Introduction
Osteoporosis (OP) is a systemic metabolic bone
disease characterized by loss of bone mass, bone microstructural
damage, decreased bone strength, and increased risk of bone
fractures. It occurs more frequently in the elderly for whom it is
one of the main causes of disability. The aging of society, living
environment, dietary habits, as well as other factors contribute to
the increased incidence of OP (1,2). The
main pathogenesis of OP is the proliferation and differentiation of
osteoclasts. The formation and inhibition of osteoclasts are
associated with multiple signaling pathways, including the
OPG/RANKL/RANK signaling pathway, tumor necrosis factor signaling
pathway, nitric oxide and estrogen, peroxisome
proliferator-activated receptor γ (PPARγ), and
microphthalmia-associated transcription factor (3,4). Among
these pathways, the PPARγ pathway has a pivotal role in osteoclast
formation and the mechanism of OP caused by its excessive
activation. Previous studies have demonstrated that activated PPARγ
could promote osteoclast differentiation and inhibit the
differentiation of mesenchymal stem cells into osteoblasts
(5,6). Furthermore, animal experiments
demonstrated that knockout of the PPARγ gene could lead to severe
osteosclerosis in mice, which indicates that differentiation and
activity of osteoclasts were significantly inhibited (7). Thus, the application of PPARγ as a
target for inhibiting osteoclast formation could be an important
strategy for OP treatment.
Over recent years, previous studies have found that
the ligand of PPARγ must first bind to heat shock protein (HSP)90α
and then polymerize with PPARγ (8,9). It
has been suggested that HSP90α may be an important molecule
affecting PPARγ. Therefore, it was theorized that HSP90α may affect
the formation and activation of osteoclasts by regulating PPARγ,
and participating in the occurrence of OP. Thus, the aim of this
study was to investigate the relationship between HSP90α and
osteoclasts, and explore their association and the effect on OP in
mice through in vivo and in vitro experiments.
Materials and methods
THP-1 human monocytic cell line was purchased from
American Type Culture Collection (cat. no. TIB-202). Mouse
anti-PPARγ antibody (product code ab41928), rabbit
anti-tartrate-resistant acid phosphatase (TRAP) antibody (product
code ab65854) and rabbit anti-HSP90α antibody (product code
ab79849) were acquired from Abcam. Rabbit anti-GAPDH (cat. no.
ab8245; Abcam) antibody, HRP-labeled goat anti-rabbit IgG antibody
(cat. no. #7074S) and goat anti-mouse IgG antibody (cat. no.
#7076S)were provided by Cell Signaling Technology, Inc.
Alvespimycin was obtained from MedChemExpress (product no.
HY-10389). High-glucose Dulbecco's modified Eagle medium
(Dulbecco's modified Eagle medium, DMEM), fetal bovine serum (FBS)
and other reagents for cell culture were provided by Gibco; Thermo
Fisher Scientific, Inc. TRAP kits were products of Sigma-Aldrich;
Merck KGaA (cat. no. 387A). Cell lysis buffer, protease inhibitors
and other reagents were purchased from Beyotime Institute of
Biotechnology. Chemiluminescence detection reagent pro-light HRP
was purchased from MilliporeSigma.
THP-1 cell-induced
differentiation
THP-1 cells were seeded in a 24-well plate
(1x106 cells/well). After cell attachment (~4 h), cells
were cultured with normal RPMI-1640 medium and a humidified
atmosphere containing 5% CO2 at 37˚C. The next day,
cells were treated with 1,000 units of M-CSF, PMA (5 ng/ml) and 50
ng/ml soluble receptor activator of NF-κB ligand (sRANKL) was added
to the culture medium. After 4 days, it was observed if the cells
had coenocytes and TRAP staining was performed after 5 days of
induction.
Anti-TRAP staining
The old culture medium in the 24-well plate was
discarded. Cells (1x105) were then gently washed two
times with sterile ice PBS, fixed with paraformaldehyde (4%) at
room temperature for 30 sec and was stained with hematoxylin for 1
h in a water bath at 37˚C. Glycerol was used for mounting, and an
inverted light microscope was used for observation.
Cell counting kit-8 (CCK-8) assay
The treated THP-1 cells (5,000/well) in RPMI-1640
medium containing 10% FBS were seeded into a 96-well plate (100
µl/well). Each well was then mixed with 10 µl of CCK-8 (cat. no.
CK04; Dojindo Molecular Technologies, Inc.) at 37˚C and 5%
CO2 in the dark for 1 h. The OD value was measured at
450 nm on a microplate reader at 0- and 1-h time-points.
Detection of proliferation by CFDA
SE-labeling and flow cytometry analysis
After the cells were treated with alvespimycin (20
nM) for 24 h, they were collected by and washed once with PBS; and
then with a 1X Buffer A kit (cat. no. C1031; Beyotime Institute of
Biotechnology) according to the manufacturer's instructions. After
collection, the cell concentration was adjusted to 1x106
cells/ml. The cells were collected by centrifugation (447 x g; 3
min; 25˚C), washed with 1X Buffer A and resuspended in 500 µl 1X
Buffer A. Then, 5 µl CFDA-SE fluorescence probe (cat. no. C1031;
Beyotime Institute of Biotechnology) was added for staining for 30
min in the dark at room temperature. Cells were then transferred to
the flow cytometer tube and detection on the instrument was
performed. CFDA SE was excited by a 488-nm argon-ion laser and
analyzed using a 630-nm band-pass filter. Finally, cells were
collected through the FSC/SSC scatter plot. Gating technology was
used to exclude adherent cells and fragments. Finally, Flowjo
software (version 10.6.0; FlowJo LLC) was used to obtain the
proportion of cells in second generation, and then a histogram was
constructed.
