Introduction
Hereditary hemorrhagic telangiectasia (HHT), also
known as Osler-Rendu-Weber disease, is an autosomal dominant
vascular disorder with an estimated prevalence of 1 in 5.000-8.000
(1,2). The disease is characterized by
mucocutaneous telangiectasias and visceral arteriovenous
malformations (AVMs). The aberrant vessels lack a capillary bed
with direct shunting between arteries and veins (1-3).
Pulmonary manifestations can cause severe cardiac output failures
and pulmonary hypertension (1-3).
However, the most common clinical symptoms are recurrent epistaxis,
which affects up to 95% of all HHT patients (3) Extreme cases can necessitate
hospitalization for blood transfusions (4-6).
Genetically, HHT is caused by mutations encoding the
ENG, ACVRL1 and Smad4 proteins. These proteins
are part of the TGF-ß1/BMP signaling pathway (7,8),
which regulates cell differentiation and angiogenesis in
endothelial cells (9).
In HHT, the TGF-ß1 signaling pathway is impaired and
the activation phase of angiogenesis tends to persist with enhanced
production of angiogenetic factors. Vascular endothelial growth
factor (VEGF) is a well-characterized pro-angiogenic cytokine that
plays a key role in angiogenesis (10,11).
Previous studies have shown significantly increased plasma
concentrations and high tissue expression levels of VEGF in HHT
patients compared with healthy controls (12), thus indicating its important role
in HHT angiodysplasia (13). Based
on these findings, Han et al (14) showed in an animal study that
blocking VEGF in ACVRL1-deficient mice with a VEGF antibody (G6-31)
prevented AVMs and even normalized already-established AVMs.
Bevacizumab is a selective recombinant human
antibody against VEGF that was designed to inhibit tumor-induced
neo-angiogenesis. It has been approved by the FDA for the treatment
of metastatic colorectal, breast, renal cell, and NSC lung cancers,
as well as glioblastomas (www.accessdata.fda.gov/drugsatfda_docs/label/2011/125085s225lbl.pdf)
(15,16). Its first off-label use was
described in Ophthalmology for the treatment of intraocular
neovascular disorders (17).
Recently, an increasing number of studies have
focused on the clinical use of Bevacizumab in HHT patients. Its
intravenous application has shown a positive effect on HHT symptoms
by reducing gastrointestinal bleeding, hepatic complications, and
epistaxis (18-20).
However, the majority of the study protocols used oncological
dosing parameters with severe systemic side effects, e.g.
hypertension, proteinuria, thromboembolic events, intestinal
perforation, and impaired wound healing (21).
To avoid these systemic side effects, current
studies have applied Bevacizumab locally in the form of nasal
sprays or submucosal injections within the nose (22,23).
Regarding the efficacy of topically applied Bevacizumab, submucosal
intranasal Bevacizumab injections seemed to have a more beneficial
effect on HHT-related epistaxis than when it was applied as a nasal
spray (24-30).
However, the main drawbacks in most of these studies are the
different dosing levels of Bevacizumab. They are very inconsistent
and vary considerably between 3.75 and 25 mg/ml, without providing
any details concerning their efficacy or even toxicity to the nasal
mucosa. Additionally, the majority of the studies do not suggest
optimal dosing guidelines for the safe and effective application of
submucosal Bevacizumab injections. Ongoing clinical and research
studies are required to define these treatment details.
The aim of this in vitro study was to
determine the efficacy and the potential toxicity level of
intranasally applied submucosal Bevacizumab injections on
endothelial cell proliferation and VEGF concentration levels in HHT
patients and healthy controls, thus allowing possible conclusions
on the optimal dosage for submucosal nasal Bevacizumab injections
in clinical practice.
Materials and methods
Tissue collection, and culturing of
HHT endothelial cells and HUVECs
Tissue samples were obtained intraoperatively from 3
HHT patients (2 female patients aged 62 and 63 years, 1 male
patient aged 67 years, altogether with an average age of 64 years)
undergoing treatment for recurrent epistaxis at the Department of
ORL-HNS of the University Hospital Mannheim (Heidelberg, Germany).
Prior to surgery, written consent to take a small tissue sample
from their telangiectatic nasal mucosa of the inferior turbinate
was obtained from all patients. This study was approved by the
Ethics Committee of the University Hospital of Mannheim (approval
no. 0238.2). The ethics approval included the use of tissue samples
taken endonasally from HHT patients to culture endothelial cells. A
commercially available HUVEC cell line served as a control.
Endothelial cells from a HUVEC cell line (PromoCell;
cat. no. C-12250) were used as the control. According to the
manufacturer's protocol, all collected nasal tissue samples were
cleaned in PBS. The samples were then cut into 0.5x0.5 cm small
pieces and incubated with 1.5 ml fibroblast growth medium (DMEM
high glucose, 200 mmol L-Glutamine, 10% fetal calf serum, 1%
Penicillin-Streptomycin solution) for 1-2 weeks. Once per week, the
medium was replaced. As soon as the T25 tissue culture flasks were
filled with endothelial cells, the cells were passaged with a
trypsin solution and 0.02% EDTA. On reaching 80% subconfluency, the
endothelial cells were once again passaged with a trypsin solution
and 0.02% EDTA. All endothelial cell cultures were used at passage
3.
Alamar Blue® Assay
Following incubation with different concentrations
of Bevacizumab (0, 2, 4, 6, 8 or 10 mg/ml), cell proliferation was
measured using an AlamarBlue® Assay (BIOZOL Diagnostica)
after 24, 48 or 72 h in fluorescence units (FU). The assay used the
cell-permeable fluorescent indicator dye resazurin as an
oxidation-reduction indicator. As the color and the intensity of
fluorescence changed proportional to the metabolic activity, the
assay indicated the degree of cell proliferation. The measurements
were repeated three times and the mean value was subsequently
calculated.
