Introduction
Coronavirus disease 2019 (COVID-19) has become a
global pandemic; as of December 2022, over 600 million confirmed
cases, including six million deaths, were reported to WHO
(https://covid19.who.int/). COVID-19 is caused by
SARS-CoV-2; the functional receptor for SARS-CoV is ACE-2, which is
highly expressed in epithelial cells in the lung (1). These cause diffuse alveolar damage
and acute respiratory distress syndrome. In addition to pneumonia
and acute respiratory distress syndrome, a wide range of
extra-pulmonary symptoms has been shown in COVID-19, including
cardiac-related symptoms (2). The
most common cardiac-related symptoms in patients with COVID-19 are
increased risk of myocardial infarction, rapidly developing
fulminant myocarditis with reduced left ventricular systolic
function, arrhythmias, venous thromboembolism, and cardiomyopathy
with STEMI-like presentation (2).
Patients with cardiovascular disease (CVD) and COVID-19 may present
with severe symptoms and a higher risk of death. Furthermore, 6~17%
COVID-19 patients were found to develop cardiac arrhythmias and the
patients in ICU had a higher prevalence (~44%) in ICU (3). 12~8% of COVID-19 patients were found
to have acute cardiac injury complications (4). And the consequences of myocarditis in
recovering COVID-19 patients have recently been found to be very
serious and potentially fatal (5).
The population is generally susceptible to COVID-19,
and some immunity can be improved after infection or vaccination
against the new coronavirus (6).
The WHO recommends the monoclonal antibody Sotrovimab for use in
non-serious COVID-19 patients but only in those at the highest risk
of hospital admission. And recommends corticosteroids in
combination with the Janus kinase (JAK) inhibitor Baricitinib to
treat patients with severe or critically ill patients (7). However, these drugs are only
sometimes effective against new variants constantly mutating. With
limited access to treatment, testing laboratory biomarkers is less
expensive, faster, and more readily available and is thought to
provide a predictor of disease severity and prognosis (8).
Myocardial damage in COVID-19 patients is closely
related to the severity of the disease and even the prognosis.
Therefore, early monitoring of cardiac damage by biomarkers is
recommended after hospitalization for COVID-19 infection in
patients with pre-existing CVD. Lactate dehydrogenase (LDH) and
aspartate aminotransferase (AST) are traditional markers of
myocardial injury. There have been several studies assessing the
utility of biomarkers indicating severe COVID-19. Fialek et
al (9) outlined the role of
elevated LDH levels in assessing the severity of COVID-19 but only
considered a single biochemical indicator rather than a
multivariate assessment of multiple biochemical parameters. Most
studies contained multiple indicators, but the indicators are too
broad without a clear target organ. The study by Malik et al
(10) involved indicators such as
lymphocytes, platelets, D-dimer, LDH, AST, alanine
aminotransferase, creatinine, procalcitonin and creatine kinase but
did not target a particular organ. To our knowledge, there has been
no meta-analysis of the association of dual indicators of cardiac
enzyme profiles LDH and AST with COVID-19 prognosis. And the
limitations of observational studies due to geographical location,
single-center experience, and small cohorts prevent these findings
from being generalized. Therefore, we conducted a comprehensive
systematic review and meta-analysis to determine whether AST and
LDH are associated with COVID-19 severity.
Materials and methods
Search strategy
Our work followed the PRISMA guidelines for
reporting systematic reviews and meta-analyses (11). The checklist is presented in Appendix S1. We
have searched Ovid-Medline (https://ovidsp.ovid.com/), PubMed (https://pubmed.ncbi.nlm.nih.gov/), Embase
(https://www.embase.com/search/quick),
and Cochrane library (https://www.cochranelibrary.com/advanced-search) for
studies published up to July 2022. We used the following search
terms and medical subject terms (MeSH): ‘COVID-19’, ‘lactate
dehydrogenase’, ‘aspartate aminotransferase’, ‘SARS-CoV-2’,
‘severity’, ‘mortality’, and ‘prognosis’. In the search process,
MeSH keywords and Boolean operators were used. The PROSPERO
registration number is CRD42022318819.
Study selection
Studies were included if they: i) Observational
studies (cohort, case-control, cross-sectional); ii) Patients were
diagnosed with COVID-19 by qPCR (quantitative real-time PCR); iii)
Reported outcomes in the form of markers to LDH and AST; iv)
Investigation of binary outcomes like ICU versus non-ICU admission,
severe versus non-severe disease, in-hospital mortality versus
discharged alive and survivors.
Exclusion criteria were: i) Lack of information on
LDH and AST levels at the initial diagnosis or follow-up; ii)
Studies investigated pregnant women or children; iii) No clear
grouping of outcome indicators; iv) Letters, reviews, conference
proceedings, guidelines, duplicate publications, or other unrelated
topics are outside the scope of this review.
Data extraction
Two investigators (ZYH and RQY) independently
reviewed the abstracts. A full-text review was conducted when a
given abstract were considered potentially relevant. If there is a
disagreement between investigators, the third author (SKY) gives
suggestions. The two investigators (ZYH and RQY) reviewed whole
papers independently against the inclusion criteria, and if
necessary, any discrepancies were decided by the third author
(SKY). Throughout the screening, the first author's name,
publication date, the number of individuals enrolled, the nation or
region, the patients' basic information (mostly gender and age),
and the LDH and AST levels were separately collected by the two
investigators from the included studies. To assess study quality,
we used the Newcastle-Ottawa Scale (NOS), with a score above six
considered high quality (12).
Statistical analysis
The forest plots of standard mean difference (SMD)
were used to analyze the differences in LDH and AST concentrations
between patients with severe and non-severe COVID-19. The raw data
has been processed to the median and IQR values, making them
acceptable for analysis (13). The
I2 statistic was used to assess inconsistency
across studies. The I2 statistic, more than 75
percent, shows high heterogeneity indicating a random-effects model
to be used (14). The pooled SMD
and associated 95% CIs were calculated. We also used the one-by-one
elimination approach to assess the contribution of each study to
the overall effect of the sensitivity analysis. Egger's test and
funnel curves were used to represent publication bias. The
heterogeneity across studies could be identified through
meta-regression and subgroup analyses. In the meta-regression
study, the following variables were investigated for heterogeneity:
age, gender, region, publication year, and specified outcomes
(severe, non-severe, survive, non-survive). Stata 15.1 (StataCorp,
TX, USA) and RevMan 5.3 (Cochrane Collaboration, Oxford, UK)
software were used for the statistical analysis.