Western blot analysis
The total protein of cells and tissues was extracted
with RIPA cell lysis buffer (Beyotime Institute of Biotechnology)
and the protein concentration was determined by the BCA method. The
protein loading amount at 40 mg/lane was subjected to SDS-PAGE
(10%). The protein was transferred to a PVDF membrane by wet
transfer at a constant voltage of 70 V for 1 h. Next, 5% skim milk
powder was used for blocking at room temperature for 2 h. Samples
were then incubated with the primary antibodies (anti-HSP90α,
1:1,000; and anti-PPARγ, 1:500) at 4˚C overnight and then with the
secondary antibodies (1:3,000) at room temperature for 2 h. ECL
chemiluminescence was used to detect protein expression.
Densitometry was quantified by ImageJ software (v1.8.0; National
Institutes of Health).
Establishment of a mouse model of OP
after ovariectomy
A total 25 Balb/c male mice, 6-8 weeks old, weighing
20-25 g, were obtained from Lingchang Biotechnology Co., Ltd. All
the animals were housed in an environment with a temperature of
22±1˚C, relative humidity of 50±1%, and a 12-h light/dark cycle.
All the animals had free access to food and water. All animal
studies (including the procedure for euthanasia of mice) were
performed in compliance with the regulations and guidelines of the
Laboratory Animal Center and Ethics Committee of Changzheng
Hospital (approval ID, #CZ2019112130) and conducted according to
the AAALAC and the IACUC guidelines.
Mice weight was monitored for more than 3 days.
Ovariectomy was performed after no significant change in weight was
detected. Before surgery, surgical instruments were disinfected
with a high temperature, including tissue scissors, tissue forceps,
tweezers, needle holders, cotton balls and rubber bands; disposable
surgical blades, sutures and surgical towels were obtained from the
Department of Instruments of Changzheng Hospital.
The laboratory mice were anesthetized by
intraperitoneal injection of 3% pentobarbital sodium solution (50
mg/kg weight). After anesthesia was completed, the mice were placed
in the prone position on the operating table, and their four limbs
and front teeth were fixed. A surgical blade was used to prepare
the skin in the lower back, after which the lower back was
disinfected with iodophor, and a surgical towel was draped.
Scissors were used to produce a longitudinal incision of ~1 cm in
the back of mice away from the lumbar spine. The lower back muscles
were cut on both sides, and the ovaries were exposed. The mouse
ovaries were red cauliflower-shaped, surrounded by adipose tissues,
containing fallopian tubes and blood vessels. For sham-operated
mice, the ovaries were removed and immediately placed back to the
abdominal cavity. For ovariectomized mice, the fallopian tubes and
blood vessels were ligated, and the ovaries and surrounding adipose
tissues were removed. After confirming that the ovaries were
completely removed, the stump of the fallopian tube was disinfected
and placed back to the abdominal cavity. The muscles were sutured
layer by layer. Disinfection was performed again to suture the
skin. Picric acid was applied to the skin wound, and the numbered
ear studs were fixed on the left ear of the mice for labeling. The
mice were then placed into cages. After the mice were fully
awakened, they were transferred to the breeding place, and the
feeding was continued.
On day 42 after the ovaries were removed, mice were
euthanized by 70% carbon dioxide inhalation, and death was
confirmed by verifying respiratory and cardiac arrest. Then their
bone tissues were fixed in 4% polyformaldehyde at room temperature
for 48 h, and decalcified in 10% EDTA at room temperature for 10-14
days. The decalcified bone tissues were embedded in paraffin and
were sectioned with mean border thickness of 1.9 mm and then
dehydrated in 70% ethanol for 30 min, 80% ethanol for 30 min, 90%
ethanol three times for 1 h and 100% ethanol twice for 2 h at room
temperature, cleared in xylene and paraffin embedded.
Cross-sections (6 µm) were prepared for staining with H&E and
Masson's trichrome at room temperature for histological evaluation
that was observed by light microscope (Eclipse 80i; Nikon
Corporation) and was analyzed by the ImageJ software (v1.8.0;
National Institutes of Health).
H&E staining
Cross-sections (6 µm) of paraffin-embedded bone
tissues were prepared. Firstly, xylene was used to remove paraffin
from the sections, and subsequently the sections were rehydrated
into ethanol (from 90-70%). After being washed with water, the
sections were placed in hematoxylin aqueous solution for a few min.
Subsequently, the sections were incubated in acid water and ammonia
water for a few secs, washed with water for 1 h, placed into
ethanol (from 70-90%) for 10 min, and finally incubated into
alcohol eosin staining solution for 2-3 min. After transparency
with xylene, the sections were mounted with Canadian gum, covered
with a cover glass and sealed, and observed under a light
microscope. All the procedures were performed at room
temperature.
Masson's trichrome staining
Bone paraffin sections were routinely deparaffinized
to water as aforementioned and stained with Weigert's iron
hematoxylin staining solution for 5-10 min, followed by acid
ethanol differentiation solution for 5-15 sec and washing with
water. Subsequently, the sections were stained with Masson's blue
solution for 3-5 min and washed with water. Then, the sections were
washed with distilled water for 1 min, and stained with Ponceau red
magenta staining solution for 5-10 min. The sections were washed
with a weak acid solution (ratio of distilled water: Weak acid
solution=2:1) for 1 min, and subsequently washed with
phosphomolybdic acid solution for 1-2 min, followed by washing with
the prepared weak acid working solution for 1 min, and directly
placed into aniline blue staining solution for 1-2 min, washed with
the prepared weak acid working solution for 1 min, quickly
dehydrated with 95% ethanol and three times with anhydrous ethanol
(5-10 sec each), followed by three times incubation in xylene (1-2
min each), and finally sealed with neutral gum and observed under a
light microscope. All the procedures were performed at room
temperature.
Immunofluorescence assay
The THP-1 cells were seeded in 24-well plates
(2x105/well), washed twice with PBS, fixed with cold
acetone/methanol (1:1) for 20 min and blocked at room temperature
with 10% normal goat serum for 1 h. The anti-HSP90Αα (1:1,000) and
anti-PPARγ (1:1,000) antibodies were separately added, and
incubated at 4˚C in a wet box overnight. A FITC-labeled goat
anti-mouse secondary antibody (1:3,000; cat. no. AP130F;
Sigma-Aldrich; Merck KGaA) was added dropwise and incubated for 1 h
at room temperature in the dark. A total of 20% glycerol was added
for mounting, and images were captured and observed on a confocal
microscope (Leica Microsystems GmbH). PBS was used as a negative
control. Green and red fluorescence were used as a positive
expression; DAPI was added and stained the nucleus at room
temperature for 30 min.
