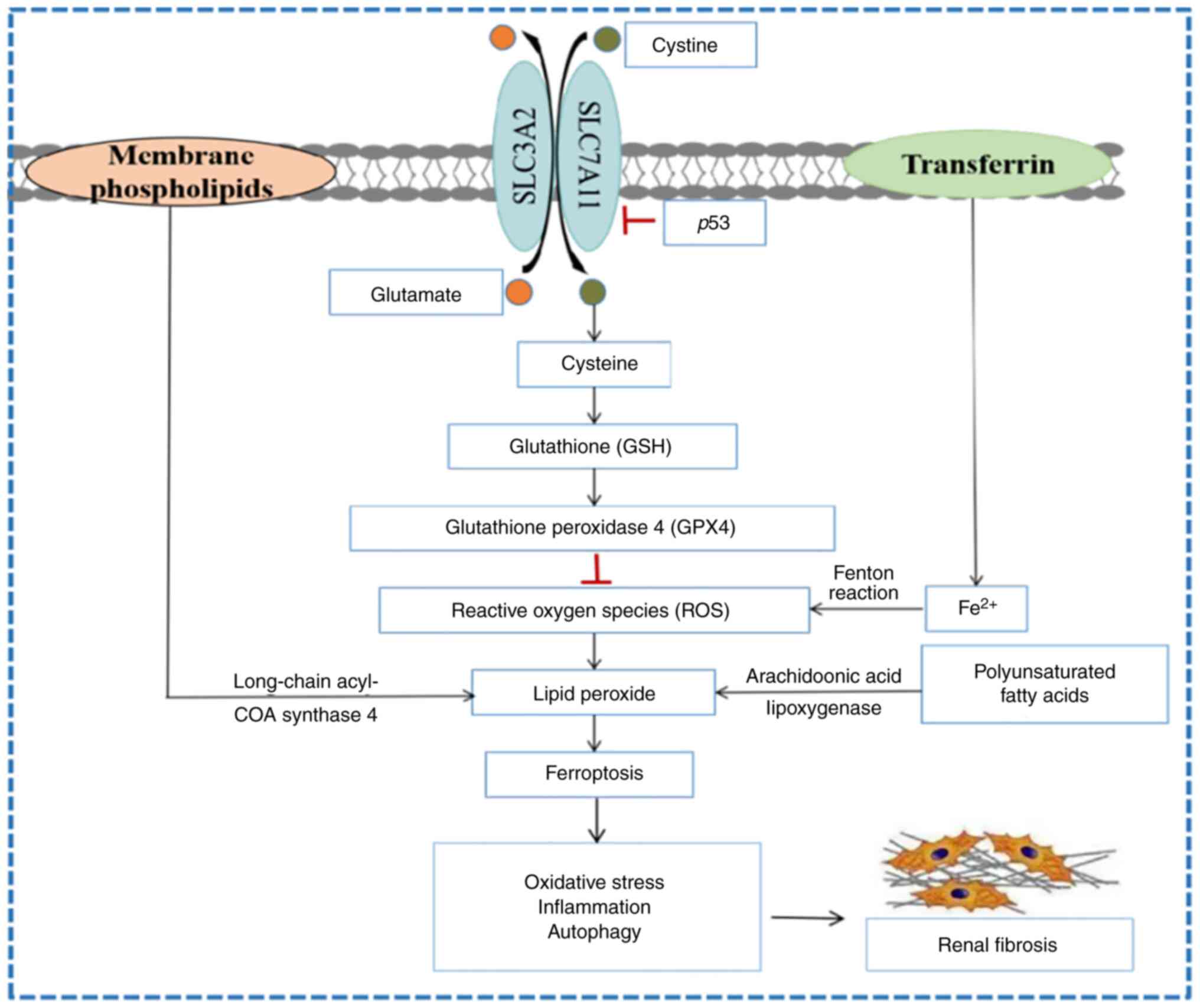
Regulated cell death (RCD) is a key biological
mechanism in the body that is required for healthy development,
homeostasis maintenance and disease prevention. RCD primarily
includes apoptosis, necroptosis, autophagy and ferroptosis
(1). Ferroptosis is fuelled by
oxidative stress and iron-dependent lipid peroxidation, which
differs from apoptosis, necroptosis and autophagy morphologically,
biochemically and genetically (2).
It is characterized by increased intracellular free iron and
accumulation of toxic lipid peroxides, leading to cell death
(3,4). Previous research has demonstrated
that renal fibrosis, which is defined by the breakdown of healthy
kidney architecture, fibroblast proliferation and excessive
extracellular matrix deposition, is a common pathological state of
almost all types of chronic and progressive kidney disorder
(5,6). Ferroptosis is closely associated with
the pathological process of numerous renal diseases and plays a key
role in numerous fibrotic diseases; however, the specific
mechanisms underlying development of renal fibrosis remain to be
fully elucidated. Ferroptosis serves a key role in the development
of renal fibrosis, and a comprehensive understanding of this
involvement may identify novel targets and approaches for the
development of disease prevention and therapy.
Ferroptosis is an iron-dependent and lipotoxic RCD.
Erastin is a cell-permeable substance that was discovered by Dolma
et al (7) in 2003 using a
high-content screening assay. In that study, it was demonstrated
that erastin selectively inhibits genetically engineered cells with
oncogenic RAS mutations without harming healthy cells. In 2012,
Dixon et al (2) named
erastin-induced iron-dependent non-apoptotic RCD as ‘ferroptosis’.
Ferroptosis is characterized by high levels of lipid peroxidation
at the cytoplasmic membrane and/or intracellular locations, such as
the mitochondria, endoplasmic reticulum or lysosome, and is a
caspase-independent type of cell death (2,8-10).
Morphologically, ferroptosis is characterized by decreased
mitochondrial density, decreased or absent mitochondrial cristae
and rupture of the outer mitochondrial membrane. Moreover, these
features are accompanied by intact membranes, normal nuclear size
and non-condensed chromatin (11).