Results
Study selection
A flowchart illustrating the filtering procedure is
shown in Fig. 1, and 592 studies
were initially identified. NoteExpress3.5.0 was used to remove the
duplicated 38 studies. After carefully reading the titles and
abstracts, we excluded 480 studies as they were all irrelevant. We
further excluded 51 articles after a full-text review of the
remaining 74 articles because they either did not report intended
outcomes or intended groups. Finally, of the 4342 patients with
COVID-19 in the 23 studies included in the meta-analysis, 3003
(54.0% male, mean age 52.1 years) had low severity and 1339 (67.3%
male, mean age 64.2 years) had high severity or died.
Characteristics of included
studies
Essential information included in the study is shown
in Table I. The clinical endpoints
for each study were split between severe vs. non-severe (15-25),
survival or discharge vs. non-survival, or ICU (26-37).
At the time of analysis, the non-survival and ICU patients were
classified as the severe group. In contrast, those patients after
hospital discharge and the survival group were considered the
non-severe group. Fifteen studies were conducted in Asia. Eleven of
the studies were in China (15,22,23,25,29,30,32,33,35-37),
one in Japan (31), one in India
(19), one in Iran (28), and one in Pakistan (24); four studies were conducted in
Europe, one in Georgia (21), one
in Belgium (17), one in the
Italian (18), one in Spain
(34); two studies were performed
in the Eurasian country of Turkey (16,26);
one study was performed in African Morocco (27); one study was performed in North
America Mexico (20). All studies
were retrospective observation studies, except one was the
case-control study (15). The
disease severity in nine studies was diagnosed according to the
‘Diagnosis and Treatment of Novel Coronavirus Pneumonia’ developed
by the National Health Care Commission of China (15,22,25,29,30,32,33,36,37);
two studies were determined according to World Health Organization
(WHO) criteria (27,31); five studies were determined based
on clinical and radiological findings (21,23,24,26,35);
seven studies were not clarified (16-20,28,34).
 | Table IBaseline characteristics of COVID-19
patients from included studies. |
Table I
Baseline characteristics of COVID-19
patients from included studies.
| | Severe group | Non-severe
group | |
|---|
