Introduction
Uterine cystic adenomyosis is an uncommon
gynecological disease (1). Its
characteristic features include ectopic endometrial glands, stromal
invasion of the myometrium, cystic part of old blood, with diffuse
or focal widening of the junctional zone and bright foci or linear
striations in the myometrium on T2-weighted magnetic resonance
imaging (MRI) scans.
Buerger and Petzing (2) pointed out that, although uterine
adenomyosis may have a cystic part of old blood, the scope of the
area is small, generally not >5 mm, and large cystic lesions are
rare, which are called adenomyoma bone cyst (adenomyotic cyst),
bursa sex gland fibroids (cystic adenomyoma) or bursa sex steroid
myopathy (cystic adenomyosis). Large adenomyotic cysts (≥1 cm in
diameter) are uncommon; to date, only ~70 cases of this type of
lesion have been reported worldwide according to a literature
search performed in MEDLINE and the Foreign Medical Literature
Retrievals Service database using search term ‘adenomyotic cyst’
from 1992 to 2022.
The serum concentration of cancer antigen 125
(CA125) has been revealed to increase during endometriosis and
measurement of the serum CA125 concentration is widely used as a
supplementary test for diagnosing the disease and evaluating
therapeutic effects (3). Another
cancer antigen, CA19-9, has recently been reported to increase
endometriosis, but it rarely increases above 1,000 U/ml (4). However, the literature has reported
only seven cases of adenomyotic cyst with elevated CA19-9 or CA125
levels (Table I).
 | Table IDetails of previous similar
studies. |
Table I
Details of previous similar
studies.
| First author,
year | Region | Patient age,
years | Elevation of CA125 or
CA19-9a(U/ml) | Case description | (Refs.) |
|---|
| Kammerer-Doak,
1996 | Maricopa, USA | 37 | CA125: >1,000 | Benign gynecologic
condition presented in a case report. After excision of the
affected areas, the CA125 levels returned to normal | (10) |
| Imaoka, 2005 | Tenri, Japan | 41 | CA125: 673; CA19-9:
846 | Cystic adenomyosis
with florid glandular mass present as a huge exophytic cystic
mimicking an ovarian malignancy | (12) |
| Cucinella, 2013 | Palermo, Italy | 25 | CA125: 38.00; CA19-9:
88.19 | A 4.5-cm adenomyotic
cyst in a 25-year-old nulliparous female with severe dysmenorrhea
and pelvic pain | (5) |
| Pontrelli, 2015 | Negrar, Italy | 27 | CA125: 96 | Rare case of giant
cystic adenomyoma (8.0 cm) mimicking an uterine malformation,
diagnosed and treated by hysteroscopy | (9) |
| Isik, 2014 | Kirikkale,
Turkey | 47 | CA125: 63 | A giant adenomyotic
cyst originating from the cervix with an elevated serum
concentration of CA125 | (4) |
| Fan, 2019 | Jilin Province,
China | 39 | CA125: 1,212.00 | Two cases of
intrauterine cystic adenomyosis were treated by hysterectomy or
hysteroscopy to release the liquid inside, one of which had
elevated serum levels of CA125 | (11) |
| Zhao, 2021 | Chongqing, China | 30 | CA125: 76.20 | A rare case of a
cystic adenomyotic lesion that was treated by laparoscopic surgery;
CA125 levels were elevated | (8) |
The present report presents a case of a uterine
adenomyotic cyst with markedly elevated serum CA19-9 and CA125
levels.
Case report
Case presentation
A 32-year-old female [gravida 3 (referred for
artificial abortion due to abnormal fetal development in 2019; one
missed abortion in 2020), para 1 (gave birth to a live healthy
full-term baby in 2011)] presented at the Northwest Women's and
Children's Hospital (Xi'an, China). Ultrasonography was performed
during the follow-up after the missed abortion, revealing an
intrauterine cystic lesion without any symptoms of menorrhagia,
chronic pelvic pain or dysmenorrhea. Following that, a hematologic
examination and tumor marker detection were performed, which
revealed CA125 levels of 175 U/ml and CA19-9 of >1,000 U/ml.
