Introduction
Lung cancer is one of the most prevalent malignant
tumors and seriously threatens human health, since it can
drastically reduce the quality of life (1). The latest statistics show that there
are about 19.3 million new lung cancer cases and nearly 10 million
mortalities worldwide (2).
Non-small cell lung cancer (NSCLC) is the most common pathological
subtype (~80%) of lung cancer globally (3). Despite the availability of various
treatment options for NSCLC, such as surgery, chemotherapy,
nanomedicines, photodynamic therapy and immune checkpoint
inhibitors, its mortality rate remains high (4-6).
It is therefore important to uncover the mechanism that can mediate
the process of NSCLC development. The occurrence and progression of
NSCLC have been reported to be influenced by genetic factors and
various environmental factors, such as smoking, air pollution and
occupational exposure (7).
Previous studies have suggested that pseudogenes
serve an important role in the development of cancer (8,9).
Pseudogenes typically arise from mutations or inactivation of
genes, which accumulate during the evolution process (10). Initially, they were dismissed as
non-functional genomic junk (11).
However, recent studies have revealed that dysregulated
pseudogenes, such as double homeobox a pseudogene 8 (DUXAP8) and
cathepsin l pseudogene 8 (CTSLP8) can affect oncogenic progression
by regulating that of particular cancer-related genes (12,13).
In particular, pseudogene-derived long non-coding RNAs (lncRNAs),
such as DUXAP8 and KRT17P3 (keratin 17 pseudogene 3), which can
function as either oncogenes or tumor suppressors, have been
previously implicated in cell proliferation, migration and
invasion, in addition to drug resistance in NSCLC (12,14).
However, the underlying molecular mechanisms of a large number of
pseudogenes remain unclear.
Chondroitin sulfate proteoglycan 4 (CSPG4)
pseudogene 12 (CSPG4P12) is a pseudogene-derived lncRNA that is
highly homologous with its parent gene CSPG4(15). CSPG4 has been reported to serve an
important role in the occurrence and development of various types
of cancer, such as anaplastic thyroid cancer, squamous cell
carcinoma of head and neck and basal breast carcinoma (16,17).
Functionally, CSPG4 is a tumor cell surface cancer antigen that can
confer phenotypic heterogeneity and mediate malignant progression
in epithelial ovarian cancer (18). It has also been previously revealed
that the overexpression of CSPG4 in anaplastic thyroid cancer (ATC)
can increase tumor size of ATC patient-derived xenografted mice
(17). In addition, a number of
studies have identified pseudogenes, such as PTENP1 (phosphatase
and tensin homolog pseudogene 1) and AGPG (actin gamma 1
pseudogene), to be important components of gene regulation networks
(19,20). Therefore, present study
hypothesized that CSPG4P12 may be involved in the occurrence and
progression of NSCLC.
In the present study, after analyzing the
differences of the expression of CSPG4P12 between NSCLC tissues and
adjacent normal tissues, the effect of CSPG4P12 on the
physiological behaviors of NSCLC was evaluated to assess its
potential mechanism.
Materials and methods
Tissue specimens
A total of 19 NSCLC tissues and corresponding
adjacent healthy tissues were obtained from Tangshan Gongren
Hospital Affiliated to North China University of Science and
Technology (NCST; Tangshan, China) from March 2021 to July 2021.
All subjects (age range, 43-81 years; median age, 63 years; 11
males and 8 females) were included with postoperative pathological
diagnosis of NSCLC and individuals who had received any
preoperative treatment were excluded. The present study was
approved by the Ethics Committee of NCST (approval no. 2021036) and
all participants or their family members signed informed consent
forms.
Differential expression analysis
Gene expression profiling interactive analysis 2
database (http://gepia2.cancer-pku.cn/#analysis) was utilized to
perform the differential expression analysis of CSPG4P12 between
cancer tissues and adjacent normal tissues in NSCLC (21). Based on datasets from The Cancer
Genome Atlas and genotype-tissue expression project, 483 lung
adenocarcinoma and 347 healthy tissues, 486 lung squamous cell
carcinoma tissues and 338 healthy tissues were involved in the
present study using a standard processing pipeline. The key search
terms used were as follows: ‘CSPG4P12’ in the search box on the
home page, then ‘Boxplots’; other parameters were the initial value
and the ‘LUAD’ and ‘LUSC’ data sets were selected.
Cell culture and plasmid
transfection
NSCLC A549 and H1299 cell lines (Procell Life
Science & Technology Co., Ltd.) were cultured in RPMI 1640
medium (Thermo Fisher Scientific, Inc.) supplemented with 10% FBS
(Zhejiang Tianhang Biotechnology Co., Ltd.) at 37˚C with 5%
CO2. The CSPG4P12-pUC57 plasmid was constructed by
Changzhou Ruibo Bio-Technology Co., Ltd. In brief, CSPG4P12
(ENST00000558282.5; CSPG4P12-201; 4,853 bp) was amplified using KOD
FX (cat. no. KFX-101; Toyobo Life Science) and the following
primers (SinoGenoMax Co., Ltd.): 5'-CTAGTCTAGACACCTGGGCACCAACCTC-3'
(XbaI) and 5'-ACGCGTCGACATAGAAAACAGCCCCAACCAG-3'
(SalI). The thermocycling conditions were: Pre-denaturation
at 95˚C for 3 min; followed by 25 cycles at 95˚C for 25 sec, at
60˚C for 20 sec and 72˚C for 40 sec; final extension at 72˚C for 1
min. The PCR product was recombined into the pUC57 vector (429-452
nt) to generate the CSPG4P12 overexpression plasmid
(CSPG4P12-pUC57), which was verified by Sanger sequencing.
Cells were seeded into six-well plates with a
density of 1x106 cells/well. Cells were transfected at
~80% confluency with 2.5 µg CSPG4P12-pUC57 or empty plasmid (pUC57)
using Lipofectamine® 2000 (Thermo Fisher Scientific,
Inc.) at 37˚C for 5 h according to the manufacturer's protocols.
Cells were then harvested for further analysis after 24 h. Cells
were transfected with (negative control) or without (blank control)
2.5 µg pUC57 plasmid for detected the effects of pUC57 plasmid on
the proliferation of NSCLC cells.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted using the TRIzol®
reagent (Invitrogen; Thermo Fisher Scientific, Inc.). According to
the manufacturer's instructions, cDNA was synthesized from 2 µg
total RNA using RevertAid First Strand cDNA Synthesis kit (cat. no.
