Introduction
Osteoarthritis (OA) is a prevalent chronic
degenerative joint disease. This is mainly manifested as joint
stiffness, swelling, pain and loss of mobility resulting from the
destruction of articular cartilage and synovial fluid inflammation
(1,2). In addition, histopathological changes
including subchondral bone resorption, sclerosis and osteophyte
formation, can be observed (1,2). In
2017, it was estimated that ~300 million individuals suffered from
OA worldwide (3). The incidence
rate of OA in China has reached 21.5% between 2000 and 2018 as the
general age of the population increased (4). Furthermore, OA is a predominant
contributor of disability among the elderly as the most frequent
form of arthritis, which adversely reduces the quality of life of
patients, imposing an economic burden on society (5). Despite the emergence of a variety of
pain-relief agents, such as MTX, side effects, including lung,
liver and kidney injury, remain an obstacle and the process of OA
cannot be prevented (6).
Articular cartilage is the hyaline cartilage that
covers the surface of the joint and is smooth in healthy
individuals. Its main function is to form the articular surface,
provide a surface with almost no friction for the joint, which is
pivotal for the smooth movement of the joint (7). As the only cell type in the articular
cartilage, chondrocytes can generate extracellular matrix (ECM)
proteins to maintain the structure, function and integrity of the
articular cartilage (8,9). Under normal physiological conditions,
a delicate balance between anabolism and catabolism in chondrocytes
is maintained, which ensures a dynamic balance between the
generation and destruction of ECM (10). However, this balance can be easily
broken by proinflammatory factors, aging, trauma, and other factors
such as physiological load and metabolism (10). When anabolism by chondrocytes is
reduced or when catabolism is increased, ECM degradation occurs,
which leads to the destruction of articular cartilage (11,12).
Chondrocyte apoptosis and cartilage ECM damage have been previously
documented to drive the initiation and progression of OA up
(13,14). Therefore, maintenance of the normal
structure and functions of chondrocytes is the key to OA
therapy.
Zinc finger proteins belong to a superfamily of
multifunctional transcription factors that are engaged in gene
regulation, cell differentiation and embryonic development
(15,16). Zinc finger and BTB
domain-containing 16 (ZBTB16), which is also referred to as
promyelocytic leukemia zinc finger and zinc finger protein 145, is
a highly conserved member of the Kruppel-like zinc finger protein
family (17). The role of ZBTB16
in human diseases has been previously highlighted. ZBTB16 has been
reported to decrease neuron apoptosis following cerebral ischemic
reperfusion injury (18). In
addition, ZBTB16 expression has been shown to be increased during
the osteoblastogenesis of human multipotent mesenchymal stromal
cells and participates in osteoblastic differentiation (19). ZBTB16 has also been reported to
display decreased expression in human OA tissues (20). ZBTB16 has been observed to promote
hypertrophic chondrocyte differentiation and accelerate
dexamethasone-stimulated cell cycle arrest (21). However, the definite role of ZBTB16
during the process of OA remains unclear.
G protein-coupled receptor kinases (GRKs) are
pivotal protein kinases that function by the phosphorylation of G
protein-coupled receptors (22).
Among all subtypes of GRKs, G protein-coupled receptor kinase type
2 (GRK2), which is widely expressed in human tissues, has been
extensively studied (23-25).
Aberrant expression of GRK2 has been observed in multiple diseases,
including cancer, and brain, cardiovascular and metabolic diseases
(24). GRK2 has been proposed to
facilitate chondrocyte hypertrophy whilst suppressing cartilage
regeneration in OA (25).
Therefore, the present study aimed to determine the
significance of ZBTB16 and the possible relationship between ZBTB16
and GRK2 in OA.
Materials and methods
Online database analysis
The GSE169077 dataset in the GEO database (26) (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE169077),
which was selected to analyze the differentially expressed genes in
OA, was used to analyze ZBTB16 expression in OA tissues. The
possible binding of ZBTB16 to the GRK2 promoter was predicted using
the Cistrome DB database (version 1.0; http://cistrome.org/db/#/).
Cell culture and treatment
The culture medium for human C-28/I2 chondrocytes
(Shanghai Yubo Biotechnology Co., Ltd.) was DMEM/Ham's F12 medium
(HyClone; Cytiva) with 10% FBS (BioWhittaker™; Lonza Group, Ltd.)
and 1% penicillin/streptomycin (Invitrogen; Thermo Fisher
Scientific, Inc.). Cells were routinely maintained in a humidified
incubator with 5% CO2 at 37˚C. To establish the OA model
in vitro, C-28/I2 cells were exposed to various
concentrations of lipopolysaccharide (LPS; 0, 1, 3 and 5 µg/ml;
Beijing Solarbio Science & Technology Co., Ltd.) for 12 h at
37˚C (27).
Reverse transcription-quantitative PCR
(RT-qPCR)
Using a RevertAid First Strand cDNA Synthesis Kit
(Fermentas; Thermo Fisher Scientific, Inc.), cDNA was produced from
total RNA prepared from C-28/I2 cells using an E.Z.N.A.®
Total RNA kit (Omega Bio-Tek, Inc.) according to the manufacturer's
protocol. PCR amplification was performed using SYBR Green PCR
Master Mix (Thermo Fisher Scientific, Inc.) using a Mx3000P PCR
system (Agilent Technologies, Inc.). The following thermocycling
conditions were used for qPCR: 95˚C for 10 min; followed by 40
cycles of denaturation at 95˚C for 10 sec and annealing/extension
at 60˚C for 60 sec. The primer pairs used in the present study were
as follows: ZBTB16 forward, 5'-CCCTCCTCGGCTCTCGG-3' and reverse,
5'-CTCAACCTTGTCCCCCATCC-3'; MMP-13 forward,
5'-GCACTTCCCACAGTGCCTAT-3' and reverse, 5'-AGTTCTTCCCTTGATGGCCG-3';
a disintegrin-like and metalloproteinase with thrombospondin type-1
motifs-5 (ADAMTS-5) forward, 5'-ACAAGAGCCTGGAAGTGAGC-3' and
reverse, 5'-TTGGACCAGGGCTTAGATGC-3'; aggrecan (ACAN) forward,
5'-AAGGGCGAGTGGAATGATGT-3' and reverse,
5'-CGTTTGTAGGTGGTGGCTGTG-3'; collagen type II a1 (COL2A1) forward,
5'-CTTCCCCCTCCTGCTCCAAG-3' and reverse, 5'-CTGGGCAGCAAAGTTTCCAC-3';
GRK2 forward, 5'-GATGGCCATGGAGAAGAGCAAG-3' and reverse,
5'-CACTGGCAAAACCGTGTGAA-3'; and GAPDH forward,
5'-GGAGCGAGATCCCTCCAAAAT-3' and reverse,
5'-GGCTGTTGTCATACTTCTCATGG-3'. Relative gene expression was
calculated using the 2-ΔΔCq method (28). GAPDH expression was used for
normalization.