| First author
(year) | Region | Number | Mean age, years
(SD) | Male, % | Mean LDH level, U/l
(SD) | Mean AST level, U/l
(SD) | Number | Mean age, years
(SD) | Male, % | Mean LDH level, U/l
(SD) | Mean AST level, U/l
(SD) | NOS | (Refs.) |
|---|
| Akdogan D
(2021) | Turkey | 57 | 51.6 (12.9) | NA | 241.5 (82.4) | 30.0 (14.9) | 118 | 38.6 (11.9) | NA | 191.4 (37.5) | 27.2 (18.1) | 6 | (26) |
| Ayten O (2020) | Turkey | 27 | 64.3 (12.0) | 74.0 | 830.4 (480.2) | 42.4 (26.3) | 46 | 52.6 (12.2) | 58.7 | 511.6 (271.7) | 28.1 (14.2) | 5 | (16) |
| Kantri A
(2021) | Morocco | 45 | 65.0 (13.0) | 77.8 | 327.3 (111.8) | 36.5 (27.2) | 89 | 42.3 (20.3) | 42.7 | 210.3 (66.3) | 20.7 (11.3) | 7 | (27) |
| Azizmohammadi S
(2021) | Iran | 63 | 59.8 (14.1) | 74.6 | 311.3 (82.7) | 33.3 (10.6) | 176 | 43.9 (14.5) | 48.9 | 213.7 (75.5) | 27.0 (10.5) | 6 | (28) |
| Wang D (2020) | China | 71 | 62.3 (12.1) | 62.0 | 349.3 (162.7) | 63.8 (32.9) | 72 | 45.3 (21.2) | 40.3 | 208.0 (78.7) | 43.2(20) | 6 | (29) |
| Sepulchre E
(2022) | Belgium | 60 | 66.5 (14.4) | 71.7 | 510.3 (208.1) | 66.7 (46.3) | 138 | 60.3 (20.2) | 52.2 | 342.7 (173.1) | 42.0 (28.5) | 8 | (17) |
| Zheng F (2020) | China | 30 | 56.5 (15.2) | 46.7 | 244.9 (94.7) | 35.6 (18.3) | 131 | 40.3 (14.2) | 50.4 | 168.0 (56.0) | 23.7 (7.3) | 5 | (30) |
| Pan F (2020) | China | 89 | 67.7 (9.0) | 75.3 | 516.0 (108.1) | 50.0 (23.4) | 35 | 63.7 (21.6) | 51.4 | 378.0 (195.6) | 48.0 (35.6) | 7 | (15) |
| Fukushima K
(2021) | Japan | 41 | 67.3 (18.4) | 90.2 | 467.7 (153.9) | 53.7 (28.4) | 193 | 44.0 (20.9) | 60.6 | 232.8 (82.9) | 28.0 (13.4) | 7 | (31) |
| Bonetti G
(2020) | Italy | 70 | 75.4 (15.0) | 64.3 | 524.0 (166.0) | 62.8 (29.5) | 75 | 62.6 (15.0) | 68.9 | 320.1 (111.8) | 43.0 (25.7) | 7 | (18) |
| Wang H (2021) | China | 24 | 55.7 (15.0) | 62.5 | 267.5 (122.6) | 32.4 (11.3) | 37 | 51.0 (17.0) | 43.2 | 181.2 (32.7) | 22.0 (11.3) | 8 | (32) |
| Huang H (2021) | China | 21 | 61.4 (16.4) | 57.1 | 356.9 (204.6) | 37.0 (30.2) | 43 | 41.2 (15.7) | 58.1 | 209.2 (52.2) | 25.0 (6.9) | 7 | (33) |
| Kumar H (2021) | India | 30 | 58.9 (13.2) | 60.0 | 617.9 (389.4) | 81.3 (51.1) | 79 | 56.1 (16.6) | 79.7 | 448.8 (185.9) | 66.5 (33.6) | 7 | (19) |
| Gómez LC
(2021) | Spain | 166 | 76.0 (14.2) | n | 377.3 (170.1) | 31.0 (16.4) | 376 | 64.0 (12.7) | n | 276.8 (110.3) | 31.0 (16.4) | 9 | (34) |
| Vidal-Cevallos P