Biochemically, ferroptosis is characterized by accumulation of
reactive oxygen species (ROS), increase of lipid peroxides and the
deposition of intracellular iron ions (2). Ferroptosis is regulated by factors
associated with metabolism, including glutathione peroxidase 4
(GPX4) dysfunction, the cystine (Cys)/glutamate (Glu) antiporter
system (system Xc-1), lipid peroxidation and iron
metabolism dysfunction (12). It
is also associated with signalling pathways, such as p53(13), ferroptosis suppressor protein 1
(FSP1)/coenzyme Q10 (COQ10)/NAD(P)H (14), PI3K/Akt/mTOR (15), sequestosome 1 (p62)/Kelch-like
ECH-associated protein 1 (Keap1)/nuclear factor erythroid 2-related
factor 2 (NRF2) (16) and
autophagy-related (ATG) 5/ATG7/nuclear receptor coactivator (NCOA)
1(17).
The antioxidant enzyme GPX4 is a member of the GPXs
family and a key target in the regulation of ferroptosis (18). GPX4 interrupts the lipid
peroxidation chain reaction by reducing complex hydroperoxides
(including phospholipid hydroperoxides and cholesterol
hydroperoxides) to their corresponding Sub-units (19). GPX4 is a selenoprotein with
selenocysteine (Sec) in its active site, and its active state
requires the catalysis of glutathione (GSH). GPX4 activity is
reduced or inactivated when GSH is depleted, and GPX4 also converts
GSH to glutathione disulfide (GSSG), thereby reducing esterified
oxidized fatty acids and cholesterol hydroperoxide, and reducing
lipid hydroperoxide (L-OOH) to a nontoxic lipid hydroxy derivative
(L-OH), thus resisting oxidative damage (13). During GPX4 maturation, Sec-tRNA is
mediated by mevalonate, one of the key regulatory factors in
positive regulation of the pathway product, isopentenyl
pyrophosphate (13). Both erastin
and RAS-selective lethal 3 (RSL3) compounds induce ferroptosis via
inactivation of GPX4, according to Dixon et al (20). A recent study also demonstrated
that erastin upregulates the expression of ATF3, which inhibits
expression of GPX4, in addition to inactivating GPX4 and inhibiting
its expression (21).
Lipid metabolism is key in the process of
ferroptosis and is a typical free radical chain reaction.
Polyunsaturated fatty acids (PUFAs) are involved in almost all
pathways of ferroptosis because they are susceptible to lipid
peroxidation due to the presence of easily extractable hydrogen
atoms at bis-allylic carbon positions (30). Any free radical that can extract
hydrogen atoms from oxidizable substrates can initiate the lipid
peroxidation process, and the abundance and location of
intracellular lipid peroxidizable substrates determines the extent
of lipid peroxidation and ferroptosis (8). Free PUFAs are substrates for
synthesis of lipid signal transduction mediators, and these are
esterified into membrane phospholipids during lipid metabolism and
oxidized into ferroptosis signals (13). Lipidomic analysis has revealed that
phosphatidylethanolamines (PEs) are key membrane phospholipids that
drive ferroptosis by oxidizing phospholipid hydroperoxides
[arachidonic acid (AA) and adrenic acid (AdA)-hydroperoxides-PE]
via a non-enzymatic process (13,16).
Acyl-CoA Synthetase is a long-chain family member 4 (ACSL4) and
lysophosphatidylcholine acyl-transferase 3 (LPCAT3) are two enzymes
involved in the biosynthesis and remodeling of PE, which activate
PUFA and affect its transmembrane properties (12). Therefore, blocking expression of
ACSL4 and LPCAT3 inhibits the esterification of AA or AdA to PE and
decreases accumulation of intracellular lipid peroxidative
substrates, thereby inhibiting ferroptosis (13). As enzymatic effectors,
lipoxygenases, of which free PUFAs are the preferred substrates,
mediate the peroxidative reaction of ferroptosis (31). SLC38A1 is a regulator of glutamate
uptake and metabolism in lipid peroxidation (32). Yang et al (33) found that lncRNA ZFAS1 could
regulate the expression of SLC38A1 through miR-150-5p and activate
the conversion of fibroblasts into myofibroblasts in lung tissue
with the development of cytosolic iron death.
Results of a recent study demonstrated that p53 is
essential for inducing ferroptosis (13). p53 responds to different stress
signals via the coordination of specific cellular responses and the
corresponding cell cycle arrest and apoptosis play important roles
in inhibiting development of cancer (13). It was found that p53 could inhibit
the uptake of System Xc- to Cys by suppressing the expression of
SLC7A11, which resulted in a large decrease in GSH production and
affected the activity of GPX4, leading to reduced antioxidant
capacity and ROS accumulation, thus promoting cellular iron death
(40). The communication between
mitochondria and other organelles is aided by voltage-dependent
anion channels (VDACs), channel proteins located in the outer
mitochondrial membranous layer (41). Yagoda et al (42) demonstrated that erastin alters the
permeability of the mitochondrial outer membrane via direct binding
to VDAC2/3, thereby decreasing the oxidation rate of NADH and
inducing ferroptosis. FSP1 is a potent ferroptosis-resistance
factor (43). Doll et al
(14) demonstrated that
myristoylation of FSP1 inhibits lipid peroxidation via NAD(P)H
reduction of COQ10, thereby inhibiting ferroptosis. Moreover,
methionine is converted to Cys under oxidative stress via the
sulphur transfer pathway to create GSH, which exerts its
antioxidant action (44). In
addition, the PI3K/Akt/mTOR (15),
p62/Keap1/NRF2(16) and
ATG5/ATG7/NCOA4(17) signalling
pathways serve regulatory roles in the occurrence of
ferroptosis.