Western blotting
Briefly, total proteins were isolated from C-28/I2
cells using the RIPA buffer (Beijing Solarbio Science &
Technology Co., Ltd.) and levels were determined using a BCA
protein assay kit (Thermo Fisher Scientific, Inc.). The proteins
were resolved by 10% SDS-PAGE (30 µg/lane) and transferred onto
PVDF membranes. The membranes were blocked with 5% non-fat milk for
2 h at room temperature to prevent non-specific interactions.
Afterwards, the membranes were immunoblotted with primary
antibodies overnight at 4˚C and the goat anti-rabbit HRP antibody
(1:5,000; cat. no. ab205718; Abcam) for 1 h at room temperature.
The bands were made visible using the ECL Western Blotting
Detection Reagent (Amersham; Cytiva) and analyzed using Image Quant
LAS 500 (Cytiva). Signal intensity was determined using Image J
software version 1.46 (National Institutes of Health). ZBTB16
(1:1,000; cat. no. ab189849; Abcam), Bcl-2 (1:1,000; cat. no.
ab32124; Abcam), Bax (1:1,000; cat. no. ab32503; Abcam), cleaved
caspase-3 (1:1,000; cat. no. ab32042; Abcam), TNF-α (1:1,000; cat.
no. ab183218; Abcam), IL-1β (1:1,000; cat. no. ab254360; Abcam),
IL-6 (1:1,000; cat. no. ab233706; Abcam), MMP-13 (1:3,000; cat. no.
ab39012; Abcam), ADAMTS-5 (1:250; cat. no. ab41037; Abcam), ACAN
(1:1,000; cat. no. NB100-74350; Novus Biologicals, LLC), COL2A1
(1:1,000; cat. no. ab188570; Abcam), GRK2 (1:1,000; cat. no.
ab227825; Abcam) and GAPDH (1:2,500; cat. no. ab9485; Abcam) were
the primary antibodies utilized for the present study.
Plasmid transfection
The pcDNA3.1 vector containing full-length ZBTB16
(Ov-ZBTB16) and GRK2 (Ov-GRK2) and the empty overexpression vector
(Ov-NC) were procured from Sino Biological, Inc. C-28/I2 cells were
subjected to plasmid transfection (20 nM) using Lipofectamine™ 3000
(Thermo Fisher Scientific, Inc.) at 37˚C for 48 h. Cells were
harvested 48 h post-transfection for subsequent experiments.
Cell Counting Kit-8 (CCK-8) assay
In brief, 10 µl CCK-8 solution (APeXBIO Technology
LLC) was added to the LPS-challenged cells (5,000 cells/well)
plated into a 96-well plate. After cultivation for an additional 1
h at 37˚C, the optical density (OD) value at 450 nm was recorded by
using a microplate reader (SPECTROstar Nano; BMG Labtech GmbH).
TUNEL
Cell apoptosis was examined using a Click-iT Plus
TUNEL Assay kit (cat. no. C10617; Invitrogen; Thermo Fisher
Scientific, Inc.) in compliance with the manufacturer's
instructions. Briefly, 4% paraformaldehyde-immobilized C-28/I2
cells (room temperature for 15 min) were immersed in 50 µl TUNEL
solution for 1 h at 37˚C and incubated with 1 mg/ml DAPI (Beijing
Solarbio Science & Technology Co., Ltd.) for 10 min at 37˚C and
mounted in an anti-fade reagent (Beijing Solarbio Science &
Technology Co., Ltd.). Finally, after rinsing with PBS, the
apoptotic rate was quantified in five fields of view selected at
random under a fluorescence microscope (Olympus Corporation) and
analyzed by ImageJ software (version 6.0; National Institutes of
Health). The apoptotic rate was calculated as follows: Apoptosis
rate=(average number of apoptotic cells/average number of total
cells) x100%.
ELISA
C-28/I2 cells were cultured in 6-well plates
(2x105 cells/ml). After LPS challenge and transfection,
the cells were centrifuged at 1,000 x g for 5 min at 4˚C. The
supernatant was collected and used for ELISA. TNF-α (human TNF-α
ELISA kit; cat. no. ab181421; Abcam), IL-1β (human IL-1β ELISA kit;
cat. no. ab214025; Abcam) and IL-6 (human IL-6 ELISA kit; cat. no.
ab178013; Abcam) levels were examined using the corresponding ELISA
kits according to the manufacturer's protocols. The OD450 nm value
was estimated using a microplate reader (SPECTROstar Nano; BMG
Labtech GmbH).
Chromatin immunoprecipitation
(ChIP)
A ChIP assay was performed using the Imprint ChIP
kit (cat. no. CHP1; Sigma-Aldrich; Merck KGaA) according to the
manufacturer's guidelines. Firstly, C-28/I2 cells were treated with
1% formaldehyde at 37˚C for 10 min followed by centrifugation at
300 x g for 3 min at 25˚C, and washed in pre-cooled PBS for 10 min
at 25˚C. A total of 300 µl SDS lysis buffer (1% SDS, 10 mM EDTA and
50 mM Tris-HCl pH 8.0) was then used to lyse the 2x106
cells at room temperature for 10 min. The chromatin fragments were
acquired after the sonication of cell lysates (20 kHz; 4 pulses of
12 sec each, followed by being sheared with 30-sec pulses on ice).
Following sonication, the samples were centrifuged at 13,000 x g
for 10 min at 4˚C. Subsequently, the supernatant (100 µg) was
pre-absorbed by 100 µl protein A/G beads and was incubated with
magnetic beads conjugated to 5 µg ZBTB16 antibody (cat. no.
sc-28319; Santa Cruz Biotechnology, Inc.) or IgG antibody (cat. no.
B900620; ProteinTech Group, Inc.) at 4˚C overnight. The magnetic
beads were then rinsed four times with lysis buffer, twice with
LiCl buffer and three times with Tris-EDTA buffer. The bound
immunocomplex was eluted by adding 300 µl of fresh elution buffer
[10 mM Tris; 1 mM EDTA, (pH 8.0)]. Subsequently, 20 µl 5 M NaCl was
mixed with the eluted product, which was incubated at 65˚C
overnight to reverse the crosslinking and the purification of
immunoprecipitated DNA was conducted using a CH-IP DNA purification
kit (cat. no. D0033; Beyotime Institute of Biotechnology). The
purified DNA fragments were then subjected to PCR analysis as
described above. The GRK2 primer sequences were as follows:
Forward, 5'-CAGTTGTCAGGTCCCAGGTT-3' and reverse,
5'-TCCTGGTTACTGACCCCAAC-3'.