(2021) | Mexico | 79 | 46.7 (25.7) | 78.5 | 577.0 (269.6) | 76.3 (80.0) | 298 | 41.8 (22.7) | 72.1 | 389.8 (191.8) | 45.3 (22.7) | 6 | (20) |
| Mo P (2021) | China | 85 | 60.7 (14.3) | 64.7 | 306.7 (181.7) | 29.0 (18.9) | 70 | 45.0 (17.4) | 44.3 | 259.0 (106.0) | 22.7 (13.6) | 9 | (35) |
| Snipelisky D
(2020) | Georgia | 42 | 61.9 (15.5) | 66.7 | 463.0 (224.0) | 66.0 (48.0) | 144 | 60.5 (17.7) | 39.6 | 361.0 (242.0) | 38.0 (22.0) | 7 | (21) |
| Qin W (2021) | China | 23 | 69.3 (7.1) | 43.5 | 458.4 (136.5) | 37.9 (23.7) | 239 | 61.3 (12.2) | 47.3 | 245.0 (80.6) | 25.3 (11.9) | 7 | (22) |
| Zhang W (2021) | China | 16 | 51.2 (14.1) | 68.8 | 759.7 (315.4) | 31.1 (17.2) | 49 | 43.6 (14.2) | 53.1 | 438.3 (71.0) | 23.7 (9.2) | 5 | (36) |
| Feng X (2020) | China | 20 | 69.2 (11.1) | 65.0 | 665.7 (388.6) | 62.8 (68.0) | 94 | 62.8 (13.7) | 61.7 | 285.3 (127.1) | 40.6 (23.5) | 6 | (37) |
| Zhu Y (2020) | China | 29 | 72.6 (13.2) | 65.5 | 785.5 (274.4) | 51.0 (50.0) | 73 | 62.2 (13.6) | 53.4 | 352.1 (140.9) | 32.0 (13.0) | 8 | (23) |
| Yousaf MN
(2022) | Pakistan | 135 | 56.5 (16.3) | n | 702.0 (375.0) | 49.0 (37.0) | 251 | 52.7 (16.0) | n | 498.0 (348.0) | 41.0 (21.0) | 9 | (24) |
| Wang Z (2020) | China | 116 | 72.7 (12.0) | 56.0 | 475.5 (203.4) | 42.0 (23.3) | 177 | 49.6 (22.2) | 41.2 | 231.3 (71.2) | 26.3 (10.5) | 9 | (25) |
Analysis of disease prognosis
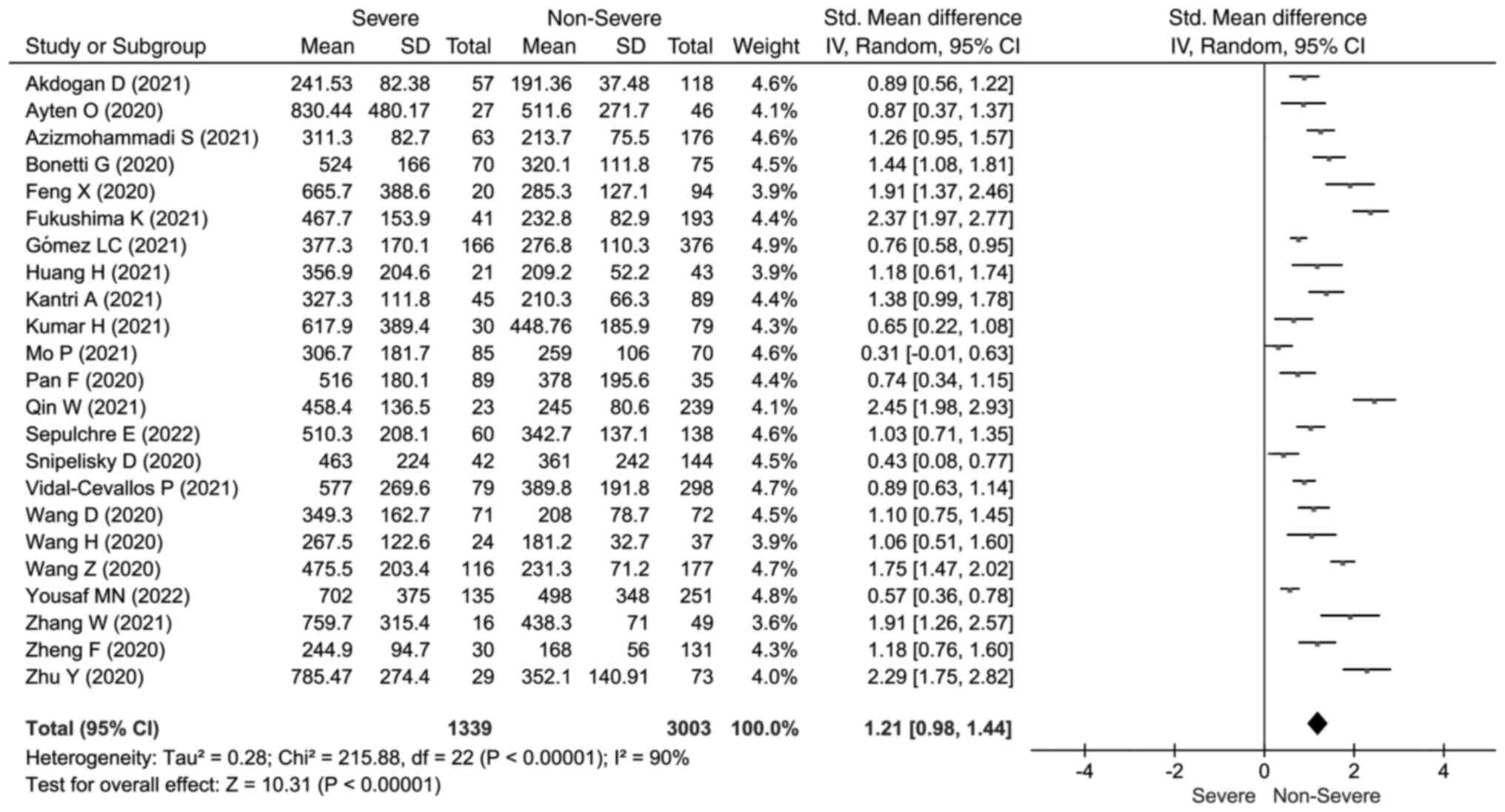
The LDH concentrations heterogeneity test results
show high heterogeneity between studies (I2=90%,
P<0.001). Hence, we used the random-effects model to calculate
the pooled SMD and 95% CIs. The meta-analysis forest plot showed
that severe outcomes presented significantly elevated LDH
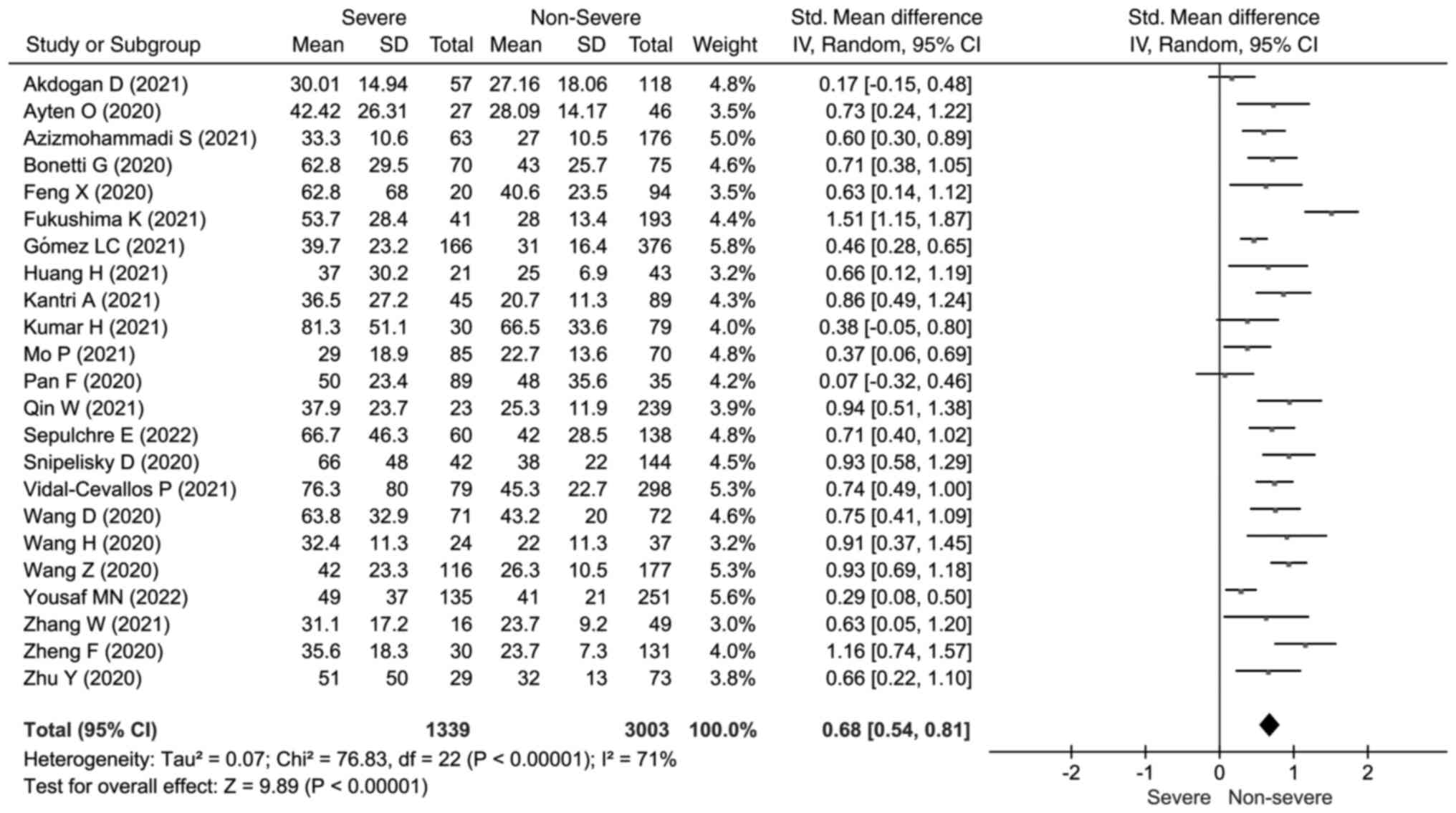
concentrations (SMD=1.21; 95% CI: 0.98, 1.44) (Fig. 2). Elevated AST concentrations were
also found to be concerned with a severe outcome (SMD=0.68; 95% CI:
0.54, 0.81; I2=71%; P<0.001) (Fig. 3).
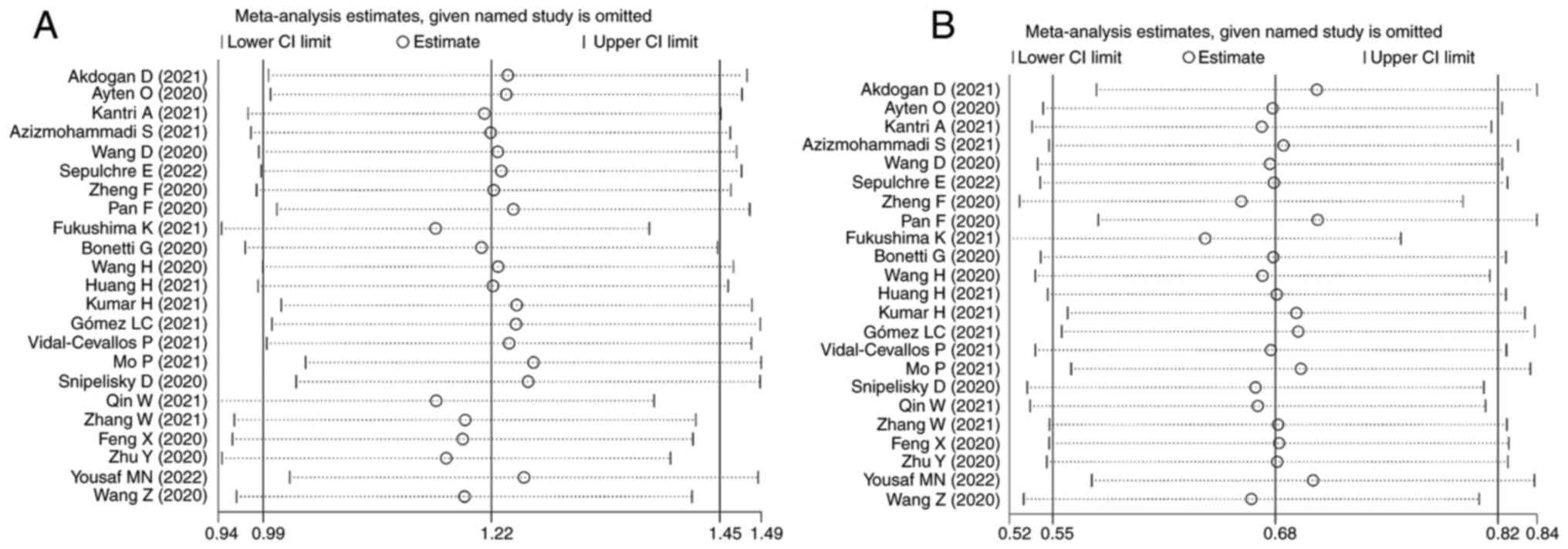
Sensitivity analysis
We did a sensitivity analysis and applied the
leave-one-out method to evaluate each study's influence. The
heterogeneity was found to be considered regardless of whatever
study was omitted. Both LDH and AST sensitivity analyses revealed
no significant differences between studies. The pooled SMD values
did not change after the sequential removal of individual studies
(Fig. 4).
