Introduction
Rheumatoid arthritis (RA) is a complex,
heterogeneous, progressive and long-term autoimmune disease
characterized by symmetrical joint inflammation and bone erosion
(1). The global prevalence of RA
is 0.3-1.0% in females, which is five times higher than that in
males (2). The risk factors for RA
include genetic susceptibility, infectious agents, oxidative stress
and inflammatory cytokines. RA is characterized by extensive
infiltration of immune cells in the synovium, synovial hyperplasia,
joint inflammation and excessive pro-inflammatory cytokine
production. Oxidative stress caused by increased production of
reactive oxygen species (ROS) and inflammatory cytokines
contributes to the pathogenesis of RA (3-5).
MicroRNAs (miRNAs or miRs) are non-coding RNAs that
are ~22 nucleotides long; they regulate the expression of target
genes by binding to the ‘seed region’, 2-8 nucleotides of the 3'
untranslated region (3'-UTR), as a post transcriptional regulator
of messenger RNA (mRNA). The perfect complementarity between miRNAs
and target mRNA sequences may lead to RNA silencing, resulting in
reduced protein expression (6-9).
A single nucleotide polymorphism (SNP) is a polymorphism of a DNA
sequence caused by single nucleotide variation at the genome level.
SNPs in the 3'-UTR of the target gene might affect its binding
affinity to the corresponding miRNA, altering gene expression,
which could change the biological functions of cells and initiate
the onset of the diseases (10).
The miRNA-related SNPs (miR-SNPs) rs2296135 and rs1131445 in
interleukin-15 receptor α (IL-15RA) and IL-16 gene are
associated with risk of RA (11,12).
SET domain containing (lysine methyltransferase) 8
(SET8), also known as PR-SET domain-containing protein
7 (PR-SET7) or Lysine methyltransferase 5a
(KMT5a), encodes histone H4 lysine 20 monomethyl
transferase, which is involved in cell cycle, DNA damage repair and
cancer development (13-16).
SET8 regulates the host immune response through the
miR-30e-3p/FoxO3a/SET8 axis during mycobacterial infection
(17). miR-502 regulates
SET8 expression via its binding site in the 3'-UTR of
SET8 mRNA and SNP rs16917496 at this site is associated with
SET8 expression (14-16).
Keratin is involved in RA; therefore, anti-keratin antibodies are
used for RA diagnosis (18).
Keratin 81 (KRT81) is a member of the keratin gene family
that encodes hair keratin, which is a key component of the
intermediate filaments and maintains mechanical stability and
integrity of epithelial cells (19). Moreover, SNP rs3660 in the miRNA
binding site of the 3'-UTR of KRT81 mediates the expression
of KRT81 (20,21). In previous studies, miR-SNPs have
been associated with the risk of RA (11,12);
therefore, the present case-control study aimed to assess the
association of miR-SNPs with risk of RA.
Materials and methods
Blood collection and DNA
extraction
A total of 2 ml blood samples were collected from 82
patients with RA from May to December 2017 at the Department of
Immunology and Rheumatology of the Second Hospital of Hebei Medical
University (Shijiazhuang, China). Blood samples were also collected
from 105 sex- and age- matched healthy controls. The diagnostic
standard for RA was based on the 2010 revised standard of the
American College of Rheumatology. Inclusion criteria were patients
diagnosed as RA who were between 18 and 80 years old (inclusive)
who had not yet started drug therapy. Patients <18 or > 80
years old, pregnant and lactating patients and patients with
infectious inflammatory diseases were excluded (22). The clinical characteristics of
patients with RA, including age, sex, disease activity score
(DAS28) and laboratory results, such as anti-cyclic citrullinated
peptide antibody (anti-CCP) and C-reactive protein (CRP) levels,
erythrocyte sedimentation rate (ESR) and rheumatoid factor (RF)
levels, were recorded. Disease activity score was based on DAS28:
≤2.6, clinical remission; >2.6 and ≤3.2, low activity; >3.2
and ≤5.1, moderate activity and >5.1, high activity (23). DNA from blood samples was extracted
using a TIANamp Blood Clot DNA Kit (Tiangen) according to the
manufacturer's instruction. All procedures were approved by the
Human Tissue Research Committee of The Second Hospital of Hebei
Medical University (approval no. 2017-P031). All participants
provided written informed consent prior to enrollment.