Luciferase reporter assay
Using Lipofectamine® 3000 (Thermo Fisher
Scientific, Inc), pGL3 vectors (100 ng; Promega Corporation)
containing the wild-type (WT) GRK2 promoter sequence or the
corresponding mutant GRK2 promoter sequence (GRK2-MUT) were
co-transfected with 2.5 µg Ov-ZBTB16 or Ov-NC into C-28/I2 cells.
After 48 h, the luciferase activity was evaluated using
Dual-Glo® Luciferase Reagent (Promega Corporation) and
normalized to Renilla luciferase activity.
Statistical analysis
All statistical analyses were performed using
GraphPad Prism 8 software (GraphPad Software, Inc.) and continuous
variables are presented as the mean ± SD from three independent
experiments. Differences between two groups was evaluated using an
unpaired Student's t-test. One-way ANOVA followed by Tukey's test
was applied for comparisons among multiple means. P<0.05 was
considered to indicate a statistically significant difference.
Results
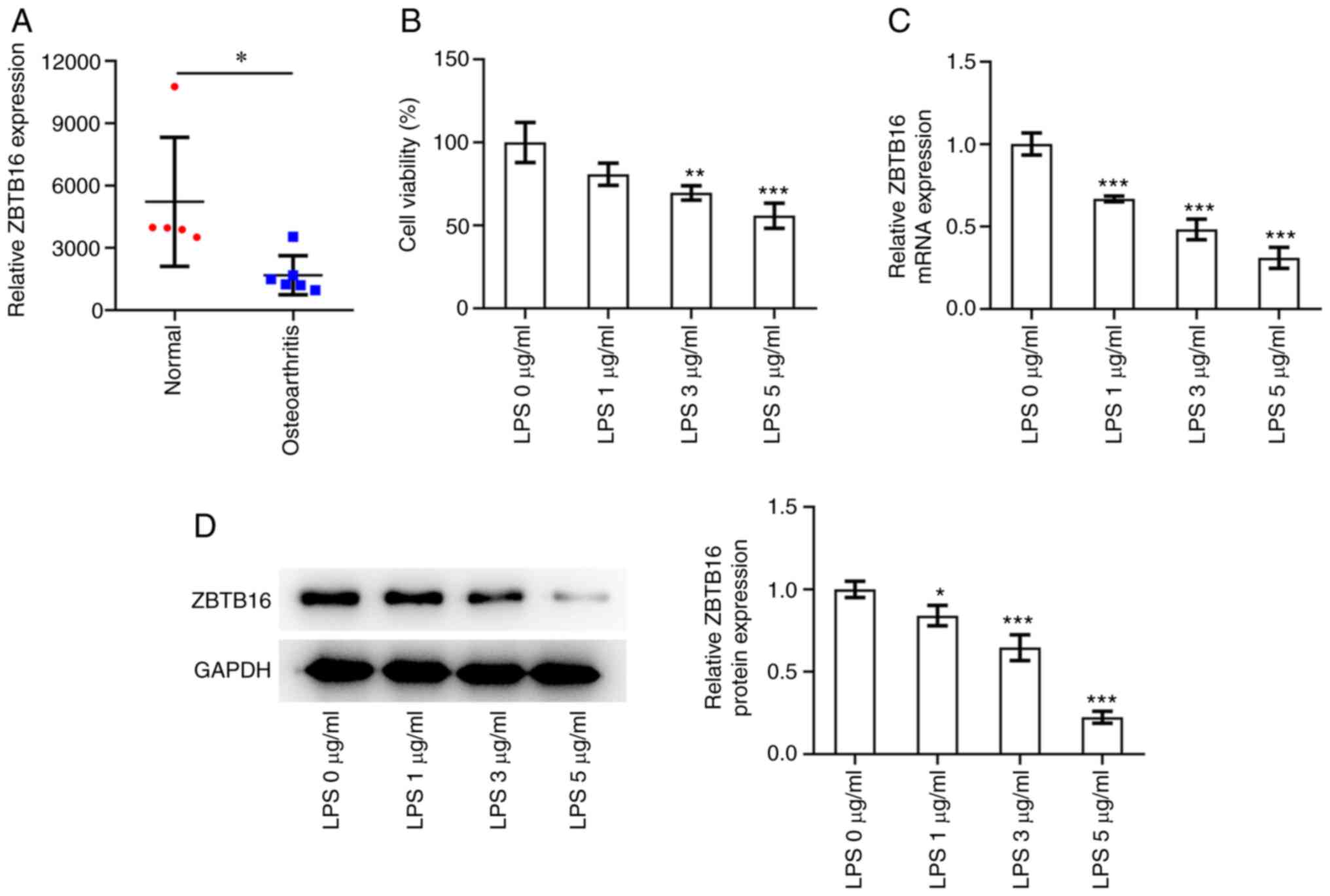
ZBTB16 expression is decreased in OA
tissues and LPS-challenged C-28/I2 cells
Based on data from the GSE169077 dataset in the GEO
database, ZBTB16 expression was significantly lower in OA tissues
compared with that in normal cartilage tissues of heakthy controls
(Fig. 1A). Experimental results of
the CCK-8 assay revealed that upon exposure to increasing
concentrations of LPS (0, 1, 3 and 5 µg/ml), the viability of
C-28/I2 cells was dose-dependently decreased (Fig. 1B). Furthermore, increasing
concentrations of LPS (0, 1, 3 and 5 µg/ml) resulted in
significantly decreased mRNA and protein expression levels of
ZBTB16 in C-28/I2 cells (Fig. 1C
and D). These observations suggest
that ZBTB16 expression is downregulated in OA tissues and
LPS-exposed C-28/I2 cells.