K1622; Thermo Fisher Scientific, Inc.). 20 ng of cDNA was then used
for qPCR with the Power SYBR-Green PCR Master Mix (cat. no. A25742;
Thermo Fisher Scientific, Inc.) via a 7900HT Fast Real-Time PCR
System (Thermo Fisher Scientific, Inc.) with GAPDH as the internal
reference. The following primer pairs, which were obtained from
SinoGenoMax Co., Ltd., were used for qPCR: CSPG4P12 forward,
5'-ATGGACCAGTACCCCACACG-3' and reverse, 5'-CCCTGCCTCTAGCCATTGAC-3'
and GAPDH forward, 5'-CTGGGCTACACTGAGGACC-3' and reverse,
5'-AAGTGGTCGTTGAGGGCAATG-3'. The thermocycling conditions were as
follows: Pre-denaturation at 95˚C for 2 min; followed by 40 cycles
at 95˚C for 15 sec and at 59˚C for 1 min; final extension at 72˚C
for 10 min. The relative expression levels of CSPG4P12 were
calculated using the 2-∆∆Cq method (22).
Cell proliferation assay
CCK-8 assay was performed to detect cell viability.
A total of 5x103 transfected cells/well were seeded into
a 96-well plate. After culturing for 24, 48 and 72 h at 37˚C, cells
were mixed with 10 µl CCK-8 reagent (Dojindo Molecular
Technologies, Inc.) for 2 h at 37˚C. The absorbance at 450 nm was
measured using the Infinite M200 PRO Microplate Reader (Tecan
Group, Ltd.).
Colony formation assay
For the colony formation assay, transfected A549 or
H1299 cells were seeded into 60-mm dishes at a density of
2x103 cells/dish and allowed to grow in RPMI 1640 medium
supplemented with 10% FBS at 37˚C for 10 days, then fixed with 4%
polyformaldehyde at 37˚C for 30 min and stained with 0.1% crystal
violet (Beijing Solarbio Science & Technology Co., Ltd.) at
37˚C for 10 min. A light microscope (magnification, x40; Olympus
Corporation) was used for observation. Each colony contained >50
cells, ranging in size from 0.3-1.0 mm. The colonies were detected
using ImageJ (v1.42q; National Institutes of Health).
Cell migration and invasion
Transwell assay was performed to measure cell
migration and invasion. A total of 7x104
plasmid-transfected cells in RPMI1640 medium were seeded into the
upper chambers (6.5-mm diameter inserts; 8-µm pore size; Costar;
cat. no. 3422; Corning, Inc.) with or without 50 mg/l Matrigel
precoating (Corning, Inc.) at 37˚C for 5 h. The lower chamber was
filled with RPMI1640 medium supplemented with 20% FBS. Cells
attached in the upper chamber were then removed after 24 (for
migration assay) or 48 h (for invasion assay) at 37˚C. The chambers
were fixed with 4% paraformaldehyde for 30 min at 37˚C and then
stained with 0.1% crystal violet at 37˚C for 10 min. Migrating or
invading cells were then observed and imaged under an inverted
light microscope (magnification, x100; Olympus Corporation). A
total of five fields (upper, lower, left, right, and middle) were
selected per chamber for quantification.
Wound healing and adhesion assay
Untransfected A549 or H1299 Cells were seeded into a
six-well plate at a density of 1x106 cells/well and
cultured in RPMI1640 medium supplemented with 1% FBS until ~80%
confluency. The cell monolayer was then scraped in a straight line
to create a ‘scratch’ with a 200 µl pipette tip. Images were
captured under an inverted light microscope (magnification, x40)
after 0, 24 and 48 h incubation at 37˚C. The width of wound was
detected using ImageJ (v1.42q; National Institutes of Health). To
measure cell adhesion, a 96-well plate was coated with 50 mg/l
Matrigel at 37˚C for 5 h, then blocked with 1% bovine serum albumin
(Thermo Fisher Scientific, Inc.) at 37˚C for 2 h. Transfected cells
were then seeded at the density of 2x104 cells/well. The
images under an inverted light microscope (x100; Olympus
Corporation) were captured at 30, 60 and 90 min timepoints at 37˚C.
CCK-8 assay was utilized to detect the cell proliferation on a
microplate reader as aforementioned.
Hoechst 33342 staining assay
Transfected cells were cultured for 48 h at 37˚C,
fixed with 4% polyformaldehyde for 30 min at 37˚C, washed with PBS
and then stained with 1 µg/ml Hoechst33342 (Beijing Solarbio
Science & Technology Co., Ltd.) in the dark for 20 min at 37˚C.
Nuclear condensation and fragmented nuclei was observed under a
fluorescent inverted microscope (magnification, x100; Olympus
Corporation).
Transmission electron microscopy
(TEM)
Cells were collected after transfection, and
centrifuged at 1,000 x g for 10 min at 37˚C to form a pellet. Cell
pellets were fixed with 2.5% glutaraldehyde at 4˚C overnight and
then washed three times with 0.1 M PBS (pH 7.4) for 15 min. The 1%
osmium tetroxide was fixed again at room temperature for 2 h in the
dark and then washed 15 min for three times with 0.1M PBS (pH 7.4).
A gradient dehydration was performed using ascending concentrations
of ethanol. The samples were embedded with the 812 resin embedding
medium (Beijing Zhongjingkeyi Technology Co., Ltd.) overnight at
37˚C and subsequently polymerized at 60˚C for 12 h. The embedded
samples were cut into 60-nm ultra-thin sections using a microtome
(EM UC6; Leica Microsystems, Inc.). Finally, the sections were
double-stained with 3% lead citrate and 1.5% uranyl acetate at 37˚C
for 20 min. Observation and acquisition of images were performed by
TEM (x10,000; H-7650; Hitachi, Ltd.).
Immunofluorescence (IF) assay
Untransfected A549 or H1299 Cells were seeded into a
six-well plate at a density of 5x105 cells/well. After
24 h at 37˚C, cells were fixed with 4% paraformaldehyde at room
temperature for 30 min, then 0.3% Triton X-100 was added (Beijing
Solarbio Science & Technology Co., Ltd.) for 15 min at 37˚C.
After blocking with 5% BSA for 1.5 h at 37˚C, cells were incubated
with the anti-Bax antibody (1:2,000; cat. no. ab32503; Abcam) and
anti-Bcl2 antibody (1:2,000; cat. no. ab32124; Abcam) overnight at
4˚C. The cells were then incubated with Alexa Fluor™ 488 goat
anti-rabbit IgG (1:5,000; cat. no. A11008; Thermo Fisher
Scientific, Inc.) at 37˚C for 1 h in the dark. Following
incubation, cells were stained with 1 µg/ml DAPI solution (BD
Biosciences) in the dark for 15 min at 37˚C. A fluorescent inverted
microscope (magnification, x200; Olympus Corporation) was utilized
to capture images of the samples.