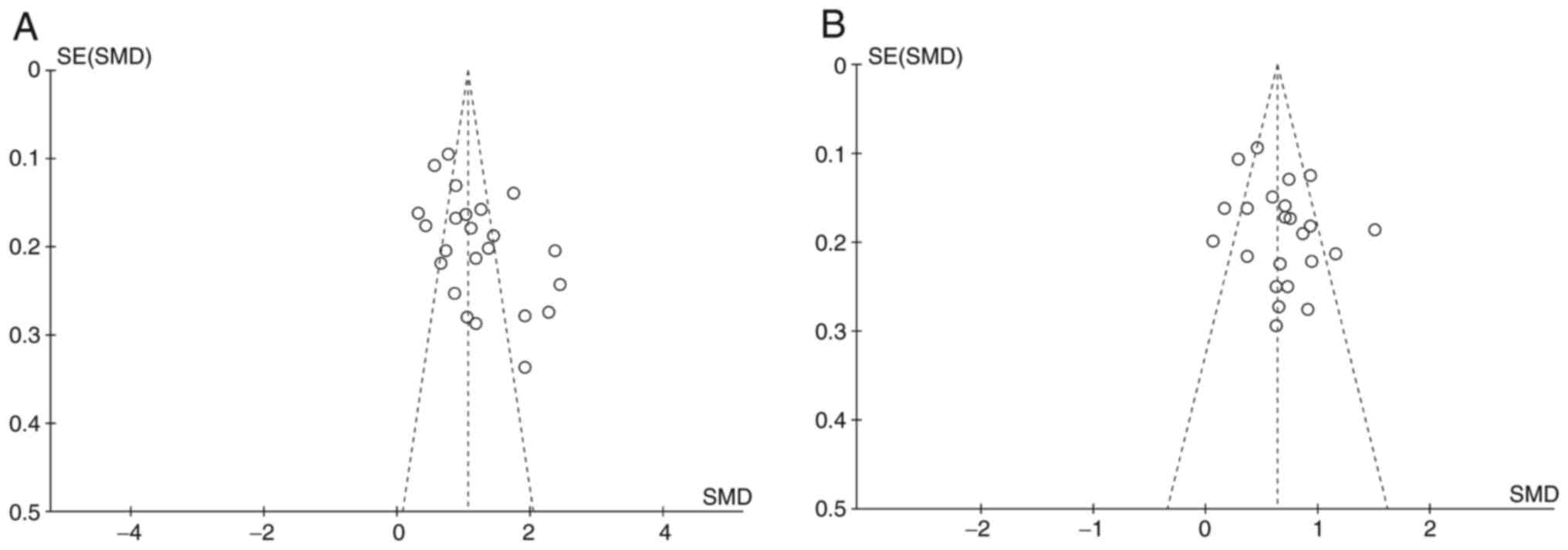
Publish bias analysis
We used funnel plots and Egger's tests to evaluate
publication bias. Using Egger's test, no significant publication
bias was found for all included AST studies (P=0.155), but LDH has
(P=0.015). The funnel plot also showed the difference between the
two groups (Fig. 5).
Meta-regression
The effect estimate did not differ substantially
with outcomes (LDH P=0.909, AST P=0.851), publication year (LDH
P=0.383, AST P=0.977), region (LDH P=0.261, AST P=0.533), and age
(LDH P=0.301, AST P=0.716) according to meta-regression (Table II).
 | Table IIMeta-regression analysis in different
subgroups. |
Table II
Meta-regression analysis in different
subgroups.
| | LDH | AST |
|---|
| Subgroup | Coef | Std. Err. |
P>|z| | Coef | Std. Err. |
P>|z| |
|---|
| Outcomes | -.0371399 | .3256605 | 0.909 | -.0333406 | .1769221 | 0.851 |
| Publication
year | .5294593 | .6070254 | 0.383 | .0099645 | .3463723 | 0.977 |
| Region | -.367411 | .3268494 | 0.261 | -.1122298 | .1800766 | 0.533 |
| Age | .3303795 | .31914 | 0.301 | -.0628999 | .1726357 | 0.716 |
Subgroup analysis
A higher proportion of men with severe COVID-19 was
found in the subgroup analysis (RD 0.21; 95% CIs: 0.06, 0.35;
P<0.001) (Fig. S1). COVID-19
patients with a high severity are also older (SMD 0.70; 95% CIs:
0.53, 0.88; P<0.001) (Fig.
S2). We also performed outcomes, published years, and areas
subgroup analyses to see whether there were any correlations
between the groups (Figs. S3 and
S4). LDH and AST levels were
higher in the experimental group, according to the subgroup
analysis of uncombined outcomes. The rise in the severe vs.
non-severe group was more significant than in the death vs.
discharge group. About half of the studies were conducted in China,
so we separated the studies into two groups, conducted in China or
other countries. The year of publication, as well as the region,
revealed a slight variance in subgroup analysis. Meta-regression
and subgroup analysis failed to identify the cause of the
heterogeneity.
Discussion
LDH catalyzes the conversion of pyruvate (the end
product of glycolysis) to lactate, reversing the liver's Cori cycle
when hypoxia or insufficient supply occurs (38). Multiple organ injury, severe
infections, reduced oxygenation, and activation of the glycolytic
pathway can cause LDH concentration increases. LDH has many
isoenzymes, and LD1 high activities are found in the heart; since
the 1960s, LDH has long been a conventional indicator of cardiac
injury (39). A pooled analysis
indicated that patients with elevated LDH had a 16-fold higher
mortality rate and a more than 6-fold increased probability of
developing severe COVID-19 illness (40). Meanwhile, according to a recent
study, COVID-19 patients' LDH and C-reactive protein levels may
predict respiratory failure. LDH and CRP should be viewed as
helpful tests for the early detection of individuals who need more
aggressive supportive therapy and tighter respiratory monitoring to
avoid poor prognosis (41).