Given the abnormally high tumor marker levels, abdominal CT
scanning and gastroscopy were all completed but did not reveal any
evidence of gastrointestinal malignant lesions. The patient
suddenly experienced pelvic pain and was referred to the department
for a thorough gynecological evaluation.
Measurement of serum CA19-9 and CA125 levels.
Blood samples were taken and serum was obtained according to
standard protocols. CA125 (normal ranges: CA125, 0-35 U/ml; CA19-9,
0-27 U/ml) were measured using the electrochemiluminescence
immunoassay on a Roche Cobas e 801 analyzer (Roche
Diagnostics).
Imaging. CT scanning parameters were
128x0.628 mm collimation with a reconstruction interval of 1 mm,
rotation time of 0.75 sec, tube voltage of 120 kV and 200 mAsec
(Brilliance iCT; Philips Healthcare).
A Color Doppler Ultrasound (U22; Philips Healthcare)
diagnostic instrument was adopted with 7.5 MHz endovaginal probe.
MRI was performed with a 1.5-T superconductive magnet unit (Picker
International Co. Ltd.). T1-weighted spin-echo images (T1-WI;
repetition time, 600 msec; echo time, 20 msec) and T2 weighted
spin-echo images (T2-WI; repetition time, 2,000 msec; echo time, 80
msec) were obtained.
Immunohistochemistry. Tissue samples were
fixed in a 10% (v/v) solution of buffered formalin for 24 h at 4˚C
and then dehydrated, cleared in xylene and embedded in paraffin.
Paraffin sections (5 mm) were mounted on silane-coated slides,
de-waxed and rehydrated. For antigen retrieval, the sections were
treated with 10 mM citrate buffer, pH 6.0 in a water bath (30 min
at 95˚C), then treated with 1.5% (v/v) H2O2
in methanol for 10 min for quenching the endogenous peroxidase
activity and finally equilibrated in 10 mM PBS-0.05% v/v Tween 20
pH 7.5. Nonspecific binding was reduced by incubation in 1% BSA
(Cytiva) for 60 min. Subsequently, the slides were incubated with
rabbit polyclonal anti-CA125 (cat. no. HPA065600; dilution, 1:100;
MilliporeSigma) or mouse monoclonal antibody anti-CA19-9 (cat. no.
399S; dilution, 1:100; MilliporeSigma) in a humidified chamber for
22 h at 4˚C. After washing in PBS-Tween 20, the sections were
incubated with biotinylated goat anti-rabbit or horse anti-mouse
immunoglobulin (cat. no. CY-4500 or CY-2500, respectively; 1:100
dilution; Vector Laboratories, Inc.; Maravai LifeSciences) for 30
min at 25˚C, and then treated with avidin-biotinylated peroxidase
complex (Vector Laboratories, Inc.; Maravai LifeSciences) for 30
min at 28˚C. The bound antibodies were visualized with
diaminobenzidine and H2O2 in PBS, pH 7.5
according to the manufacturer's instructions (Vector Laboratories,
Inc.; Maravai LifeSciences). Finally, the tissues were stained with
Gill's hematoxylin, dehydrated and mounted with coverslips.
Negative controls were prepared by substituting the primary
antibodies with rabbit IgG (cat. no. 0023305; dilution, 1:20;
Thermo Fisher Scientific, Inc.) or mouse IgG (cat. no. 12371;
dilution, 1:20; EMD Millipore).
History of menstruation. The patient had
attained menarche at the age of 14 years. The patient had a history
of a 28-day regular menstrual cycle without hypermenorrhea.
Pelvic examination. The uterus was displaced
to the right of the median line and was estimated to be larger than
a goose egg. There were no pelvic tenderness or nodules. Pelvic
examination revealed a normal vagina, vulva and adnexa and an
enlarged retroflexed uterus.
Hematologic examination. Laboratory tests
revealed anemia (hemoglobin concentration, 102 g/l (normal range,
115-150 g/l). The biochemical profile was otherwise unremarkable.
The serum concentration of CA125 and CA19-9 was elevated at 642
U/ml and >1,000 U/ml, respectively.