VEGF-ELISA
The exact concentration of VEGF was evaluated using
the VEGF-Immunoassay from Bio-Techne (R&D Systems, cat. no DVE
00). The assay employed the quantitative sandwich enzyme
immunoassay technique. A monoclonal antibody specific to VEGF was
pre-coated onto a microplate. According to the manufacturer's
instructions, standards and samples were pipetted into wells and
the present VEGF was bound by the immobilized antibody. The unbound
substances were then washed out, and an enzyme-linked polyclonal
antibody specific to VEGF was added. Again, unbound substances were
washed out. Finally, a substrate solution was added to the wells,
inducing color development in proportion to the amount of VEGF
bound in the initial step. The color development was stopped, and
the intensity of the color was measured. The exact concentration of
VEGF was determined using a standard curve.
After 24, 48 or 72 h of incubation with 0, 2, 4, 6,
8 or 10 mg/ml Bevacizumab (kindly provided by the Pharmacy
supplying the Oncological Department, University Hospital Mannheim,
Medical Faculty of the University of Heidelberg, Germany), the
expression of VEGF was analyzed in the supernatants of the HHT cell
cultures and the HUVECs (Fig.
1).
In this study, experiments were performed with even
concentration levels of Bevacizumab of 2, 4, 6, 8 or 10 mg/ml as
these concentration levels were obtained directly from the
pharmaceutical and cytostatic laboratory of the hospital already in
ready to be used ‘pre-filled syringes’ same as they were used in
the clinic for patient treatment in the Hematooncology department.
With these provided concentration levels of Bevacizumab we were
wanting to evaluate the concentration levels and the means how the
effects differed on endothelial cell proliferation and VEGF
expression in HHT cell cultures.
Statistical analysis
Quantitative variables have been presented by their
mean values and standard deviations. For qualitative factors,
absolute and relative frequencies are given.
To compare the mean values of the three groups, a
one-way ANOVA was used. In the case of a significant test result, a
post-hoc Scheffé test was performed. P<0.05 was considered to
indicate a statistically significant difference. For statistical
evaluation, the SAS software version 9.4 (SAS Institute Inc.) was
used.
Results
Cell proliferation following
incubation with different Bevacizumab concentrations. HUVECs
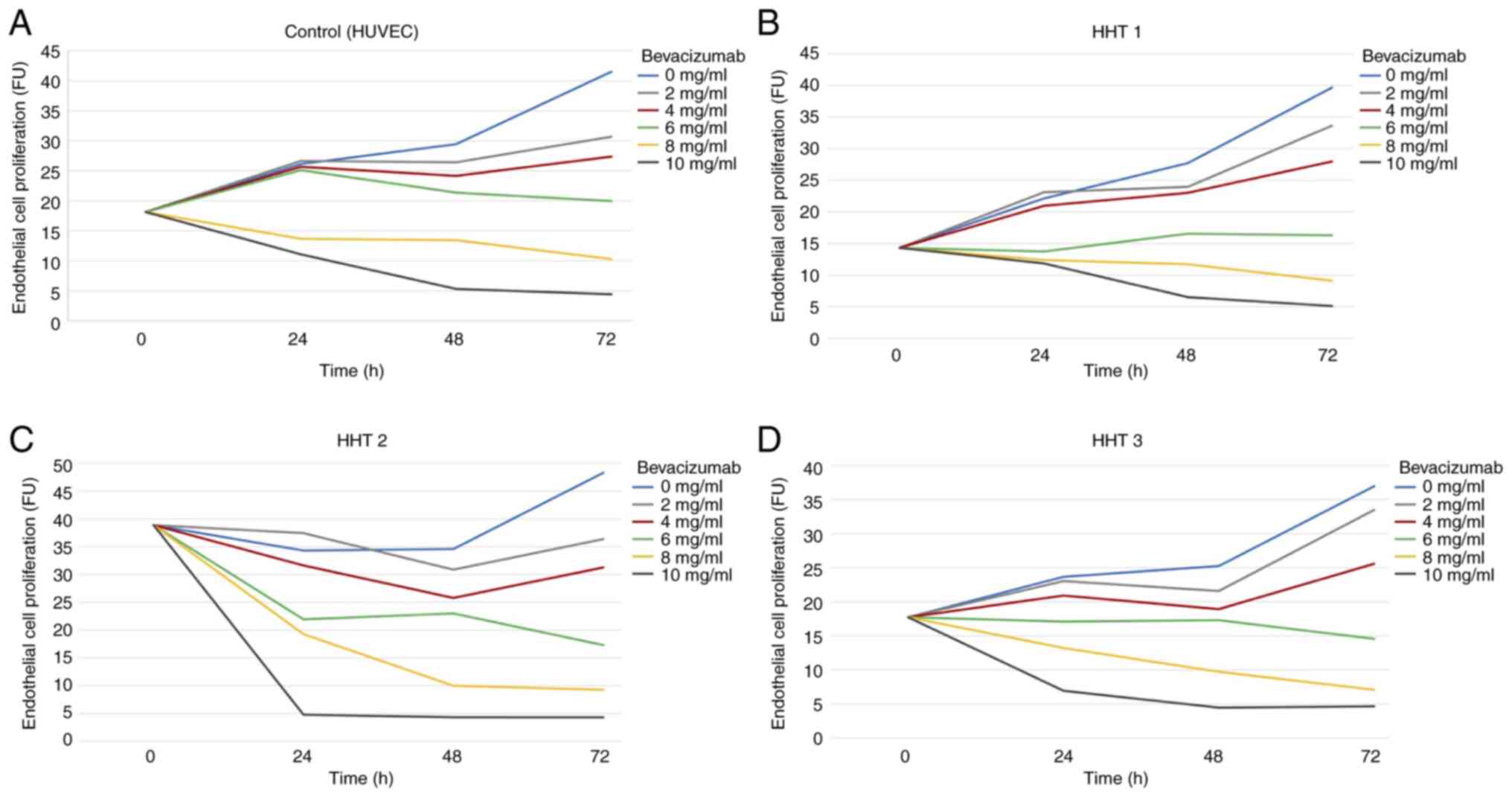
In the HUVECs, untreated endothelial cells showed an
increase in cell proliferation throughout the entire 72 h period.