The time-dependent concentration of AST also has
strong sensitivity and specificity to acute myocardial injury. A
multi-center retrospective study showed that the mortality rate of
AST abnormalities in COVID-19 patients was higher than that of
other patients. This result recommended using AST to monitor
COVID-19 patients immediately (42). Interestingly, not all studies
showed that AST abnormalities were associated with higher
in-hospital mortality. For example, Aloisio and Panteghini thinks
the practical significance of AST in COVID-19 patients is affected
by data sources, lack of standardization of commercial assays, and
interference from unqualified specimens (43). AST and LDH are both markers of
myocardial injury, and whether they simultaneously increase and
interact with each other in COVID-19 patients is ambiguous. In a
Chinese cohort study, it has been observed that the increased AST
has a dependence on the LDH of hospitalized COVID-19 patients
(44). A systematic review found
that elevated AST and LDH were independently associated with the
risk of adverse clinical outcomes for COVID-19 patients (10). Our findings were consistent with
the meta-analysis published by Battaglini et al (45) and the review by Stegeman et
al (46). Battaglini's group
researched the possibility of multi-organ impaired in COVID-19
patients and Stegeman explored the accuracy of routine laboratory
tests (including hematological, inflammatory, and other laboratory
biomarkers) to diagnose COVID-19, they both found that LDH and AST
were increased in COVID-19 patients. Hence, the study adds to the
existing knowledge of biomarkers that can be used as a predictor in
the risk stratification model of severe COVID-19.
It is worth noting that both LDH and AST have
widespread activities in numerous body tissues, and they are also
typical markers of sepsis. What is certain is that they are indeed
elevated in patients with severe COVID-19. The potential causes of
cardiac injury in COVID-19 patients are diverse, such as direct
viral damage to cardiomyocytes, cytokines and interferon-induced
inflammation, myocardial interstitial fibrosis, T-cell responses,
disruption of ACE-2 receptors. Moreover, the lung injury may cause
damage to cardiac muscle cells due to hypoxia, and continued
disruption of endothelial function negatively affect the
thrombotic/fibrinolytic balance (47). Additionally, numerous studies have
demonstrated that COVID-19 infections and severe symptoms are more
frequent in individuals with cardiovascular disease (48-50).
The study aimed to investigate the prognostic role of conventional
myocardial enzyme profiles in COVID-19. LDH and AST are
characterized by their long duration at the time of myocardial
injury and better reflect COVID-19 severity. The study also tested
the simultaneous detection of LDH and AST, which is more dominant
than a single indicator indicating myocardial injury. The study
also has certain limitations, including the following. Firstly,
there were considerable discrepancies in effect size estimates due
to differences in the number of included studies and sample sizes.
Second, since no RCTs were published, only observational
retrospective studies were included, which conduct a high risk of
bias. In the future, more randomized controlled studies should be
included. Despite the considerable heterogeneity between studies,
the sensitivity analysis was unaffected by deleting each study. The
explanations for the large to extreme heterogeneity between studies
may be: i) As an emerging infectious disease, the studies included
in this meta-analysis lack a single criterion for grading COVID-19
severity, which could impact the final results. ii) Because of the
varying severity of COVID-19 individuals included in this study and
the considerable age gap between them, there is a significant
difference in the prognostic index values of patients of different
age groups, which could be a source of high heterogeneity. iii)
This is likely owing to a lack of standardization of analytical
methodologies, including various measurement methods. More
standardized diagnostics and test studies, including RCTs, are
needed in the future.
The systematic review and meta-analysis showed that
LDH and AST serum concentrations were considerably higher in
COVID-19 patients with severe disease than in non-severe. These
findings show that LDH and AST could be employed as possible
predictors of prognosis and risk of death in patients with
COVID-19. However, as most of the studies in the current review
were retrospective and had significant heterogeneity, bigger sample
sizes and high-quality prospective cohort studies are required to
confirm this finding.
Supplementary Material
Forest plot of studies reporting
gender in patients with coronavirus disease 2019. M-H,
Mantel-Haenszel; CI, confidence interval; SD, standard deviation;
Std. Mean Difference, standardized mean difference; df, degree of
freedom.
Forest plot of studies reporting age
in patients with coronavirus disease 2019. SD, standard deviation;
Std. Mean Difference, standardized mean difference; IV, inverse
variance; CI, confidence interval; df, degree of freedom.
Forest plot of Subgroup analysis
reporting lactate dehydrogenase concentrations in patients with
coronavirus disease 2019. SD, standard deviation; Std. Mean
Difference, standardized mean difference; IV, inverse variance; CI,
confidence interval; df, degree of freedom.
Forest plot of Subgroup analysis
reporting aspartate aminotransferase concentrations in patients
with coronavirus disease 2019. SD, standard deviation; Std. Mean
Difference, standardized mean difference; IV, inverse variance; CI,
confidence interval; df, degree of freedom.
PRISMA 2020 Checklist
Acknowledgements
The authors would like to thank Professor Richard
Pang for checking the language usage, grammar, punctuation and
spelling of this article.
Funding
Funding: The present study was funded by the National Natural
Science Foundation of China (grant no. 82070916).
Availability of data and material
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SKY, LZ and FZ conceptualized and designed the
study. ZYH and RQY collected data and drafted the manuscript. JSL,
HBD and YLZ analyzed and interpreted data. SKY gave final approval
of the version to be published. ZYH and RQY confirm the
authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Parasher A: COVID-19: Current
understanding of its pathophysiology, clinical presentation and
treatment. Postgrad Med J. 97:312–320. 2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Abobaker A, Raba A and Alzwi A:
Extrapulmonary and atypical clinical presentations of COVID-19. J
Med Virol. 92:2458–2464. 2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J,
Wang B, Xiang H, Cheng Z, Xiong Y, et al: Clinical characteristics
of 138 hospitalized patients with 2019 novel coronavirus-infected
pneumonia in Wuhan, China. JAMA. 323:1061–1069. 2020.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Huang C, Wang Y, Li X, Ren L, Zhao J, Hu
Y, Zhang L, Fan G, Xu J, Gu X, et al: Clinical features of patients
infected with 2019 novel coronavirus in Wuhan, China. Lancet.