When the redox system is damaged, ROS and reactive
nitrogen species are excessively produced and oxidative stress
occurs (45). Well-established
mechanisms of oxidative stress-mediated renal injury include
production of ROS and the ensuing disruption of the antioxidant
system, which result in apoptosis, ferroptosis and necrosis
(46,47). There are numerous causes of renal
fibrosis, including oxidative stress (48). Specific inhibitors, including
ferrostatin-1 (Fer-1), which is characterized by lipid-dependent
peroxidation, prevent ferroptosis (2). Roxadustat is an emerging therapeutic
option for treatment of anaemia in patients with chronic kidney
disease (CKD). It is an oral inhibitor of hypoxia-inducible factor
(HIF) prolyl hydroxylase, which stimulates erythropoiesis and
regulates iron metabolism. Using a folic acid-induced kidney injury
model, Li et al (49)
demonstrated that roxadustat pre-treatment decreases ferroptosis
and inhibits inflammation by stabilizing HIF-1α and activating the
nuclear factor erythroid 2-related factor 2 (Nrf2) signalling
pathway, thereby inhibiting renal fibrosis. Ide et al
(50) demonstrated that lipid
peroxidation induces ferroptotic stress and ferroptosis. Following
injury, renal proximal tubule (PT) cells may exhibit a
pro-inflammatory state and genes involved in high ferroptosis
stress may trigger accumulation of inflammatory PT cells, thereby
enhancing inflammation and fibrosis. Feng et al (51) demonstrated that diabetes increases
HIF-1α and HO-1 in the kidney of mice, resulting in increased lipid
peroxidation due to increased ROS production and tubular iron
deposition. This leads to renal tubular damage and fibrosis in
mice. In a diabetic mouse model, inhibition of ferroptosis prevents
lipid peroxidation by decreasing ROS production in the kidneys and
reducing iron deposition in the renal tubules (51). This alleviates diabetes mellitus,
renal tubular injury and renal fibrosis progression in mice
(51). Nobiletin (Nob), an
important active flavonoid found in citrus fruits, exhibits
antioxidant, anti-inflammatory, antifibrotic and antiapoptotic
properties (52). Lo et al
(53) demonstrated that Nob
partially decreases oxidative stress and ferroptosis or apoptosis
in unilateral ureteral obstruction (UUO) mice, which decreases
inflammatory responses and subsequently inhibits development of
renal fibrosis. In conclusion, the inhibition of ferroptosis via
regulation of oxidative stress may represent a novel method for
treatment of renal fibrosis.
Inflammation is an immune response to exogenous or
endogenous injury and contributes to the maintenance of tissue
homeostasis under stressful conditions (54). A high inflammatory burden is
associated with kidney damage. Results of a clinical study on type
2 diabetes demonstrated that the ratio of C-reactive protein
expression to serum albumin is increased in patients with diabetic
nephropathy compared with those without diabetic nephropathy
(55). Using an adenine-induced
mouse model of aging, the model group demonstrated increased levels
of extensive tubular damage and fibrosis, as well as increased
inflammatory responses, compared with groups of control (56). Inflammation is the main
pathogenesis of diabetic kidney injury (DKI), and the monocyte to
lymphocyte ratio (MLR) is considered a marker of inflammatory
disease. microalbuminuria (MA) is the Microalbuminuria (MA) is the
last reversible stage of DKI treatment, and in type 2 diabetic
patients, MLR expression levels are significantly higher in the MA
group compared to the normoalbuminuria (NA) group (57). Monocyte chemoattractant protein-1,
macrophage colony-stimulating factor and neopterin levels are
markedly increased in patients with chronic renal disease compared
with controls (58). Moreover,
numerous inflammatory cytokines, such as neuregulin (59), kidney injury molecule-1 (KIM-1)
(60) and omentin (61) are associated with degree of kidney
damage. In addition, interleukin (IL)-10 exerts anti-inflammatory
effects. In a renal ischemia-reperfusion injury model, IL-10
knockout mice demonstrated decreased levels of renal function,
upregulation of renal injury biomarkers, such as KIM-1, and
increased expression of certain pro-inflammatory cytokines,
compared with the control group (62). Therefore, renal damage is
associated with inflammation.
Damage to renal tissue induces the inflammatory and
fibrotic processes that aid in regeneration and repair (63). Results of previous studies
demonstrated that macrophages are a potential therapeutic target
for renal injury and fibrosis and play a significant role in the
pathophysiology of kidney disease (64-66).
Notably, renal fibrosis may be reversed as different subpopulations
of macrophages in the kidney can either promote or inhibit
deposition of extracellular matrix in the kidney (64). Inflammatory cell infiltration is a
key characteristic of renal fibrosis (67). Renal tubular injury is considered a
proinflammatory driving force in fibrosis. Following renal tubular
injury, renal tubular epithelial cells (TECs) produce immune
responses and release inflammatory mediators. The aggravation of
inflammation leads to cell death, while cell death also has a
strong pro-inflammatory effect, further worsening tubular injury,
and continued inflammation and injury can lead to
tubulointerstitial fibrosis (68).
In conclusion, renal fibrosis and inflammation are associated with
renal damage.
Due to increased permeability and rupture of the
cell membrane during ferroptosis, associated contents, including
damage-associated molecular pattern (DAMP) may be released, causing
an inflammatory response and activation of the innate immune
response. However, the specific mechanism requires further
investigation (69).
Necroinflammation associated with ferroptosis is observed acute
kidney injury model in mice, as well as in GPX4-deficient knockout
mouse models (70,71). In the kidneys of GPX4 knockout mice
induced by tamoxifen, a large number of renal tubular cells died,
and the release of cellular debris, mitochondria and even nuclei
from ruptured cells into the tubular lumen could be observed at the
histological level; this may be associated with DAMP (72). In ferroptotic tissues, F4/80
immunofluorescent staining has demonstrated that macrophages are
markedly activated (73),
releasing pro-inflammatory substances, thus triggering inflammatory
responses.