HSP90α inhibitor application in vivo
and in vitro
Alvespimycin is a common inhibitor of HSP90α that
has been reported in numerous studies (10-12),
and it was used in further experiments in vivo and in
vitro. Briefly, for the mouse model of OP, alvespimycin was
injected intraperitoneally with a dosage of 25 mg/kg five times a
week according to a study by Mellatyar et al (11). For experiments in vitro,
alvespimycin at 20 nM was used to inhibit HSP90α (13,14).
Statistical analysis
All experiments were repeated in triplicate to
ensure the reliability of the results. All data were presented as
the mean ± SD. SPSS 19.0 (IBM Corp.) software package was used for
statistical analysis. Comparisons between two groups were performed
using unpaired t-tests. Multiple groups were analyzed using one-way
analysis of variance and post hoc unpaired Tukey's tests. P<0.05
was considered to indicate a statistically significant
difference.
Results
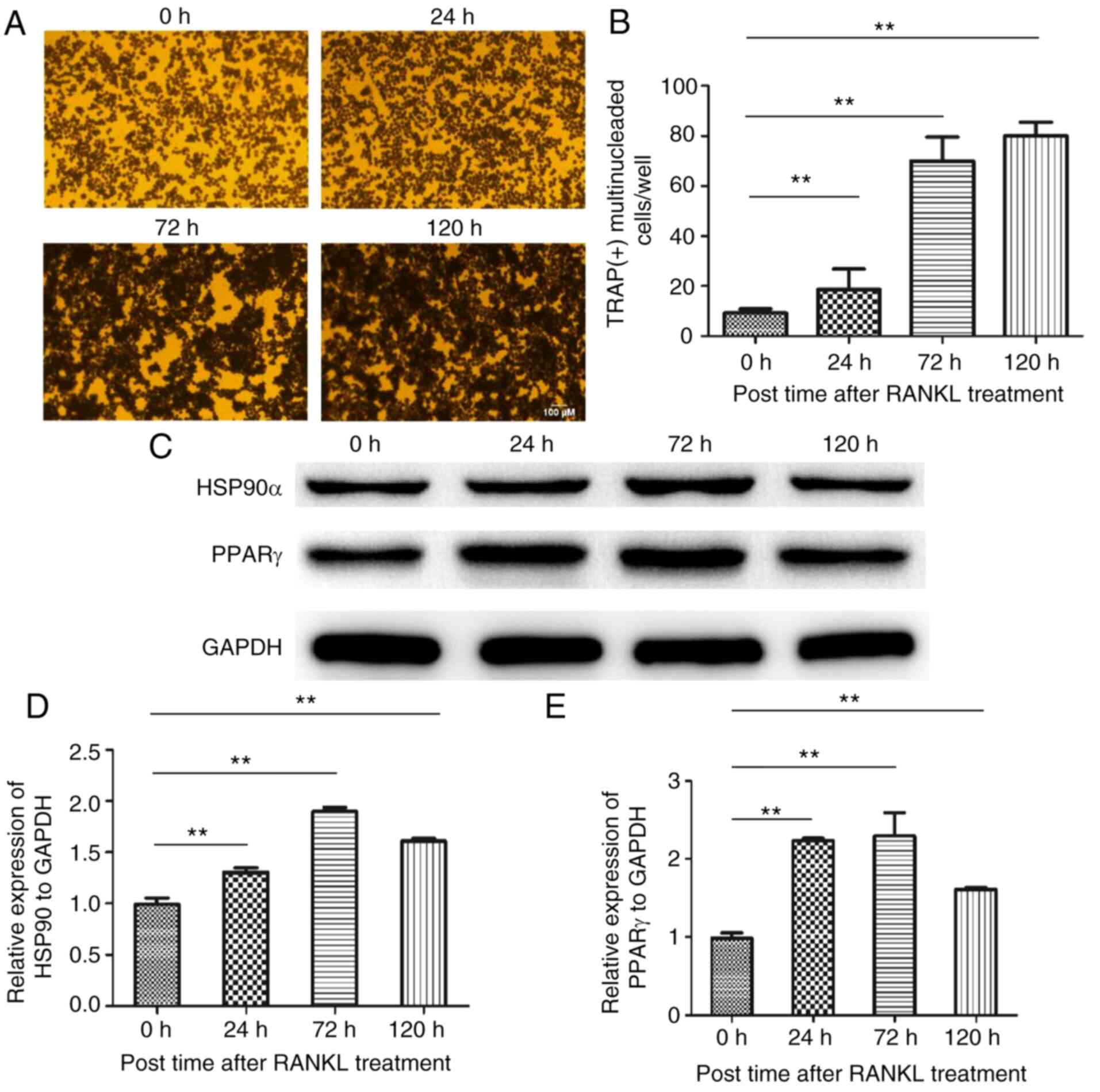
Expression of HSP90α in osteoclast
differentiation
To explore the role of HSP90α in osteoclast
differentiation, western blotting was used to detect the
expression. THP-1 monocytes, which were precursors of osteoclasts,
were cultured for 5 days under the induction of PMA, M-CSF, and
RANKL. All THP-1 monocytes differentiated into TRAP(+) multinuclear
osteoclasts (Fig. 1A and B). Further analysis confirmed that in the
RANKL-induced osteoclast differentiation, the expression level of
HSP90α reached a peak at 72 h, and then gradually decreased. At 120
h, the expression level of HSP90α was decreased compared with the
72-h treatment. Concurrently, the expression level of PPARγ was
increased, and that of HSP90α was also upregulated. (Fig. 1C-E).
