Introduction
Mucinous epithelial tumors originating from the
kidney are uncommon. Most reported cases are malignant. Only
isolated cases have been reported where the epithelial lining of
the tumor was benign (1). In the
present study, clinical data of a patient with mucinous cystadenoma
of the kidney, who was treated at hospital in December 2011, were
analyzed. A literature review was also performed to investigate
clinicopathologic features and treatment of mucinous cystadenoma of
the kidney.
Case report
A 52-year-old Chinese woman complained of angina
capitis, dizziness, and intermittent pain in the abdomen for 6 days
with a 3-year history of hypertension. Physical examination on
admission showed nothing remarkable. The laboratory data revealed
normal values. From abdominal computed tomography, an ovoid cyst
with a diameter of 12 cm and partial calcifications in the cystic
wall was found in the inferior pole of the left kidney.
Additionally, two cystic masses measuring 2 cm in diameter were
found in the left kidney. The patient was clinically diagnosed with
multiple renal cysts with thickened walls and hemorrhage. An
intravenous pyelogram revealed that a large, round, high-density
mass had developed in the left kidney and part of the pelvic area,
and the calyx was absent. A diagnosis of a large cyst in the left
kidney and left hydronephrosis was made. No relevant past
interventions were implemented.
The diameter of the renal cysts of the present case
reached 12 cm. The patient was clinically diagnosed with multiple
renal cysts with thickened walls and bleeding. As the disease has