PCR and sequence analysis
SNPs of the miRNA binding sites of SET8
(rs16917496) and KRT81 (rs3660) were genotyped based on the
sequences available in the National Center for biotechnology
information SNP database (ncbi.nlm.nih.gov/snp/) using a PCR-ligase detection
reaction assay that amplified the DNA fragment flanking the
miR-SNPs. The probes used for miR-SNPs genotyping were as follows:
Probe S1 and S2 located upstream of the SNPs matched alleles of the
miR-SNPs with length difference of three base pairs and probe S3
located downstream of SNPs with the complementary sequence.
Following ligation with the PCR products using probes S1 + S3 or S2
+ S3, SNPs were detected and verified based on length of ligated
products. The sequences of primers and probes are listed in
Table I. PCR was performed using a
DreamTaq Green PCR Master Mix (2X) (K1081) (Shanghai Yisheng
Biotechnology Co., Ltd.). The thermocycling conditions were as
follows: 5 min denaturation at 95˚C followed by 35 cycles of 20 sec
denaturation at 94˚C, 20 sec annealing at 55˚C and 40 sec extension
at 72˚C and final extension at 72˚C for 10 min. Ligation was
performed using different probes and the ligated products were
separated using an ABI PRISM genetic analyzer 3730XL (Applied
Biosystems; Thermo Fisher Scientific, Inc.). Polymorphisms were
confirmed by repeating the analysis of two DNA strands.
 | Table IPrimers and probes for
microRNA-associated SNP genotyping. |
Table I
Primers and probes for
microRNA-associated SNP genotyping.
| Gene | SNP | Primer sequence | Probe sequence |
|---|
| SET8 | rs16917496 | F:
5'-TCAGCAAATTGGA | S1:
5'-ctgactgaTTGTGGTTTAGCTTTGTATTTAAAC-3' |
| | (C/T) | AGAGGAT-3’ | S2:
5'-ctgactgactgTTGTGGTTTAGCTTTGTATTTAAAT-3' |
| | | R:
5’-GTGGCCTTGCCTG | S3:
5'-AAGGAAATAAACTTGAAAATTATTTGTCATC-3' |
| | | GCAAT-3’ | |
| KRT81 | rs3660 | F:
5'-GGGGATCACACAG | S1:
5'-ctgactgactgaGAGTGGGAGGGGTCTTTCAAAGTGC-3' |
| | (C/G) | AGAAATGT-3’ | S2:
5'-ctgactgactgactgGAGTGGGAGGGGTCTTTCAAAGTGG-3' |
| | | R:
5’-ATCTTTCCTGCCC | S3:
5'-AGGAGAAGTAGCTGAGCACTTGCTCctgactgac-3' |
| | | TGCCTTG-3’ | |
Western blotting
Western blotting was performed to confirm the
association between SET8 expression and SNPs rs16917496.
Total protein was isolated from human peripheral blood mononuclear
cells using a ReadyPrep protein extraction kit (Bio-Rad
Laboratories, Inc.) according to the manufacturer's instructions.
BCA assay was used to determine protein concentration. A total of
40 µg total protein/lane was loaded in a 10% denaturing
polyacrylamide gel for separation and transferred to polyvinylidene
difluoride membranes. The membranes were blocked using 5% skimmed
milk powder with TBST containing 0.1% Tween-20 for 1 h at room
temperature, then incubated at 4˚C overnight with the following
primary antibodies: Mouse monoclonal anti-SET8 (Abcam; cat.
no. ab3798; 1:500) or anti-β-actin antibody (Santa Cruz
Biotechnology, Inc.; cat. no. SC-47778A; 1:20,000). The membranes
were incubated with horseradish peroxidase-conjugated anti-mouse
IgG secondary antibody (Thermo Fisher Scientific, Inc.; cat. no.