Western blot analysis
Total protein was extracted from cells using RIPA
lysis buffer (Pierce; Thermo Fisher Scientific, Inc.). Protein
concentration was measured using the Pierce BCA Protein Assay kit
(Thermo Fisher Scientific, Inc.). Protein samples (50 µg) were
separated by SDS-PAGE on an 8% gel and then transferred onto
nitrocellulose membranes (Merck KGaA). The separated proteins were
subsequently blocked with 5% milk at room temperature for 2 h. The
membranes were incubated at 4˚C overnight with the following
primary antibodies: Anti-β-actin (1:2,000; cat. no. 66009-1-Ig;
ProteinTech Group, Inc.); anti-Bax (1:2,000; cat. no. ab32503;
Abcam); anti-Bcl2 (1:2,000; cat. no ab32124; Abcam); and the p53
polyclonal antibody (1:2,000; cat. no. 10442-1-AP; ProteinTech
Group, Inc.). Following incubation with primary antibodies, the
membranes were incubated with HRP-conjugated goat anti-rabbit IgG
(1:5,000; cat. no. ZB2301; OriGene Technologies, Inc.) or goat
anti-mouse IgG (1:5,000; cat. no. S0002; Affinity Biosciences,
Ltd.) secondary antibodies for 1.5 h at 37˚C. Protein bands were
visualized using an ECL kit (cat. no. RPN2232; Cytiva) and a gel
imaging system (Bio-Rad Laboratories, Inc.), then analyzed using
ImageJ (v1.42q; National Institutes of Health).
Statistical analysis
Statistical analysis was performed using SPSS
(version 23.0; IBM Corp.). Each assay was repeated three times. The
data are presented as mean ± standard deviation. The difference in
CSPG4P12 expression between cancer tissues and adjacent normal
tissues was analyzed using paired Student's t-test. The one-way
ANOVA test followed by Bonferroni's post hoc correction was
utilized to evaluate the result of wound healing assay. The
two-tailed Student's t-test was utilized for the analysis of the
other experimental data. P<0.05 was considered to indicate a
statistically significant difference. The inspection level α
was 0.05.
Results
Expression of CSPG4P12 is lower in
NSCLC tissues compared with that in the adjacent healthy
tissues
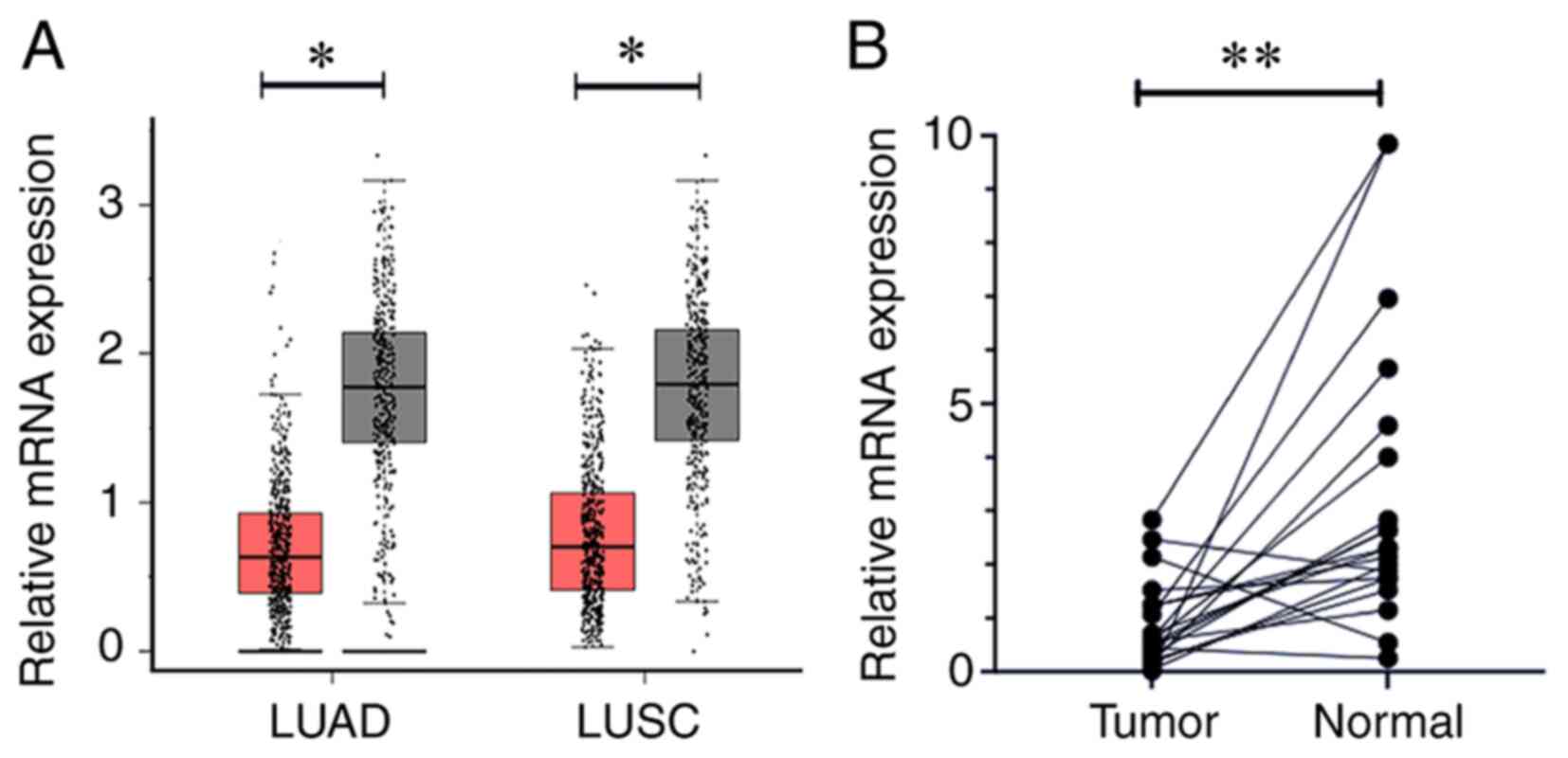
GEPIA data demonstrated that the expression level of
CSPG4P12 in NSCLC tissues was significantly lower compared with
that in the adjacent healthy tissues (P<0.05; Fig. 1A). To validate this finding, the
expression of CSPG4P12 was detected in 19 pairs of NSCLC tissues
and adjacent healthy tissues, which yielded the same outcome
(P<0.01; Fig. 1B).