malignant potential, a left nephrectomy was performed. Only a few
ml of cyst material were obtained during the surgery via renal cyst
aspiration to determine cyst fluid characteristics. Due to the
mucinous and characteristic thickness of the cyst content, it was
impossible to empty the cyst.
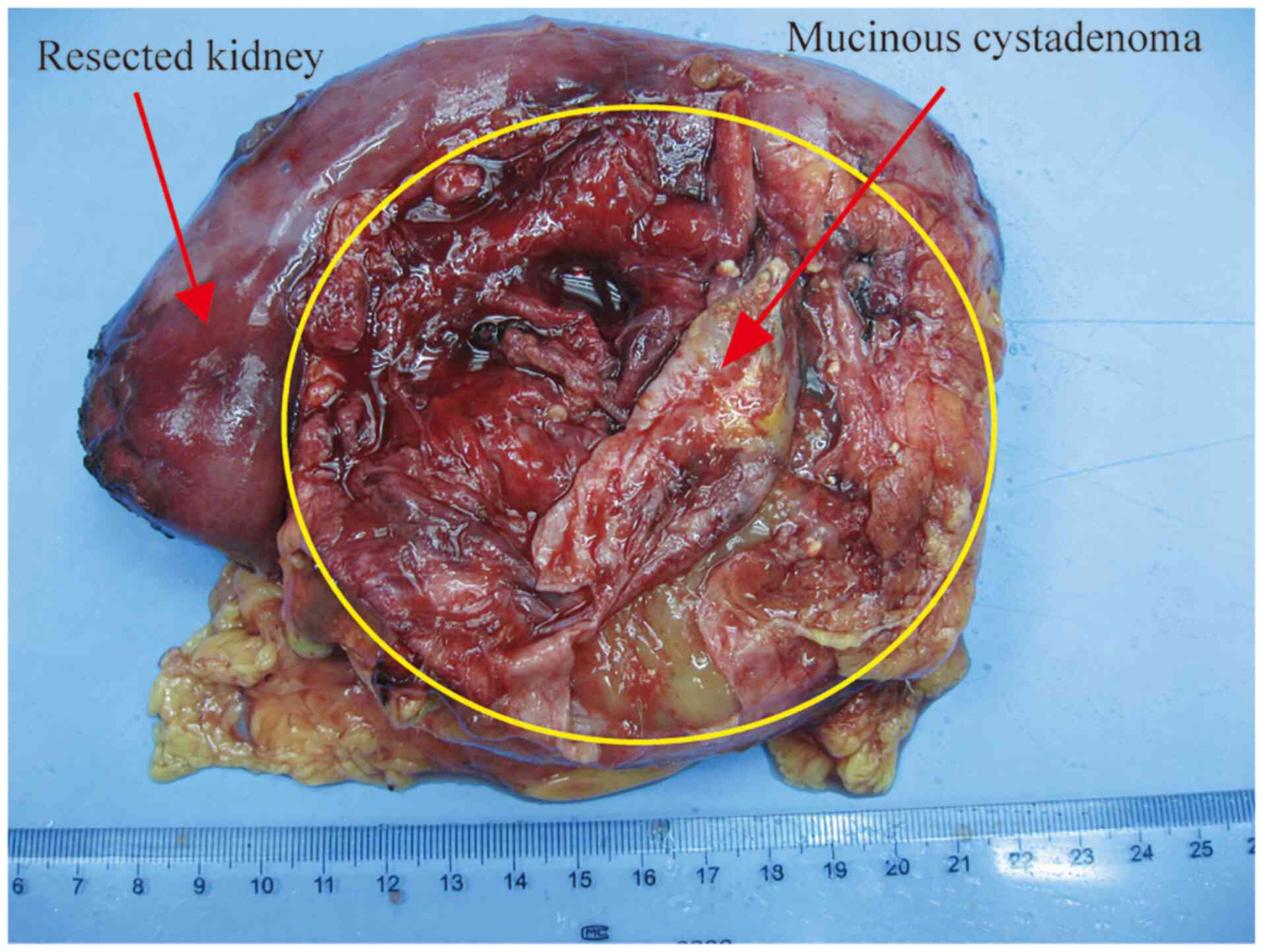
From sections, the specimen showed grossly distorted
kidney and pelvic tissue. The well-defined monolocular cyst was 10
cm in diameter, filled with gray-red jelly mucin, and located in
the upper half of the kidney (Fig.
1). The inner surface was gray-white, turbid, and rather smooth
and showed no apparent tumorous protrusion. The cyst wall was
0.2-0.4 cm thick and accompanied by focal calcifications, which led
to pressure atrophy of the renal parenchyma. The border between the
cyst and residual renal pelvis formed a firm septum without any
communication through the lumen. In addition, two cysts of 2 cm in
diameter were located around the large cyst and contained dark red
liquid. The two cysts were 0.2 cm thick. No calculi had formed in
the urinary tracts or cyst. No tumorous lesion was found in the
renal parenchyma.
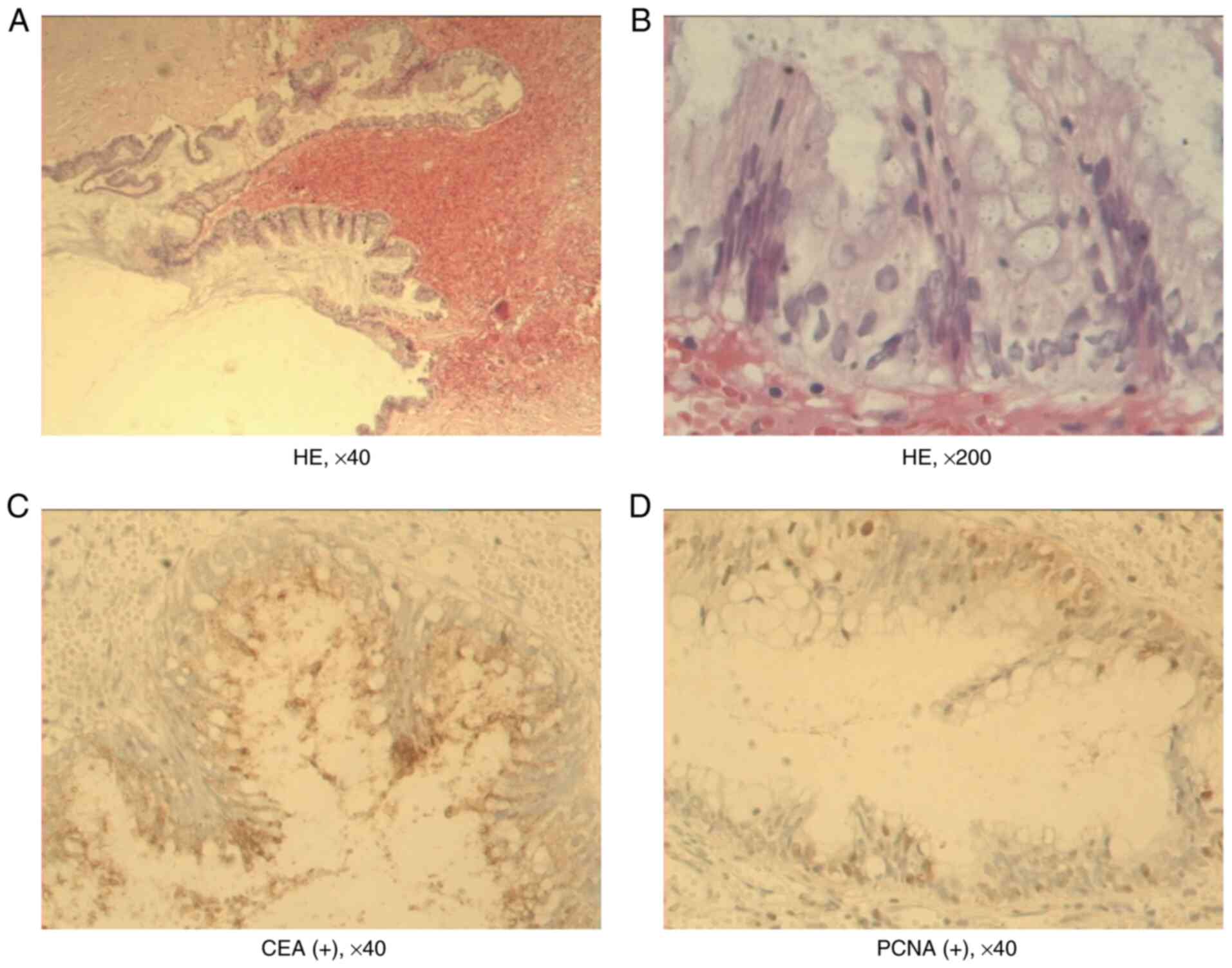
Histopathological examination of the renal cyst wall
revealed that it consisted of mucinous epithelium with supporting
fibrous tissue. The epithelial lining was characterized by a single
layer of columnar cells with sporadic papillary proliferation
(Fig. 2A and B). Atrophy in the renal glomerulus and
tubules were found in some sections of the cyst wall. Therefore,
the cyst was believed to arise from the renal parenchyma, not from