31430, 1:10,000) at room temperature for 2 h. FluorChem HD2
(ProteinSimple) was used to detect signals.
Determination of ROS levels
BBOXiProbe® serum active oxygen detection
kit (BestBio Technology) was used to determine total serum ROS
levels. Briefly, blood was centrifuged at 1,000 x g for 5 min at
25˚C to obtain serum, and 100 µl serum with 10 µl O12 probe were
incubated at 37˚C for 30 min. ROS levels were measured using a
fluorescence microplate reader (BioTek Instruments, Inc.) at an
excitation wavelength of 488 nm and an emission wavelength of 520
nm.
Determination of cytokine levels
Human TH1/TH2 panel (8-plex) in a filter plate V02
(BioLegend, Inc.) was used to measure the levels of IL-5, 13, 2, 6,
10 and 4, interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α).
A total of 25 µl serum sample (two-fold diluted with assay buffer)
was incubated for 30 min at 25˚C with 25 µl Human Th Panel
Detection antibody (BioLegend, Inc.; cat. no. 741041, stock
solution), mixed with 25 µl Capture beads (BioLegend) for 1 h in
the dark at room temperature and shaken using an oscillator at ~800
rpm for 1 h at 25˚C. The sample was supplemented with 25 µl
streptavidin-phycoerythrin and shaken using an oscillator at ~800
rpm in the dark for 30 min at room temperature. The PE fluorescence
signal of the analyte-specific bead region was quantitatively
analyzed using MACSQuant analyzer 10 (Miltenyi Biotec GmbH) and the
analyte concentration was determined using LEGENDplex™ software
V8.0 (BioLegend, Inc.).
Statistical analysis
Unpaired student's t test was used for the analysis
of continuous variables of independent samples. Data are presented
as the mean and standard deviation. The independent experiments
were repeated three times. Wilcoxon rank-sum test was used if the
assumption of normality was incomplete. χ2 or Fisher's
exact test was used to evaluate categorical variables in the
contingency tables. All statistical analysis was performed using