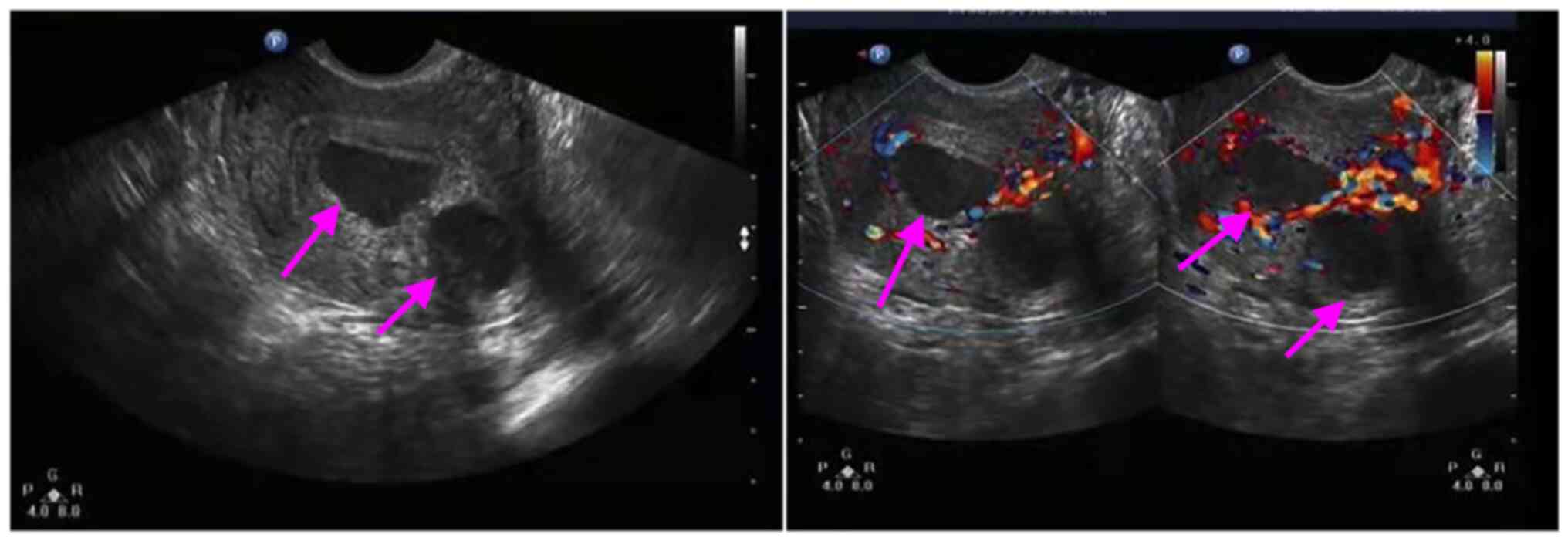
Ultrasonic examination. Ultrasonography
displayed a multi-locular cystic mass on the posterior wall of the
uterus. Color power Doppler revealed a difference from a leiomyoma.
The mass was diagnosed as uterine adenomyoma (Fig. 1). The ultrasound of the
hepatobiliary duct, pancreas and spleen exhibited no abnormalities.
An abdominal CT scan indicated no abnormal density lesions in the
liver, bile duct, pancreas, spleen or gastrointestinal tract.