Overexpression of CSPG4P12 inhibits
NSCLC cell proliferation
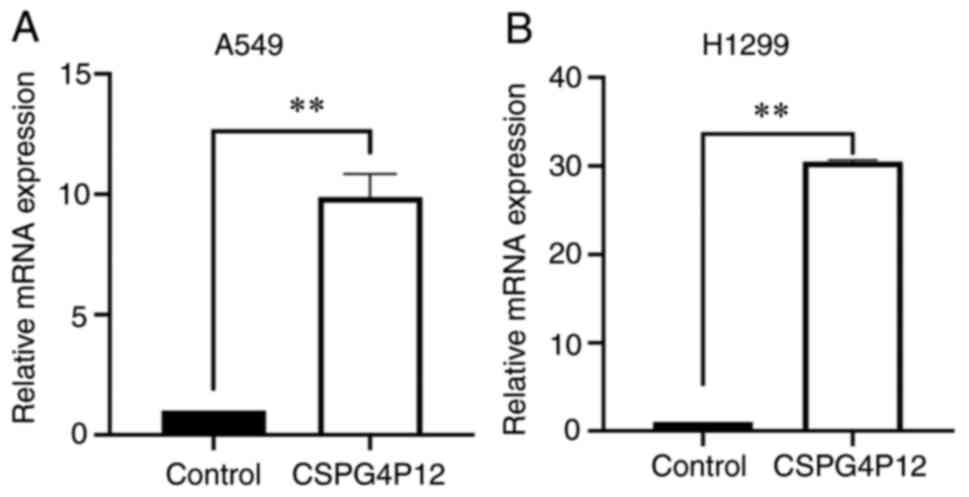
After transfection with CSPG4P12-pUC57, the
expression levels of CSPG4P12 were significantly higher compared
with those in the control pUC57 plasmid group in both A549
(P<0.01; Fig. 2A) and H1299
cells (P<0.01; Fig. 2B). This
suggests that CSPG4P12-pUC57 was successfully transfected into the
NSCLC cells tested in the present study. Fig. S1 indicated that transfection of
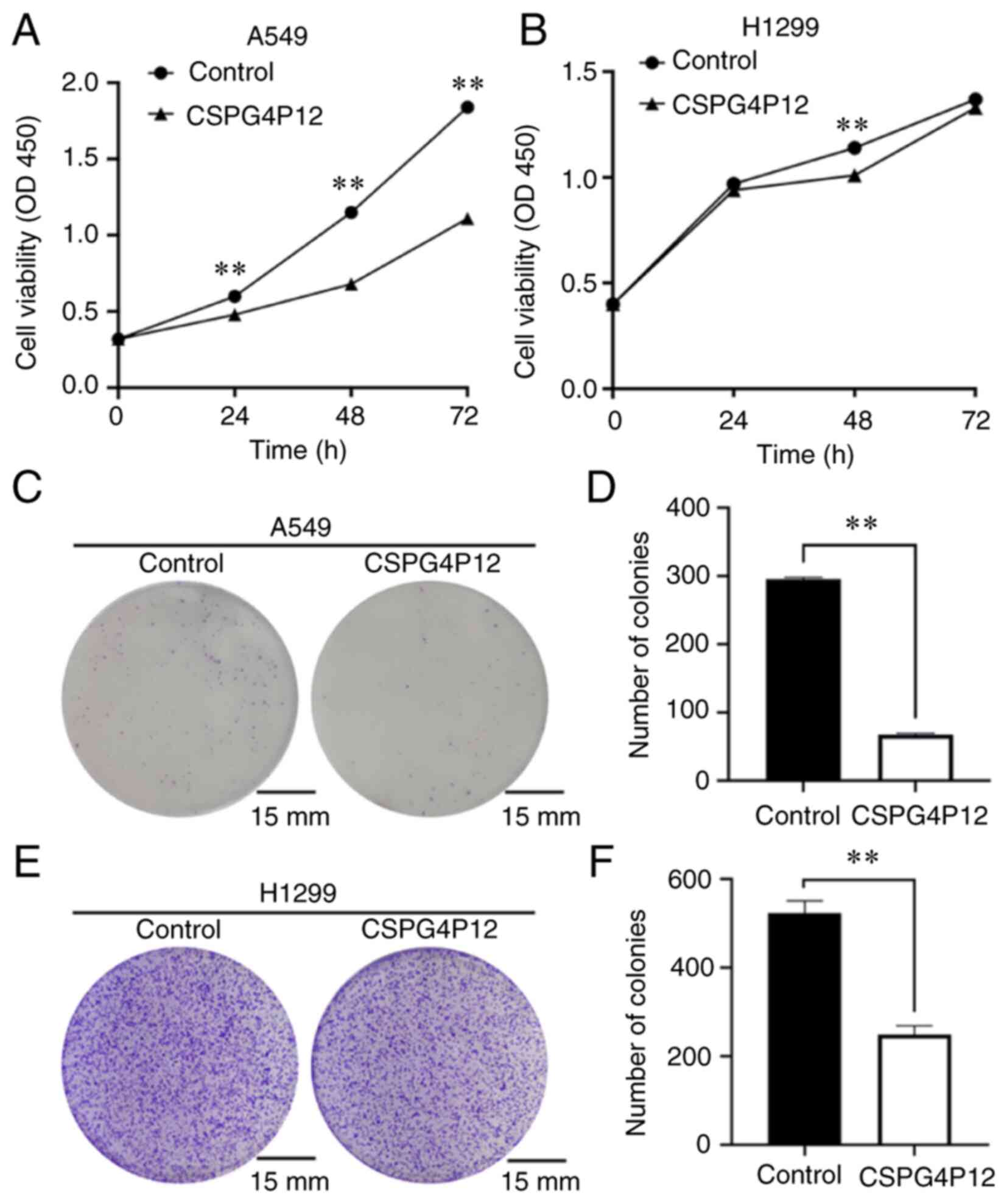
pUC57 plasmid had no effect on cell proliferation. CCK-8 assay
results showed that, after CSPG4P12 overexpression for 24, 48 and
72 h, the A549 cell viability was significantly reduced (P<0.01;
Fig. 3A). However, it was only at
the 48 h timepoint that H1299 cell viability was significantly
reduced compared with that in the control pUC57 plasmid group
(P<0.01; Fig. 3B). Colony
formation assay revealed that the number of cell clones in the
CSPG4P12 overexpression group was significantly reduced compared
with that in the control plasmid group (P<0.01; Fig. 3C and D).