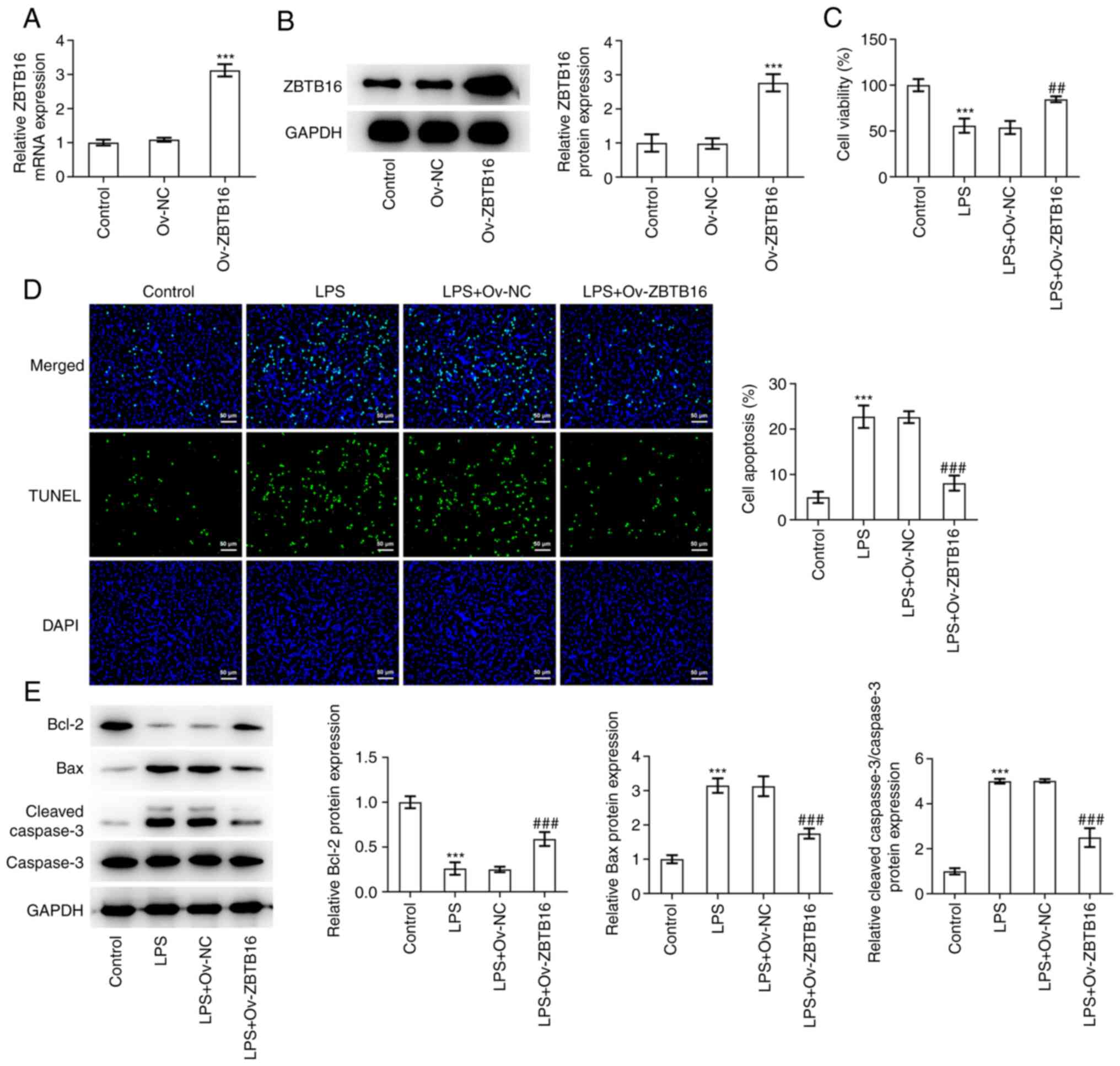
ZBTB16 overexpression reverses
LPS-elicited reductions in cell viability and apoptosis in C-28/I2
cells
To assess the impact of ZBTB16 on the phenotype of
LPS-challenged C-28/I2 cells, ZBTB16 overexpression plasmids were
transfected into C-28/I2 cells before the transfection efficacy was
verified by RT-qPCR and western blotting (Fig. 2A and B). CCK-8 assay revealed that LPS exposure
significantly inhibited C-28/I2 cell viability, which was then
significantly reversed following ZBTB16 overexpression (Fig. 2C). Furthermore, TUNEL assay
demonstrated that the apoptosis of C-28/I2 cells was significantly
increased following LPS treatment. Under this condition, ZBTB16
overexpression significantly impeded the apoptosis of LPS-treated
C-28/I2 cells (Fig. 2D).
Subsequent western blot analysis revealed that ZBTB16
overexpression significantly reversed the decreased Bcl-2
expression, whilst also significantly reversing the increased Bax
expression and cleaved caspase-3/caspase-3 ratio, originally
induced by LPS treatment in C-28/I2 cells (Fig. 2E). Collectively, these data
indicate that ZBTB16 overexpression increased the viability but
decreased the apoptosis of C-28/I2 cells that were exposed to
LPS.
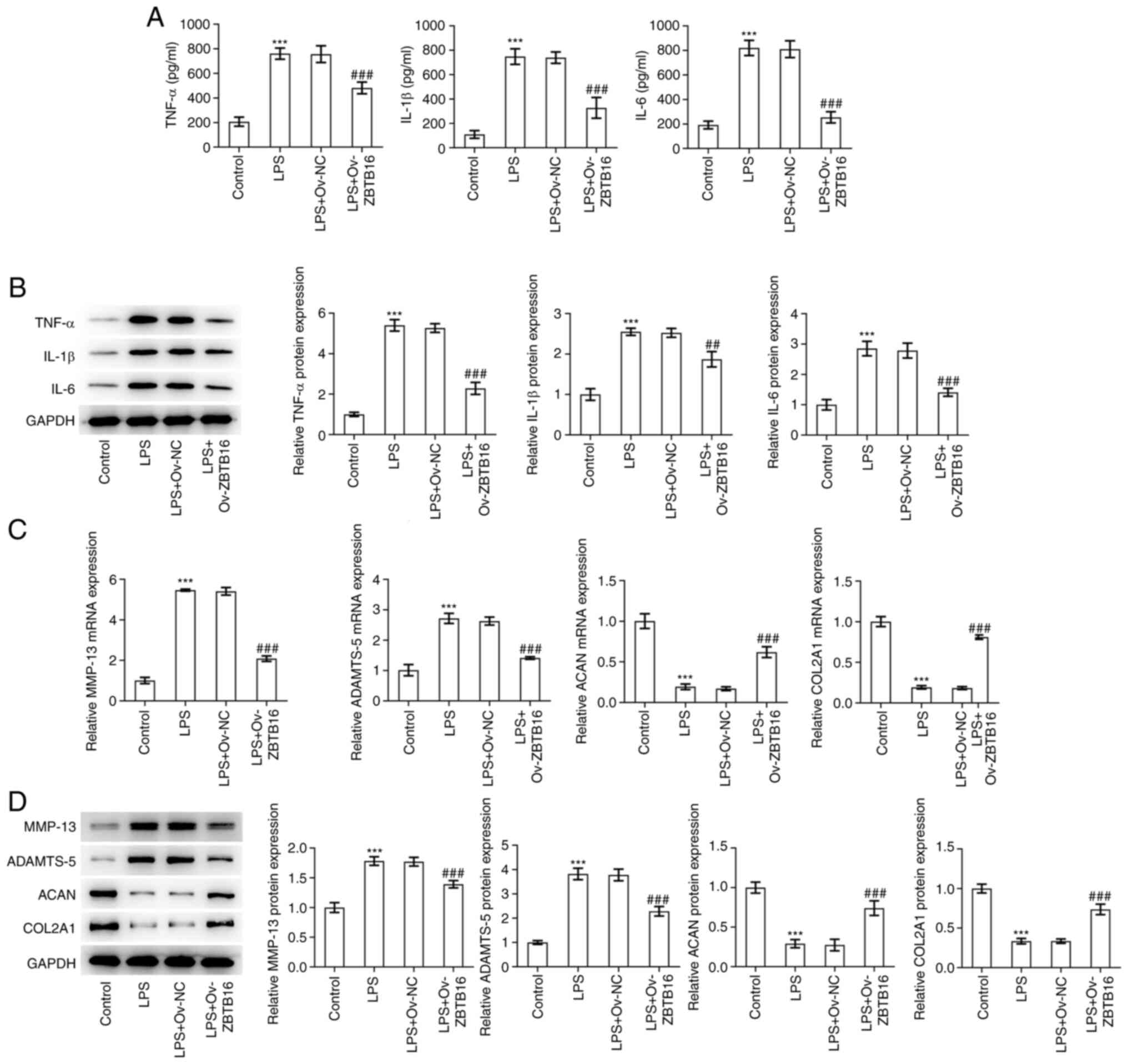
ZBTB16 overexpression alleviates the
LPS-stimulated inflammatory response and ECM degradation by C-28/I2
cells
The levels and expression of inflammatory factors
TNF-α, IL-1β and IL-6 were measured by ELISA and western blotting,
respectively. The expression levels of TNF-α, IL-1β and IL-6 were
found to be significantly increased in LPS-challenged C-28/I2
cells. These were then significantly reduced following ZBTB16
overexpression (Fig. 3A and
B). Subsequently, RT-qPCR and
western blotting were used to examine the expression levels of ECM
degradation-associated proteins MMP-13, ADAMTS-5, ACAN and COL2A1.