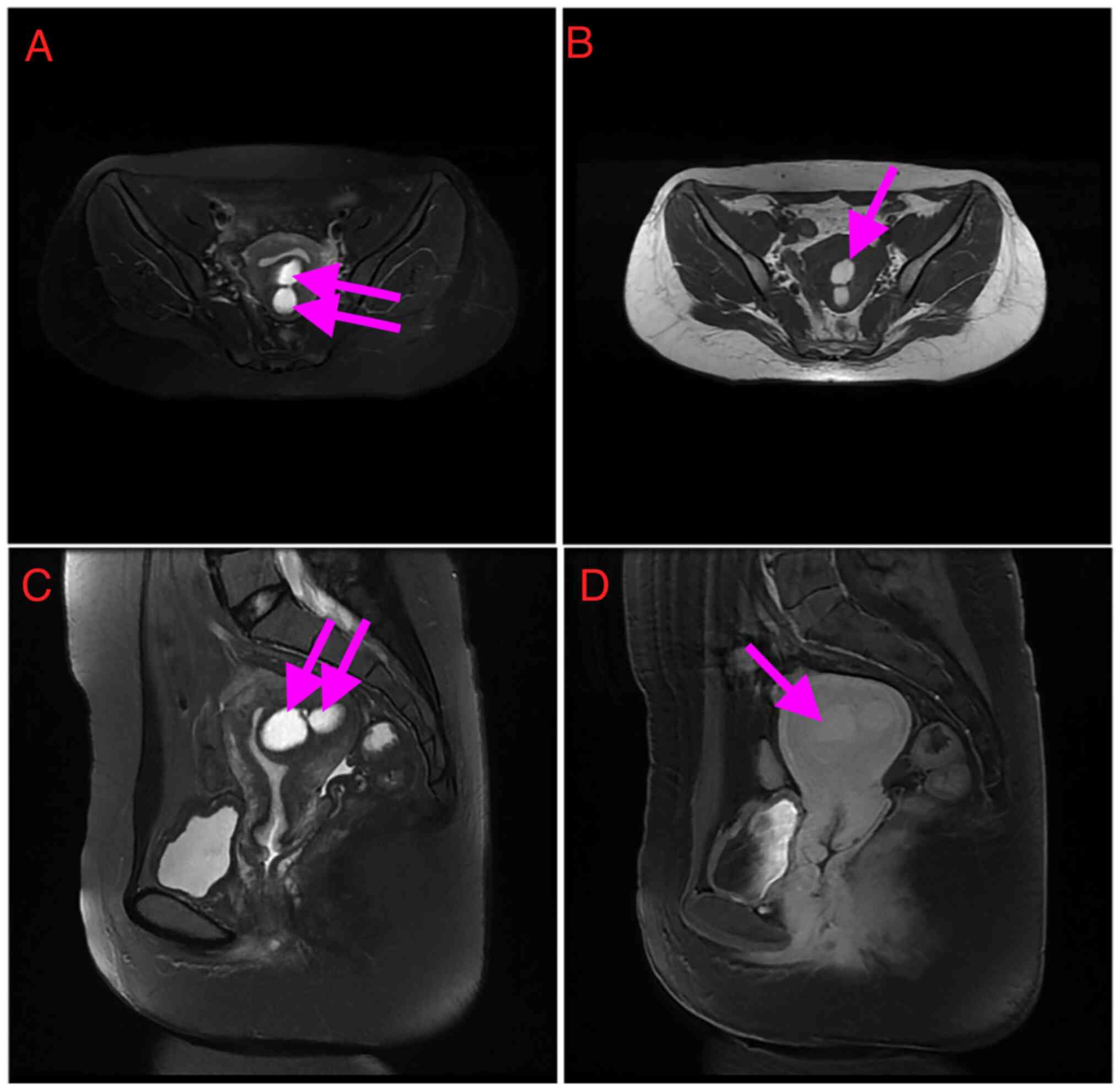
MRI findings. From the MRI findings (Fig. 2), the abnormal signal shadow and
central hemorrhagic cystic in the posterior compartment of the
uterus were considered adenomyoma. The mass measured 53x48x41 mm
and T2-WI displayed the cyst as an area of high intensity. The
normal uterine cavity was visualized as a line near the cyst. It
was slightly hyperintense on T1- and T2-WI, with an internal fluid
level, typical for the layering of simple and hemorrhagic or
proteinaceous fluids.