However, to the best of our knowledge, the
interaction between ferroptosis and inflammation in renal fibrosis
remains unclear. Tectorigenin, a compound derived from the iris
plant Belamcanda chinensis, is an active ingredient used in
Traditional Chinese Medicine (74). Tectorigenin exhibits numerous
pharmacological activities, such as anti-inflammatory and
antioxidant properties, liver protection and diabetes control
(75,76). Li et al (77) demonstrated that tectorigenin
inhibits ferroptosis and fibrosis induced by external stimuli in
primary TECs. Moreover, Fer-1, a ferroptosis inhibitor, inhibits
the pro-fibrotic effect of TGF-β1-stimulated TECs, suggesting that
tectorigenin may alleviate renal fibrosis via inhibition of
ferroptosis in TECs (77). Results
of a recent study also demonstrated that Fer-1 attenuates
oxalate-induced TEC damage and renal fibrosis via inhibition of
ferroptosis (78). Zhang et
al (79) demonstrated that
ferroptosis of TECs may be induced following UUO in mice, while
liproxstatin-1 (Lip-1), a ferroptosis inhibitor, inhibits
downregulation of GPX4 expression and ferroptosis in TECs and
attenuates expression of pro-fibrotic factors in UUO mice. These
results suggested that Lip-1 alleviates renal fibrosis in UUO mice
via inhibition of ferroptosis in TECs. Moreover, Luo et al
(80) demonstrated that obesity
induces ferroptosis in the kidney while Fer-1 inhibits the
development of high-fat diet-induced inflammation and fibrosis in
renal tissue. Tocilizumab is an emerging interleukin-6 (IL6)
receptor-targeting drug Yang et al (81) demonstrated that tocilizumab
attenuates renal fibrosis in mice via inhibition of ferroptosis.
Renal fibrosis is a common pathological process in diabetic
nephropathy (82). Results of
previous studies have demonstrated that expression levels of ACSL4
are increased in diabetic nephropathy mice and expression levels of
GPX4 are decreased (83). Using
the ACSL4 inhibitor rosiglitazone, both ferroptosis and production
of pro-inflammatory cytokines are inhibited in TECs, preventing
development of diabetic nephropathy (83). Zhou et al (84) confirmed that inhibiting ferroptosis
in TECs reduces interstitial inflammation and renal fibrosis.
Collectively, these results suggested that attenuating cellular
inflammation development via inhibition of ferroptosis may be a
novel approach for the treatment of renal fibrosis.
Autophagy refers to self-phagocytosis of cells,
which removes misfolded proteins and damaged organelles in cells
awaiting degradation, thereby maintaining cell homeostasis
(85). Autophagy is a
self-protection mechanism of eukaryotic cells (85). A previous study demonstrated that
changes in autophagy activity are associated with renal fibrosis
(86). The regulatory function of
autophagy in fibrosis is associated with coordinated regulation of
tubular cell death, interstitial inflammation and, in particular,
production of pro-fibrotic secretory proteins (87). Results of a previous study
demonstrated that, as a relatively recently discovered regulatory
mode of cell death, ferroptosis differs from other regulatory modes
such as autophagy, apoptosis, necrosis (2). Nonetheless, a more recent study
demonstrated that ferroptosis and autophagy exhibit common
regulators such as SLC7A11, GPX4, Nrf2 and heat shock protein
β-1(88). The autophagy-related
protein beclin 1 (BECN1) inhibits the function of system
Xc- via formation of the BECN1/SLC7A11 complex and
induces ferroptosis under the action of erastin and RSL3(84). Ferritinophagy is a type of
cell-selective autophagy mediated by NCOA4. To facilitate the
movement of intracellular ferritin to autophagy lysosomes and
liberate free iron, NCOA4 functions as a selective autophagy
receptor and binds to FTH1 of ferritin (89). Overexpression of NCOA4 increases
ferritin degradation in cancer cells and fibroblasts, thereby
promoting ferroptosis (90). Wang
et al (91) demonstrated
that expression of NCOA4 is increased in a 5/6 nephrectomy-induced
CKD rat model Following the addition of ferroptosis inducer
cisplatin or the ferroptosis inhibitor desferrioxamine mesylate,
expression of NCOA4 is enhanced or attenuated, respectively. This
treatment alters the progression of renal fibrosis. Therefore,
ferritinophagy may induce ferroptosis in CKD and promote
development of renal fibrosis. Consequently, inhibition of
ferroptosis via regulation of autophagy may act as a novel
therapeutic method in treatment of renal fibrosis.
Renal fibrosis is a common pathological state in
almost all chronic and progressive kidney diseases, but effective
measures for its clinical prevention and treatment are still not
available. Ferroptosis is a novel regulatory cell death modality,
and by summarizing the association between renal fibrosis and
ferroptosis, we found that ferroptosis is involved in various
biological processes such as oxidative stress, inflammation, and
autophagy during renal fibrosis (Fig.
1). However, the specific molecular mechanism is still unclear,
and further research is needed to investigate the role of
ferroptosis in the development of renal fibrosis and to explore
effective and highly targeted therapeutic measures against
ferroptosis to provide new targets and more valuable therapeutic
approaches for renal fibrosis research.
Not applicable.
Funding: The present study was funded by the National Natural
Science Foundation of China (grant no. 82004165) and the
Collaborative Public Relations Project Plan of Chinese and Western
Medicine of Major and Difficult Diseases in Anhui Province (grant
no. 2021-70).
Not applicable.
HYZ, MC, LZ and YPW were responsible for the
conceptualization of the present review. HYZ and MC were
responsible for the original draft preparation. LZ and YPW were
responsible for reviewing and editing the manuscript. LZ and YPW
were responsible for funding acquisition. All authors have read and
approved the final manuscript. Data authentication is not
applicable.
Not applicable.
Not applicable.
The authors declare that they have no competing
interests.
|
1
|
Zhang X and Li X: Abnormal iron and lipid
metabolism mediated ferroptosis in kidney diseases and its
therapeutic potential. Metabolites. 12(58)2022.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta
R, Zaitsev EM, Gleason CE, Patel DN, Bauer AJ, Cantley AM, Yang WS,
et al: Ferroptosis: An iron-dependent form of nonapoptotic cell
death. Cell. 149:1060–1072. 2012.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Dixon SJ and Stockwell BR: The role of
iron and reactive oxygen species in cell death. Nat Chem Biol.
10:9–17. 2014.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Tuo QZ, Lei P, Jackman KA, Li XL, Xiong H,
Li XL, Liuyang ZY, Roisman L, Zhang ST, Ayton S, et al:
Tau-mediated iron export prevents ferroptotic damage after ischemic
stroke. Mol Psychiatry. 22:1520–1530. 2017.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lu X, Rudemiller NP, Ren J, Wen Y, Yang B,
Griffiths R, Privratsky JR, Madan B, Virshup DM and Crowley SD:
Opposing actions of renal tubular- and myeloid-derived porcupine in
obstruction-induced kidney fibrosis. Kidney Int. 96:1308–1319.
