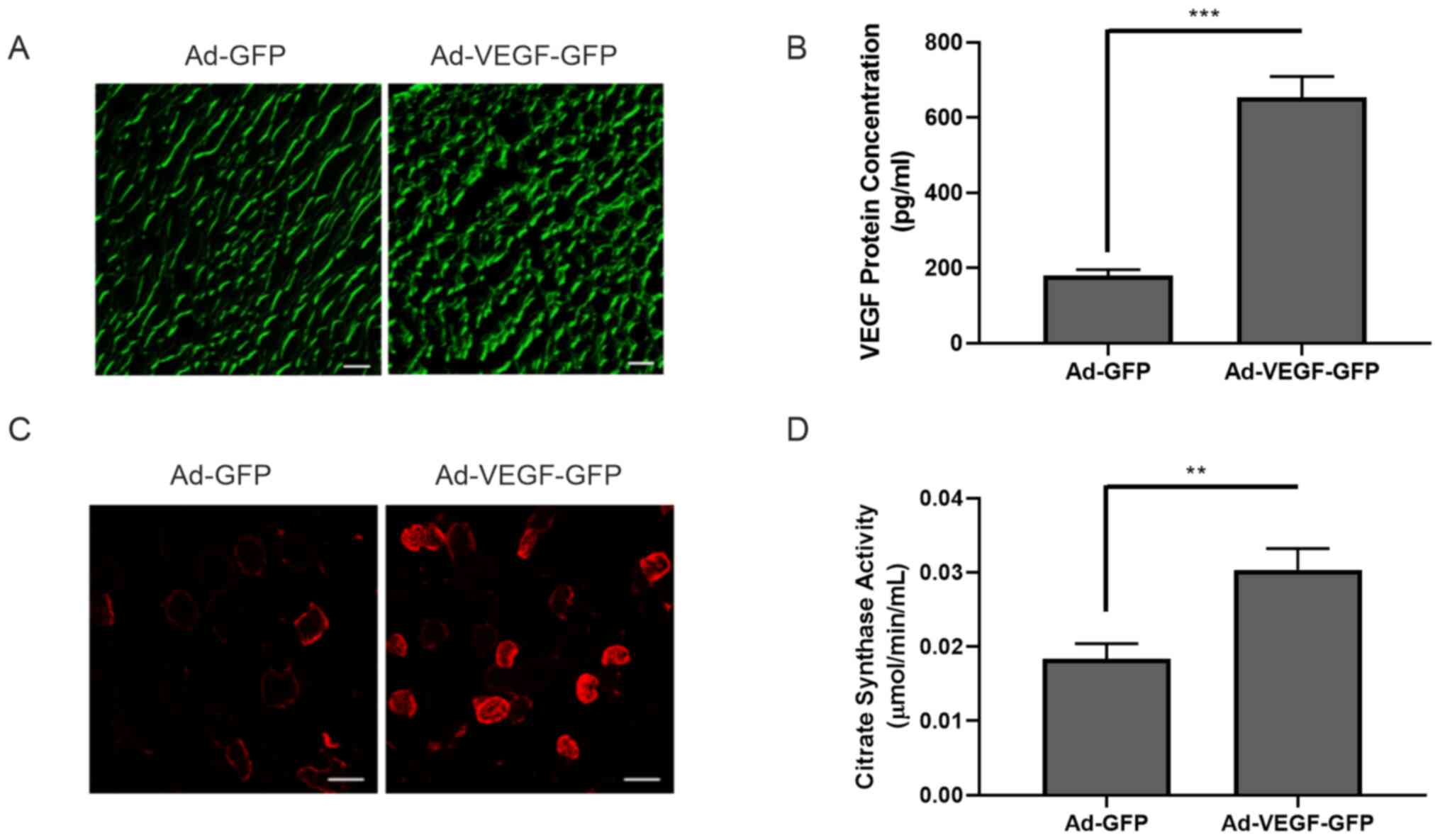
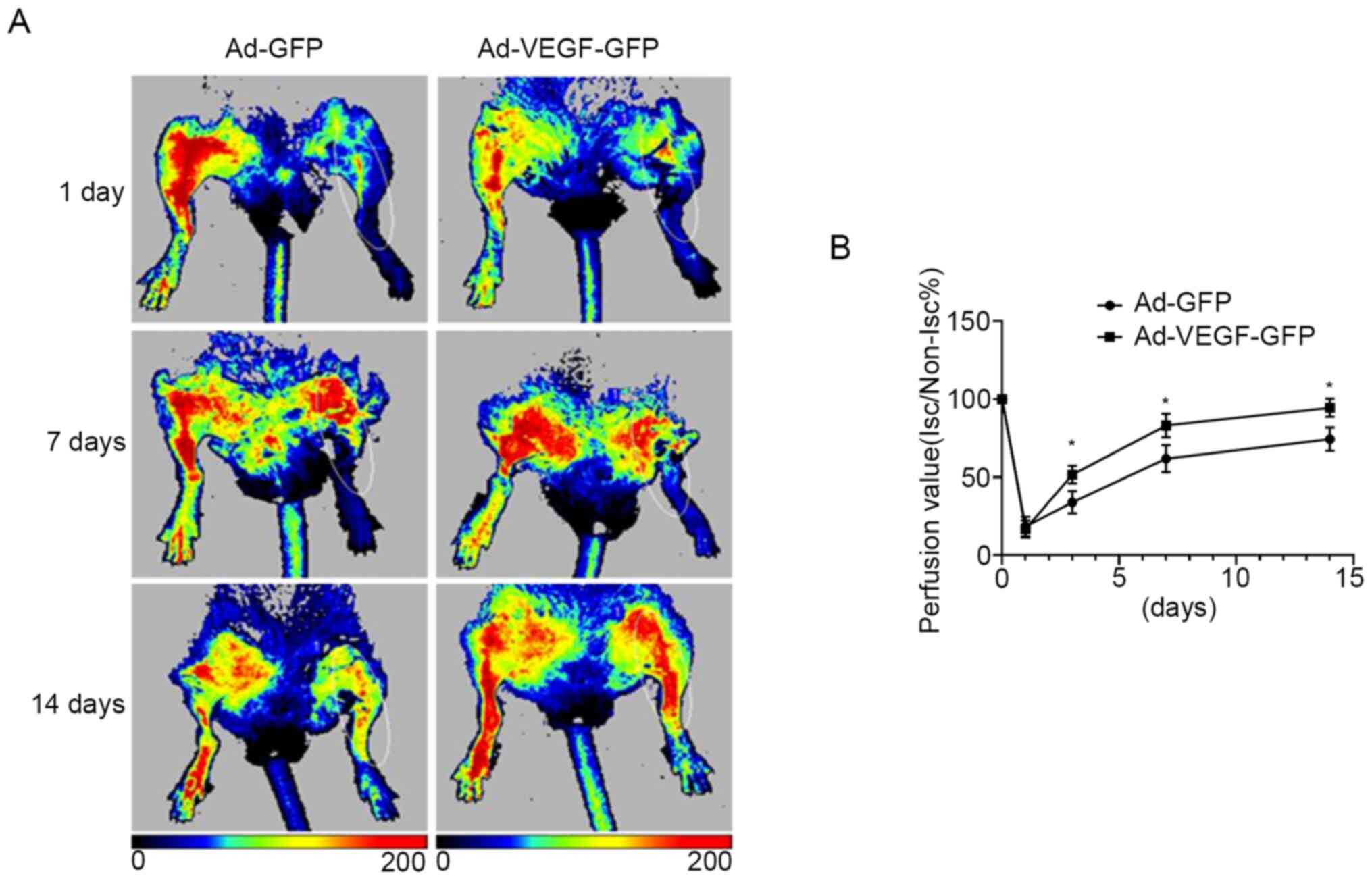
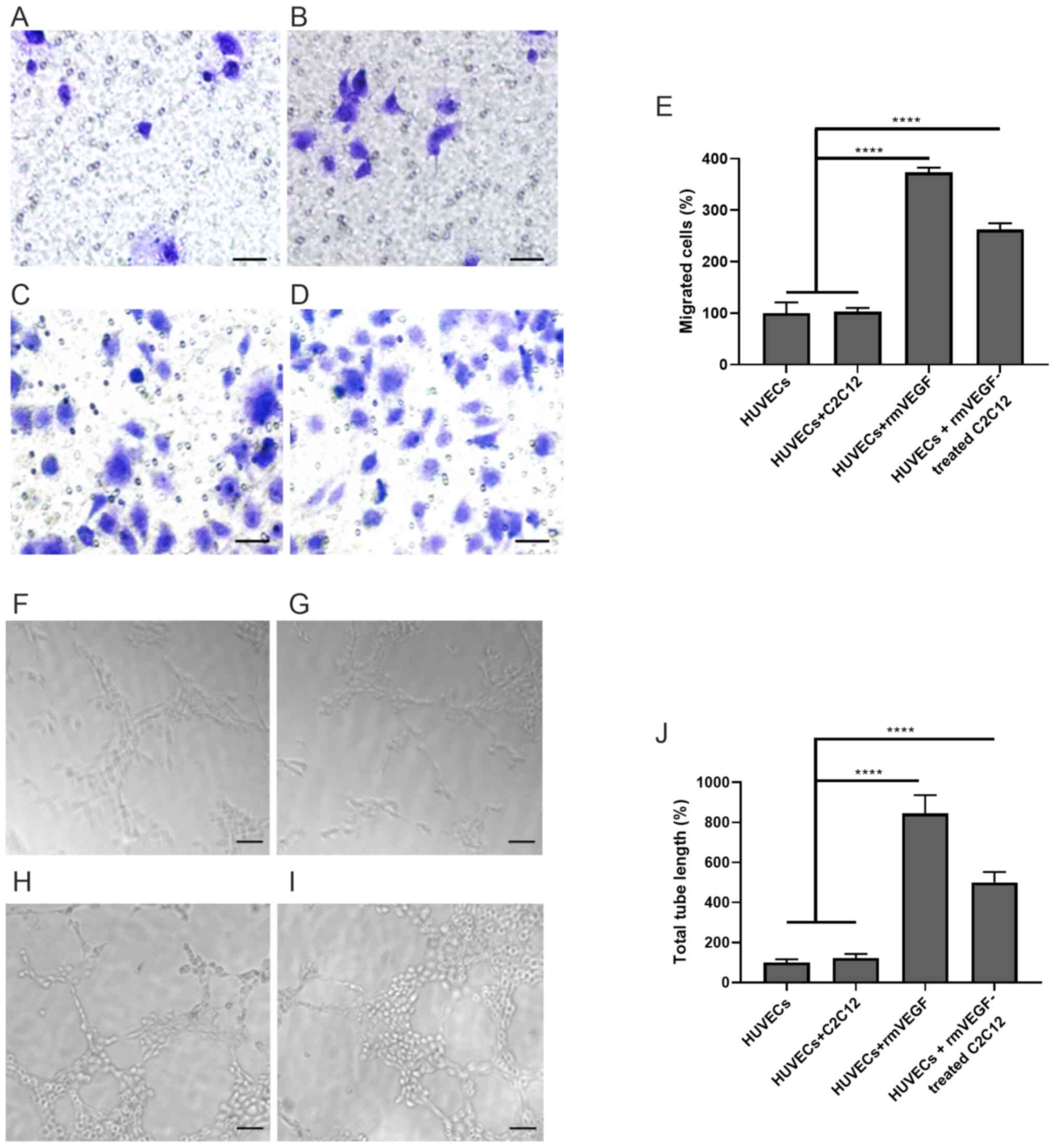
|
1
|
Boulton AJ, Vileikyte L,
Ragnarson-Tennvall G and Apelqvist J: The global burden of diabetic
foot disease. Lancet. 366:1719–1724. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Johannesson A, Larsson GU, Ramstrand N,
Turkiewicz A, Wiréhn AB and Atroshi I: Incidence of lower-limb
amputation in the diabetic and nondiabetic general population: A
10-year population-based cohort study of initial unilateral and
contralateral amputations and reamputations. Diabetes Care.
