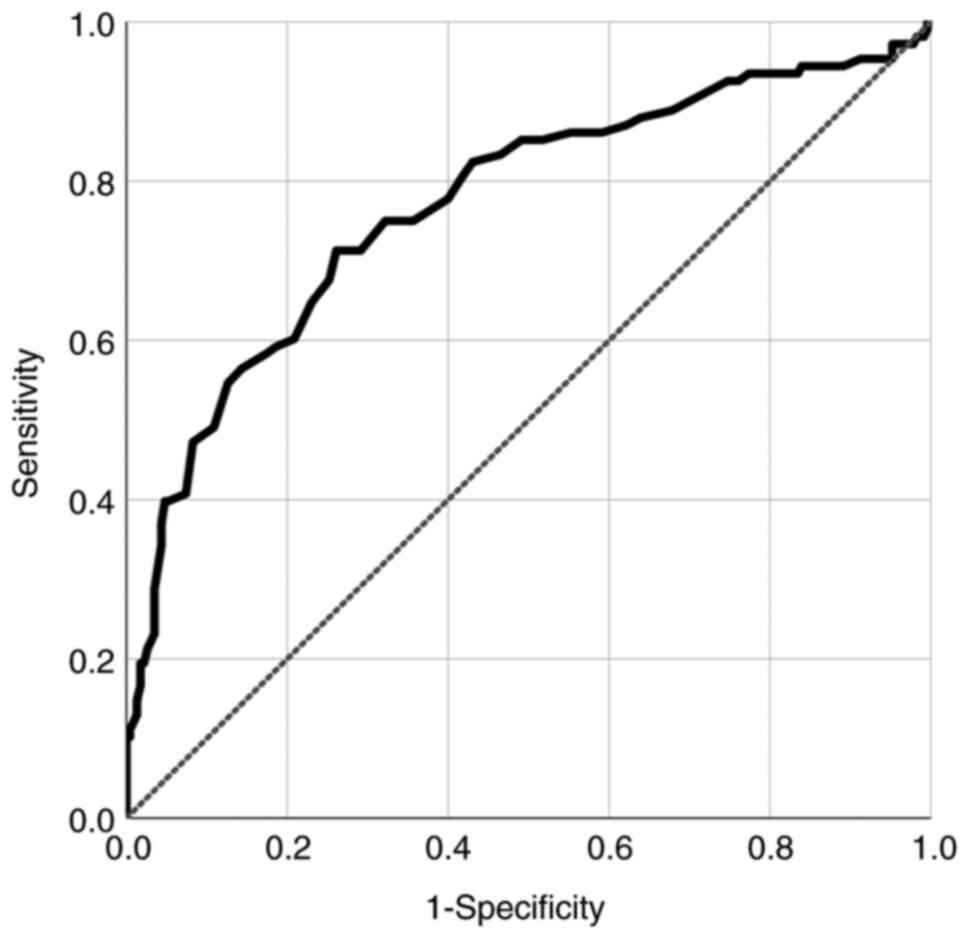
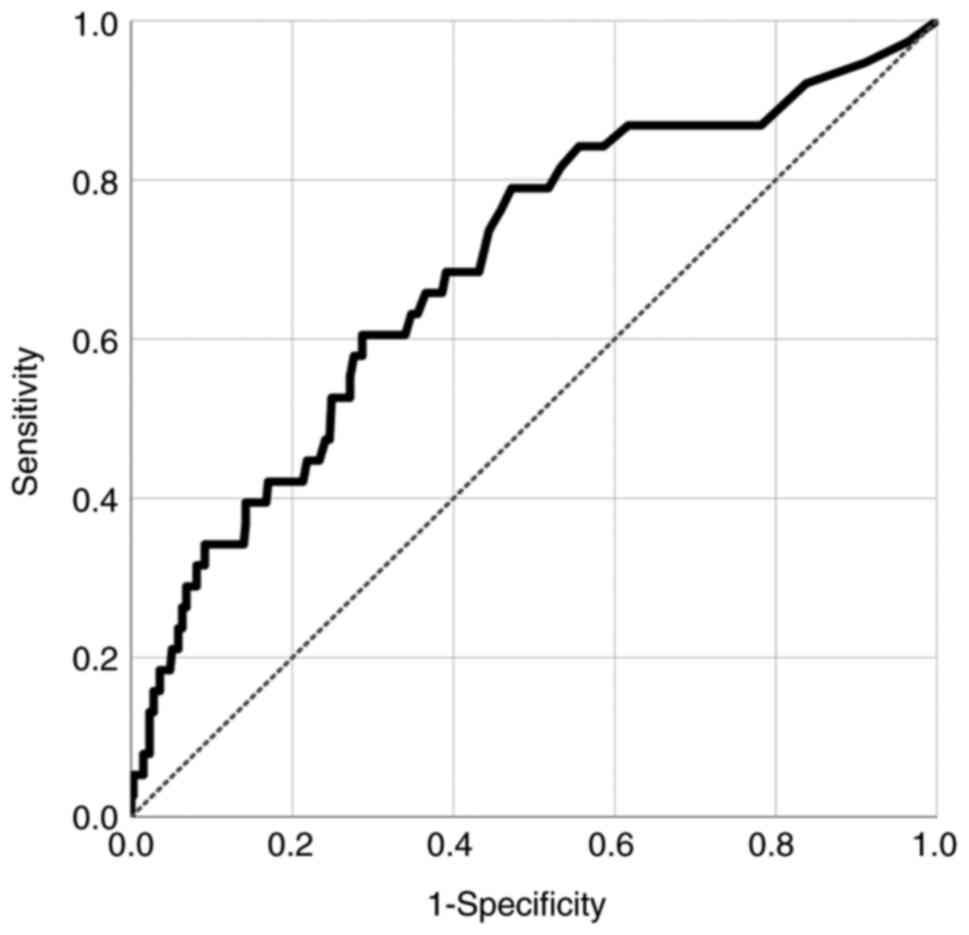
|
1
|
Rinkel GJ and Algra A: Long-term outcomes
of patients with aneurysmal subarachnoid haemorrhage. Lancet
Neurol. 10:349–356. 2011.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dijkland SA, Jaja BNR, van der Jagt M,
Roozenbeek B, Vergouwen MDI, Suarez JI, Torner JC, Todd MM, van den
Bergh WM, Saposnik G, et al: Between-center and between-country
differences in outcome after aneurysmal subarachnoid hemorrhage in
the subarachnoid hemorrhage international trialists (SAHIT)
repository. J Neurosurg. 1–9. 2019.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
3
|
Li K, Barras CD, Chandra RV, Kok HK,
Maingard JT, Carter NS, Russell JH, Lai L, Brooks M and Asadi H: A
review of the management of cerebral vasospasm after aneurysmal
subarachnoid hemorrhage. World Neurosurg. 126:513–527.
2019.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Veldeman M, Lepore D, Hollig A, Clusmann
H, Stoppe C, Schubert GA and Albanna W: Procalcitonin in the
context of delayed cerebral ischemia after aneurysmal subarachnoid
hemorrhage. J Neurosurg. 1-9:2020.PubMed/NCBI View Article : Google Scholar : (Epub ahead of
print).
|
|
5
|
Connolly ES Jr, Rabinstein AA, Carhuapoma
JR, Derdeyn CP, Dion J, Higashida RT, Hoh BL, Kirkness CJ, Naidech
AM, Ogilvy CS, et al: Guidelines for the management of aneurysmal
subarachnoid hemorrhage: A guideline for healthcare professionals
from the American heart association/american stroke association.
Stroke. 43:1711–1737. 2012.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Committee for Guidelines for Management of
Aneurysmal Subarachnoid Hemorrhage, Japanese Society on Surgery for
Cerebral Stroke. Evidence-based guidelines for the management of
aneurysmal subarachnoid hemorrhage. English edition. Neurol Med