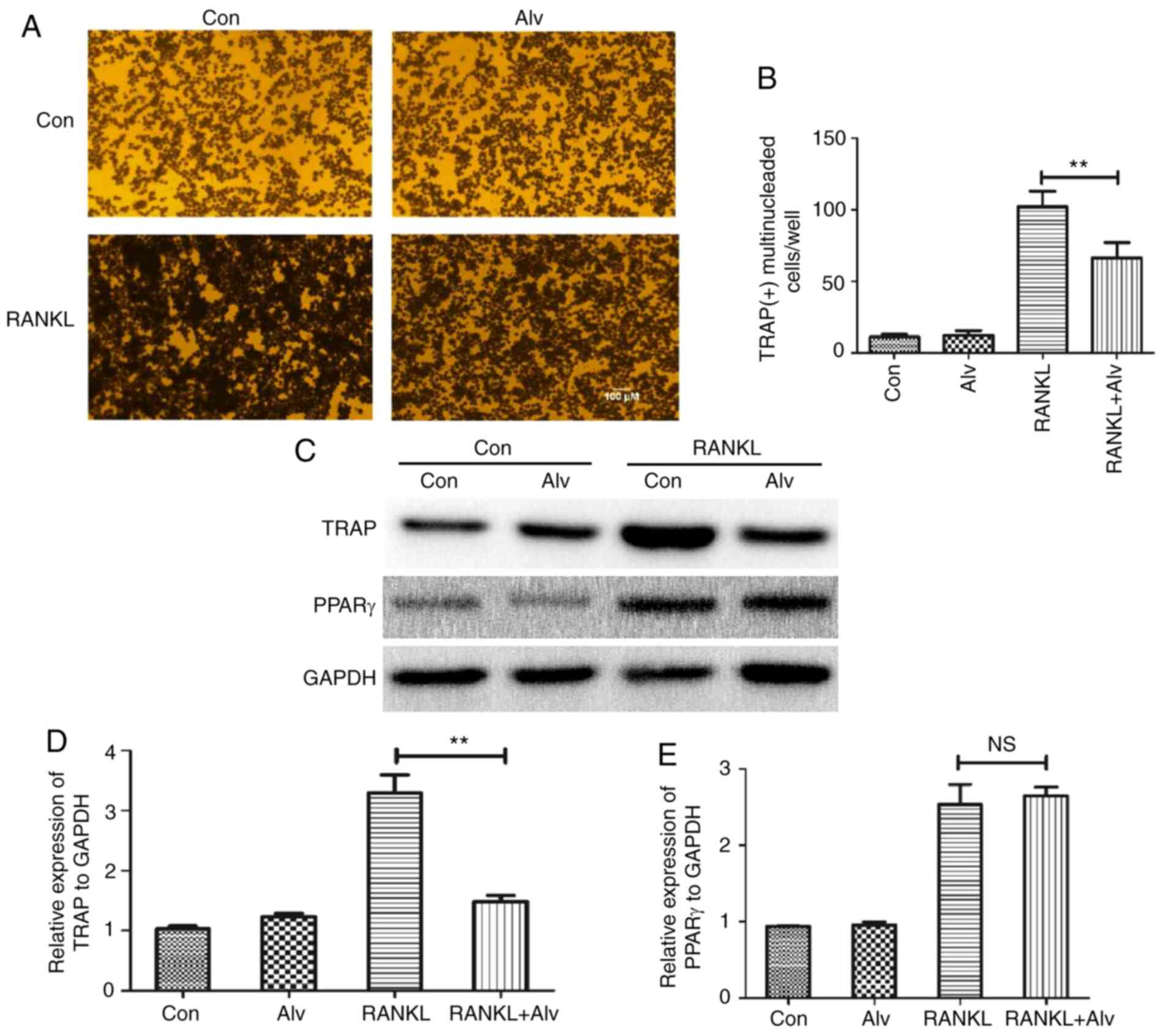
Inhibition of HSP90α can block the
formation and differentiation of osteoclasts
To further confirm the effect of HSP90α on
osteoclast formation and differentiation, alvespimycin was used to
inhibit HSP90α. The results revealed that alvespimycin had no
significant effect on the number of TRAP(+) multinuclear
osteoclasts in the uninduced culture. When induced, alvespimycin
significantly reduced the number of TRAP(+) multinuclear
osteoclasts, indicating that inhibition of HSP90α may inhibit the
activation of osteoclasts (Fig. 2A
and B). In order to further verify
the aforementioned results, the expression levels of TRAP and PPARγ
in THP-1 monocytes were detected by western blotting. The results
were consistent with those of TRAP staining. Alvespimycin
significantly reduced the expression level of TRAP after induction,
but had no significant effect on the expression of PPARγ (Fig. 2C-E). The aforementioned findings
indicated that inhibition of HSP90α could block the formation and
differentiation of osteoclasts, but had no significant effect on
the expression of PPARγ.