Overexpression of CSPG4P12 suppresses
migration, invasion and adhesion of NSCLC cells
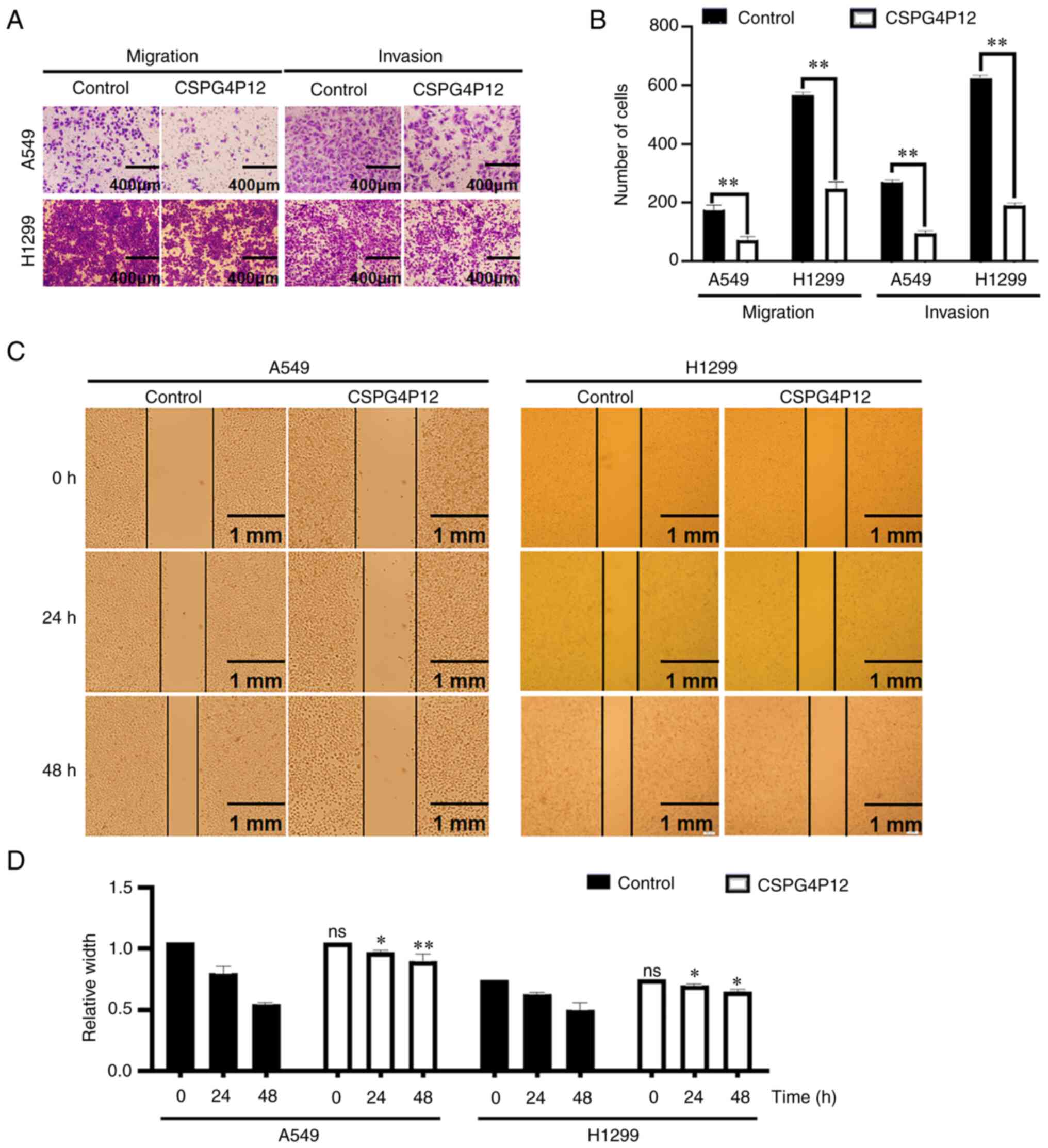
Transwell migration and invasion assays showed that
the cell migration and invasion in the CSPG4P12 overexpression
group were significantly lower compared with that in the control
plasmid group in both cell lines tested (P<0.01; Fig. 4A and B). Wound healing assay showed that, after
CSPG4P12 overexpression for 24 and 48 h, wound healing was
significantly reduced, which suggested that CSPG4P12 overexpression
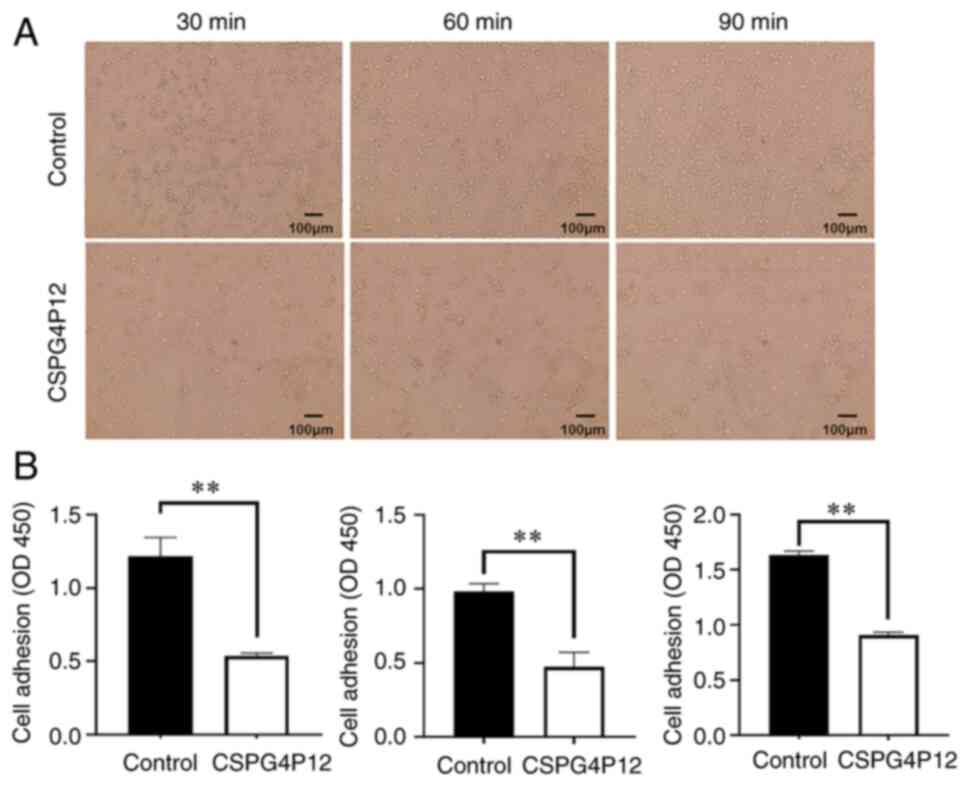
impaired cell migration (P<0.01; Fig. 4C and D). Cell adhesion experiments indicated
that the overexpression of CSPG4P12 significantly decreased A549
cell adhesion 30, 60 and 90 min after transfection with
CSPG4P12-pUC57 (P<0.01; Fig.
5).