2019.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Humphreys BD: Mechanisms of renal
fibrosis. Annu Rev Physiol. 80:309–326. 2018.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Dolma S, Lessnick SL, Hahn WC and
Stockwell BR: Identification of genotype-selective antitumor agents
using synthetic lethal chemical screening in engineered human tumor
cells. Cancer Cell. 3:285–296. 2003.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Gao M, Yi J, Zhu J, Minikes AM, Monian P,
Thompson CB and Jiang X: Role of mitochondria in ferroptosis. Mol
Cell. 73:354–363.e3. 2019.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Lee YS, Lee DH, Choudry HA, Bartlett DL
and Lee YJ: Ferroptosis-induced endoplasmic reticulum stress:
Cross-talk between ferroptosis and apoptosis. Mol Cancer Res.
16:1073–1076. 2018.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Hirayama T, Miki A and Nagasawa H:
Organelle-specific analysis of labile Fe(ii) during ferroptosis by
using a cocktail of various colour organelle-targeted fluorescent
probes. Metallomics. 11:111–117. 2019.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Lin X, Ping J, Wen Y and Wu Y: The
mechanism of ferroptosis and applications in tumor treatment. Front
Pharmacol. 11(1061)2020.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Stockwell BR, Friedmann Angeli JP, Bayir
H, Bush AI, Conrad M, Dixon SJ, Fulda S, Gascón S, Hatzios SK,
Kagan VE, et al: Ferroptosis: A regulated cell death nexus linking
metabolism, redox biology, and disease. Cell. 171:273–285.
2017.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Li J, Cao F, Yin HL, Huang ZJ, Lin ZT, Mao
N, Sun B and Wang G: Ferroptosis: Past, present and future. Cell
Death Dis. 11(88)2020.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Doll S, Freitas FP, Shah R, Aldrovandi M,
da Silva MC, Ingold I, Goya Grocin A, Xavier da Silva TN, Panzilius
E, Scheel CH, et al: FSP1 is a glutathione-independent ferroptosis
suppressor. Nature. 575:693–698. 2019.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Yi J, Zhu J, Wu J, Thompson CB and Jiang
X: Oncogenic activation of PI3K-AKT-mTOR signaling suppresses
ferroptosis via SREBP-mediated lipogenesis. Proc Natl Acad Sci USA.
117:31189–31197. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Doll S, Proneth B, Tyurina YY, Panzilius
E, Kobayashi S, Ingold I, Irmler M, Beckers J, Aichler M, Walch A,
et al: ACSL4 dictates ferroptosis sensitivity by shaping cellular
lipid composition. Nat Chem Biol. 13:91–98. 2017.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Wen J, Chen H, Ren Z, Zhang P, Chen J and
Jiang S: Ultrasmall iron oxide nanoparticles induced ferroptosis
via beclin1/ATG5-dependent autophagy pathway. Nano Converg.
8(10)2021.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Galaris D, Barbouti A and Pantopoulos K:
Iron homeostasis and oxidative stress: An intimate relationship.
Biochim Biophys Acta Mol Cell Res. 1866(118535)2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Wang S, Luo J, Zhang Z, Dong D, Shen Y,
Fang Y, Hu L, Liu M, Dai C, Peng S, et al: Iron and magnetic: New
research direction of the ferroptosis-based cancer therapy. Am J
Cancer Res. 8:1933–1946. 2018.PubMed/NCBI
|
|
20
|
Dixon SJ, Winter GE, Musavi LS, Lee ED,
Snijder B, Rebsamen M, Superti-Furga G and Stockwell BR: Human
haploid cell genetics reveals roles for lipid metabolism genes in
nonapoptotic cell death. ACS Chem Biol. 10:1604–1609.
2015.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Dai C, Chen X, Li J, Comish P, Kang R and
Tang D: Transcription factors in ferroptotic cell death. Cancer
Gene Ther. 27:645–656. 2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Lewerenz J, Hewett SJ, Huang Y, Lambros M,
Gout PW, Kalivas PW, Massie A, Smolders I, Methner A, Pergande M,
et al: The cystine/glutamate antiporter system x(c)(-) in health
and disease: From molecular mechanisms to novel therapeutic
opportunities. Antioxid Redox Signal. 18:522–555. 2013.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Bannai S: Exchange of cystine and
glutamate across plasma membrane of human fibroblasts. J Biol Chem.
261:2256–2263. 1986.PubMed/NCBI
|
|
24
|
Gao M, Monian P, Quadri N, Ramasamy R and
Jiang X: Glutaminolysis and transferrin regulate ferroptosis. Mol
Cell. 59:298–308. 2015.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Lang X, Green MD, Wang W, Yu J, Choi JE,
Jiang L, Liao P, Zhou J, Zhang Q, Dow A, et al: Radiotherapy and
immunotherapy promote tumoral lipid oxidation and ferroptosis via
synergistic repression of SLC7A11. Cancer Discov. 9:1673–1685.
2019.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Koppula P, Zhuang L and Gan B: Cystine
transporter SLC7A11/xCT in cancer: Ferroptosis, nutrient
dependency, and cancer therapy. Protein Cell. 12:599–620.
2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Sato H, Shiiya A, Kimata M, Maebara K,
Tamba M, Sakakura Y, Makino N, Sugiyama F, Yagami K, Moriguchi T,
et al: Redox imbalance in cystine/glutamate transporter-deficient
mice. J Biol Chem. 280:37423–37429. 2005.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Badgley MA, Kremer DM, Maurer HC,
DelGiorno KE, Lee HJ, Purohit V, Sagalovskiy IR, Ma A, Kapilian J,
Firl CEM, et al: Cysteine depletion induces pancreatic tumor
ferroptosis in mice. Science. 368:85–89. 2020.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Chang LC, Chiang SK, Chen SE, Yu YL, Chou
RH and Chang WC: Heme oxygenase-1 mediates BAY 11-7085 induced
ferroptosis. Cancer Lett. 416:124–137. 2018.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Gan B: Mitochondrial regulation of
ferroptosis. J Cell Biol. 220(e202105043)2021.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Kuhn H, Banthiya S and van Leyen K:
Mammalian lipoxygenases and their biological relevance. Biochim
Biophys Acta. 1851:308–330. 2015.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Qureshi T, Sørensen C, Berghuis P, Jensen
V, Dobszay MB, Farkas T, Dalen KT, Guo C, Hassel B, Utheim TP, et
al: The glutamine transporter Slc38a1 regulates GABAergic
neurotransmission and synaptic plasticity. Cereb Cortex.