The proliferation rate after 24 h increased from 18.14 to 26.13 FU.
After 48 h the proliferation rate was 29.46 FU and after 72 h it
was 41.54 FU.
A positive trend in cell proliferation could also be
seen in endothelial cells incubated with 2 and 4 mg/ml Bevacizumab.
After 24 h, cell proliferation increased from 18.14 to 26.67 and
25.7 FU, respectively, and was nearly identical to that of
untreated cells. Cell proliferation then almost plateaued until 48
h and started to increase again after 72 h to 30.7 and 27.38 FU,
respectively.
In general, incubation with 6, 8 and 10 mg/ml
Bevacizumab led to a decrease in cell proliferation with a
significant decline in cell numbers after 72 h. This negative trend
was already obvious 24 h after incubation with 10 mg/ml
Bevacizumab. The considerable decline in cell numbers reached
values <10 FU and continued to decrease after 48 and 72 h.
Values <10 FU were considered as full apoptosis of the cell
culture and therefore considered toxic in this study. A similar
trend, though more prolonged in the decrease of cell proliferation,
was seen after incubation with 8 mg/ml Bevacizumab. A toxicity
level of <10 FU was reached after 72 h without any recovery of
cell proliferation. Incubation with 6 mg/ml Bevacizumab first
increased after 24 h from 18.14 to 25.12 FU, but after 48 and 72 h,
the cell number decreased to 21.38 and 19.98 FU, respectively.
However, the 6 mg/ml Bevacizumab concentration level did not reach
the toxic cut-off level of 10 FU (Fig.
2A).
HHT cell cultures
The cell proliferation of the HHT 1 cell culture was
similar to that of the HUVECs. Whereas concentration levels of 0, 2
and 4 mg/ml Bevacizumab had a positive impact on cell
proliferation, concentration levels of 8 and 10 mg/ml Bevacizumab
induced a toxic effect leading to cell apoptosis after 48 and 72 h.
The number of endothelial cells incubated with 6 mg/ml Bevacizumab
almost stagnated for 24 h at a rate between 13.74 and 14.32 FU
before they started to increase again to a value of 16 FU (Fig. 2B).
The findings for endothelial cell proliferation in
the HHT 2 and HHT 3 cell cultures were similar to those of the HHT
1 group. The proliferation rate at 24 and 48 h after incubation
with 2 and 4 mg/ml Bevacizumab first decreased slightly before it
increased again at 72 h. Endothelial cells incubated with 10 mg/ml
Bevacizumab exhibited negative proliferation after only 24 h of
incubation, with a massive decrease in cell numbers reaching values
below the toxic cut-off level of 10 FU. After 48 and 72 h, a
further decrease in cell numbers was observed without any signs of
recovery. Apoptosis was also observed in cell cultures incubated
with 8 mg/ml Bevacizumab even though the toxic level of 10 FU was
not reached until after 48 h. Endothelial cells of the HHT 2 and 3
cell cultures which were incubated with 6 mg/ml Bevacizumab showed
a slight decrease in cell proliferation after a period of 72 h to
17.29 and 14.57 FU, respectively (Fig.
2C and D).
A detailed overview of the data regarding the cell
proliferation rates 24, 48 and 72 h after incubation of the HUVECs
and the HHT cell cultures with different concentration levels of
Bevacizumab are provided in Table
I.
 | Table IDetailed overview of cell
proliferation rates 24, 48 and 72 h after incubation of the HUVEC
cell line (control) and the HHT cell cultures with different
concentration levels of Bevacizumab. |
Table I
Detailed overview of cell
proliferation rates 24, 48 and 72 h after incubation of the HUVEC
cell line (control) and the HHT cell cultures with different
concentration levels of Bevacizumab.
| | Bevacizumab |
|---|
| Incubation time
(h) | 0 (mg/ml) | 2 (mg/ml) | 4 (mg/ml) | 6 (mg/ml) | 8 (mg/ml) | 10 (mg/ml) |
|---|
| HUVEC
(Control) | | | | | | |
|
0 | 18.14 | 18.14 | 18.14 | 18.14 | 18.14 | 18.14 |
|
24 | 26.13 | 26.67 | 25.7 | 25.12 | 13.69 | 11.06 |
|
48 | 29.46 | 26.42 | 24.15 | 21.38 | 13.45 | 5.31 |
|
72 | 41.54 | 30.7 | 27.38 | 19.98 | 10.29 | 4.43 |
| HHT 1 | | | | | | |
|
0 | 14.32 | 14.32 | 14.32 | 14.32 | 14.32 | 14.32 |
|
24 | 22.11 | 23.08 | 20.95 | 13.74 | 12.38 | 11.85 |
|
48 | 27.7 | 23.97 | 23.01 | 16.55 | 11.47 | 6.54 |
|
72 | 39.64 | 33.63 | 27.97 | 16.28 | 9.12 | 5.12 |
| HHT 2 | | | | | | |
|
0 | 38.88 | 38.88 | 38.88 | 38.88 | 38.88 | 38.88 |
|
24 | 34.3 | 37.43 | 31.61 | 21.93 | 19.27 | 4.76 |
|
48 | 34.55 | 30.86 | 25.78 | 23.01 | 9.99 | 4.31 |
|
72 | 48.29 | 36.37 | 31.26 | 17.29 | 9.24 | 4.29 |
| HHT 3 | | | | | | |
|
0 | 17.74 | 17.74 | 17.74 | 17.74 | 17.74 | 17.74 |
|
24 | 23.66 | 23.05 | 20.94 | 17.09 | 13.24 | 6.96 |
|
48 | 25.27 | 21.59 | 18.93 | 17.32 | 9.76 | 4.48 |
|
72 | 36.96 | 33.48 | 25.58 | 14.57 | 7.1 | 4.69 |
The mean values and P-values for the cell
proliferation rate of the HUVEC, HHT 1, HHT 2 and HHT 3 endothelial
cells incubated with different concentration levels of Bevacizumab
are given in Table II.