Overexpression of CSPG4P12 promotes
apoptosis by the mitochondria apoptosis pathway
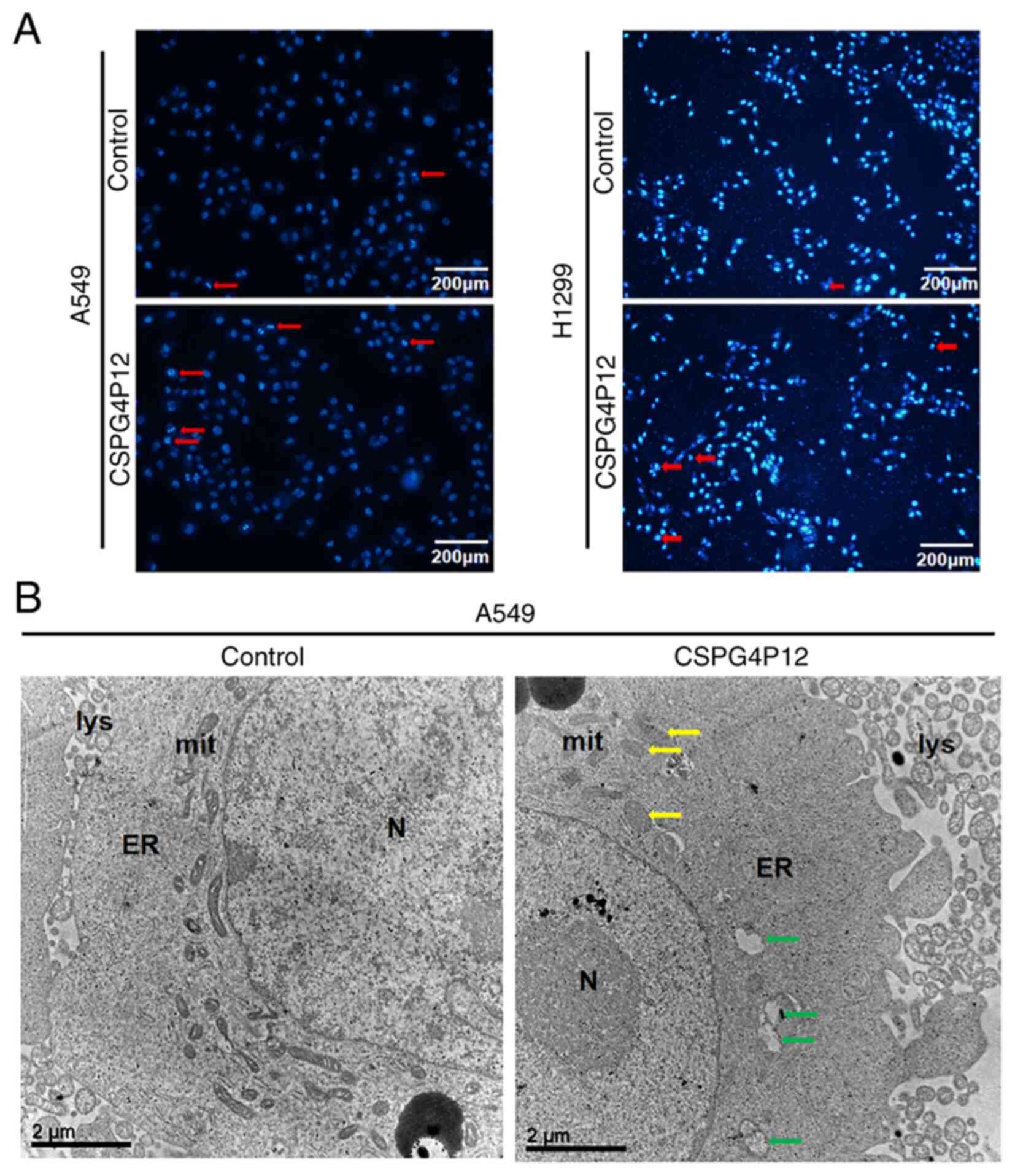
Hoechst33342 fluorescence assay showed that the
number of apoptotic cells (dark blue with pyknosis or division) in
the control group was markedly lower compared with that in the
CSPG4P12 overexpression group (Fig.
6A). In addition, under TEM, some mitochondrial cristaes in the
CSPG4P12 overexpression group were either reduced or disappeared,
where vacuoles were even formed (Fig.
6B). To explore how CSPG4P12 modulated apoptosis, the
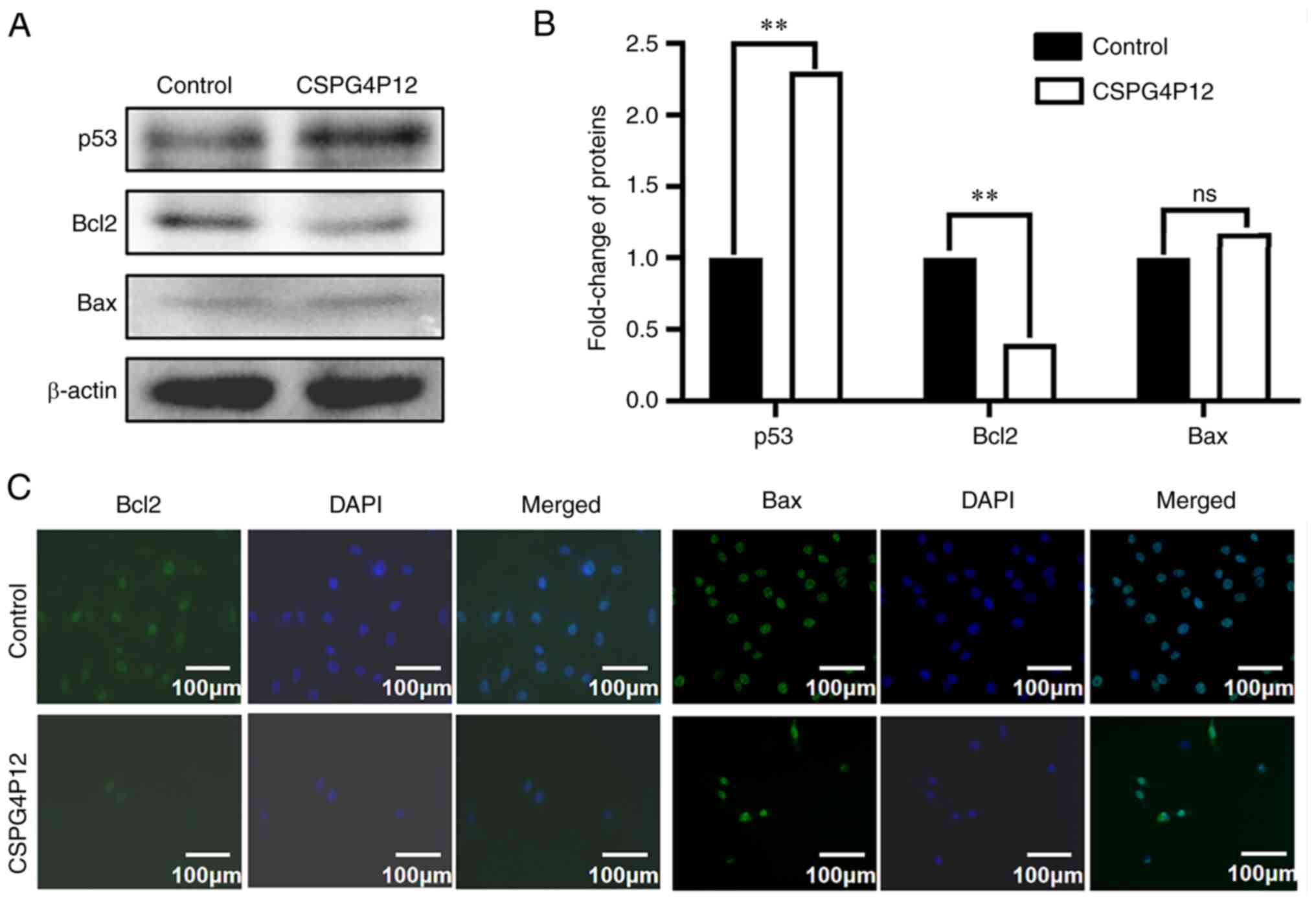
expression of apoptosis markers was next measured. Western blot
analysis demonstrated that CSPG4P12 overexpression significantly
increased the expression of p53 (Fig.