SPSS software (version 25.0; IBM Corp.). P<0.05 was considered
to indicate a statistically significant difference.
Results
SET8 genotype is associated with risk
of RA
A total of 82 patients with RA were compared with
105 sex- and age- matched healthy controls. The clinical
characteristics of patients and the control group are listed in
Table II.
 | Table IIClinical characteristics of patients
with rheumatoid arthritis and controls. |
Table II
Clinical characteristics of patients
with rheumatoid arthritis and controls.
| Characteristic | Patients
(n=82) | Controls
(n=105) |
t/χ2 | P-value |
|---|
| Mean age,
years | 53.71±11.59 | 52.02±8.49 | 1.11 | 0.27 |
| Sex,
male/female | 16/66 | 24/81 | 0.31 | 0.58 |
| Other immunological
disease, yes/no | 37/45 | NA | NA | NA |
| Anti-CCP, +/- | 53/29 | NA | NA | NA |
| CRP, +/- | 61/21 | NA | NA | NA |
| ESR, +/- | 70/12 | NA | NA | NA |
| RF, +/- | 61/21 | NA | NA | NA |
| DAS28 | | | | |
|
≤2.6 | 2 | NA | NA | NA |
|
2.6<DAS28≤3.2 | 3 | NA | NA | NA |
|
3.2<DAS28≤5.1 | 18 | NA | NA | NA |
|
>5.1 | 59 | NA | NA | NA |
SNPs in the miRNA binding sites of SET8
(rs16917496) and KRT81 (rs3660) in patients with RA and
healthy controls were genotyped to evaluate their impact on the
risk of RA. There was no significant difference in the genotype
distribution frequency of SNP rs3660 in KRT81 (P=0.091). For
SNP rs16917496 in SET8, the frequencies of genotypes CC and
CT + TT were 10.98 and 89.02 in patients with RA and 2.86 and
97.14% in controls, respectively. The CC genotype was associated
with a 4.192-fold increased risk of RA compared with that of CT +
TT (odds ratio, 4.192; 95% CI: 1.097-16.021; P=0.025; Table III). The CC genotype of
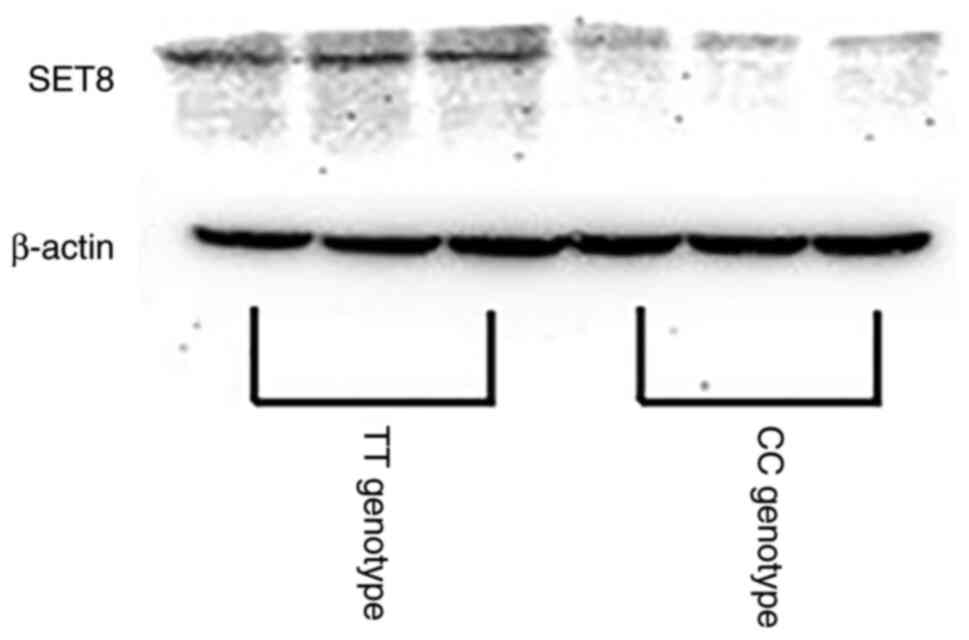
SET8 was also associated with lower protein expression than
TT (Fig. 1). These data implied
that SNP rs16917496 was associated with SET8 expression and
regulated RA onset.
 | Table IIIFrequency distribution of SNP sites