Chir (Tokyo). 52:355–429. 2012.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rass V and Helbok R: Early brain injury
after poor-grade subarachnoid hemorrhage. Curr Neurol Neurosci Rep.
19(78)2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yun S, Yi HJ, Lee DH and Sung JH: Systemic
inflammation response index and systemic immune-inflammation index
for predicting the prognosis of patients with aneurysmal
subarachnoid hemorrhage. J Stroke Cerebrovasc Dis.
30(105861)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
McBride DW, Blackburn SL, Peeyush KT,
Matsumura K and Zhang JH: The role of thromboinflammation in
delayed cerebral ischemia after subarachnoid hemorrhage. Front
Neurol. 8(555)2017.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kasius KM, Frijns CJ, Algra A and Rinkel
GJ: Association of platelet and leukocyte counts with delayed
cerebral ischemia in aneurysmal subarachnoid hemorrhage.
Cerebrovasc Dis. 29:576–583. 2010.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Ogden M, Bakar B, Karagedik MI, Bulut IU,
Cetin C, Aydin G, Kisa U and Ozveren MF: Analysis of biochemical
laboratory values to determine etiology and prognosis in patients
with subarachnoid hemorrhage: A clinical study. Neurol Res.
41:156–167. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Güresir E, Coch C, Fimmers R, Ilic I,
Hadjiathanasiou A, Kern T, Brandecker S, Güresir Á, Velten M,
Vatter H and Schuss P: Initial inflammatory response is an
independent predictor of unfavorable outcome in patients with
good-grade aneurysmal subarachnoid hemorrhage. J Crit Care.
60:45–49. 2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chamling B, Gross S, Stoffel-Wagner B,
Schubert GA, Clusmann H, Coburn M and Höllig A: Early diagnosis of
delayed cerebral ischemia: possible relevance for inflammatory
biomarkers in routine clinical practice? World Neurosurg.