7A and B). Western blotting
showed that overexpression of CSPG4P12 markedly decreased the
expression of Bcl2; but had no effect on the expression of Bax in
A549 cells (Fig. 7A and B). IF assay presented that CSPG4P12
decreased the expression of Bcl2; however, due to the different
cell density, we still could not draw the solid conclusion from
this assay (Fig. 7C).
Discussion
NSCLC remains to be a malignancy with one of the
highest rates of morbidity and mortality in the world (7,23).
Pseudogene-derived lncRNAs have been reported to be involved in the
occurrence and development of NSCLC (12,24).
The lncRNA double homeobox A pseudogene 8, which is partially
duplicated from myosin light chain kinase (MYLK), is highly
expressed in lung adenocarcinoma and can promote cell progression
(25). In addition, a previous
study revealed that overexpression of MYLK pseudogene 1 can promote
the proliferation of NSCLC (26).
Therefore, present study focused on CSPG4P12, a lncRNA-derived
pseudogene.
CSPG4, the derived gene of CSPG4P12, has been
previously found to be overexpressed in triple-negative breast
cancer cells (27). Application of
the CSPG4 antibody was able to significantly inhibit the
proliferation, adhesion and migration of breast cancer cells
(27). Furthermore, CSPG4 has been
proposed to be a viable immunotherapy target for patients with
melanoma (28,29). The present data revealed that
overexpressing CSPG4P12 significantly inhibited the cell
proliferation, migration, invasion and adhesion whilst promoting
apoptosis in NSCLC cells. These results suggest that CSPG4P12 may
be involved in the tumorigenesis and progression of NSCLC.
Apoptosis is fundamental in maintaining the balance
between cell division and death. Evasion of apoptosis results in
the uncontrolled multiplication of cells that leads to different
diseases, including cancer (30).
Therefore, apoptosis analysis was performed in the present study to
assess the effect of CSPG4P12 on NSCLC. TEM imaging indicated that
the overexpression of CSPG4P12 resulted in disappearance of
mitochondrial cristae. The damage in the cristae changes the
permeability of the mitochondrial membrane, which may induce
apoptosis by affecting the expression of mitochondrial proteins,
such as Bax and Bcl2(31). Western
blot assay demonstrated that the overexpression of CSPG4P12
facilitated the expression of p53. Previous studies have indicated
that p53 induces apoptosis by inhibiting the expression of the
anti-apoptotic protein Bcl2 (32,33).
By contrast, other studies have previously shown that Bcl-2 and Bax
exist as heterodimers where they cooperate with each other to
regulate apoptosis (34,35). In the present study, western
blotting results supported the hypothesis that the overexpression
of CSPG4P12 promoted apoptosis through the p53/Bcl2/Bax pathway.
However, IF assay could not fully demonstrate the effect of
CSPG4P12 on the expression of Bcl2 and Bax due to the different
cell density. Further experiments still need to be conducted to
fully understand this mechanism. Another previous study has
demonstrated that CSPG4 immunotoxins combined with a panel of Bcl-2
inhibitors exerted a synergistic effect in mouse xenograft models
of glioblastoma, melanoma and breast cancer (36), which indirectly supports the
present findings.
There are several limitations that remain associated
with the present study. The small sample size of NSCLC tissues
hindered the analysis of the association of CSPG4P12 with clinical
characteristics of patients with NSCLC. In addition, due to
insufficient TEM magnification, the present study did not
accurately show the results of mitochondrial damage. However, it
was demonstrated that CSPG4P12 functions as a suppressor gene in
NSCLC.
In conclusion, CSPG4P12 was found to be
downregulated in NSCLC tissues, whilst the overexpression of
CSPG4P12 inhibited NSCLC development by activating the p53/Bcl2/Bax
axis.
Supplementary Material
Effects of transfection with the pUC57
plasmid on the proliferation of NSCLC. BC, blank control (without
pUC57 transfection); NC, negative control (with pUC57
transfection); ns, not significant.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Key Project of
Natural Science Foundation of Hebei province of China (grant no.
H2017209233).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
XueZ designed the study. WH, HW, AL and WZ performed
the experiments and confirm the authenticity of all the raw data.
XuaZ and QT analyzed and interpreted the data. All authors read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of North China University of Science and Technology
(approval no. 2021036) and all patients or their family members
signed informed consent.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer Statistics, 2021. CA Cancer J Clin. 71:7–33.
2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
GLOBOCAN estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249.
2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Gadgeel SM, Severson RK, Kau Y, Graff J,
Weiss LK and Kalemkerian GP: Impact of race in lung cancer:
Analysis of temporal trends from a surveillance, epidemiology and
end results database. Chest. 120:55–63. 2001.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Norouzi M and Hardy P: Clinical
applications of nanomedicines in lung cancer treatment. Acta
Biomater. 121:134–142. 2021.PubMed/NCBI View Article : Google Scholar
|
|
5
|
El-Hussein A, Manoto SL, Ombinda-Lemboumba
S, Alrowaili ZA and Mthunzi-Kufa P: A review of chemotherapy and
photodynamic therapy for lung cancer treatment. Anticancer Agents
Med Chem. 21:149–161. 2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Ortega-Franco A, Calvo V, Franco F,
Provencio M and Califano R: Integrating immune checkpoint
inhibitors and targeted therapies in the treatment of early stage
non-small cell lung cancer: A narrative review. Transl Lung Cancer
Res. 9:2656–2673. 2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Duma N, Santana-Davila R and Molina JR:
Non-small cell lung cancer: Epidemiology, screening, diagnosis and
treatment. Mayo Clin Proc. 94:1623–1640. 2019.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Hu X, Yang L and Mo YY: Role of
Pseudogenes in tumorigenesis. Cancers (Basel). 10:45–61.
2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Sisu C: Pseudogenes as biomarkers and
therapeutic targets in human cancers. Methods Mol Biol.