 | Table IIMean values and P-values for the cell
proliferation rate of the HUVEC, HHT 1, HHT 2 and HHT 3 endothelial
cells incubated with different concentration levels of
bevacizumab. |
Table II
Mean values and P-values for the cell
proliferation rate of the HUVEC, HHT 1, HHT 2 and HHT 3 endothelial
cells incubated with different concentration levels of
bevacizumab.
| | Concentration
levels | |
|---|
| Incubation time
(h) | Low (2 mg/ml, 4
mg/ml) | Medium (6
mg/ml) | High (8 mg/ml, 10
mg/ml) | Comparisons |
P-valuea |
|---|
| HUVEC
(Control) | | | | | |
|
0 | 18.14 | 18.14 | 18.14 | | |
|
24 | 26.19 | 25.12 | 12.38 | Low vs. High | <0.0001 |
|
48 | 25.29 | 21.38 | 9.38 | Low vs. Medium | 0.0265 |
|
72 | 29.04 | 19.98 | 7.36 | Medium vs.
High | 0.1413 |
| HHT 1 | | | | | |
|
0 | 14.32 | 14.32 | 14.32 | | |
|
24 | 22.01 | 13.74 | 12.11 | Low vs. High | <0.0001 |
|
48 | 23.49 | 16.55 | 9.14 | Low vs. Medium | 0.0002 |
|
72 | 30.8 | 16.28 | 7.12 | Medium vs.
High | <0.0001 |
| HHT 2 | | | | | |
|
0 | 38.88 | 38.88 | 38.88 | | |
|
24 | 34.52 | 21.93 | 12.02 | Low vs. High | <0.0001 |
|
48 | 28.32 | 23.01 | 7.15 | Low vs. Medium | <0.0001 |
|
72 | 33.99 | 17.29 | 6.77 | Medium vs.
High | 0.0163 |
| HHT 3 | | | | | |
|
0 | 17.74 | 17.74 | 17.74 | | |
|
24 | 22 | 17.09 | 10.1 | Low vs. High | <0.0001 |
|
48 | 20.26 | 17.32 | 7.12 | Low vs. Medium | <0.0001 |
|
72 | 29.53 | 14.57 | 5.9 | Medium vs.
High | 0.045 |
VEGF tissue expression after
incubation with different Bevacizumab concentrations. HUVECs
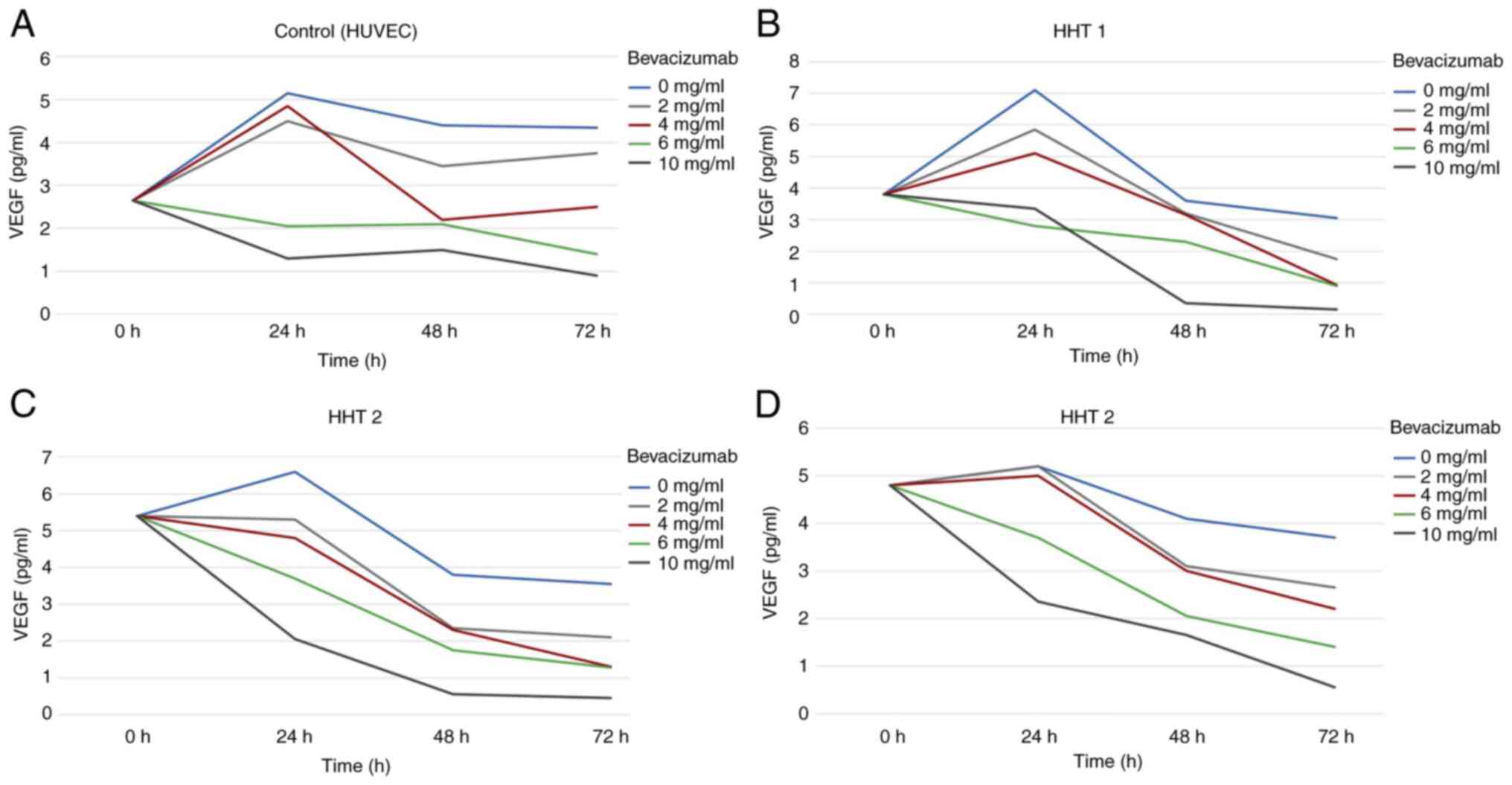
VEGF tissue expression in the HUVECs initially
showed a slight increase after 24 h of incubation with 2 mg/ml
concentration. However, it then decreased after 48 h and increased
again after 72 h. The same effect was observed with 4 mg/ml
Bevacizumab, although the suppression in VEGF tissue expression was
considerably stronger than with 2 mg/ml Bevacizumab. However, this
effect did not seem to be long-lasting, as the VEGF tissue
expression increased again after 72 h of incubation.