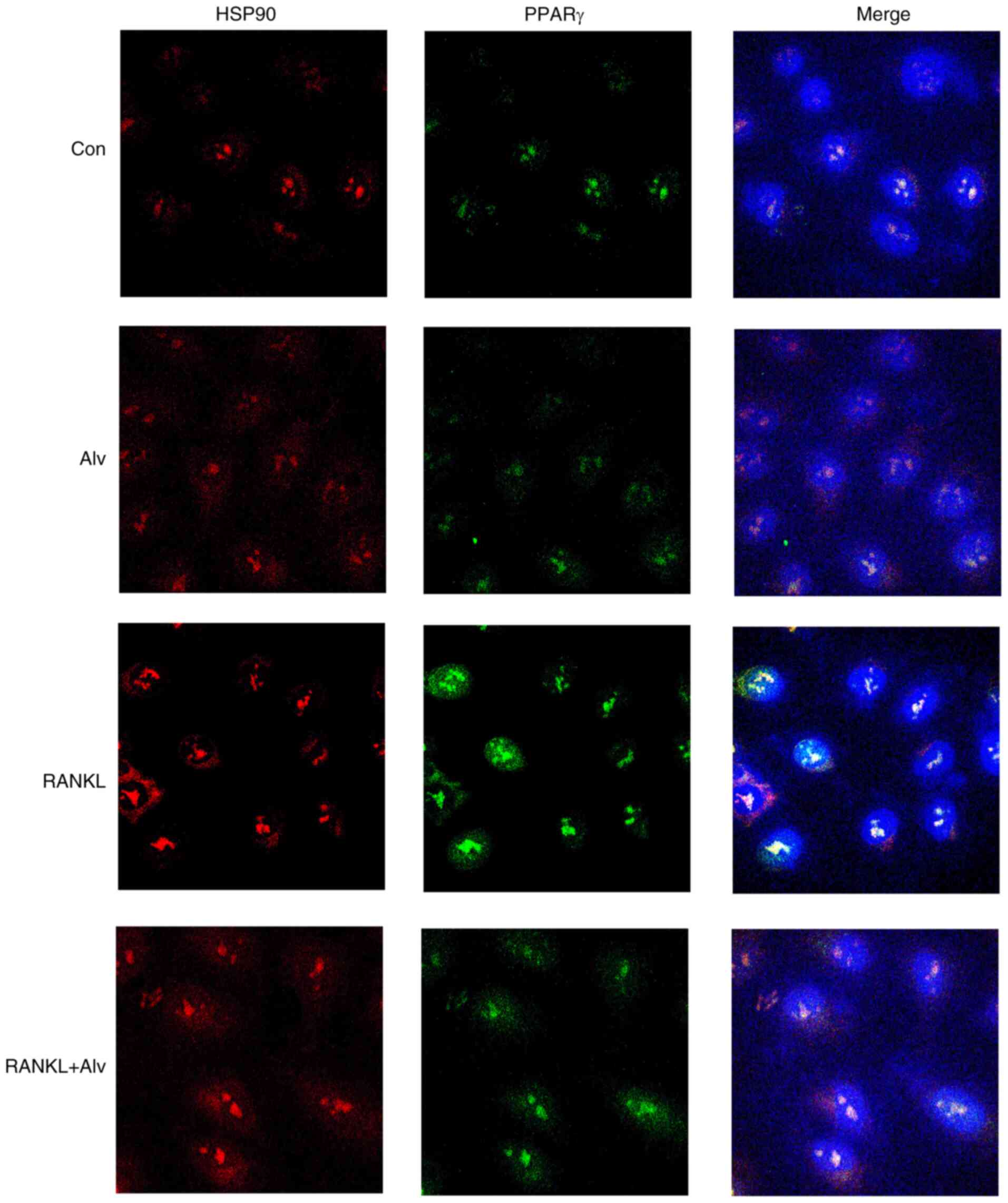
Inhibition of HSP90α can exert an
inhibitory effect on osteoclasts by blocking nuclear import of
PPARγ
Previous experiments revealed that inhibition of
HSP90α significantly inhibited osteoclast differentiation, but had
no effect on PPARγ expression. It has been indicated that PPARγ
enters the nucleus and initiates the transcription of the
corresponding genes (15).
Therefore, it was theorized in the present study that HSP90α may
regulate the differentiation of osteoclasts by affecting the
nuclear import of PPARγ. Thus, a confocal microscope was used to
observe the nuclear import of PPARγ (red) and HSP90α (green) after
RANKL induction. The results demonstrated that after induction by
PMA, M-CSF, and RANKL for 1 h, nuclear import of HSP90α (red) and
PPARγ (green) in THP-1 monocytes was markedly increased. However,
after inhibition of HSP90α by alvespimycin, the nuclear import was
markedly decreased (Fig. 3). These
data indicated that the inhibition of HSP90α exerted an inhibitory
effect on osteoclasts by blocking the nuclear import of PPARγ.
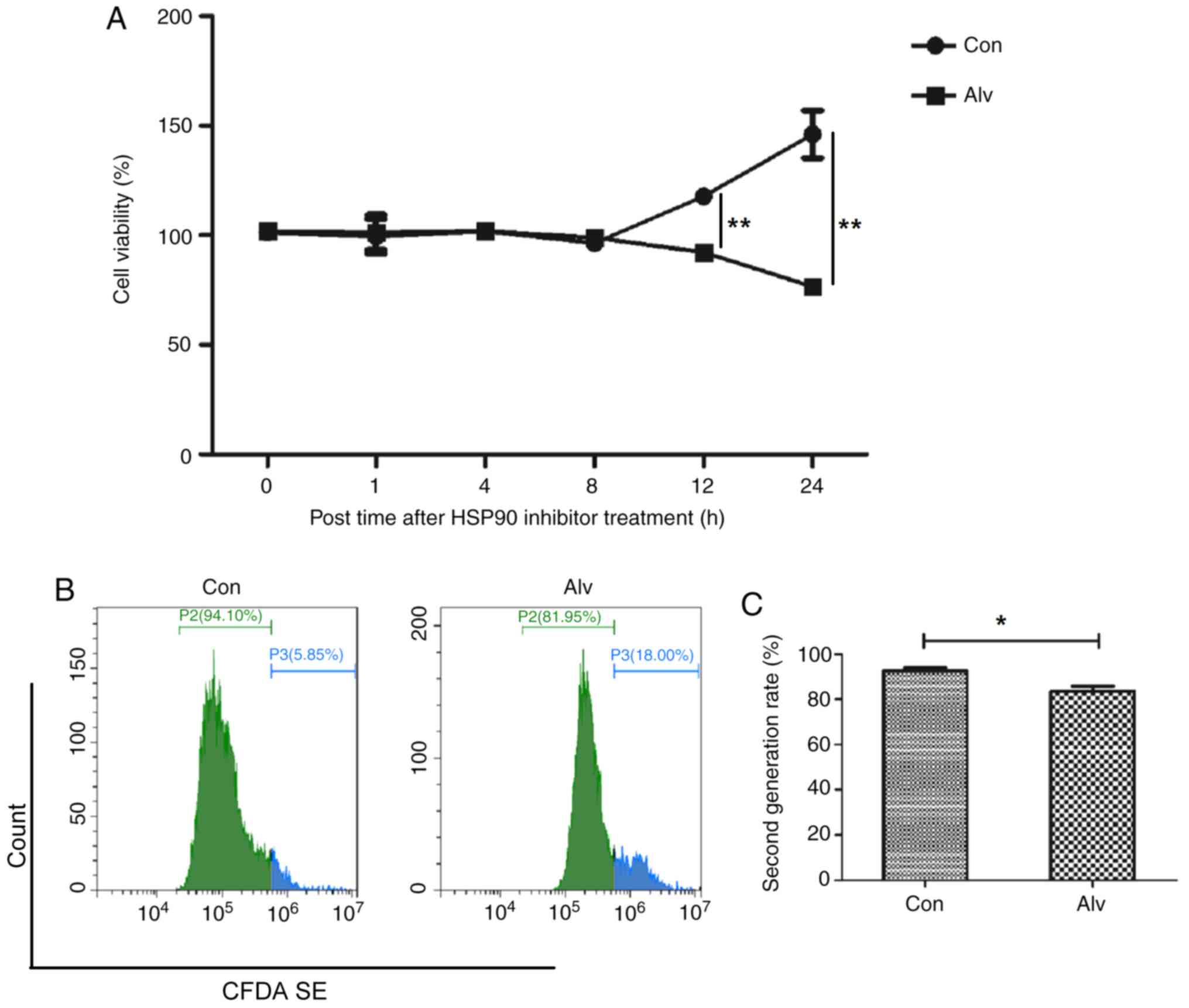
Effect of HSP90α on the biological
proliferative ability of osteoclasts
The aforementioned experiments demonstrated that
inhibition of HSP90α could inhibit osteoclast differentiation, and
the effect of inhibition of HSP90α on the proliferation of
osteoclast precursor monocytes was further observed. THP-1
monocytes were treated with alvespimycin. Results of the CCK-8
assay revealed that THP-1 monocytes had a significant decrease in
viability 12 h after alvespimycin treatment, and continued to
decrease over time (Fig. 4A).
Furthermore, flow cytometric results revealed that after 24 h of