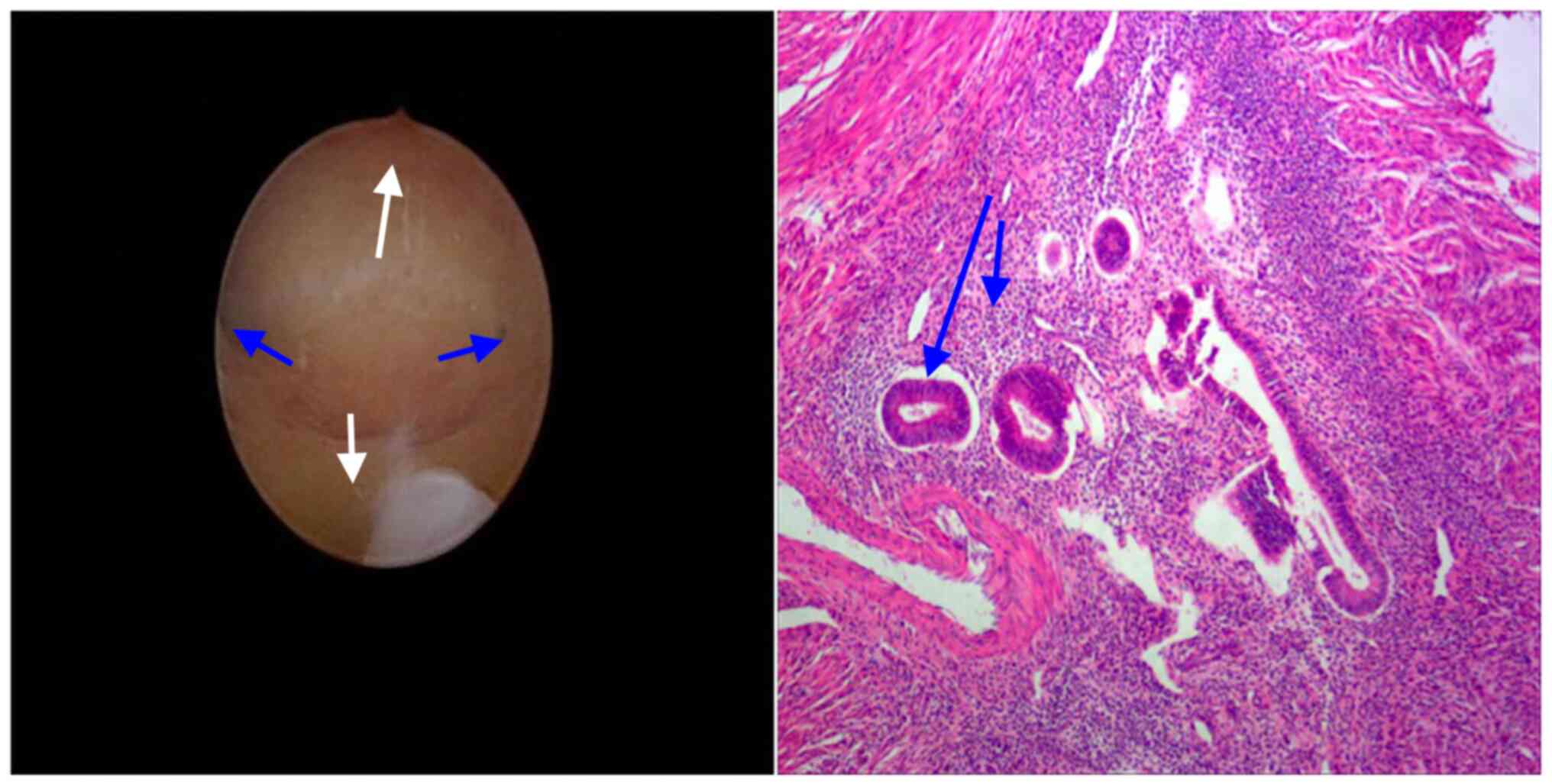
Hysteroscopy check. Hysteroscopy visualized a
normal uterine cavity and bilaterally patent tubal ostia (Fig. 3).
Intraoperative findings. Laparotomy surgery
was performed. The surgery was very similar to that in the case
report of Cucinella et al (5). Enlargement of the posterior wall of
the uterus was seen and chocolate-colored fluid flowed out of the
posterior wall after incision. No abnormality was found in the
appearance of bilateral fallopian tubes and ovaries. The excised
lesion was sent for pathological examination (Fig. 3).
Tissue distribution of CA19-9 and CA125. The
resected specimens were fixed in 10% formalin, embedded in paraffin
and examined by immunohistochemical staining with anti-CA125 and
anti-CA19-9 to determine the tissue distribution of each tumor
marker. CA19-9 and CA125 were not expressed in the adenomyotic cyst
which results not shown.