Higher concentration levels of 6 and 10 mg/ml
Bevacizumab showed an immediate suppression of VEGF tissue
expression after only 24 h of incubation, which continued even
after 72 h (Fig. 3A).
HHT cell cultures
In all three HHT cell cultures, Bevacizumab had a
suppressive effect on VEGF expression in endothelial cells
throughout the entire 72 h period of incubation. However, lower
concentration levels of Bevacizumab (0, 2 and 4 mg/ml) had a more
delayed effect and appeared to exert its suppressive action after
24 h of incubation. Higher concentration levels of 6 and 10 mg/ml
Bevacizumab had an immediate effect after only 24 h of incubation
and continued to decrease the VEGF expression levels consistently
over the entire 72 h period of incubation without any recovery
(Fig. 3B-D).
A detailed overview of the data regarding the VEGF
expression levels 24, 48 and 72 h after incubation of the HUVECs
and the HHT cell cultures with different concentration levels of
Bevacizumab are given in Table
III.
 | Table IIIDetailed overview of VEGF expression
levels 24, 48 and 72 h after incubation of the HUVEC cell line
(control) and the HHT cell cultures with different concentration
levels of Bevacizumab. |
Table III
Detailed overview of VEGF expression
levels 24, 48 and 72 h after incubation of the HUVEC cell line
(control) and the HHT cell cultures with different concentration
levels of Bevacizumab.
| | Bevacizumab |
|---|
| Incubation time
(h) | 0 (mg/ml) | 2 (mg/ml) | 4 (mg/ml) | 6 (mg/ml) | 10 (mg/ml) |
|---|
| HUVEC
(Control) | | | | | |
|
0 | 2.65 | 2.65 | 2.65 | 2.65 | 2.65 |
|
24 | 5.15 | 4.5 | 4.85 | 2.05 | 1.3 |
|
48 | 4.4 | 3.45 | 2.2 | 2.1 | 1.5 |
|
72 | 4.35 | 3.75 | 2.5 | 1.4 | 0.9 |
| HHT 1 | | | | | |
|
0 | 3.8 | 3.8 | 3.8 | 3.8 | 3.8 |
|
24 | 7.1 | 5.85 | 5.1 | 2.8 | 3.35 |
|
48 | 3.6 | 3.2 | 3.15 | 2.3 | 0.35 |
|
72 | 3.05 | 1.75 | 0.93 | 0.9 | 0.155 |
| HHT 2 | | | | | |
|
0 | 5.4 | 5.4 | 5.4 | 5.4 | 5.4 |
|
24 | 6.6 | 5.3 | 4.8 | 3.7 | 2.05 |
|
48 | 3.8 | 2.35 | 2.3 | 1.75 | 0.55 |
|
72 | 3.55 | 2.1 | 1.3 | 1.28 | 0.45 |
| HHT 3 | | | | | |
|
0 | 4.8 | 4.8 | 4.8 | 4.8 | 4.8 |
|
24 | 5.2 | 5.2 | 5 | 3.7 | 2.35 |
|
48 | 4.1 | 3.1 | 3 | 2.05 | 1.65 |
|
72 | 3.7 | 2.65 | 2.2 | 1.4 | 0.55 |
Discussion
The monoclonal VEGF-antibody Bevacizumab appears to
be a promising therapy for patients with HHT (12,22,23).
Its systemic intravenous application in HHT patients has been
abandoned in favor of topical applications of Bevacizumab. Very few
studies describe the topical use of Bevacizumab in HHT patients
(22,23). Most studies that have assessed this
are small heterogeneous case series with notable differences in the
dosing regimens, which makes the analysis of outcomes extremely
difficult.
Endonasal submucosal injections of Bevacizumab seem
to be more effective regarding the epistaxis severity score (ESS)
and the hemoglobin level than when it is used as a topical nose
spray (22). They seem to be well
tolerated by HHT patients without major signs of severe systemic
side effects (26-30).
However, nasal septal perforations are a potential side effect of
the intranasal delivery of Bevacizumab, especially when injected
bilateral to the cartilaginous septum. Therefore, injections should
favor non-cartilaginous areas within the nose to avoid septal
perforations (22-24).
In the literature, the dosing levels of Bevacizumab
for endonasal injections in HHT patients vary considerably between
3.75 and 25 mg/ml (24-30).