the collecting system. Histopathology was consistent with mucinous
cystadenoma. Immunohistochemistry showed that the cyst was positive
for carcinoembryonic antigen (CEA) and proliferating cell nuclear
antigen (PCNA; Fig. 2C and
D). No adverse and unanticipated
events occurred following surgery. Furthermore, an 8-year follow-up
was conducted. The patient was well, and there was no evidence of
recurrence or metastasis. The patient was highly satisfied with the
treatment methods and outcomes.
Literature review
To further elucidate the clinical manifestations,
treatment, and prognosis of mucinous cystadenoma of the kidney, a
literature review was conducted. The search was performed using
PubMed and the following search terms were used in all databases:
((kidney[Title/Abstract]) OR (renal[Title/Abstract])) AND
(cystadenoma[Title/Abstract]). A total of five articles were
retrieved, including six cases with mucinous cystadenoma of the
kidney. A total of six cases (1-5)
were reported between 1985 and 2015 (Table I). of these, two cases arose from
the left kidney, three cases from the horseshoe kidney, and one
case from the right kidney. The onset age was between 27 and 69
years. Three cysts occurred in female patients and three in male
patients. Of these six patients, three patients with capillary
mucinous cystadenomas had horseshoe kidney deformities, suggesting
that renal tumor may be associated with congenital kidney
malformation.
 | Table IReview of reported mucinous
cystadenoma of the kidney. |
Table I
Review of reported mucinous
cystadenoma of the kidney.