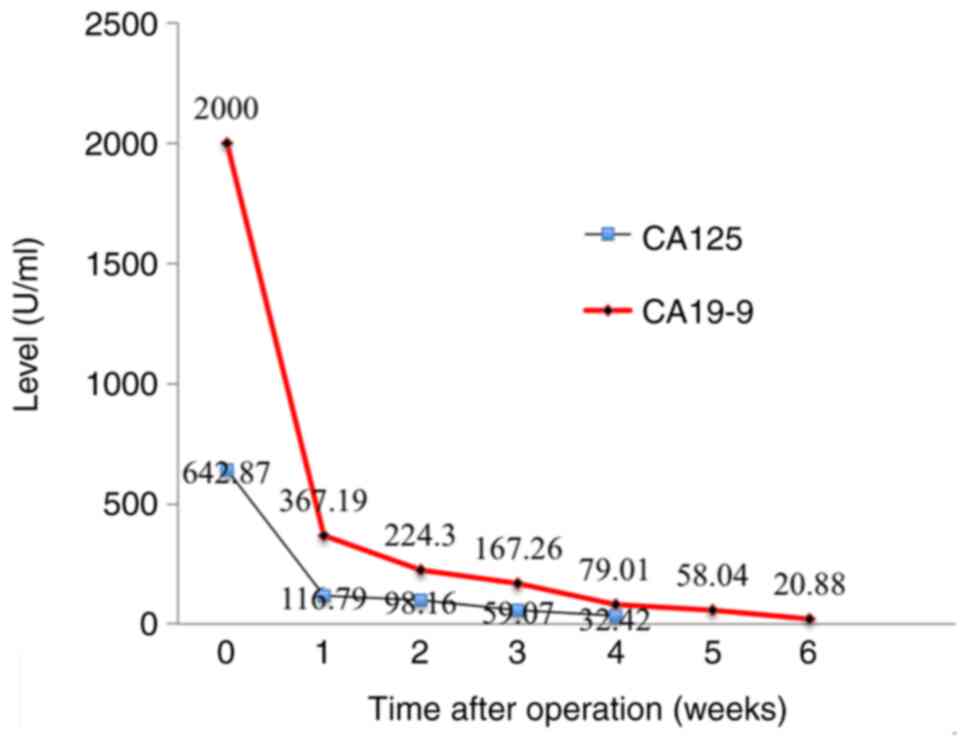
Changes in serum CA19-9 and CA125 levels.
Both tumor markers were assayed using commercially available
radioimmunoassay kits (Centocorl) once per week. CA125 decreased
below the cut-off level (0-35 U/ml) at ≥4 weeks after the
operation, but CA19-9 did not decrease below the cut-off level
(0-37 U/ml) until ≥6 weeks after the operation (Fig. 4). The patient underwent GnRha
therapy for six months and there was no evidence of recurrence
throughout the follow-up.
Discussion
CA19-9, first reported by Koprowski et al
(6), is mainly found in the fetal
stomach, intestinal epithelial cells and pancreas, and as a
carbohydrate antigen recognized by a monoclonal antibody in a human
colon cancer cell line. In addition to being a highly sensitive
tumor marker for pancreatic and bile duct cancers, it has been
reported to be highly sensitive for gynecologic tumors, such as
endometrial carcinoma or ovarian cancer. Recently, CA19-9 was also
reported to be increased in patients with endometriosis and
adenomyosis (7), but it rarely
exceeds 1,000 U/ml, as it did in the present case.
Increased serum CA125 levels have also been proposed
as a diagnostic tool for cystic adenomyosis. Cucinella et al
(5) and Zhao et al
(8) respectively described a large
adenomyotic cyst with slightly elevated CA125. Furthermore, two
cases of giant adenomyotic cyst originating from the cervix with
raised CA125 levels were also reported by Pontrelli et al
(9) and Isik et al
(4). Certain studies reported that
preoperative CA125 levels as high as 1,212.00 U/ml returned to
normal after excision of these areas (10,11).
Serum CA125 levels are generally elevated in patients with giant
adenomyotic cyst.