in patients with rheumatoid arthritis and controls. |
Table III
Frequency distribution of SNP sites
in patients with rheumatoid arthritis and controls.
| Gene (SNP) | Genotype | Patients
(n=82) | Controls
(n=105) | χ2 | P-value | OR | 95%CI |
|---|
| SET8 | CC | 9 | 3 | 5.053 | 0.025 | 4.192 | 1.097-16.021 |
| (rs16917496) | CT + TT | 73 | 102 | | | | |
| KRT81 | CC | 56 | 59 | 2.848 | 0.091 | 1.679 | 0.918-3.072 |
| (rs3660) | CG + GG | 26 | 46 | | | | |
Association of SET8 genotype with ROS
and cytokines levels
In previous studies, increased ROS levels have been
reported in patients with RA (24,25);
therefore, the present study investigated the relationship between
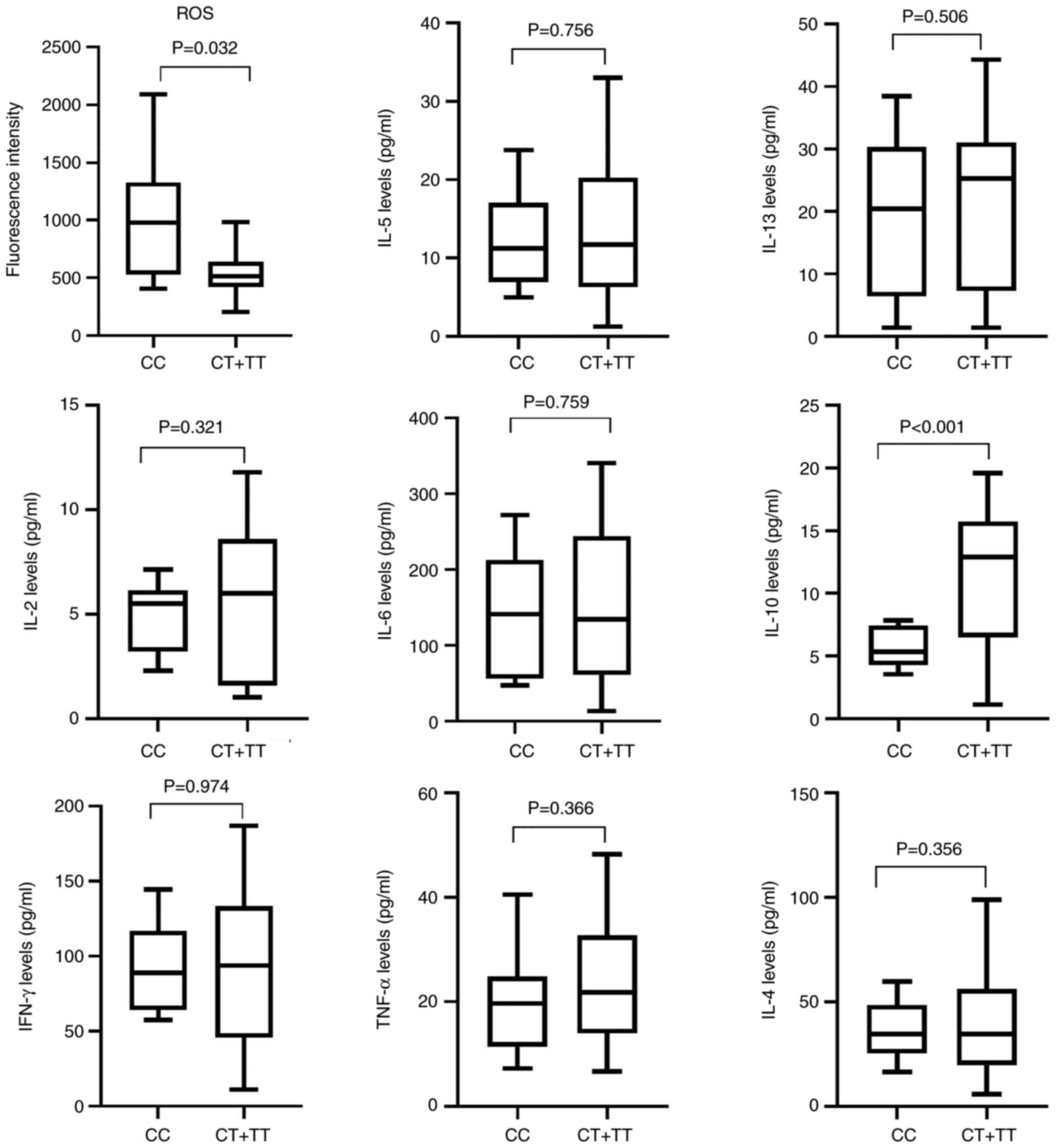
SNP rs16917496 and ROS levels in patients with RA. ROS levels were
higher in the CC genotype carriers (1011.500±536.426 vs.
548.616±190.508, P=0.032, Fig. 2)
than in the CT + TT genotype carriers. CC genotype was
significantly associated with lower IL-10 levels (P<0.001;
Fig. 2). These data suggested that
SNPs regulated RA development by mediating ROS production and
cytokine expression. There was no significant association between
SNP rs16917496 and clinical characteristics of the participants,
including age, sex, levels of anti-CCP and CRP, ESR, RF levels and
DAS28 (data not shown).
Discussion
miRNAs serve important roles in the
post-transcriptional regulation by controlling mRNA translation
(26,27). SNPs in the miRNA binding sites can
alter miRNA-mRNA binding affinity, which alters expression of the
corresponding genes, thereby affecting cell proliferation,
apoptosis and development. This process is associated with
susceptibility to diseases, including cancer and autoimmune
diseases (20,28,29).
Here, SNP rs16917496 in the miR-502 binding site of the 3'-UTR of
SET8 mRNA was associated with the occurrence of RA; low
SET8 and higher ROS levels and decreased IL-10 expression
were associated with CC genotype and increased risk of RA.