| Author, year | Sex | Age, years | Chief complaint | Tumor size, cm | Treatment | Pathology | Follow-up time | Recurrence | (Refs.) |
|---|
| Gangane et al,
2008 | Female | 35 | Intermittent pain in
the abdomen | NA | Left nephrectomy | Mucinous
cystadenoma | 2 years | None | (2) |
| Gangane et al,
2008 | Male | 65 | Pain in the abdomen,
weakness and loss of appetite | NA | Left nephrectomy | Mucinous cystadenoma
arising from the renal pelvis with changes of pyonephrosis | 8 months | None | (2) |
| Toyoda et al,
1997 | Male | 69 | Dysuria | 5 | Right
nephrectomy | Mucinous cystadenoma
with malignant transformation | 2 years | None | (5) |
| Ross and D'Amato,
1985 | Female | 59 | Experiencing
abdominal pain | 7 | Partial
nephrectomy | Horseshoe kidney,
mucous papillary cystadenoma of renal pelvis | 4 years | None | (4) |
| Akan et al,
2005 | Female | 27 | A mass was
incidentally discovered in the right upper quadrant of the
abdomen | 12 | Cyst excision | Horseshoe kidney,
mucinous cystadenoma | 22 months | None | (1) |
| Mitome et al,
2015 | Male | 45 | A palpable mass in
the left abdomen | 15 | Cyst excision | Horseshoe kidney,
mucinous cystadenoma | 6 months | None | (3) |
Discussion
Benign primary mucinous cystadenomas arising from
the kidney are extremely rare. Some reports have shown that
mucinous cystadenomas originate from the renal pelvis (2,4,5),
while others have shown that they originate from the renal
parenchyma (1). In the present
case, the cyst originated from the renal parenchyma. The cause of
this disease remains elusive. Mardi et al (6) suggest the possibility that an
adenoma-carcinoma sequence might exist among glandular neoplasms
arising from the renal pelvis. According to this theory, the normal
transitional epithelium may undergo squamous, columnar, or cuboidal
metaplasia. Gangane et al (2) hypothesize that mucinous metaplasia
may progress into an adenoma, which may then progress into a
malignancy. Kobayashi et al (7) hypothesize that kidney calculi and
long-standing chronic infection are possible etiologic factors for
mucinous adenocarcinoma of the pelvis. However, not all patients
with mucinous cystadenoma arising from the kidney had such
preceding lesions. It is unclear whether the same hypothesis also
holds for adenomas. A total of six cases (1-5)
were reported, of which three patients with capillary mucinous
cystadenomas had horseshoe kidney deformities, which suggests that
the renal tumor may be associated with congenital kidney
malformation. Patients suffering from mucinous epithelial tumors
originating from the kidney have no specific symptoms, and most of
these tumors are found from physical examination or based on
nonspecific symptoms, such as abdominal pain, abdominal lumps, and
other symptoms. Generally, the tumor is 5-12 cm across. In the
present case, the patient presented with headache and dizziness,
and a tumor of 10 cm across was found.
Renal mucinous cystadenoma is a tumor with a single
capsule or polycystic tumor containing jelly-like mucus. During
microscopic examinations, a single-layered tall columnar mucinous
epithelium, and occasionally papillary hyperplasia, can be found
inside the wall. Thus, it is not difficult to make a pathological
diagnosis. Toyoda et al (5)
hypothesize that the diagnosis of malignant/benign tumors can be
safely made only if the following criteria are met: i) Macroscopic
or microscopic evidence of invasion, metastasis, or recurrence; and
ii) histological evidence of structural or cellular atypia.
However, despite the first criterion, the second criterion alone is
the basis for judgments. This case does not meet the above criteria
for histology; thus, the diagnosis was a benign tumor. The patient
was symptom-free during the 8-year follow-up period. Ross and
D'Amato (4) hypothesize that renal
mucinous cystadenoma and ovarian mucinous cystadenoma tissues are
very similar, suggesting that the criteria used to evaluate
mucinous ovarian tumors might also be appropriate for mucinous
tumors of the kidney.
As for renal mucinous cystadenoma treatment, Akan
et al (1) suggest that as
there are no definite diagnostic radiological criteria, the
treatment should be individualized. If a malignancy is suspected,
partial nephrectomy or cyst excision can be performed. Benign cysts
found from imaging can be followed. Fine-needle aspiration biopsy
should be performed first if the cyst compresses the collecting
system. If needle aspiration fails due to viscous cyst content or
if the malignant material is extracted, surgical specimens can be
obtained through the operation. Thus, a diagnosis can be made and
confirmed. Kawahara et al (8) present a case of primary renal
carcinoid coexistent with a mucinous cystadenomatous element.