A large-magnitude increase in the serum levels of
both CA125 and CA19-9 in patients with cystic adenomyosis is rare
(12,13). In the present case, serum CA125 was
642 U/ml and CA19-9 was as high as >1,000 U/ml prior to surgery,
which decreased after tumor removal, consistent with previous
reports. The present study expands the knowledge on cases with
significantly increased CA19-9. However, it was not possible to
determine the reason for the abnormally high serum level of CA19-9.
Similarly, MRI and other imaging studies (14,15)
suggest that hemorrhage in uterine adenomyosis lesions in the
present case may damage the ovaries, uterus and nearby tissues,
causing surrounding tissue adhesion, damaging the peritoneal
barrier and possibly causing CA19-9 leakage into the circulation.
In benign biliary tract diseases such as acute cholangitis, the
bile duct epithelium is reported to be damaged by inflammation and
biliary CA19-9 and CA125 may leak into the circulation (3). The differential diagnosis (such as
biliary tract diseases, ovarian disease and enteropatia), was
excluded by ultrasonic examination, MRI findings, CT scan,
hysteroscopy check and pathological results.
MRI imaging and 3D ultrasound have recently been
clinically applied in the gynecologic field, with reports that may
be useful for the diagnosis of adenomyosis (16). MRI may now detect the normal
junctional zone and adenomyotic cysts (17). The Myometrial location (intramural,
submucous, subserous), Uterine site (midline, paramedian, lateral);
Structure (cystic, mixed, polypoid), Contents (clear, hemorrhagic),
Level (fundus, body, cervix) and Endometrial or inner lining
(endometrium, metaplastic) (MUSCLE) classification was suggested
for evaluating cystic adenomyosis (18). The patient of the present study may
be classified as an intramural cystadenoma, namely M1 U1 S1 C2 L2
E2. MRI is considered clinically valuable in this respect. To
distinguish the condition from other types of intramural cyst,
histological diagnosis is necessary to identify the endometrial
inner layering and the presence of outer myometrium.
To prevent malignant tumor spread, a laparotomy
operation was performed. There is doubt regarding the nature of
such rare voluminous cysts in young females prior to surgery,
considering patient age at symptom occurrence (8).
The postoperative decrease of CA19-9 was slower than
that of CA125. It remains unclear whether this difference was
caused by a difference in the half-life of these two tumor markers
in the circulation. However, unlike CA125 in adenomyotic cyst,
CA19-9 appears to not be a sensitive marker of adenomyotic
cyst.
In conclusion, larger adenomyotic cysts are rare,
while small adenomyotic cysts of <5 mm in diameter have been
detected in hysterectomy specimens. In addition, 70 cases of
adenomyotic cyst have so far been reported in the literature. In
the case of the present study, serum CA19-9 and CA125 levels were
significantly increased, which has been rarely reported. Due to
these laboratory test results, it was not possible to make a
confident differentiation of benign vs. malignant tumors. The
ultrasound results and MRI suggested a diagnosis of a mass of
uterine origin. In addition, the operation and pathology
examination confirmed cystic lesions. However, additional case
reports are required to further elucidate the pathogenesis and
clinical characteristics of this rare disorder.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Shanghai Yangpu
District Health and Family Planning Commission Fund for Hao Yi Shi
Training Project (grant nos. 201742 and 2020-2023), the Natural
Science Foundation of Shanghai (grant no. 18ZR1436000) and by the
clinical research project fund for Northwest Women's and Children's
Hospital (grant no. 2022YN08).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
LZ substantially contributed to the conception or
design of the work, as well as the acquisition, analysis and
interpretation of data for the work. LS performed the experiments,
analyzed the data and drafted the manuscript. FC made contributions
to the analysis and interpretation of data. All authors checked and
approved the authenticity of all the raw data. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The study protocol was reviewed and approved by the
Medical Ethics committee of Northwest Women's and Children's
Hospital (Xi'an, China; approval no. 21-039).
Patient consent for publication
Written informed consent was obtained from the
patient for the publication of the details of her medical case and
any accompanying images.
Competing interests
The authors have no competing interests to
declare.