Consistent with the results of a previous study, the
present study elucidated that the CC genotype of SET8 was
associated with low SET8 expression (30). SET8 overexpression inhibits
high glucose-mediated ROS accumulation in human umbilical vein
endothelial cells (31) and a
decrease in SET8 levels could enhance intracellular ROS
levels. Oxidative stress is involved in the development of
angiogenesis, synovial proliferation and inflammatory infiltration
in RA (32,33). ROS can damage cartilage and
extracellular matrix components by promoting the formation of
rheumatoid factors and reducing the synthesis of collagen and
proteoglycans in the pathogenesis of RA (34). Moreover, ROS oxidize lipids,
nucleic acids and proteins to accelerate RA development (33). Therefore, functional analysis is
required to assess the impact of enhanced ROS production induced by
low SET8 levels during RA onset.
IL-10 inhibits expression of inflammatory factors,
such as TNF-α, IL-17 and IL-1 β via the NF-κB signaling pathway
(35,36). Moreover, IL-10 overexpression in
bone marrow mesenchymal stem cells significantly alleviates RA
symptoms by decreasing proliferation of synovium and promotes the
repair of articular cartilage in collagen-induced arthritis (CIA)
rats (36). It also decreases
synovial inflammation, bone destruction and articular cartilage
damage in CIA rats (37). The
underlying mechanisms for RA risk-associated SNPs and IL-10, and
that between SET8 and IL-10, should be evaluated.
The present findings suggested that the SNPs in the
miR-502 binding site of the 3'-UTR of SET8 mRNA were
predictors of RA risk and may regulate RA pathogenesis by mediating
expression of SET8 to regulate ROS and IL-10 levels.
Acknowledgements
Not applicable.
Funding
Funding: The present study was supported by the Key Science and
Technology Research Program from Health Commission of Hebei
Province (grant no. 20221248).
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
SZ designed the experiments. YZ and ZS collected
tissue specimens. XZ and JZ performed the experiments. CP and SZ
interpreted the data and drafted the manuscript. SZ and CP confirm
the authenticity of all the raw data. All authors have read and
approved the final manuscript.
Ethics approval and consent to
participate
The present study involving human participants was
approved by The Human Tissue Research Committee at the Second
Hospital of Hebei Medical University (approval no. 2017-P031).
Patient consent for publication
All subjects provided written informed consent to
participate in this study.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Behl T, Upadhyay T, Singh S, Chigurupati
S, Alsubayiel AM, Mani V, Vargas-De-La-Cruz C, Uivarosan D, Bustea
C, Sava C, et al: Polyphenols targeting MAPK mediated oxidative
stress and inflammation in rheumatoid arthritis. Molecules.
26(6570)2021.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Dar WR, Mir IA, Siddiq S, Nadeem M and
Singh G: The assessment of fatigue in rheumatoid arthritis patients
and its impact on their quality of life. Clin Pract. 12:591–598.
2022.PubMed/NCBI View Article : Google Scholar
|
|
3
|
Alturaiki W, Alhamad A, Alturaiqy M, Mir
SA, Iqbal D, Bin Dukhyil AA, Alaidarous M, Alshehri B, Alsagaby SA,
Almalki SG, et al: Assessment of IL-1β, IL-6, TNF-α, IL-8, and CCL
5 levels in newly diagnosed Saudi patients with rheumatoid
arthritis. Int J Rheum Dise. 25:1013–1019. 2022.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Wang X, Fan D, Cao X, Ye Q, Wang Q, Zhang
M and Xiao C: The role of reactive oxygen species in the rheumatoid
arthritis-associated synovial microenvironment. Antioxidants
(Basel). 11(1153)2022.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Mateen S, Zafar A, Moin S, Khan AQ and
Zubair S: Understanding the role of cytokines in the pathogenesis
of rheumatoid arthritis. Clin Chim Acta. 455:161–171.
2016.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297.