2324:319–337. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Groen JN, Capraro D and Morris KV: The
emerging role of pseudogene expressed non-coding RNAs in cellular
functions. Int J Biochem Cell Biol. 54:350–355. 2014.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Balakirev ES and Ayala FJ: Pseudogenes:
Are they ‘junk’ or functional DNA. Annu Rev Genet. 37:123–151.
2003.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Wu C, Song W, Wang Z and Wang B: Functions
of lncRNA DUXAP8 in non-small cell lung cancer. Mol Biol Rep.
17:78–92. 2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wang XJ, Li XD, Lin FK, Sun HZ, Lin YY,
Wang ZL and Wang XP: The lnc-CTSLP8 upregulates CTSL1 as a
competitive endogenous RNA and promotes ovarian cancer metastasis.
J Exp Clin Cancer Res. 40:151–167. 2021.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Hou Z, Wang Y, Xia N, Lv T, Yuan X and
Song Y: Pseudogene KRT17P3 drives cisplatin resistance of human
NSCLC cells by modulating miR-497-5p/mTOR. Cancer Sci. 112:275–286.
2021.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Wiest T, Hyrenbach S, Bambul P, Erker B,
Pezzini A, Hausser I, Arnold M, Martin JJ, Engelter S, Lyrer P, et
al: Genetic analysis of familial connective tissue alterations
associated with cervical artery dissections suggests locus
heterogeneity. Stroke. 37:1697–1702. 2006.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wang X, Wang Y, Yu L, Sakakura K, Visus C,
Schwab JH, Ferrone CR, Favoino E, Koya Y, Campoli MR, et al: CSPG4
in cancer: Multiple roles. Curr Mol Med. 10:419–429.
2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Egan CE, Stefanova D, Ahmed A, Raja VJ,
Thiemeyer JW, Chen KJ, Greenberg JA, Zhang TT, He B, Finnerty BM,
et al: CSPG4 is a potential therapeutic target in anaplastic
thyroid cancer. Thyroid. 31:1481–1493. 2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Yang J, Liao Q, Price M, Moriarity B, Wolf
N, Felices M, Miller JS, Geller MA, Bendzick L, Hopps R, et al:
Chondroitin sulfate proteoglycan 4, a targetable oncoantigen that
promotes ovarian cancer growth, invasion, cisplatin resistance and
spheroid formation. Transl Oncol. 16(101318)2022.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Poliseno L, Salmena L, Zhang J, Carver B,
Haveman WJ and Pandolfi PP: A coding-independent function of gene
and pseudogene mRNAs regulates tumour biology. Nature.
465:1033–1038. 2010.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Liu J, Liu ZX, Wu QN, Lu YX, Wong CW, Miao
L, Wang Y, Wang ZX, Jin Y, He MM, et al: Long noncoding RNA AGPG
regulates PFKFB3-mediated tumor glycolytic reprogramming. Nat
Commun. 11:1507–1523. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Tang ZF, Kang BX, Li CW, Chen TX and Zhang
ZM: GEPIA2: An enhanced web server for large-scale expression
profiling and interactive analysis. Nucleic Acids Res.
47:W556–W560. 2019.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Parkin DM: Global cancer statistics in the
year 2000. Lancet Oncol. 2:533–543. 2001.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Lou W, Ding B and Fu P: Pseudogene-derived
lncRNAs and their miRNA sponging mechanism in human cancer. Front
Cell Dev Biol. 8:85–87. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Yin D, Hua L, Wang J, Liu Y and Li X: Long
non-coding RNA DUXAP8 facilitates cell viability, migration and
glycolysis in non-small-cell lung cancer via regulating HK2 and
LDHA by inhibition of miR-409-3p. Onco Targets Ther. 13:7111–7123.
2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Han YJ, Ma SF, Yourek G, Park YD and
Garcia JG: A transcribed pseudogene of MYLK promotes cell
proliferation. FASEB J. 25:2305–2312. 2011.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Wang XH, Osada T, Wang YY, Yu L, Sakakura
K, Katayama A, McCarthy JB, Brufsky A, Chivukula M, Khoury T, et
al: CSPG4 protein as a new target for the antibody-based
immunotherapy of triple-negative breast cancer. J Natl Cancer Inst.
102:1496–1512. 2010.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Mittelman A, Chen ZJ, Kageshita T, Yang H,
Yamada P, Baskind P, Goldbery N, Puccio C, Ahmed T, et al: Active
specific immunotherapy in patients with melanoma. A clinical trial
with mouse antiidiotypic monoclonal antibodies elicited with
syngeneic anti-high-molecular-weight-melanoma-associated antigen
monoclonal antibodies. J Clin Invest. 86:2136–2144. 1990.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Mittelman A, Chen ZJ, Yang H, Wong GY and
Ferrone S: Human high molecular weight melanoma-associated antigen
(HMW-MAA) mimicry by mouse anti-idiotypic monoclonal antibody
MK2-23: Induction of humoral anti-HMW-MAA immunity and prolongation
of survival in patients with stage IV melanoma. Proc Natl Acad Sci
USA. 89:466–470. 1992.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Jan R and Chaudhry GE: Understanding
apoptosis and apoptotic pathways targeted cancer therapeutics. Adv
Pharm Bull. 9:205–218. 2019.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Precht TA, Phelps RA, Linseman DA, Butts
BD, Le SS, Laessig TA, Bouchard RJ and Heidenreich KA: The
permeability transition pore triggers Bax translocation to
mitochondria during neuronal apoptosis. Cell Death Differ.
12:255–265. 2005.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Dashzeveg N and Yoshida K: Cell death
decision by p53 via control of the mitochondrial membrane. Cancer
Lett. 367:108–112. 2015.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Bai HL, Kang CM, Sun ZQ, Li XH, Dai XY,
Huang RY, Zhao JJ, Bei YR, Huang XZ, Lu ZF, et al: TTDA inhibited
apoptosis by regulating the p53-Bax/Bcl2 axis in glioma. Exp
Neurol. 331:113380–113398. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Campbell KJ and Tait SWG: Targeting BCL-2
regulated apoptosis in cancer. Open Biol. 8:203–219.
2018.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Zhang J, Xie Y, Fan Q and Wang C: Effects
of karanjin on dimethylhydrazine induced colon carcinoma and
aberrant crypt foci are facilitated by alteration of the
p53/Bcl2/BAX pathway for apoptosis. Biotech Histochem. 96:202–212.
2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Yu X, Dobrikov M, Keir ST, Gromeier M,
Pastan IH, Reisfeld R, Bigner DD and Chandramohan V: Synergistic
antitumor effects of 9.2.27-PE38KDEL and ABT-737 in primary and
metastatic brain tumors. PLoS One. 14:38–54. 2019.PubMed/NCBI View Article : Google Scholar
|