As depicted in Fig. 3C, LPS
treatment significantly increased MMP-13 and ADAMTS-5 expression
whilst it significantly reduced ACAN and COL2A1 expression. By
contrast, overexpression of ZBTB16 significantly decreased MMP-13
and ADAMTS-5 expression whilst significantly increasing ACAN and
COL2A1 expression in LPS-treated C-28/I2 cells (Fig. 3C and D). Overall, these findings suggest that
ZBTB16 overexpression can suppress the LPS-stimulated inflammatory
response and ECM degradation by C-28/I2 cells.
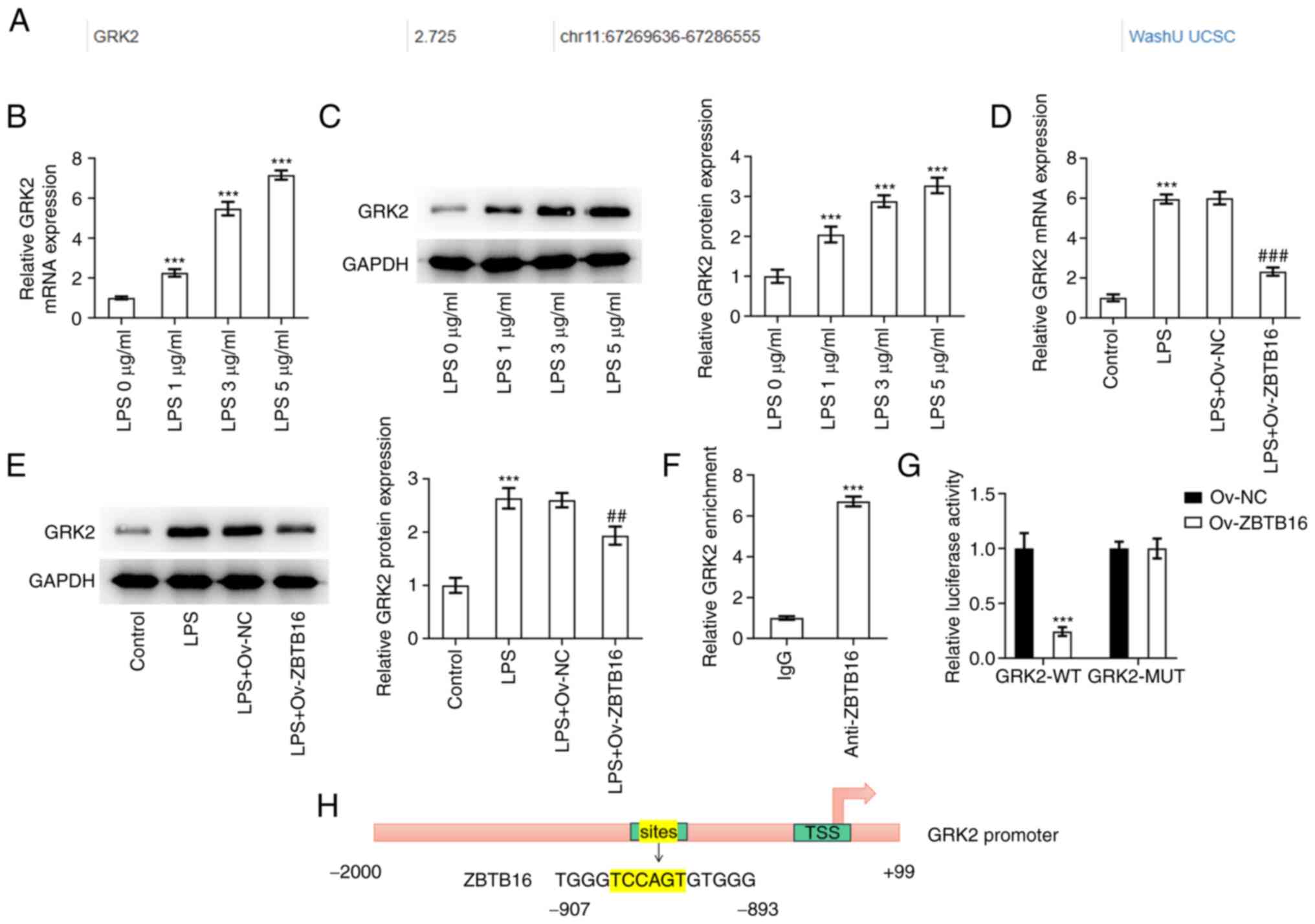
ZBTB16 transcriptionally suppresses
GRK2 expression
According to the Cistrome DB database, it was
predicted that ZBTB16 can potentially bind to the GRK2 promoter
(Fig. 4A). RT-qPCR and western
blotting then revealed that GRK2 expression was significantly
increased in C-28/I2 cells exposed to increasing concentrations of
LPS (0, 1, 3 and 5 µg/ml; Fig. 4B
and C). Additionally, the
significantly augmented GRK2 expression levels in LPS-treated
C-28/I2 cells were then significantly reversed by ZBTB16
overexpression (Fig. 4D and
E). Subsequently, ChIP assay
demonstrated significant accumulation of the GRK2 promoter sequence
in protein complexes pulled down by the ZBTB16 antibody (Fig. 4F). Furthermore, the luciferase
reporter assay revealed that the luciferase activity of the GRK2-WT
promoter was significantly diminished following the overexpression
of ZBTB16 compared with the Ov-NC group, but no apparent changes
were observed in the luciferase activity of the GRK2-MUT promoter
(Fig. 4G). The potential binding
site for ZBTB16 on the GRK2 promoter is shown in Fig. 4H. Overall, these findings are
indicative that GRK2 can be transcriptionally inactivated by
ZBTB16.