29:5166–5179. 2019.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Yang Y, Tai W, Lu N, Li T, Liu Y, Wu W, Li
Z, Pu L, Zhao X, Zhang T and Dong Z: lncRNA ZFAS1 promotes lung
fibroblast-to-myofibroblast transition and ferroptosis via
functioning as a ceRNA through miR-150-5p/SLC38A1 axis. Aging
(Albany NY). 12:9085–9102. 2020.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Chen X, Yu C, Kang R and Tang D: Iron
metabolism in ferroptosis. Front Cell Dev Biol.
8(590226)2020.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Doll S and Conrad M: Iron and ferroptosis:
A still ill-defined liaison. IUBMB Life. 69:423–434.
2017.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Tang D, Chen X, Kang R and Kroemer G:
Ferroptosis: Molecular mechanisms and health implications. Cell
Res. 31:107–125. 2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Alvarez SW, Sviderskiy VO, Terzi EM,
Papagiannakopoulos T, Moreira AL, Adams S, Sabatini DM, Birsoy K
and Possemato R: NFS1 undergoes positive selection in lung tumours
and protects cells from ferroptosis. Nature. 551:639–643.
2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Fang X, Wang H, Han D, Xie E, Yang X, Wei
J, Gu S, Gao F, Zhu N, Yin X, et al: Ferroptosis as a target for
protection against cardiomyopathy. Proc Natl Acad Sci USA.
116:2672–2680. 2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chen PH, Wu J, Ding CC, Lin CC, Pan S,
Bossa N, Xu Y, Yang WH, Mathey-Prevot B and Chi JT: Kinome screen
of ferroptosis reveals a novel role of ATM in regulating iron
metabolism. Cell Death Differ. 27:1008–1022. 2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Jiang L, Kon N, Li T, Wang SJ, Su T,
Hibshoosh H, Baer R and Gu W: Ferroptosis as a p53-mediated
activity during tumour suppression. Nature. 520:57–62.
2015.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Mazure NM: VDAC in cancer. Biochim Biophys
Acta Bioenerg. 1858:665–673. 2017.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Yagoda N, von Rechenberg M, Zaganjor E,
Bauer AJ, Yang WS, Fridman DJ, Wolpaw AJ, Smukste I, Peltier JM,
Boniface JJ, et al: RAS-RAF-MEK-dependent oxidative cell death
involving voltage-dependent anion channels. Nature. 447:864–868.
2007.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Bersuker K, Hendricks JM, Li Z, Magtanong
L, Ford B, Tang PH, Roberts MA, Tong B, Maimone TJ, Zoncu R, et al:
The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit
ferroptosis. Nature. 575:688–692. 2019.PubMed/NCBI View Article : Google Scholar
|
|
44
|
McBean GJ: The transsulfuration pathway: A
source of cysteine for glutathione in astrocytes. Amino Acids.
42:199–205. 2012.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Sedeek M, Nasrallah R, Touyz RM and Hébert
RL: NADPH oxidases, reactive oxygen species, and the kidney: Friend
and foe. J Am Soc Nephrol. 24:1512–1518. 2013.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Guerrero-Hue M, García-Caballero C,
Palomino-Antolín A, Rubio-Navarro A, Vázquez-Carballo C, Herencia
C, Martín-Sanchez D, Farré-Alins V, Egea J, Cannata P, et al:
Curcumin reduces renal damage associated with rhabdomyolysis by
decreasing ferroptosis-mediated cell death. FASEB J. 33:8961–8975.
2019.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Linkermann A, Chen G, Dong G, Kunzendorf
U, Krautwald S and Dong Z: Regulated cell death in AKI. J Am Soc
Nephrol. 25:2689–2701. 2014.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Su H, Wan C, Song A, Qiu Y, Xiong W and
Zhang C: Oxidative stress and renal fibrosis: Mechanisms and
therapies. Adv Exp Med Biol. 1165:585–604. 2019.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Li X, Zou Y, Xing J, Fu YY, Wang KY, Wan
PZ and Zhai XY: Pretreatment with roxadustat (FG-4592) attenuates
folic acid-induced kidney injury through antiferroptosis via
Akt/GSK-3 β/Nrf2 pathway. Oxid Med Cell Longev.
2020(6286984)2020.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Ide S, Kobayashi Y, Ide K, Strausser SA,
Abe K, Herbek S, O'Brien LL, Crowley SD, Barisoni L, Tata A, et al:
Ferroptotic stress promotes the accumulation of pro-inflammatory
proximal tubular cells in maladaptive renal repair. Elife.
10(e68603)2021.PubMed/NCBI View Article : Google Scholar
|
|
51
|
Feng X, Wang S, Sun Z, Dong H, Yu H, Huang
M and Gao X: Ferroptosis enhanced diabetic renal tubular injury via
HIF-1α/HO-1 pathway in db/db mice. Front Endocrinol (Lausanne).
12(626390)2021.PubMed/NCBI View Article : Google Scholar
|
|
52
|
Liu B, Deng Q, Zhang L and Zhu W:
Nobiletin alleviates ischemia/reperfusion injury in the kidney by
activating PI3K/AKT pathway. Mol Med Rep. 22:4655–4662.
2020.PubMed/NCBI View Article : Google Scholar
|
|
53
|
Lo YH, Yang SF, Cheng CC, Hsu KC, Chen YS,
Chen YY, Wang CW, Guan SS and Wu CT: Nobiletin alleviates
ferroptosis-associated renal injury, inflammation, and fibrosis in
a unilateral ureteral obstruction mouse model. Biomedicines.