32:275–280. 2009.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Prompers L, Schaper N, Apelqvist J,
Edmonds M, Jude E, Mauricio D, Uccioli L, Urbancic V, Bakker K,
Holstein P, et al: Prediction of outcome in individuals with
diabetic foot ulcers: Focus on the differences between individuals
with and without peripheral arterial disease. The EURODIALE study.
Diabetologia. 51:747–755. 2008.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Prompers L, Huijberts M, Apelqvist J, Jude
E, Piaggesi A, Bakker K, Edmonds M, Holstein P, Jirkovska A,
Mauricio D, et al: High prevalence of ischaemia, infection and
serious comorbidity in patients with diabetic foot disease in
Europe. Baseline results from the Eurodiale study. Diabetologia.
50:18–25. 2007.PubMed/NCBI View Article : Google Scholar
|
|
5
|
American Diabetes Association. Peripheral
arterial disease in people with diabetes. Diabetes Care.
26:3333–3341. 2003.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Guo X, Shi Y, Huang X, Ye M, Xue G and
Zhang J: Features analysis of lower extremity arterial lesions in
162 diabetes patients. J Diabetes Res. 2013(781360)2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Adams V and Linke A: Impact of exercise
training on cardiovascular disease and risk. Biochim Biophys Acta
Mol Basis Dis. 1865:728–734. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Brevetti G, Silvestro A, Schiano V and
Chiariello M: Endothelial dysfunction and cardiovascular risk
prediction in peripheral arterial disease: Additive value of
flow-mediated dilation to ankle-brachial pressure index.
Circulation. 108:2093–2098. 2003.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Daiber A, Steven S, Weber A, Shuvaev VV,
Muzykantov VR, Laher I, Li H, Lamas S and Münzel T: Targeting
vascular (endothelial) dysfunction. Br J Pharmacol. 174:1591–1619.
2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Senger DR, Galli SJ, Dvorak AM, Perruzzi
CA, Harvey VS and Dvorak HF: Tumor cells secrete a vascular
permeability factor that promotes accumulation of ascites fluid.
Science. 219:983–985. 1983.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Germani A, Di Carlo A, Mangoni A, Straino
S, Giacinti C, Turrini P, Biglioli P and Capogrossi MC: Vascular
endothelial growth factor modulates skeletal myoblast function. Am
J Pathol. 163:1417–1428. 2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yan Z, Okutsu M, Akhtar YN and Lira VA:
Regulation of exercise-induced fiber type transformation,
mitochondrial biogenesis, and angiogenesis in skeletal muscle. J
Appl Physiol (1985). 110:264–274. 2011.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Oberbach A, Bossenz Y, Lehmann S, Niebauer
J, Adams V, Paschke R, Schön MR, Blüher M and Punkt K: Altered
fiber distribution and fiber-specific glycolytic and oxidative
enzyme activity in skeletal muscle of patients with type 2
diabetes. Diabetes Care. 29:895–900. 2006.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Picard M, Hepple RT and Burelle Y:
Mitochondrial functional specialization in glycolytic and oxidative
muscle fibers: Tailoring the organelle for optimal function. Am J
Physiol Cell Physiol. 302:C629–C641. 2012.PubMed/NCBI View Article : Google Scholar
|
|
15
|
van Weel V, Deckers MML, Grimbergen JM,
van Leuven KJ, Lardenoye JH, Schlingemann RO, van Nieuw Amerongen
GP, van Bockel JH, van Hinsbergh VW and Quax PH: Vascular
endothelial growth factor overexpression in ischemic skeletal
muscle enhances myoglobin expression in vivo. Circ Res. 95:58–66.
2004.PubMed/NCBI View Article : Google Scholar
|
|
16
|
McGrath JC and Lilley E: Implementing
guidelines on reporting research using animals (ARRIVE etc.): New
requirements for publication in BJP. Br J Pharmacol. 172:3189–3193.