Takashi et al (9) report
the first documented composite tumor of mucinous cystadenoma and
somatostatinoma of the kidney. They consider that excision is the
first choice for treatment due to the unspecific clinical
manifestations of this case, difficult preoperative qualitative
diagnosis, and probable malignant tumor. Moreover, the cyst wall is
thin and may rupture during surgery. Total nephrectomy is advocated
to prevent metastasis. Of the six cases reported, three cases were
treated with nephrectomy, one case with partial renal nephrectomy,
and two cases with cyst excision. Complete laparoscopic resection
is not advocated as it may be difficult to separate the tumor, or
the tumor may rupture due to adhesions between renal mucinous
papillary cystadenoma and the surrounding tissue. Therefore, manual
laparoscopic nephrectomy or open nephrectomy is recommended.
Although the cyst is possibly malignant in this case, the patient
was in good condition 8 years after surgery, and there was no
evidence of recurrence or metastasis. The patient was highly
satisfied with the treatment methods and outcomes. The limitation
of the present study is that there is no corresponding
pre-operative imaging data.
In the present study, a case of mucinous cystadenoma
of the kidney was reported. The left nephrectomy was performed and
no tumor recurrence was found during the 8-year follow-up. The
clinical feature of this disease was atypical, making clinical
diagnosis difficult. Histopathological examination revealed that
the cyst consisted of mucinous epithelium with supporting fibrous
tissue and immunohistochemistry showed that the cyst was positive
for CEA and PCNA. The main treatment is partial nephrectomy or
nephrectomy, and postoperative follow-up is required. Patients with
this disease had an improved prognosis.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LX and YZ designed the study and revised the
manuscript. JL and QZ collected and analyzed the patient data. YZ
contributed to data extraction, quality assessment and drafted the
paper. QZ and JL checked and confirm the authenticity of the raw
data. All authors contributed to the manuscript and all authors
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
The patient gave written informed consent for the
publication of this case report and all accompanying images.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Akan H, Dalva I, Yildiz O, Kutluay L,
Gündoğdu S and Güngen Y: Mucinous cystadenoma mimicking simple
renal parenchymal cyst in a horseshoe kidney. Int J Urol.
12:493–496. 2005.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Gangane N, Anshu Shende N and Sharma SM:
Mucinous cystadenoma arising from renal pelvis: A report of 2
cases. Urol J. 5:197–199. 2008.PubMed/NCBI
|
|
3
|
Mitome T, Yao M, Udaka N, Fusayasu S,
Izumi K, Osaka K, Hayashi N, Nakaigawa N, Nagashima Y and Kubota Y:
Mucinous cystadenoma of a horseshoe kidney: A case report and
literature review. Can Urol Assoc J. 9:E30–E32. 2015.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Ross DG and D'Amato NA: Papillary mucinous
cystadenoma of probable renal pelvic origin in a horseshoe kidney.
Arch Pathol Lab Med. 109:954–955. 1985.PubMed/NCBI
|
|
5
|
Toyoda H, Mabuchi T and Fukuda K: Mucinous
cystadenoma with malignant transformation arising in the renal
pelvis. Pathol Int. 47:174–178. 1997.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Mardi K, Sharma J and Mahajan P: Mucinous
cystadenoma of the renal pelvis with malignant transformation: A
case report. Indian J Pathol Microbiol. 49:595–596. 2006.PubMed/NCBI
|
|
7
|
Kobayashi S, Ohmori M, Akaeda T, Ohmori H
and Miyaji Y: Primary adenocarcinoma of the renal pelvis. Report of
two cases and brief review of literature. Acta Pathol Jpn.
33:589–597. 1983.PubMed/NCBI
|
|
8
|
Kawahara T, Nagashima Y and Misaki H:
Primary renal carcinoid tumor with a mucinous cystadenoma element.
Int J Urol. 16:920–921. 2009.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Takashi M, Matsuyama M, Furuhashi K,
Kodama Y, Shinzato M, Shamoto M and Nakashima N: Composite tumor of
mucinous cystadenoma and somatostatinoma of the kidney. Int J Urol.
10:603–606. 2003.PubMed/NCBI View Article : Google Scholar
|