References
|
1
|
Lacheta J: Uterine adenomyosis:
Pathogenesis, diagnostics, symptomatology and treatment. Ceska
Gynekol. 84:240–246. 2019.PubMed/NCBI
|
|
2
|
Buerger PT and Petzing He: Congenital
cysts of the corpus uteri. Am J Obstet Gynecol. 67:143–151.
1954.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Takemori M and Sugimura K: Ovarian
chocolate cyst with markedly elevated serum CA19-9 level: A case
report. Eur J Obstet Gynecol Reprod Biol. 42:241–244.
1991.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Isik Y, Dag ZO and Celik H: A giant
adenomyotic cyst originating from the cervix. Int J Gynaecol
Obstet. 131:205–206. 2015.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Cucinella G, Billone V, Pitruzzella I, Lo
Monte AI, Palumbo VD and Perino A: Adenomyotic cyst in a
25-year-old woman: Case report. J Minim Invasive Gynecol.
20:894–898. 2013.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Koprowski H, Steplewski Z, Mitchell K,
Herlyn M, Herlyn D and Fuhrer P: Colorectal carcinoma antigens
detected by hybridoma antibodies. Somatic Cell Genet. 5:957–971.
1979.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rokhgireh S, Mehdizadeh Kashi A, Chaichian
S, Delbandi AA, Allahqoli L, Ahmadi-Pishkuhi M, Khodaverdi S and
Alkatout I: The Diagnostic accuracy of combined enolase/Cr, CA125,
and CA19-9 in the detection of endometriosis. Biomed Res Int.
2020(5208279)2020.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zhao CZ, Wang B, Zhong CY, Lu ST and Lei
L: Management of uterine cystic adenomyosis by laparoscopic
surgery: Case report. BMC Womens Health. 21(263)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Pontrelli G, Bounous VE, Scarperi S,
Minelli L, Di Spiezio Sardo A and Florio P: Rare case of giant
cystic adenomyoma mimicking a uterine malformation, diagnosed and
treated by hysteroscopy. J Obstet Gynaecol Res. 41:1300–1304.
2015.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Kammerer-Doak DN, Magrina JF, Nemiro JS
and Lidner TK: Benign gynecologic conditions associated with a
CA-125 level > 1,000 U/mL. A case report. J Reprod Med.
41:179–182. 1996.PubMed/NCBI
|
|
11
|
Fan YY, Liu YN, Li J and Fu Y:
Intrauterine cystic adenomyosis: Report of two cases. World J Clin
Cases. 7:676–683. 2019.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Imaoka I, Kaji Y, Kobashi Y, Wada A, Honjo
G, Hayashi M, Yoshida M and Matsuo M: Cystic adenomyosis with
florid glandular differentiation mimicking ovarian malignancy. Br J
Radiol. 78:558–561. 2005.PubMed/NCBI View Article : Google Scholar
|
|
13
|
AL-Tai TH, AL-Hadithi HS and Abdulsalam
HS: CA19-9 versus CA-125 in Endometriosis. J Dent Med Sci.
13:27–30. 2014.
|
|
14
|
Kataoka ML, Togashi K, Konishi I, Hatabu
H, Morikawa K, Kojima N, Kuroda H, Fujimoto R, Kataoka N and
Konishi J: MRI of adenomyotic cyst of the uterus. J Comput Assist
Tomogr. 22:555–559. 1998.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Vinci V, Saldari M, Sergi ME, Bernardo S,
Rizzo G, Porpora MG, Catalano C and Manganaro L: MRI, US or
real-time virtual sonography in the evaluation of adenomyosis?
Radiol Med. 122:361–368. 2017.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Gordts S, Campo R and Brosens I:
Hysteroscopic diagnosis and excision of myometrial cystic
adenomyosis. Gynecol Surg. 11:273–278. 2014.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Ho ML, Raptis C, Hulett R, McAlister WH,
Moran K and Bhalla S: Adenomyotic cyst of the uterus in an
adolescent. Pediatr Radiol. 38:1239–1242. 2008.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bazot M and Daraï E: Role of transvaginal
sonography and magnetic resonance imaging in the diagnosis of
uterine adenomyosis. Fertil Steril. 109:389–397. 2018.PubMed/NCBI View Article : Google Scholar
|