2015.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Novak CM, Burghardt PR and Levine JA: The
use of a running wheel to measure activity in rodents: Relationship
to energy balance, general activity, and reward. Neurosci Biobehav
Rev. 36:1001–1014. 2012.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Waters RE, Rotevatn S, Li P, Annex BH and
Yan Z: Voluntary running induces fiber type-specific angiogenesis
in mouse skeletal muscle. Am J Physiol Cell Physiol.
287:C1342–C1348. 2004.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Thinakaran G, Ojala J and Bag J:
Expression of c-jun/AP-1 during myogenic differentiation in mouse
C2C12 myoblasts. FEBS Lett. 319:271–276. 1993.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Jensen EC: Quantitative analysis of
histological staining and fluorescence using ImageJ. Anat Rec
(Hoboken). 296:378–381. 2013.PubMed/NCBI View
Article : Google Scholar
|
|
21
|
Padgett ME, McCord TJ, McClung JM and
Kontos CD: methods for acute and subacute murine hindlimb Ischemia.
J Vis Exp. 112(54166)2016.PubMed/NCBI View
Article : Google Scholar
|
|
22
|
Tokudome T, Kishimoto I, Yamahara K, Osaki
T, Minamino N, Horio T, Sawai K, Kawano Y, Miyazato M, Sata M, et
al: Impaired recovery of blood flow after hind-limb ischemia in
mice lacking guanylyl cyclase-A, a receptor for atrial and brain
natriuretic peptides. Arterioscler Thromb Vasc Biol. 29:1516–1521.
2009.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Jockusch H and Eberhard D: Green
fluorescent protein as a tracer in chimeric tissues: The power of
vapor fixation. Methods Mol Biol. 411:145–154. 2007.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Ariyanti AD, Sisjayawan J, Zhang J, Zhang
JQ, Wang GX, Miyagishi M, Wu SR and Kasim V: Elevating VEGF-A and
PDGF-BB secretion by salidroside enhances neoangiogenesis in
diabetic hind-limb ischemia. Oncotarget. 8:97187–97205.
2017.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Liu N, Ding D, Hao W, Yang F, Wu X, Wang
M, Xu X, Ju Z, Liu JP, Song Z, et al: hTERT promotes tumor
angiogenesis by activating VEGF via interactions with the Sp1
transcription factor. Nucleic Acids Res. 44:8693–8703.
2016.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Delavar H, Nogueira L, Wagner PD, Hogan
MC, Metzger D and Breen EC: Skeletal myofiber VEGF is essential for
the exercise training response in adult mice. Am J Physiol Regul
Integr Comp Physiol. 306:R586–R595. 2014.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Huh JY: The role of exercise-induced
myokines in regulating metabolism. Arch Pharm Res. 41:14–29.
2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Pedersen BK and Febbraio MA: Muscles,
exercise and obesity: Skeletal muscle as a secretory organ. Nat Rev
Endocrinol. 8:457–465. 2012.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Wrann CD, White JP, Salogiannnis J,
Laznik-Bogoslavski D, Wu J, Ma D, Lin JD, Greenberg ME and
Spiegelman BM: Exercise induces hippocampal BDNF through a
PGC-1α/FNDC5 pathway. Cell Metab. 18:649–659. 2013.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Birot OJG, Koulmann N, Peinnequin A and
Bigard XA: Exercise-induced expression of vascular endothelial
growth factor mRNA in rat skeletal muscle is dependent on fibre
type. J Physiol. 552:213–221. 2003.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Cannon DT, Rodewohl L, Adams V, Breen EC
and Bowen TS: Skeletal myofiber VEGF deficiency leads to
mitochondrial, structural, and contractile alterations in mouse
diaphragm. J Appl Physiol (1985). 127:1360–1369. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Leick L, Hellsten Y, Fentz J, Lyngby SS,
Wojtaszewski JF, Hidalgo J and Pilegaard H: PGC-1alpha mediates
exercise-induced skeletal muscle VEGF expression in mice. Am J
Physiol Endocrinol Metab. 297:E92–E103. 2009.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Matsakas A, Yadav V, Lorca S, Evans RM and
Narkar VA: Revascularization of ischemic skeletal muscle by
estrogen-related receptor-γ. Circ Res. 110:1087–1096.