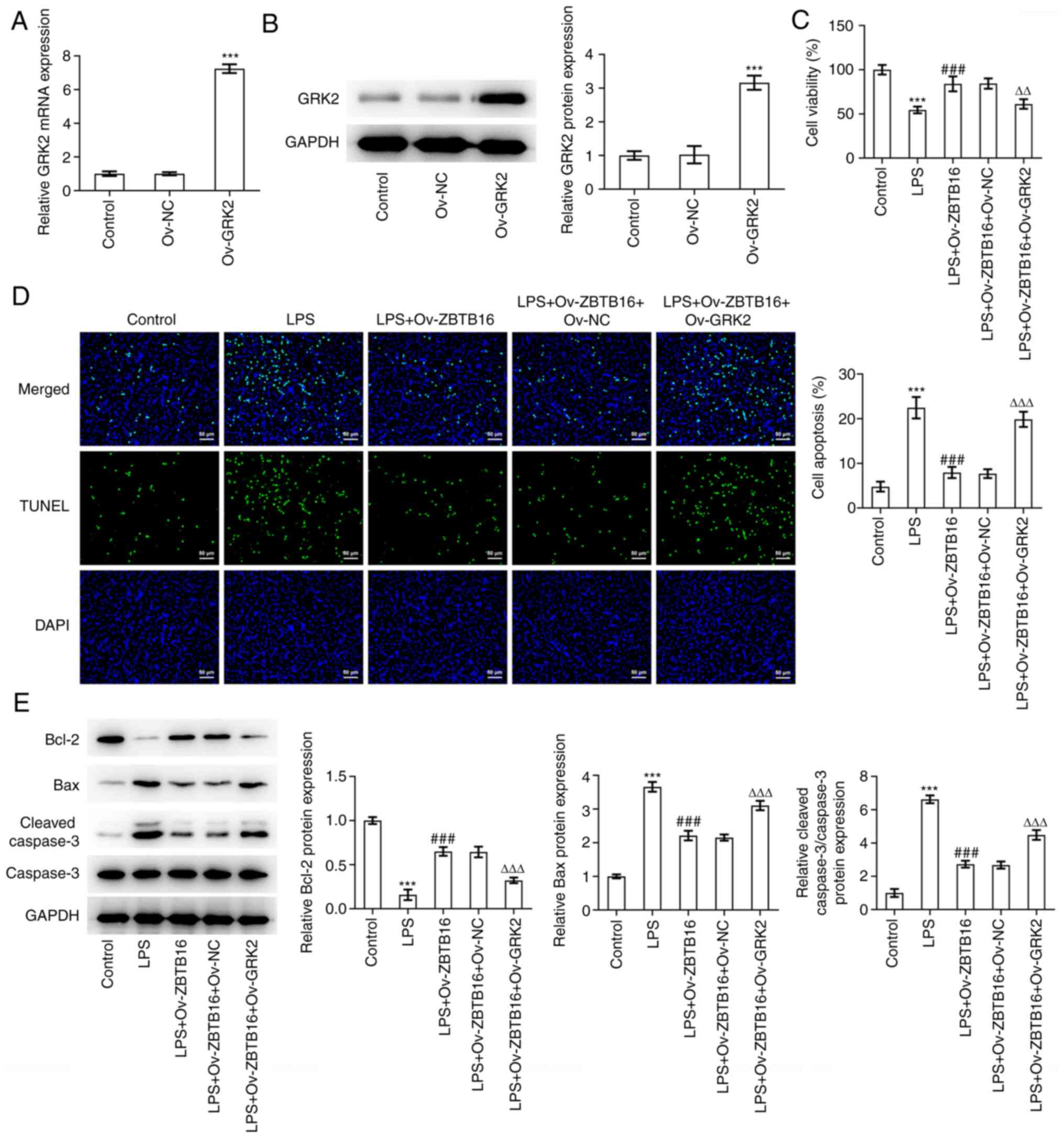
GRK2 overexpression reverses the
inhibitory effects of ZBTB16 on LPS-induced C-28/I2 cell viability
inhibition and apoptosis
To assess the role of ZBTB16 overexpression on OA
and the transcriptional regulation of GRK2, GRK2 overexpression
plasmids were transfected into C-28/I2 cells before the
transfection efficacy was verified by RT-qPCR and western blotting
(Fig. 5A and B). As shown in Fig. 5C, results from the CCK-8 assay
corroborated that the increased viability of LPS-challenged C-28/I2
cells induced by ZBTB16 overexpression was significantly reversed
when GRK2 was also overexpressed. Conversely, the apoptosis of
LPS-exposed C-28/I2 cells was significantly reduced due to ZBTB16
overexpression, which was coupled with significantly augmented Bcl2
expression and significantly diminished Bax and cleaved
caspase-3/caspase-3 expression. However, following GRK2
co-overexpression, these aforementioned effects were all
significantly reversed (Fig. 5D
and E). Taken together, these
results suggest that ZBTB16 overexpression protected against
LPS-triggered C-28/I2 cell viability inhibition and apoptosis by
transcriptionally suppressing GRK2 expression.
GRK2 overexpression reverses the
inhibitory effect of ZBTB16 on the LPS-evoked inflammatory response
and ECM degradation by C-28/I2 cells
The levels and expression of TNF-α, IL-1β and IL-6
in LPS-treated C-28/I2 cells that were significantly decreased by
ZBTB16 overexpression were significantly increased by GRK2
co-overexpression (Fig. 6A and
B). Western blotting revealed that
the significantly decreased MMP-13 and ADAMTS-5 expression and
significantly increased ACAN and COL2A1 expression in
LPS-challenged C-28/I2 cells previously overexpressing ZBTB16 were
all significantly reversed following GRK2 co-overexpression
(Fig. 6C and D). Taken together, these data suggest
that ZBTB16 protected against the LPS-evoked inflammatory responses
and ECM degradation by C-28/I2 cells through transcriptionally
suppressing GRK2 expression.