10(595)2022.PubMed/NCBI View Article : Google Scholar
|
|
54
|
Yang L and Xia H: TRIM proteins in
inflammation: From expression to emerging regulatory mechanisms.
Inflammation. 44:811–820. 2021.PubMed/NCBI View Article : Google Scholar
|
|
55
|
Bilgin S, Kurtkulagi O, Atak BM, Duman TT,
Kahveci G, Khalid A and Aktas G: Does C-reactive protein to serum
albumin ratio correlate with diabEtic nephropathy in patients with
Type 2 dIabetes MEllitus? The CARE TIME study. Prim Care Diabetes.
15:1071–1074. 2021.PubMed/NCBI View Article : Google Scholar
|
|
56
|
Jung SW, Kim DJ, Kim YG, Moon JY, Jeong KH
and Lee SH: Renal aging resembles a continuum between normal and
diseased kidneys that potentiates inflammatory response to injury.
J Gerontol A Biol Sci Med Sci. 76:385–392. 2021.PubMed/NCBI View Article : Google Scholar
|
|
57
|
Kocak MZ, Aktas G, Duman TT, Atak BM,
Kurtkulagi O, Tekce H, Bilgin S and Alaca B: Monocyte lymphocyte
ratio as a predictor of diabetic kidney injury in type 2 diabetes
mellitus; The MADKID study. J Diabetes Metab Disord. 19:997–1002.
2020.PubMed/NCBI View Article : Google Scholar
|
|
58
|
Musiał K and Zwolińska D: New markers of
cell migration and inflammation in children with chronic kidney
disease. Biomarkers. 24:295–302. 2019.PubMed/NCBI View Article : Google Scholar
|
|
59
|
Kocak MZ, Aktas G, Atak BM, Duman TT, Yis
OM, Erkus E and Savli H: Is neuregulin-4 a predictive marker of
microvascular complications in type 2 diabetes mellitus? Eur J Clin
Invest. 50(e13206)2020.PubMed/NCBI View Article : Google Scholar
|
|
60
|
Kin Tekce B, Tekce H, Aktas G and Sit M:
Evaluation of the urinary kidney injury molecule-1 levels in
patients with diabetic nephropathy. Clin Invest Med. 37:E377–E383.
2014.PubMed/NCBI View Article : Google Scholar
|
|
61
|
Tekce H, Tekce BK, Aktas G, Alcelik A and
Sengul E: Serum omentin-1 levels in diabetic and nondiabetic
patients with chronic kidney disease. Exp Clin Endocrinol Diabetes.
122:451–456. 2014.PubMed/NCBI View Article : Google Scholar
|
|
62
|
Sakai K, Nozaki Y, Murao Y, Yano T, Ri J,
Niki K, Kinoshita K, Funauchi M and Matsumura I: Protective effect
and mechanism of IL-10 on renal ischemia-reperfusion injury. Lab
Invest. 99:671–683. 2019.PubMed/NCBI View Article : Google Scholar
|
|
63
|
Black LM, Lever JM and Agarwal A: Renal
inflammation and fibrosis: A double-edged sword. J Histochem
Cytochem. 67:663–681. 2019.PubMed/NCBI View Article : Google Scholar
|
|
64
|
Tang PM, Nikolic-Paterson DJ and Lan HY:
Macrophages: Versatile players in renal inflammation and fibrosis.
Nat Rev Nephrol. 15:144–158. 2019.PubMed/NCBI View Article : Google Scholar
|
|
65
|
Meng XM, Wang S, Huang XR, Yang C, Xiao J,
Zhang Y, To KF, Nikolic-Paterson DJ and Lan HY: Inflammatory
macrophages can transdifferentiate into myofibroblasts during renal
fibrosis. Cell Death Dis. 7(e2495)2016.PubMed/NCBI View Article : Google Scholar
|
|
66
|
Wei J, Xu Z and Yan X: The role of the
macrophage-to-myofibroblast transition in renal fibrosis. Front
Immunol. 13(934377)2022.PubMed/NCBI View Article : Google Scholar
|
|
67
|
Kang HM, Ahn SH, Choi P, Ko YA, Han SH,
Chinga F, Park AS, Tao J, Sharma K, Pullman J, et al: Defective
fatty acid oxidation in renal tubular epithelial cells has a key
role in kidney fibrosis development. Nat Med. 21:37–46.
2015.PubMed/NCBI View Article : Google Scholar
|
|
68
|
Liu BC, Tang TT, Lv LL and Lan HY: Renal
tubule injury: A driving force toward chronic kidney disease.
Kidney Int. 93:568–579. 2018.PubMed/NCBI View Article : Google Scholar
|
|
69
|
Wen Q, Liu J, Kang R, Zhou B and Tang D:
The release and activity of HMGB1 in ferroptosis. Biochem Biophys
Res Commun. 510:278–283. 2019.PubMed/NCBI View Article : Google Scholar
|
|
70
|
Von Mässenhausen A, Tonnus W and
Linkermann A: Cell death pathways drive necroinflammation during
acute kidney injury. Nephron. 140:144–147. 2018.PubMed/NCBI View Article : Google Scholar
|
|
71
|
Friedmann Angeli JP, Schneider M, Proneth
B, Tyurina YY, Tyurin VA, Hammond VJ, Herbach N, Aichler M, Walch
A, Eggenhofer E, et al: Inactivation of the ferroptosis regulator
Gpx4 triggers acute renal failure in mice. Nat Cell Biol.
16:1180–1191. 2014.PubMed/NCBI View Article : Google Scholar
|
|
72
|
Martin-Sanchez D, Ruiz-Andres O, Poveda J,
Carrasco S, Cannata-Ortiz P, Sanchez-Niño MD, Ruiz Ortega M, Egido
J, Linkermann A, Ortiz A and Sanz AB: Ferroptosis, but not
necroptosis, is important in nephrotoxic folic acid-induced AKI. J
Am Soc Nephrol. 28:218–229. 2017.PubMed/NCBI View Article : Google Scholar
|
|
73
|
Shah R, Shchepinov MS and Pratt DA:
Resolving the role of lipoxygenases in the initiation and execution
of ferroptosis. ACS Cent Sci. 4:387–396. 2018.PubMed/NCBI View Article : Google Scholar
|
|
74
|
Ha le M, Que do TN, Huyen do TT, Long PQ
and Dat NT: Toxicity, analgesic and anti-inflammatory activities of
tectorigenin. Immunopharmacol Immunotoxicol. 35:336–340.