2012.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Mishra P, Varuzhanyan G, Pham AH and Chan
DC: Mitochondrial dynamics is a distinguishing feature of skeletal
muscle fiber types and regulates organellar compartmentalization.
Cell Metab. 22:1033–1044. 2015.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Hood DA, Memme JM, Oliveira AN and Triolo
M: Maintenance of skeletal muscle mitochondria in health, exercise,
and aging. Annu Rev Physiol. 81:19–41. 2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Fernandez-Marcos PJ and Auwerx J:
Regulation of PGC-1α, a nodal regulator of mitochondrial
biogenesis. Am J Clin Nutr. 93 (Suppl):884S–890S. 2011.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Chinsomboon J, Ruas J, Gupta RK, Thom R,
Shoag J, Rowe GC, Sawada N, Raghuram S and Arany Z: The
transcriptional coactivator PGC-1alpha mediates exercise-induced
angiogenesis in skeletal muscle. Proc Natl Acad Sci USA.
106:21401–21406. 2009.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Handschin C, Chin S, Li P, Liu F,
Maratos-Flier E, Lebrasseur NK, Yan Z and Spiegelman BM: Skeletal
muscle fiber-type switching, exercise intolerance, and myopathy in
PGC-1alpha muscle-specific knock-out animals. J Biol Chem.
282:30014–30021. 2007.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Lin J, Wu H, Tarr PT, Zhang CY, Wu Z, Boss
O, Michael LF, Puigserver P, Isotani E, Olson EN, et al:
Transcriptional co-activator PGC-1 alpha drives the formation of
slow-twitch muscle fibres. Nature. 418:797–801. 2002.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Zhao Y, Li X, Yang L, Eckel-Mahan K, Tong
Q, Gu X, Kolonin MG and Sun K: Transient overexpression of vascular
endothelial growth factor a in adipose tissue promotes energy
expenditure via activation of the sympathetic nervous system. Mol
Cell Biol. 38:e00242–18. 2018.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Liu X, Chu B, Jin S, Li M, Xu Y, Yang H,
Feng Z, Bi J and Wang P: Vascular endothelial growth factor
alleviates mitochondrial dysfunction and suppression of
mitochondrial biogenesis in models of Alzheimer's disease. Int J
Neurosci. 131:154–162. 2021.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Wright GL, Maroulakou IG, Eldridge J, Liby
TL, Sridharan V, Tsichlis PN and Muise-Helmericks RC: VEGF
stimulation of mitochondrial biogenesis: Requirement of AKT3
kinase. FASEB J. 22:3264–3275. 2008.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Mészáros K, Lang CH, Bagby GJ and Spitzer
JJ: Contribution of different organs to increased glucose
consumption after endotoxin administration. J Biol Chem.
262:10965–10970. 1987.PubMed/NCBI
|
|
45
|
Evans PL, McMillin SL, Weyrauch LA and
Witczak CA: Regulation of skeletal muscle glucose transport and
glucose metabolism by exercise training. Nutrients.
11(2432)2019.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Gaster M, Beck-Nielsen H and Schroder H:
Regenerating human muscle fibres express GLUT3 protein. Pflügers
Arch. 445:105–114. 2002.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Gaster M, Franch J, Staehr P, Beck-Nielsen
H, Smith T and Schroder H: Induction of GLUT-1 protein in adult
human skeletal muscle fibers. Am J Physiol Endocrinol Metab.
279:E1191–E1195. 2000.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Goodyear LJ, Hirshman MF, Smith RJ and
Horton ES: Glucose transporter number, activity, and isoform
content in plasma membranes of red and white skeletal muscle. Am J
Physiol Endocrinol Metab. 261:E556–E561. 1991.PubMed/NCBI View Article : Google Scholar
|