104:152–157. 2017.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Turner CL, Budohoski K, Smith C,
Hutchinson PJ, Kirkpatrick PJ and Murray GD: STASH collaborators.
Elevated baseline C-reactive protein as a predictor of outcome
after aneurysmal subarachnoid hemorrhage: Data from the simvastatin
in aneurysmal subarachnoid hemorrhage (STASH) trial. Neurosurgery.
77:786–793. 2015.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Li Q and Gong X: Clinical significance of
the detection of procalcitonin and C-reactive protein in the
intensive care unit. Exp Ther Med. 15:4265–4270. 2018.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Li Y, Chen L, Fang W and Chen H:
Application value of procalcitonin, C-reactive protein and
interleukin-6 in the evaluation of traumatic shock. Exp Ther Med.
17:4586–4592. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Shi G, Li M, Zhou R, Wang X, Xu W, Yang F
and Xue S: Procalcitonin related to stroke-associated pneumonia and
clinical outcomes of acute ischemic stroke after IV rt-PA
treatment. Cell Mol Neurobiol. 42:1419–1427. 2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Schuetz P, Birkhahn R, Sherwin R, Jones
AE, Singer A, Kline JA, Runyon MS, Self WH, Courtney DM, Nowak RM,
et al: Serial procalcitonin predicts mortality in severe sepsis
patients: Results from the multicenter procalcitonin monitoring
SEpsis (MOSES) study. Crit Care Med. 45:781–789. 2017.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang R, He M, Ou XF, Xie XQ and Kang Y:
Serum procalcitonin level predicts acute kidney injury after
traumatic brain injury. World Neurosurg. 141:e112–e117.
2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Sakran JV, Michetti CP, Sheridan MJ,
Richmond R, Waked T, Aldaghlas T, Rizzo A, Griffen M and Fakhry SM:
The utility of procalcitonin in critically ill trauma patients. J
Trauma Acute Care Surg. 73:413–418. 2012.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Cabral L, Afreixo V, Meireles R, Vaz M,
Chaves C, Caetano M, Almeida L and Paiva JA: Checking procalcitonin
suitability for prognosis and antimicrobial therapy monitoring in
burn patients. Burns Trauma. 6(10)2018.PubMed/NCBI View Article : Google Scholar
|
|
22
|
van Gijn J, Kerr RS and Rinkel GJ:
Subarachnoid haemorrhage. Lancet. 369:306–318. 2007.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Thompson K, Venkatesh B and Finfer S:
Sepsis and septic shock: Current approaches to management. Intern
Med J. 49:160–170. 2019.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Frontera JA, Fernandez A, Schmidt JM,
Claassen J, Wartenberg KE, Badjatia N, Connolly ES and Mayer SA:
Defining vasospasm after subarachnoid hemorrhage: What is the most
clinically relevant definition? Stroke. 40:1963–1968.
2009.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Vergouwen MD, Vermeulen M, van Gijn J,
Rinkel GJ, Wijdicks EF, Muizelaar JP, Mendelow AD, Juvela S, Yonas
H, Terbrugge KG, et al: Definition of delayed cerebral ischemia
after aneurysmal subarachnoid hemorrhage as an outcome event in
clinical trials and observational studies: Proposal of a
multidisciplinary research group. Stroke. 41:2391–2395.
2010.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Höllig A, Remmel D, Stoffel-Wagner B,
Schubert GA, Coburn M and Clusmann H: Association of early
inflammatory parameters after subarachnoid hemorrhage with
functional outcome: A prospective cohort study. Clin Neurol
Neurosurg. 138:177–183. 2015.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Morga R, Dziedzic T, Moskala M, Slowik A
and Pera J: Clinical relevance of changes in peripheral blood cells
after intracranial aneurysm rupture. J Stroke Cerebrovasc Dis.
29(105293)2020.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Karlsson S, Heikkinen M, Pettila V, Alila
S, Väisänen S, Pulkki K, Kolho E and Ruokonen E: Finnsepsis Study
G. Predictive value of procalcitonin decrease in patients with
severe sepsis: A prospective observational study. Crit Care.
14(R205)2010.PubMed/NCBI View
Article : Google Scholar
|
|
29
|
Oconnor E, Venkatesh B, Mashongonyika C,
Lipman J, Hall J and Thomas P: Serum procalcitonin and C-reactive
protein as markers of sepsis and outcome in patients with
neurotrauma and subarachnoid haemorrhage. Anaesth Intensive Care.
32:465–470. 2004.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Festic E, Siegel J, Stritt M and Freeman
WD: The utility of serum procalcitonin in distinguishing systemic
inflammatory response syndrome from infection after aneurysmal
subarachnoid hemorrhage. Neurocrit Care. 20:375–381.
2014.PubMed/NCBI View Article : Google Scholar
|