To date, no Otorhinolaryngological studies have provided ideal
dosing guidelines for Bevacizumab administration. For the
practicing clinician, details on the efficacy and damaging potency
of different Bevacizumab concentration levels to the nasal mucosa
are missing. An inconsiderate, reckless application of high
concentration levels of Bevacizumab should be avoided.
In Ophthalmology, studies have described the
well-established use of 1.25 and 2.5 mg Bevacizumab in patients
with retinal neovascularization for local intravitreal injections
every 4-12 weeks (31-33).
The results indicate a very low complication rate of ≤0.21%
(31). These complications include
blood pressure elevation, corneal abrasion, mild discomfort, and
inflammation uveitis.
Studies are needed to show the efficacy of lower
doses of Bevacizumab on the severity of epistaxis and visceral AVM
involvement and their potentially lower profile of side effects
(34).
To the best of our knowledge, the results presented
in this paper are the first in vitro data assessing the
effects of different concentration levels of Bevacizumab on
endothelial cell cultures from HHT patients and the VEGF
concentration in their supernatant with the aim of identifying a
safe dose. The proliferation rate of endothelial cells was affected
by different concentration levels of Bevacizumab. The higher the
concentration level of Bevacizumab, the greater the decrease in the
proliferation rate was. Concentration levels of 8 mg/ml Bevacizumab
and higher were shown to harm cell proliferation to the point of
induction of cell apoptosis in all specimens as an indicator for
cell toxicity (values <10 FU).
The results of the present study showed the
inhibitory effect of Bevacizumab on endothelial cell proliferation.
Even though we did not undertake any specific TUNEL staining of
cells, it was assumed that the cytotoxic effects of Bevacizumab
enhanced endogenous apoptosis in primary endothelial cell lines as
well as established HHT cell cultures as already described in other
studies for different cell lines and tumor entities in the
literature (35,36).
These results stand in contrast to the doses used in
submucosal injections in previous studies, which mainly ranged
between 10-25 mg/ml with an overall application of 50-100 mg
Bevacizumab to each nasal cavity (24,26,27,29).
After Simonds et al (24)
and Chen et al (22)
described the risk of septal perforations associated with
submucosal injections of Bevacizumab, subsequent studies avoided
injections in the cartilaginous septum.
The results of the present study suggest that the
risk of systemic adverse events and septal perforations may be
reduced by using considerably lower doses. Lower concentration
levels of up to 4 mg/ml Bevacizumab initially decelerated
endothelial cell proliferation within the first 48 h but had a
tendency towards recovery after 72 h of incubation.
Concentration levels between 4 and 6 mg/ml
Bevacizumab should be investigated more carefully. Concentration
levels of 4 mg/ml Bevacizumab seem to be effective in slowing down
endonasal cell proliferation but allow the cells to recover after
48 h of incubation. Concentration levels of 6 mg/ml Bevacizumab
impair cell recovery by inducing stagnation or even deceleration of
endonasal cell proliferation. These findings highlight 4 mg/ml
Bevacizumab as a cut-off concentration level that should not be
exceeded when applied as a submucosal injection to the nose, merely
to be on the safe side and not destroy endothelial cell
function.
A potential drawback of this study was the
one-to-one translation of the described in vitro data to the
endonasal in vivo injections of Bevacizumab in HHT patients.
The local delivery of lower doses of Bevacizumab may still result
in a high tissue load. Potential damage to the nasal endothelial
cells after the application of ≥6 mg/ml Bevacizumab need to be more
seriously considered (34).
However, conclusions drawn from these results should be treated
with care due to the small sample size.
To the best of our knowledge, this in vitro
study is the first Rhinological study that aims to suggest a dosing
guideline for the safe and effective application of Bevacizumab
when applied intranasally as a submucosal injection in HHT
patients. This data may provide valuable information to assess the
risk/benefit ratio of Bevacizumab on the endothelial cells of the
nasal mucosa. This study also clearly demonstrates that oncological
dosing parameters are not indicated to improve medically refractory
epistaxis in patients with HHT (37).
However, further clinical research is required to
define these treatment details and optimize the dosing and delivery
routes for Bevacizumab in the treatment of epistaxis caused by HHT
(29,30,34).
Regarding the efficacy of Bevacizumab on endonasal
endothelial cells, low concentration levels of 2 and 4 mg/ml
effected a considerable decline in VEGF expression within the first
48 h. After 48 h, VEGF concentration increased again when compared
to higher concentration levels of 6 and 10 mg/ml, which indicated
no signs of recovery. These results indicate that incubation of
endonasal endothelial cells with low concentration levels does have
an effect, even though it might be short-lasting. Our hypothesis of
a sufficient efficacy of low-dose Bevacizumab is also supported by
the observations of Rohrmeier et al, who showed a benefit of
combined treatment with submucosal injections of 3.75 mg/ml (in
total, 3.75 mg per nasal cavity) and YAC-Laser vs. laser therapy
alone (28). However, conclusions
drawn from these results should be treated with care due to the
small sample size.
Other than in the control group, the results of the
HHT endothelial cell cultures indicated that incubation up to 72 h
with Bevacizumab concentration levels of 4 mg/ml or less seemed to
be effective enough to suppress the VEGF level without jeopardizing
cell proliferation. However, in this study, we could not see a
significant correlation between cell proliferation and a decreased
VEGF concentration level after 24 h of incubation of the HHT cell
lines with Bevacizumab.
Further research studies with odd concentration
levels of Bevacizumab are necessary to determine even more
accurately the safety concentration level of Bevacizumab in the
treatment of epistaxis in HHT patients. In addition, further
investigations would help to get a better insight into the
pathophysiology and inhibitory mechanism of Bevacizumab. Of special
relevance would be a more precise answer to the question whether
Bevacizumab inhibits cell proliferation or induces endogenous
apoptosis.