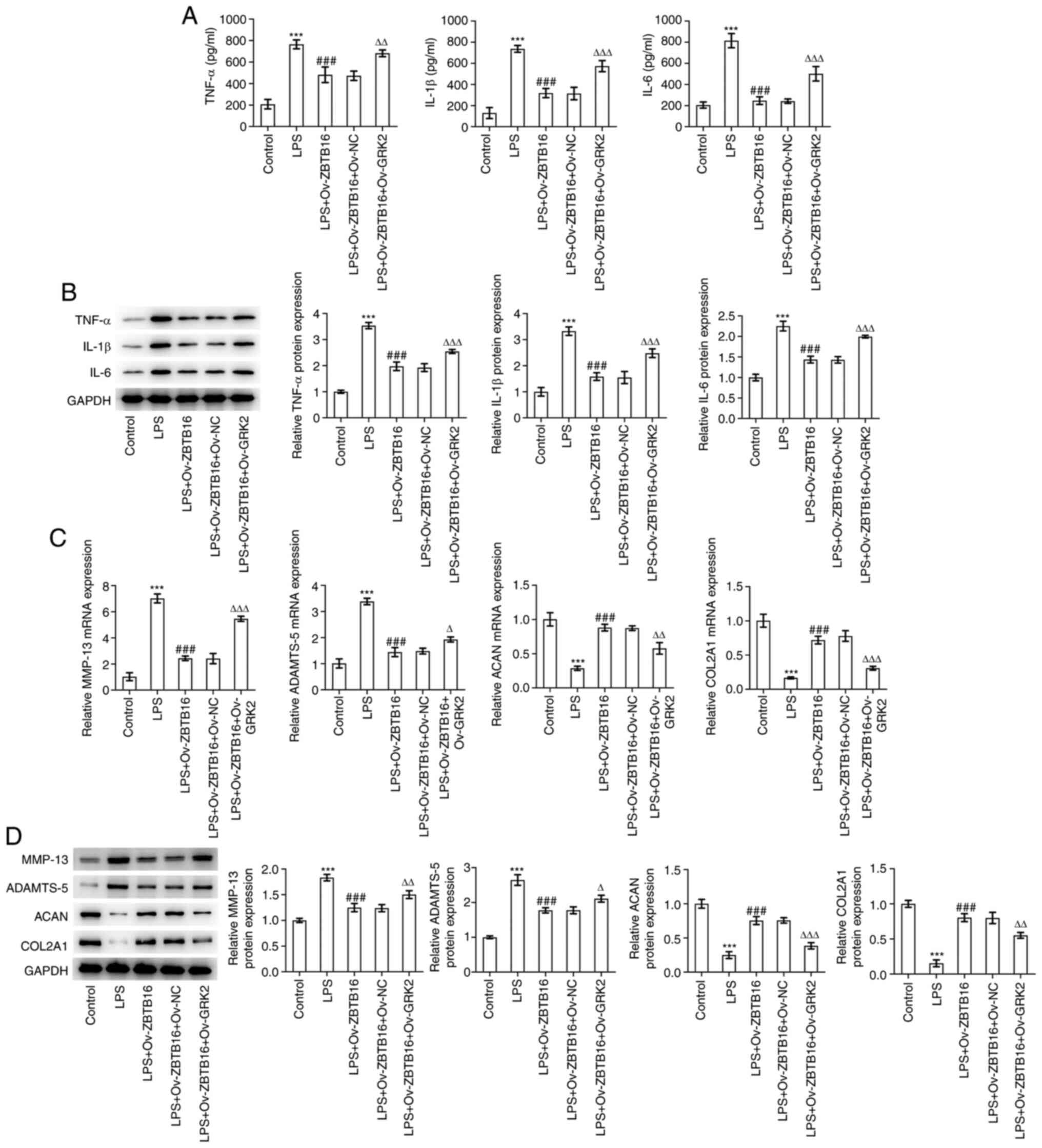 | Figure 6GRK2 overexpression reverses the
inhibitory effects of ZBTB16 overexpression on the LPS-evoked
inflammatory response and ECM degradation by C-28/I2 cells. (A)
ELISA and (B) western blotting were used to measure inflammatory
factor levels and expression in the cell culture supernatant of
LPS-treated C-28/I2 cells. (C) Reverse transcription-quantitative
PCR and (D) western blotting were used to examine the expression
levels of ECM degradation-associated proteins in LPS-treated
C-28/I2 cells. ***P<0.001 vs. Control.
###P<0.001 vs. LPS. ∆P<0.05,
∆∆P<0.01 and ∆∆∆P<0.001 vs. LPS +
Ov-ZBTB16 + Ov-NC. GRK2, G protein coupled receptor kinase type 2;
ZBTB16, zinc finger and BTB domain containing 16; LPS,
lipopolysaccharide; ECM, extracellular matrix; Ov-ZBTB16, ZBTB16
overexpression vector; Ov-NC, empty overexpression vector; Ov-GRK2,
GRK2 overexpression vector; ADAMTS-5, a disintegrin-like and
metalloproteinase with thrombospondin type-1 motifs-5; ACAN,
aggrecan; COL2A1, collagen type II α1. |
Discussion
OA is a highly prevalent chronic degenerative joint
disease, the occurrence and development of which are closely
associated with chondrocyte loss or damage (29). Under normal physiological
conditions, chondrocytes, which are distributed in ECM in articular
cartilage all over the body, maintain cartilage structure and
functions by secreting large quantities of ECM (30). Proteolytic enzymes released by
chondrocytes may mediate ECM degradation to induce articular
cartilage destruction (12).
Furthermore, inhibition of chondrocyte apoptosis is considered to
be an essential means for preventing OA (13). Therefore, protecting against
chondrocyte damage may be critical for OA therapy. ZBTB16 is an
epigenetically-regulated transcription factor that has been
previously revealed to be aberrantly expressed in OA (20). Using the GSE169077 dataset, the
significantly reduced ZBTB16 expression in OA tissues was also
highlighted in the present study. LPS is commonly deemed to be a
proinflammatory cell-wall component of Gram-negative bacteria in
cartilage tissues, which may drive the progression of OA by
stimulating the generation of MMPs from chondrocytes and other
inflammatory cytokines, including TNF-α and IL-1β (31). Therefore, the present study
utilized LPS to establish an in vitro inflammatory cell
model in C-28/I2 cells as OA is considered to be an inflammatory
disease. The experimental results revealed that LPS
dose-dependently suppressed cell viability and exacerbated the
apoptosis of C-28/I2 cells, which are accompanied by decreased Bcl2
expression, increased Bax and cleaved caspase-3/caspase-3
expression. These findings are consistent with the previous
research conducted by Luo et al (32). In addition, ZBTB16 expression was
found to be reduced in C-28/I2 cells exposed to LPS in a
concentration-dependent manner. Overexpression of ZBTB16 improved
cell viability whilst hindering the apoptosis of LPS-challenged
C-28/I2 cells, as evidenced by the augmented Bcl2 expression,
decreased Bax and cleaved caspase-3/caspase-3 expression. These
findings suggest that ZBTB16 mediates a protective role against
OA.