2004.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Ambros V: The functions of animal
microRNAs. Nature. 431:350–355. 2004.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Zeng Y, Yi R and Cullen BR: MicroRNAs and
small interfering RNAs can inhibit mRNA expression by similar
mechanisms. Proc Natl Acad Sci USA. 100:9779–9784. 2003.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Zeng Y, Wagner EJ and Cullen BR: Both
natural and designed micro RNAs can inhibit the expression of
cognate mRNAs when expressed in human cells. Mol Cell. 9:1327–1333.
2002.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Wang X, Tomso DJ, Liu X and Bell DA:
Single nucleotide polymorphism in transcriptional regulatory
regions and expression of environmentally responsive genes. Toxicol
App Pharmacol. 207:84–90. 2005.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Jadidi N, Alesaeidi S, Arab F, Pakzad B,
Siasi E and Esmaeilzadeh E: MiRNA-binding site polymorphism in
IL-15RA gene in rheumatoid arthritis and systemic lupus
erythematosus: Correlation with disease risk and clinical
characteristics. Clin Rheumatol. 41:3487–3494. 2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Zeinalzadeh S, Kheradmand N, Rasouli G,
Esmaeilzadeh E, Pakzad B, Behroozi J, Chamanara M, Zoshk MY,
Ehtesham N and Sabet MN: Association of a miRNA-binding site
polymorphism in IL-16 gene with disease risk and clinical
characteristics of rheumatoid arthritis and systemic lupus
erythematosus. Clin Rheumatol. 41:2189–2196. 2022.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Chen X, Ding X, Wu Q, Qi J, Zhu M and Miao
C: Monomethyltransferase SET8 facilitates hepatocellular carcinoma
growth by enhancing aerobic glycolysis. Cell Death Dis.
10(312)2019.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Fang J, Feng Q, Ketel CS, Wang H, Cao R,
Xia L, Erdjument-Bromage H, Tempst P, Simon JA and Zhang Y:
Purification and functional characterization of SET8, a nucleosomal
histone H4-lysine 20-specific methyltransferase. Curr Biol.
12:1086–1099. 2002.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Nishioka K, Rice JC, Sarma K,
Erdjument-Bromage H, Werner J, Wang Y, Chuikov S, Valenzuela P,
Tempst P, Steward R, et al: PR-Set7 is a nucleosome-specific
methyltransferase that modifies lysine 20 of histone H4 and is
associated with silent chromatin. Mol Cell. 9:1201–1213.
2002.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Wu S, Wang W, Kong X, Congdon LM, Yokomori
K, Kirschner MW and Rice JC: Dynamic regulation of the PR-Set7
histone methyltransferase is required for normal cell cycle
progression. Genes Dev. 24:2531–2542. 2010.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Singh V, Prakhar P, Rajmani RS, Mahadik K,
Borbora SM and Balaji KN: Histone methyltransferase SET8
epigenetically reprograms host immune responses to assist
mycobacterial survival. J Infect Dis. 216:477–488. 2017.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Wang XP, Cheng QY, Gu MM, Leng RX, Fan YG,
Li BZ and Ye DQ: Diagnostic accuracy of anti-keratin antibody for
rheumatoid arthritis: A meta-analysis. Clin Rheumatol.
38:1841–1849. 2019.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Moll R, Divo M and Langbein L: The human
keratins: Biology and pathology. Histochem Cell Biol. 129:705–733.
2008.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Sha Z, Lai R, Zhang X, Zhao Y, Wu J, Geng
C and Guo Z: A polymorphism at the microRNA binding Site in the 3'
untranslated region of KRT81 is associated with breast cancer. DNA
and Cell Biol. 39:1886–1894. 2020.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Zhang K, Liang Y, Zhang W, Zeng N, Tang S
and Tian R: KRT81 knockdown inhibits malignant progression of
melanoma through regulating interleukin-8. DNA Cell Biol.
40:1290–1297. 2021.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Aletaha D, Neogi T, Silman AJ, Funovits J,
Felson DT, Bingham CO III, Birnbaum NS, Burmester GR, Bykerk VP,
Cohen MD, et al: 2010 Rheumatoid arthritis classification criteria:
An American college of rheumatology/European league against
rheumatism collaborative initiative. Arthritis Rheum. 62:2569–2581.