alvespimycin treatment, the proportion of second-generation for
CFDA SE-labeled cells significantly decreased (92.51±1.39 vs.
83.55±2.30), indicating that the proliferation ability was reduced
(Fig. 4B and C). The aforementioned results indicated
that inhibition of HSP90α could block the proliferation of
osteoclast precursor monocytes.
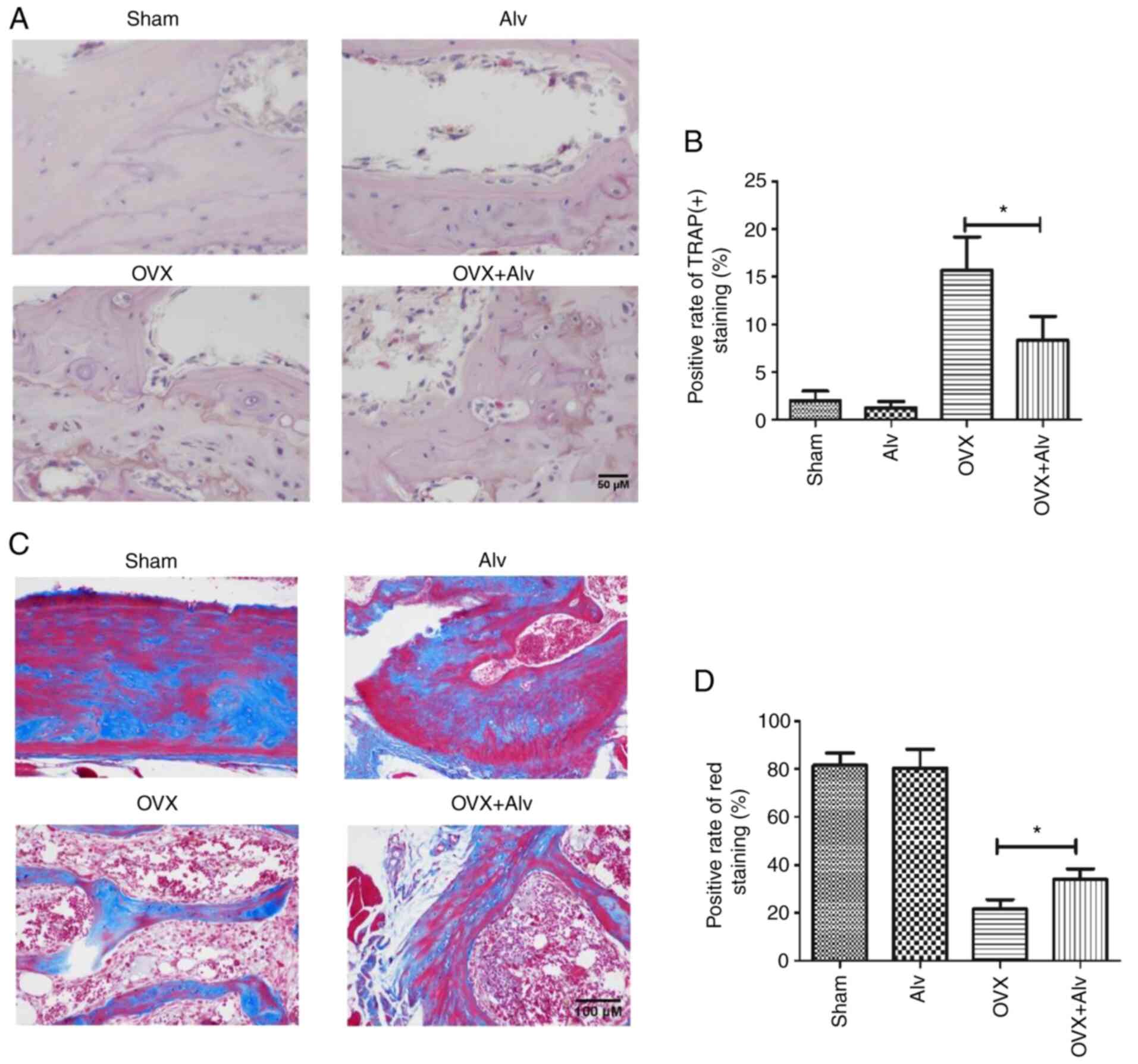
Inhibition of HSP90α can improve OP in
mice
The aforementioned experiments demonstrated that
inhibition of HSP90α could inhibit the differentiation and
proliferation of osteoclasts, which are the bases of OP. Therefore,
the effect of inhibition of HSP90α on OP in mice was further
studied. A mouse model of OP was constructed by removing mouse
ovaries. The results revealed that on day 42 after the ovaries were
removed, the bone mass of mice decreased, the bone cortex became
thinner, the trabecular bone was decreased, deformed and thinned,
and arrangement was disordered. In addition, the bones were porous
and deficient in calcium, which suggested the presence of OP. After
treatment with alvespimycin (25 mg/kg), the bone mass became
thicker and the trabeculae was increased compared with the
sham-operated group. Moreover, a lower TRAP(+) rate in mice was
found in the alvespimycin treatment group (Fig. 5A and B), indicating less osteoclast
differentiation. Concurrently, the results revealed that the red
area of Masson staining of the bone in the model group was
decreased, indicating the reduced bone maturity. After treatment
with alvespimycin, the red area increased, suggesting that
inhibition of HSP90α by alvespimycin can inhibit osteoclast
differentiation, promote bone maturation, thus improving OP
(Fig. 5C and D).