2013.PubMed/NCBI View Article : Google Scholar
|
|
75
|
Lee HU, Bae EA and Kim DH:
Hepatoprotective effect of tectoridin and tectorigenin on
tert-butyl hyperoxide-induced liver injury. J Pharmacol Sci.
97:541–544. 2005.PubMed/NCBI View Article : Google Scholar
|
|
76
|
Pan CH, Kim ES, Jung SH, Nho CW and Lee
JK: Tectorigenin inhibits IFN-gamma/LPS-induced inflammatory
responses in murine macrophage RAW 264.7 cells. Arch Pharm Res.
31:1447–1456. 2008.PubMed/NCBI View Article : Google Scholar
|
|
77
|
Li J, Yang J, Zhu B, Fan J, Hu Q and Wang
L: Tectorigenin protects against unilateral ureteral obstruction by
inhibiting Smad3-mediated ferroptosis and fibrosis. Phytother Res.
36:475–487. 2022.PubMed/NCBI View Article : Google Scholar
|
|
78
|
Xie J, Ye Z, Li L, Xia Y, Yuan R, Ruan Y
and Zhou X: Ferrostatin-1 alleviates oxalate-induced renal tubular
epithelial cell injury, fibrosis and calcium oxalate stone
formation by inhibiting ferroptosis. Mol Med Rep.
26(256)2022.PubMed/NCBI View Article : Google Scholar
|
|
79
|
Zhang B, Chen X, Ru F, Gan Y, Li B, Xia W,
Dai G, He Y and Chen Z: Liproxstatin-1 attenuates unilateral
ureteral obstruction-induced renal fibrosis by inhibiting renal
tubular epithelial cells ferroptosis. Cell Death Dis.
12(843)2021.PubMed/NCBI View Article : Google Scholar
|
|
80
|
Luo Y, Chen H, Liu H, Jia W, Yan J, Ding
W, Zhang Y, Xiao Z and Zhu Z: Protective effects of ferroptosis
inhibition on high fat diet-induced liver and renal injury in mice.
Int J Clin Exp Pathol. 13:2041–2049. 2020.PubMed/NCBI
|
|
81
|
Yang L, Guo J, Yu N, Liu Y, Song H, Niu J
and Gu Y: Tocilizumab mimotope alleviates kidney injury and
fibrosis by inhibiting IL-6 signaling and ferroptosis in UUO model.
Life Sci. 261(118487)2020.PubMed/NCBI View Article : Google Scholar
|
|
82
|
Zhang Y, Jin D, Kang X, Zhou R, Sun Y,
Lian F and Tong X: Signaling pathways involved in diabetic renal
fibrosis. Front Cell Dev Biol. 9(696542)2021.PubMed/NCBI View Article : Google Scholar
|
|
83
|
Wang Y, Bi R, Quan F, Cao Q, Lin Y, Yue C,
Cui X, Yang H, Gao X and Zhang D: Ferroptosis involves in renal
tubular cell death in diabetic nephropathy. Eur J Pharmacol.
888(173574)2020.PubMed/NCBI View Article : Google Scholar
|
|
84
|
Zhou L, Xue X, Hou Q and Dai C: Targeting
ferroptosis attenuates interstitial inflammation and kidney
fibrosis. Kidney Dis (Basel). 8:57–71. 2021.PubMed/NCBI View Article : Google Scholar
|
|
85
|
Mancias JD, Wang X, Gygi SP, Harper JW and
Kimmelman AC: Quantitative proteomics identifies NCOA4 as the cargo
receptor mediating ferritinophagy. Nature. 509:105–109.
2014.PubMed/NCBI View Article : Google Scholar
|
|
86
|
Zhang Z, Yao Z, Wang L, Ding H, Shao J,
Chen A, Zhang F and Zheng S: Activation of ferritinophagy is
required for the RNA-binding protein ELAVL1/HuR to regulate
ferroptosis in hepatic stellate cells. Autophagy. 14:2083–2103.
2018.PubMed/NCBI View Article : Google Scholar
|
|
87
|
Livingston MJ, Ding HF, Huang S, Hill JA,
Yin XM and Dong Z: Persistent activation of autophagy in kidney
tubular cells promotes renal interstitial fibrosis during
unilateral ureteral obstruction. Autophagy. 12:976–998.
2016.PubMed/NCBI View Article : Google Scholar
|
|
88
|
Li W, Feng G, Gauthier JM, Lokshina I,
Higashikubo R, Evans S, Liu X, Hassan A, Tanaka S, Cicka M, et al:
Ferroptotic cell death and TLR4/Trif signaling initiate neutrophil
recruitment after heart transplantation. J Clin Invest.
129:2293–2304. 2019.PubMed/NCBI View Article : Google Scholar
|
|
89
|
Masaldan S, Clatworthy SAS, Gamell C,
Meggyesy PM, Rigopoulos AT, Haupt S, Haupt Y, Denoyer D, Adlard PA,
Bush AI and Cater MA: Iron accumulation in senescent cells is
coupled with impaired ferritinophagy and inhibition of ferroptosis.
Redox Biol. 14:100–115. 2018.PubMed/NCBI View Article : Google Scholar
|
|
90
|
Hou W, Xie Y, Song X, Sun X, Lotze MT, Zeh
HJ III, Kang R and Tang D: Autophagy promotes ferroptosis by
degradation of ferritin. Autophagy. 12:1425–1428. 2016.PubMed/NCBI View Article : Google Scholar
|
|
91
|
Wang J, Wang Y, Liu Y, Cai X, Huang X, Fu
W, Wang L, Qiu L, Li J and Sun L: Ferroptosis, a new target for
treatment of renal injury and fibrosis in a 5/6 nephrectomy-induced
CKD rat model. Cell Death Discov. 8(127)2022.PubMed/NCBI View Article : Google Scholar
|