2010.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Fransen J and van Riel PL: The disease
activity score and the EULAR response criteria. Rheum Dis Clin
North Am. 35:745–757, vii-viii. 2009.PubMed/NCBI
|
|
24
|
Mukhopadhyay K, De S, Kundu S, Ghosh P,
Chatterjee S and Chatterjee M: Evaluation of levels of oxidative
stress as a potential biomarker in patients with rheumatoid
arthritis. J Family Med Prim Care. 10:1981–1986. 2021.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Mateen S, Moin S, Khan AQ, Zafar A and
Fatima N: Increased reactive oxygen species formation and oxidative
stress in rheumatoid arthritis. PLoS One.
11(e0152925)2016.PubMed/NCBI View Article : Google Scholar
|
|
26
|
He L and Hannon GJ: MicroRNAs: Small RNAs
with a big role in gene regulation. Nat Rev Genet. 5:522–531.
2004.PubMed/NCBI View
Article : Google Scholar
|
|
27
|
Esteller M: Non-coding RNAs in human
disease. Nat Rev Gene. 12:861–874. 2011.PubMed/NCBI View
Article : Google Scholar
|
|
28
|
Zhou M, Jiang B, Xiong M and Zhu X: An
updated meta-analysis of the associations between microRNA
polymorphisms and susceptibility to rheumatoid arthritis. Front
Physiol. 9(1604)2018.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Papathanasiou I, Mourmoura E, Balis C and
Tsezou A: Impact of miR-SNP rs2910164 on miR-146a expression in
osteoarthritic chondrocytes. Adv Med Sci. 65:78–85. 2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Song F, Zheng H, Liu B, Wei S, Dai H,
Zhang L, Calin GA, Hao X, Wei Q, Zhang W and Chen K: An
miR-502-binding site single-nucleotide polymorphism in the
3'-untranslated region of the SET8 gene is associated with early
age of breast cancer onset. Clin Cancer Res. 15:6292–6300.
2009.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Chen X, Wu Q, Jiang H, Wang J, Zhao Y, Xu
Y and Zhu M: SET8 is involved in the regulation of hyperglycemic
memory in human umbilical endothelial cells. Acta Biochim Biophys
Sin(Shanghai). 50:635–642. 2018.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Balogh E, Veale DJ, McGarry T, Orr C,
Szekanecz Z, Ng CT, Fearon U and Biniecka M: Oxidative stress
impairs energy metabolism in primary cells and synovial tissue of
patients with rheumatoid arthritis. Arth Res Ther.
20(95)2018.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Mateen S, Moin S, Zafar A and Khan AQ:
Redox signaling in rheumatoid arthritis and the preventive role of
polyphenols. Clin Chim Acta. 463:4–10. 2016.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Bauerová K and Bezek A: Role of reactive
oxygen and nitrogen species in etiopathogenesis of rheumatoid
arthritis. Gen Physiol Biophys. 18:15–20. 1999.PubMed/NCBI
|
|
35
|
Broeren MG, de Vries M, Bennink MB, Arntz
OJ, Blom AB, Koenders MI, van Lent PL, van der Kraan PM, van den
Berg WB and van de Loo FA: Disease-regulated gene therapy with
anti-inflammatory interleukin-10 under the control of the CXCL10
promoter for the treatment of rheumatoid arthritis. Hum Gene Ther.
27:244–254. 2016.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Tian S, Yan Y, Qi X, Li X and Li Z:
Treatment of type II collagen-induced rat rheumatoid arthritis
model by interleukin 10 (IL10)-mesenchymal stem cells (BMSCs). Med
Sci Monit. 25:2923–2934. 2019.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Henningsson L, Eneljung T, Jirholt P,
Tengvall S, Lidberg U, van den Berg WB, van de Loo FA and Gjertsson
I: Disease-dependent local IL-10 production ameliorates collagen
induced arthritis in mice. PLoS One. 7(e49731)2012.PubMed/NCBI View Article : Google Scholar
|