395:497–506. 2020.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Liu J, Deswal A and Khalid U: COVID-19
myocarditis and long-term heart failure sequelae. Curr Opin
Cardiol. 36:234–240. 2021.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Islam MT, Nasiruddin M, Khan IN, Mishra
SK, Kudrat-E-Zahan M, Riaz TA, Ali ES, Rahman MS, Mubarak MS,
Martorell M, et al: A perspective on emerging therapeutic
interventions for COVID-19. Front Public Health.
8(281)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Kmietowicz Z: Covid-19: WHO recommends
baricitinib and sotrovimab to treat patients. BMJ.
376(097)2022.PubMed/NCBI View
Article : Google Scholar
|
|
8
|
Wu X, Deng KD, Li C, Yang Z, Hu H, Cai H,
Zhang C, He T, Zheng F, Wang H, et al: Cardiac involvement in
recovered patients from COVID-19: A preliminary 6-month follow-up
study. Front Cardiovasc Med. 8(654405)2021.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Fialek B, Pruc M, Smereka J, Jas R,
Rahnama-Hezavah M, Denegri A, Szarpak A, Jaguszewski MJ, Peacock FW
and Szarpak L: Diagnostic value of lactate dehydrogenase in
COVID-19: A systematic review and meta-analysis. Cardiol J.
29:751–758. 2022.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Malik P, Patel U, Mehta D, Patel N, Kelkar
R, Akrmah M, Gabrilove JL and Sacks H: Biomarkers and outcomes of
COVID-19 hospitalisations: Systematic review and meta-analysis. BMJ
Evid Based Med. 26:107–108. 2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Liberati A, Altman DG, Tetzlaff J, Mulrow
C, Gøtzsche PC, Ioannidis JP, Clarke M, Devereaux PJ, Kleijnen J
and Moher D: The PRISMA statement for reporting systematic reviews
and meta-analyses of studies that evaluate healthcare
interventions: Explanation and elaboration. BMJ.
339(b2700)2009.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Stang A: Critical evaluation of the
Newcastle-Ottawa scale for the assessment of thequality of
nonrandomized studies in meta-analyses. Eur J Epidemiol.
25:603–605. 2010.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Wan X, Wang W, Liu J and Tong T:
Estimating the sample mean and standard deviation from the
samplesize, median, range and/or interquartile range. BMC Med Res
Methodol. 14(135)2014.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Bowden J, Tierney JF, Copas AJ and Burdett
S: Quantifying, displaying and accounting for heterogeneity in the
meta-analysis of RCTs using standard and generalised Q statistics.
BMC Med Res Methodol. 11(41)2011.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Pan F, Yang L, Li Y, Liang B, Li L, Ye T,
Li L, Liu D, Gui S, Hu Y and Zheng C: Factors associated with death
outcome in patients with severe coronavirus disease-19 (COVID-19):
A case-control study. Int J Med Sci. 17:1281–1292. 2020.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ayten O and Saylan B: Retrospective
analysis of severe COVID-19 pneumonia patients treated with
lopinavir/ritonavir: A comparison with survivor and non-survivor
patients. S Afr J Infect Dis. 35(233)2020.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Sepulchre E, Pittie G, Stojkovic V,
Haesbroek G, Crama Y, Schyns M, Paridaens H, de Marchin J, Degesves
S, Biemar C, et al: Covid-19: Contribution of clinical
characteristics and laboratory features for early detection of
patients with high risk of severe evolution. Acta Clin Belg.
77:261–267. 2022.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Bonetti G, Manelli F, Patroni A,
Bettinardi A, Borrelli G, Fiordalisi G, Marino A, Menolfi A,
Saggini S, Volpi R, et al: Laboratory predictors of death from
coronavirus disease 2019 (COVID-19) in the area of Valcamonica,
Italy. Clin Chem Lab Med. 58:1100–1105. 2020.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Kumar H, Gupta P and Shakya S: Predictors
of mortality in patients of COVID-19 pneumonia in intensive care
unit: An observational study in a tertiary care hospital, Lucknow,
India. J Clin Diagnostic Res. 15:C11–C15. 2021.
|
|
20
|
Vidal-Cevallos P, Higuera-De-La-Tijera F,
Chávez-Tapia NC, Sanchez-Giron F, Cerda-Reyes E, Rosales-Salyano
VH, Servin-Caamaño A, Vázquez-Medina MU and Méndez-Sánchez N:
Lactate-dehydrogenase associated with mortality in hospitalized
patients with COVID-19 in Mexico: A multi-centre retrospective
cohort study. Ann Hepatol. 24(100338)2021.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Snipelisky D, Johnson R, Prasad R, Lakhani
B and Ellington J: Characteristics and outcomes based on perceived
illness severity in SARS-CoV-2. South Med J. 113:618–622.
2020.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Qin W, Bai W, Liu K, Liu Y, Meng X, Zhang
K and Zhang M: Clinical course and risk factors of disease
deterioration in critically Ill patients with COVID-19. Hum Gene
Ther. 32:310–315. 2021.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Zhu Y, Du Z, Zhu Y, Li W, Miao H and Li Z:
Evaluation of organ function in patients with severe COVID-19
infections. Med Clin (Barc). 155:191–196. 2020.PubMed/NCBI View Article : Google Scholar : (In English,
Spanish).
|
|
24
|
Yousaf MN, Sarwar S, Tarique S, Ahmed M
and Tahir H: Mortality in patients of COVID-19 infection:
Biochemical markers and its cut-off values for predicting outcome.
J Coll Physicians Surg Pak. 32:37–41. 2022.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Wang Z, Ye D, Wang M, Zhao M, Li D, Ye J,
Liu J, Xu Y, Zhang J, Pan W, et al: Clinical features of COVID-19
patients with different outcomes in Wuhan: A retrospective
observational study. Biomed Res Int. 2020(2138387)2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Akdogan D, Guzel M, Tosun D and Akpinar O:
Diagnostic and early prognostic value of serum CRP and LDH levels
in patients with possible COVID-19 at the first admission. J Infect
Dev Ctries. 15:766–772. 2021.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Kantri A, Ziati J, Khalis M, Haoudar A, El
Aidaoui K, Daoudi Y, Chikhaoui I, El Yamani K, Mouhaoui M, El
Bakkouri J, et al: Hematological and biochemical abnormalities
associated with severe forms of COVID-19: A retrospective
single-centre study from Morocco. PLoS One.