Discussion
Heat shock proteins (HSPs) are potent antioxidative
and anti-inflammatory proteins that protect cells from numerous
acute and chronic stress conditions; HSPs can act as molecular
chaperones that prevent the aggregation of misfolded proteins,
rendering these proteins refolded or removed (16). Studies have revealed that PPARγ can
exert its biological role by regulating a variety of HSPs (such as
HSP90, HSP70, HSP60, and HSP32) (17). Nguyen et al (18) found that the function of HSP90α was
essential for PPARγ transcription output and survival of mature
adipose cells. Moreover, it was revealed that HSP90α can bind to
PPARγ to form an HSP-PPARγ compound, controlling the stability of
PPARγ and cell differentiation (18). Lee et al (19) discovered that binding of ox-LDL to
the receptor CD36 could inhibit HSP expression at the translation
level through PPARγ activation. Moreover, Chen et al
(20) revealed that nifedipine
could inhibit the phosphorylation of p38MAPK, ERK1/2 and HSP90 by
promoting the expression of PPARβ/γ, thereby inhibiting the
production of MMP-2 and CD40L and exerting its anti-AS effect. The
aforementioned studies demonstrated that HSP90α has a pivotal role
in the regulation of PPARγ function, and that PPARγ is an important
driving factor for osteoclast formation and differentiation.
Therefore, in the present study, it was further examined whether
HSP90α could affect the function of osteoclasts, and further
regulate the formation of OP.
Firstly, the expression level of HSP90α during
osteoclast formation and differentiation in vitro was
examined, and it was determined that after induction by PMA, M-CSF,
and RANKL, the number of TRAP(+) multinuclear osteoclasts
differentiated by THP-1 monocytes was gradually increased.
Concurrently, the expression level of HSP90α was also gradually
increased. These results indicated that there may be an association
between the two. On this basis, HSP90α inhibitor alvespimycin was
used to intervene on THP-1 monocytes, and the results revealed that
alvespimycin could significantly inhibit the differentiation of
THP-1 monocytes into osteoclasts, suggesting that the inhibition
HSP90α was crucial in the differentiation and formation of
osteoclasts. Nevertheless, the inhibition of HSP90α had no
significant effect on PPARγ expression, suggesting a synergistic
relationship between HSP90α and PPARγ. These data were consistent
with Kim et al (17), who
revealed that ligand of PPARγ did not immediately activate the
downstream pathway after binding to PPARγ, but instead, it bound to
HSP90α before exerting its biological effect. However, the
underlying molecular mechanism remained unclear. Considering that
PPARγ has the function of nuclear transcription, its function is
based on binding to response elements. In the present study, it was
theorized that the binding of HSP90α to PPARγ may promote its
transcription into the nucleus. Thus, the nuclear import of PPARγ
was observed with a fluorescence microscope, revealing that nuclear
import of PPARγ was increased after THP-1 monocytes were induced,
and the nuclear import was decreased after inhibition of HSP90α.
This indicated that the inhibition of HSP90α led to the blocking of
osteoclast differentiation, which was achieved by inhibiting the
nuclear import of PPARγ.
Finally, the effects of HSP90α inhibition on OP in
mice were further verified through in vivo experiments.
First, a mouse model of OP was established by ovariectomy, after
which it was used to verify the role of HSP90α. The obtained
results revealed that alvespimycin could reduce TRAP staining of
bone and relieve OP in mice.
The present study also has a few limitations.
Firstly, the specific binding between HSP90α and PPARγ was not
analyzed. Secondly, the effects of other HSPs were not explored.
Thus, additional studies are required to confirm these
findings.
To sum up, this is the first study, to the best of
our knowledge, that reported how inhibition of HSP90α could block
PPARγ and in turn inhibit differentiation in mice osteoclasts. It
is considered that inhibition of HSP90α can block the nuclear
import of PPARγ, which in turn affects the transcription of
corresponding downstream molecules, thereby inhibiting osteoclast
differentiation. The aforementioned findings can provide a new
direction for the treatment of OP, using HSP90α as a target and
then interfering with the formation of osteoclasts may be the
direction of future research.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
JW and YG designed the study. JM, CY and KZ
performed the experiments. CW performed the histological
examination. XL performed the confocal microscopy analysis. JM, CY
and HZ wrote the paper. HZ and CY analyzed and interpreted the
data. JW and YG confirm the authenticity of all the raw data. All
authors read and approved the final manuscript.