In summary, this study suggests that incubation of
endonasal endothelial cells with Bevacizumab in HHT patients should
not exceed the concentration level of 4 mg/ml. Higher concentration
levels risk a more toxic effect on endothelial cells, as they
jeopardize cell proliferation. Further studies are now necessary to
investigate the toxicity of 6 mg/ml Bevacizumab over a longer
period, as well as in vivo studies addressing the efficacy
of intranasal submucosal injections of low-dose Bevacizumab between
2 and 6 mg/ml.
Acknowledgements
The authors gratefully acknowledge the excellent
technical support of Ms. Petra Prohaska (Department of
Otorhinolaryngology, Head and Neck Surgery, University Hospital
Mannheim, Medical Faculty Mannheim of the University of Heidelberg,
D-68135 Mannheim, Germany). We also thank Ms. Sylvia Büttner
(Department of Medical Statistics, University Hospital Mannheim,
Medical Faculty Mannheim of the University of Heidelberg, D-68135
Mannheim, Germany) for her help with statistical analysis.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HS, ES and DH made substantial contributions to
concept, design and data acquisition and were major contributors in
writing the manuscript. CW performed the statistical analysis and
helped with data analysis and interpretation. MS, NR, CEM and RB
helped drafting the manuscript and revising it critically after
initial analysis and interpretation of the data. HS and ES confirm
the authenticity of all the raw data. All authors read and approved
the final manuscript.
Ethics approval and consent to
participate
Prior to surgery, written consent to take a small
tissue sample from their telangiectatic nasal mucosa of the
inferior turbinate was obtained from all patients. This study was
approved by the Ethics Committee of the University Hospital of
Mannheim (approval no. 0238.2).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Donaldson JW, McKeever TM, Hall IP,
Hubbard RB and Fogarty AW: The UK prevalence of hereditary
haemorrhagic telangiectasia and its association with sex,
socioeconomic status and region of residence: A population-based
study. Thorax. 69:161–167. 2014.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Kjeldsen AD, Vase P and Green A:
Hereditary haemorrhagic telangiectasia: A population-based study of
prevalence and mortality in Danish patients. J Intern Med.
245:31–39. 1999.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Sautter NB and Smith TL: Hereditary
hemorrhagic telangiectasia-related epistaxis: Innovations in
understanding and management. Int Forum Allergy Rhinol. 2:422–431.
2012.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Plauchu H, de Chadarévian JP, Bideau A and
Robert JM: Age-related clinical profile of hereditary hemorrhagic
telangiectasia in an epidemiologically recruited population. Am J
Med Genet. 32:291–297. 1989.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Ingrand I, Ingrand P, Gilbert-Dussardier
B, Defossez G, Jouhet V, Migeot V, Dufour X and Klossek JM: Altered
quality of life in Rendu-Osler-Weber disease related to recurrent
epistaxis. Rhinology. 49:155–162. 2011.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Kritharis A, Al-Samkari H and Kuter DJ:
Hereditary hemorrhagic telangiectasia: diagnosis and management
from the hematologist's perspective. Haematologica. 103:1433–1443.
2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Jakobsson L and van Meeteren LA:
Transforming growth factor β family members in regulation of
vascular function: in the light of vascular conditional knockouts.
Exp Cell Res. 319:1264–1270. 2013.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Steineger J, Geirdal AØ, Osnes T, Heimdal
KR and Dheyauldeen S: Intranasal bevacizumab injections improve
quality of life in HHT patients. Laryngoscope. 130:E284–E288.
2020.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Prigoda NL, Savas S, Abdalla SA, Piovesan
B, Rushlow D, Vandezande K, Zhang E, Ozcelik H, Gallie BL and
Letarte M: Hereditary haemorrhagic telangiectasia: Mutation
detection, test sensitivity and novel mutations. J Med Genet.
43:722–728. 2006.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Oh SP, Seki T, Goss KA, Imamura T, Yi Y,
Donahoe PK, Li L, Miyazono K, ten Dijke P, Kim S and Li E: Activin
receptor-like kinase 1 modulates transforming growth factor-beta 1
signaling in the regulation of angiogenesis. Proc Natl Acad Sci
USA. 97:2626–2631. 2000.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Sadick H, Hage J, Goessler U, Bran G,
Riedel F, Bugert P and Hoermann K: Does the genotype of HHT
patients with mutations of the ENG and ACVRL1 gene correlate to
different expression levels of the angiogenic factor VEGF? Int J
Mol Med. 22:575–580. 2008.PubMed/NCBI
|
|
12
|
Sadick H, Riedel F, Naim R, Goessler U,
Hörmann K, Hafner M and Lux A: Patients with hereditary hemorrhagic
telangiectasia have increased plasma levels of vascular endothelial
growth factor and transforming growth factor-beta1 as well as high
ALK1 tissue expression. Haematologica. 90:818–828. 2005.PubMed/NCBI
|
|
13
|
Sadick H, Naim R, Sadick M, Hörmann K and
Riedel F: Plasma level and tissue expression of angiogenic factors
in patients with hereditary hemorrhagic telangiectasia. Int J Mol
Med. 15:591–596. 2005.PubMed/NCBI
|
|
14
|
Han C, Choe SW, Kim YH, Acharya AP,
Keselowsky BG, Sorg BS, Lee YJ and Oh SP: VEGF neutralization can
prevent and normalize arteriovenous malformations in an animal
model for hereditary hemorrhagic telangiectasia 2. Angiogenesis.
17:823–830. 2014.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Ferrara N, Hillan KJ and Novotny W:
Bevacizumab (Avastin), a humanized anti-VEGF monoclonal antibody
for cancer therapy. Biochem Biophys Res Commun. 333:328–335.
2005.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Shih T and Lindley C: Bevacizumab: An
angiogenesis inhibitor for the treatment of solid malignancies.