16(e246295)2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Azizmohammadi S, Azizmohammadi S,
Dahmardeh S, Azargashb H, Shokouh SJH, Mohajeri-Iravani M, Mobasher
M, Soleiman-Meigooni S and Zabihi M: Analysis of 239 ordinary and
severe cases of COVID-19:Clinical features and treatment. Eur J
Transl Myol. 31(9579)2021.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wang D, Li R, Wang J, Jiang Q, Gao C, Yang
J, Ge L and Hu Q: Correlation analysis between disease severity and
clinical and biochemical characteristics of 143 cases of COVID-19
in Wuhan, China: A descriptive study. BMC Infect Dis.
20(519)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Zheng F, Tang W, Li H, Huang YX, Xie YL
and Zhou ZG: Clinical characteristics of 161 cases of corona virus
disease 2019 (COVID-19) in Changsha. Eur Rev Med Pharmacol Sci.
24:3404–3410. 2020.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Fukushima K, Yamada Y, Fujiwara S, Tanaka
M, Kobayashi T, Yajima K, Tanaka K, Sekiya N and Imamura A:
Development of a risk prediction score to identify high-risk groups
for the critical coronavirus disease 2019 (COVID-19) in Japan. Jpn
J Infect Dis. 74:344–351. 2021.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Wang H, Xing Y, Yao X, Li Y, Huang J, Tang
J, Zhu S, Zhang Y and Xiao J: Retrospective study of clinical
features of COVID-19 in inpatients and their association with
disease severity. Med Sci Monit. 26(e927674)2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Huang H, Song B, Xu Z, Jiao Y, Huang L,
Zhao P, Huang J, Zhou Z, Zhao Z, Tian J, et al: Predictors of
coronavirus disease 2019 severity: A retrospective study of 64
cases. Jpn J Infect Dis. 74:54–60. 2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Gómez LC, Curto SV, Sebastian MBP, Jiménez
BF and Duniol MD: Predictive model of severity in SARS CoV-2
patients at hospital admission using blood-related parameters.
EJIFCC. 32:255–264. 2021.PubMed/NCBI
|
|
35
|
Mo P, Xing Y, Xiao Y, Deng L, Zhao Q, Wang
H, Xiong Y, Cheng Z, Gao S, Liang K, et al: Clinical
characteristics of refractory coronavirus disease 2019 in Wuhan,
China. Clin Infect Dis. 73:e4208–e4213. 2021.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhang W, Ban Y, Wu YH, Liu JY, Li XH, Wu
H, Li H, Chen R, Yu XX and Zheng R: Epidemiological and clinical
characteristics of 65 hospitalized patients with COVID-19 in
Liaoning, China. World J Clin Cases. 9:2205–2217. 2021.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Feng X, Li P, Ma L, Liang H, Lei J, Li W,
Wang K, Song Y, Li S, Yang W and Yang C: Clinical characteristics
and short-term outcomes of severe patients with COVID-19 in Wuhan,
China. Front Med (Lausanne). 7(491)2020.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Wu Y, Lu C, Pan N, Zhang M, An Y, Xu M,
Zhang L, Guo Y and Tan L: Serum lactate dehydrogenase activities as
systems biomarkers for 48 types of human diseases. Sci Rep.
11(12997)2021.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Chen XY, Huang MY, Xiao ZW, Yang S and
Chen XQ: Lactate dehydrogenase elevations is associated with
severity of COVID-19: A meta-analysis. Crit Care.
24(459)2020.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Henry BM, Aggarwal G, Wong J, Benoit S,
Vikse J, Plebani M and Lippi G: Lactate dehydrogenase levels
predict coronavirus disease 2019 (COVID-19) severity and mortality:
A pooled analysis. Am J Emerg Med. 38:1722–1726. 2020.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Poggiali E, Zaino D, Immovilli P, Rovero
L, Losi G, Dacrema A, Nuccetelli M, Vadacca GB, Guidetti D,
Vercelli A, et al: Lactate dehydrogenase and C-reactive protein as
predictors of respiratory failure in CoVID-19 patients. Clin Chim
Acta. 509:135–138. 2020.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Lei F, Liu YM, Zhou F, Qin JJ, Zhang P,
Zhu L, Zhang XJ, Cai J, Lin L, Ouyang S, et al: Longitudinal
association between markers of liver injury and mortality in
COVID-19 in China. Hepatology. 72:389–398. 2020.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Aloisio E and Panteghini M: Aspartate
aminotransferase in COVID-19: A probably overrated marker. Liver
Int. 41:2809–2810. 2021.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Zhang C, Shi L and Wang FS: Liver injury
in COVID-19: Management and challenges. Lancet Gastroenterol
Hepatol. 5:428–430. 2020.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Battaglini D, Lopes-Pacheco M,
Castro-Faria-Neto HC, Pelosi P and Rocco PRM: Laboratory biomarkers
for diagnosis and prognosis in COVID-19. Front Immunol.
13(857573)2022.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Stegeman I, Ochodo EA, Guleid F, Holtman
GA, Yang B, Davenport C, Deeks JJ, Dinnes J, Dittrich S, Emperador
D, et al: Routine laboratory testing to determine if a patient has
COVID-19. Cochrane Database Syst Rev. 11(CD013787)2020.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Babapoor-Farrokhran S, Gill D, Walker J,
Rasekhi RT, Bozorgnia B and Amanullah A: Myocardial injury and
COVID-19: Possible mechanisms. Life Sci. 253(117723)2020.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Evans PC, Rainger GE, Mason JC, Guzik TJ,
Osto E, Stamataki Z, Neil D, Hoefer IE, Fragiadaki M, Waltenberger
J, et al: Endothelial dysfunction in COVID-19: A position paper of
the ESC working group for atherosclerosis and vascular biology, and
the ESC council of basic cardiovascular science. Cardiovasc Res.
116:2177–2184. 2020.PubMed/NCBI View Article : Google Scholar
|
|
49
|
Chung MK, Zidar DA, Bristow MR, Cameron
SJ, Chan T, Harding CV III, Kwon DH, Singh T, Tilton JC, Tsai EJ,
et al: COVID-19 and cardiovascular disease: From bench to bedside.
Circ Res. 128:1214–1236. 2021.PubMed/NCBI View Article : Google Scholar
|
|
50
|
Weckbach LT, Curta A, Bieber S, Kraechan
A, Brado J, Hellmuth JC, Muenchhoff M, Scherer C, Schroeder I,
Irlbeck M, et al: Myocardial inflammation and dysfunction in
COVID-19-associated myocardial injury. Circ Cardiovasc Imaging.
14(e12220)2021.PubMed/NCBI View Article : Google Scholar
|