Ethics approval and consent to
participate
All animal experiments were undertaken in accordance
with the National Institute of Health ‘Guide for the Care and Use
of Laboratory Animals’ (NIH Publication no. 85-23, National Academy
Press, Washington, DC, revised 1996), with the approval of the
Laboratory Animal Center and Ethics Committee of Changzheng
Hospital (approval ID: #CZ2019112130).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Shanb AA and Youssef EF: The impact of
adding weight-bearing exercise versus nonweight bearing programs to
the medical treatment of elderly patients with osteoporosis. J
Family Community Med. 21:176–181. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Lin J, Zhu J, Wang Y, Zhang N, Gober HJ,
Qiu X, Li D and Wang L: Chinese single herbs and active ingredients
for postmenopausal osteoporosis: From preclinical evidence to
action mechanism. Biosci Trends. 11:496–506. 2017.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Hamdy NA: Targeting the RANK/RANKL/OPG
signaling pathway: A novel approach in the management of
osteoporosis. Curr Opin Investig Drugs. 8:299–303. 2007.PubMed/NCBI
|
|
4
|
Li X, Jie Q, Zhang H, Zhao Y, Lin Y, Du J,
Shi J, Wang L, Guo K, Li Y, et al: Disturbed MEK/ERK signaling
increases osteoclast activity via the Hedgehog-Gli pathway in
postmenopausal osteoporosis. Prog Biophys Mol Biol. 122:101–111.
2016.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Granéli C, Karlsson C, Brisby H, Lindahl A
and Thomsen P: The effects of PPAR-γ inhibition on gene expression
and the progression of induced osteogenic differentiation of human
mesenchymal stem cells. Connect Tissue Res. 55:262–274.
2014.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wang J, Wang G, Gong L, Sun G, Shi B, Bao
H and Duan Y: Isopsoralen regulates PPAR-γ/WNT to inhibit oxidative
stress in osteoporosis. Mol Med Rep. 17:1125–1131. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Wan Y, Chong LW and Evans RM: PPAR-gamma
regulates osteoclastogenesis in mice. Nat Med. 13:1496–1503.
2007.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Rossi A, Kapahi P, Natoli G, Takahashi T,
Chen Y, Karin M and Santoro MG: Anti-inflammatory cyclopentenone
prostaglandins are direct inhibitors of IkappaB kinase. Nature.
403:103–108. 2000.PubMed/NCBI View
Article : Google Scholar
|
|
9
|
Zhang M, Qian C, Zheng ZG, Qian F, Wang Y,
Thu PM, Zhang X, Zhou Y, Tu L, Liu Q, et al: Jujuboside A promotes
Aβ clearance and ameliorates cognitive deficiency in Alzheimer's
disease through activating Axl/HSP90/PPARγ pathway. Theranostics.
8:4262–4278. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hu Y, Bobb D, He J, Hill DA and Dome JS:
The HSP90 inhibitor alvespimycin enhances the potency of telomerase
inhibition by imetelstat in human osteosarcoma. Cancer Biol Ther.
16:949–957. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Mellatyar H, Talaei S,
Pilehvar-Soltanahmadi Y, Barzegar A, Akbarzadeh A, Shahabi A,
Barekati-Mowahed M and Zarghami N: Targeted cancer therapy through
17-DMAG as an Hsp90 inhibitor: Overview and current state of the
art. Biomed Pharmacother. 102:608–617. 2018.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Tsai YC, Leu SY, Chen SY, Kung CW, Lee YM,
Liu YP, Yen MH and Cheng PY: 17-DMAG, an Hsp90 inhibitor,
ameliorates ovariectomy-induced obesity in rats. Life Sci.
232(116672)2019.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Hertlein E, Wagner AJ, Jones J, Lin TS,
Maddocks KJ, Towns WH III, Goettl VM, Zhang X, Jarjoura D, Raymond
CA, et al: 17-DMAG targets the nuclear factor-kappaB family of
proteins to induce apoptosis in chronic lymphocytic leukemia:
Clinical implications of HSP90 inhibition. Blood. 116:45–53.
2010.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Ge J, Normant E, Porter JR, Ali JA,
Dembski MS, Gao Y, Georges AT, Grenier L, Pak RH, Patterson J, et
al: Design, synthesis, and biological evaluation of hydroquinone
derivatives of 17-amino-17-demethoxygeldanamycin as potent,
water-soluble inhibitors of Hsp90. J Med Chem. 49:4606–4615.
2006.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wu JS, Tsai HD, Cheung WM, Hsu CY and Lin
TN: PPAR-γ ameliorates neuronal apoptosis and ischemic brain injury
via suppressing NF-κB-driven p22phox transcription. Mol Neurobiol.
53:3626–3645. 2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Zininga T, Ramatsui L and Shonhai A: Heat
shock proteins as immunomodulants. Molecules.
23(2846)2018.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Kim HJ, Hwang NR and Lee KJ: Heat shock
responses for understanding diseases of protein denaturation. Mol
Cells. 23:123–131. 2007.PubMed/NCBI
|
|
18
|
Nguyen MT, Csermely P and Sőti C: Hsp90
chaperones PPARγ and regulates differentiation and survival of
3T3-L1 adipocytes. Cell Death Differ. 20:1654–1663. 2013.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Lee KJ, Ha ES, Kim MK, Lee SH, Suh JS, Lee
SH, Park KH, Park JH, Kim DJ, Kang D, et al: CD36 signaling
inhibits the translation of heat shock protein 70 induced by
oxidized low density lipoprotein through activation of peroxisome
proliferators-activated receptor gamma. Exp Mol Med. 40:658–668.
2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Chen TH, Shih CY, Hsu WL and Chou TC:
Mechanisms of nifedipine-downregulated CD40L/sCD40L signaling in
collagen stimulated human platelets. PLoS One.
10(0127054)2015.PubMed/NCBI View Article : Google Scholar
|