Clin Ther. 28:1779–1802. 2006.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Amarakoon S, Martinez-Ciriano JP, van den
Born LI, Baarsma S and Missotten T: Bevacizumab in age-related
macular degeneration: A randomized controlled trial on the effect
of on-demand therapy every 4 or 8 weeks. Acta Ophthalmol.
97:107–112. 2019.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Iyer VN, Apala DR, Pannu BS, Kotecha A,
Brinjikji W, Leise MD, Kamath PS, Misra S, Begna KH, Cartin-Ceba R,
et al: Intravenous Bevacizumab for refractory hereditary
hemorrhagic telangiectasia-related epistaxis and gastrointestinal
bleeding. Mayo Clin Proc. 93:155–166. 2018.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Andrejecsk JW, Hosman AE, Botella LM,
Shovlin CL, Arthur HM, Dupuis-Girod S, Buscarini E, Hughes CCW,
Lebrin F, Mummery CL, et al: Executive summary of the 12th HHT
international scientific conference. Angiogenesis. 21:169–181.
2018.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Dupuis-Girod S: Anti-VEGF: A new
therapeutic option in hereditary hemorrhagic telangiectasia. Presse
Med. 42:385–387. 2013.PubMed/NCBI View Article : Google Scholar : (In French).
|
|
21
|
Cohen MH, Gootenberg J, Keegan P and
Pazdur R: FDA drug approval summary: Bevacizumab (Avastin) plus
Carboplatin and Paclitaxel as first-line treatment of
advanced/metastatic recurrent nonsquamous non-small cell lung
cancer. Oncologist. 12:713–718. 2007.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Chen S IV, Karnezis T and Davidson TM:
Safety of intranasal Bevacizumab (Avastin) treatment in patients
with hereditary hemorrhagic telangiectasia-associated epistaxis.
Laryngoscope. 121:644–646. 2011.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Stokes P and Rimmer J: Intranasal
bevacizumab in the treatment of HHT -related epistaxis: A
systematic review. Rhinology. 56:3–10. 2018.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Simonds J, Miller F, Mandel J and Davidson
TM: The effect of bevacizumab (Avastin) treatment on epistaxis in
hereditary hemorrhagic telangiectasia. Laryngoscope. 119:988–992.
2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Dupuis-Girod S, Ambrun A, Decullier E,
Fargeton AE, Roux A, Bréant V, Colombet B, Rivière S, Cartier C,
Lacombe P, et al: Effect of bevacizumab nasal spray on epistaxis
duration in hereditary hemorrhagic telangectasia: A randomized
clinical trial. JAMA. 316:934–942. 2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Steineger J, Osnes T, Heimdal K and
Dheyauldeen S: Long-term experience with intranasal bevacizumab
therapy. Laryngoscope. 128:2237–2244. 2018.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Riss D, Burian M, Wolf A, Kranebitter V,
Kaider A and Arnoldner C: Intranasal submucosal bevacizumab for
epistaxis in hereditary hemorrhagic telangiectasia: A double-blind,
randomized, placebo-controlled trial. Head Neck. 37:783–787.
2015.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Rohrmeier C, Sachs HG and Kuehnel TS: . A
retrospective analysis of low dose, intranasal injected bevacizumab
(Avastin) in hereditary haemorrhagic telangiectasia. Eur Arch
Otorhinolaryngol. 269:531–536. 2012.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Karnezis TT and Davidson TM: Efficacy of
intranasal Bevacizumab (Avastin) treatment in patients with
hereditary hemorrhagic telangiectasia-associated epistaxis.
Laryngoscope. 121:636–638. 2011.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Karnezis TT and Davidson TM: Treatment of
hereditary hemorrhagic telangiectasia with submucosal and topical
bevacizumab therapy. Laryngoscope. 122:495–497. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Lynch SS and Cheng CM: Bevacizumab for
neovascular ocular diseases. Ann Pharmacother. 41:614–625.
2007.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Gordon MS, Margolin K, Talpaz M, Sledge GW
Jr, Holmgren E, Benjamin R, Stalter S, Shak S and Adelman D: Phase
I safety and pharmacokinetic study of recombinant human
anti-vascular endothelial growth factor in patients with advanced
cancer. J Clin Oncol. 19:843–850. 2001.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Michels S: Is intravitreal bevacizumab
(Avastin) safe? Br J Ophthalmol. 90:1333–1334. 2006.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Thompson AB, Ross DA, Berard P,
Figueroa-Bodine J, Livada N and Richer SL: Very low dose
bevacizumab for the treatment of epistaxis in patients with
hereditary hemorrhagic telangiectasia. Allergy Rhinol. 5:91–95.
2014.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Färkkilä A, Pihlajoki M, Tauriala H,
Bützow R, Leminen A, Unkila-Kallio L, Heikinheimo M and Anttonen M:
Serum vascular endothelial growth factor A (VEGF) is elevated in
patients with ovarian granulosa cell tumor (GCT), and VEGF
inhibition by Bevacizumab induces apoptosis in GCT in vitro. J Clin
Endocrinol Metab. 96:E1973–E1981. 2011.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Ramezani S, Vousooghi N, Kapourchali FR,
Hadjighasem M, Hayat P, Amini N and Joghataei MT: Rolipram
potentiates bevacizumab-induced cell death in human glioblastoma
stem-like cells. Life Sci. 173:11–19. 2017.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Guilhem A, Fargeton AE, Simon AC, Duffau
P, Harle JR, Lavigne C, Carette MF, Bletry O, Kaminsky P, Leguy V,
et al: Intra-venous bevacizumab in hereditary hemorrhagic
telangiectasia (HHT): A retrospective study of 46 patients. PLoS
One. 12(e0188943)2017.PubMed/NCBI View Article : Google Scholar
|