The inflammatory response occupies an important
position in the pathology of joint destruction during OA and is
considered to be an important process mediating cartilage
degeneration in OA (33,34). During the inflammatory response,
the excessive release of inflammatory factors, including TNF-α,
IL-1β and IL-6, may disrupt chondrocyte metabolism, inhibit the
synthesis of ECM proteins and eventually induce chondrocyte
apoptosis and ECM degradation (35,36).
A previous study reported that ZBTB16 can reverse advanced
glycation end product-induced inflammation in vascular endothelial
cells (37). In accordance with
this, the present study demonstrated that the LPS-enhanced TNF-α,
IL-1β and IL-6 levels and expression were decreased by ZBTB16
overexpression in C-28/I2 cells. The imbalance between the
synthesis and degradation of ECM is a primary pathological change
during the early stages of OA and serves a vital role in the
cartilage degeneration process in OA (38). MMP-13 and ADAMTS-5 are the main
proteases that mediate ECM degradation during OA and were
previously found to be upregulated during cartilage damage
(39,40). ACAN and COL2A1 form the major
components of the ECM (41). In
the present study, it was found that the increased MMP-13, ADAMTS-5
expression and the decreased ACAN and COL2A1 expression in
LPS-treated C-28/I2 cells were reversed after ZBTB16 was
overexpressed.
ZBTB16 has been suggested to serve as a
transcriptional suppressor through DNA binding (42,43).
According to analysis using the Cistrome DB database, ZBTB16 could
potentially bind to the GRK2 promoter. In addition, GRK2 expression
was found to be dose-dependently augmented in LPS-treated C-28/I2
cells but was then reversed by ZBTB16 overexpression. The affinity
of ZBTB16 to the GRK2 promoter was next validated by mechanistic
assays. The enhanced viability and attenuated apoptosis of C-28/I2
cells exposed to LPS mediated by ZBTB16 overexpression were both
reversed when GRK2 was co-overexpressed. Additionally, ZBTB16
overexpression-induced alterations in the expression of apoptotic
factors Bcl2, Bax and cleaved caspase-3 were all reversed by GRK2
overexpression. GRK2 has been previously revealed to be implicated
in inflammation (44). In
addition, GRK2 can halt cartilage regeneration and contribute to OA
development (25). Consistent with
these previous findings, the decreased TNF-α, IL-1β, IL-6, MMP-13
and ADAMTS-5 expression and the increased ACAN and COL2A1
expression caused by ZBTB16 overexpression in LPS-challenged
C-28/I2 cells were all reversed by GRK2 co-overexpression.
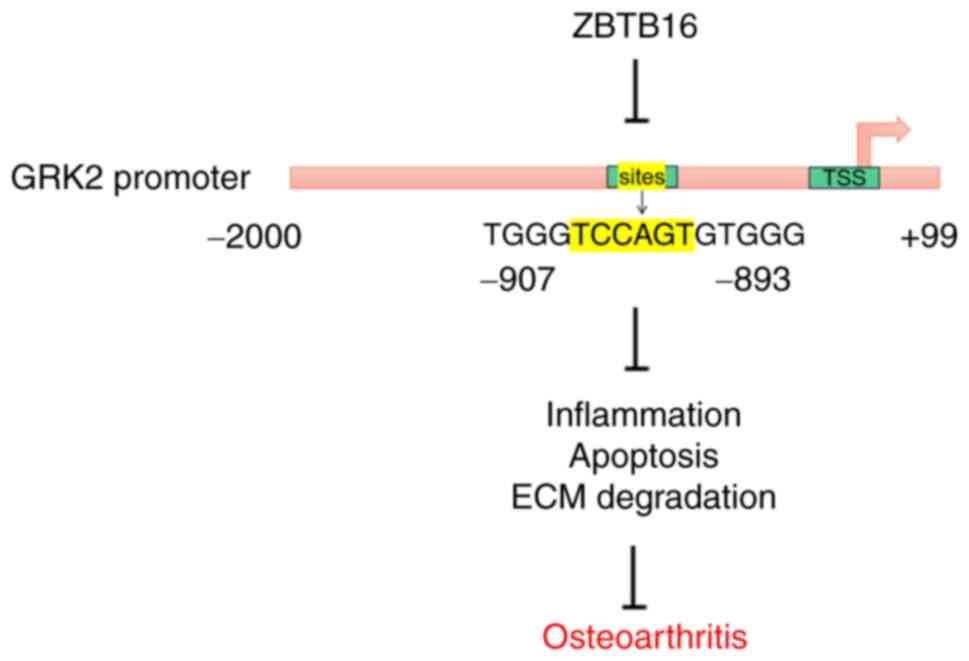
In conclusion, ZBTB16 overexpression reversed
LPS-mediated viability inhibition, apoptosis, inflammation and ECM
degradation in chondrocytes by possibly binding to the GRK2
promoter to transcriptionally inactivate GRK2 (Fig. 7). To the best of our knowledge, the
present study was the first to demonstrate the suppressive role of
ZBTB16 in OA and present a novel negative regulatory relationship
between ZBTB16 and GRK2 in OA. Overall, the present observations
may provide a potentially novel therapeutic modality for OA.
However, future studies are required to expound the role of ZBTB16
in OA in vivo. Additionally, whether ZBTB16 can regulate the
transcription of other genes in the OA setting will also need to be
explored in future experiments.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current
study are available from the corresponding author on reasonable
request.
Authors' contributions
HP designed and conceived the study. BX, LC, YH and
GL conducted the experiments. CC and JN helped to analyze the data.
BX and LC drafted the manuscript, which was revised by HP. HP and
BX confirm the authenticity of all the raw data. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Mei X, Villamagna IJ, Nguyen T, Beier F,
Appleton CT and Gillies ER: Polymer particles for the
intra-articular delivery of drugs to treat osteoarthritis. Biomed
Mater: 16, 2021. doi: 10.1088/1748-605X/abee62.
|
|
2
|
Sohn R, Rösch G, Junker M, Meurer A,
Zaucke F and Jenei-Lanzl Z: Adrenergic signalling in
osteoarthritis. Cell Signal. 82(109948)2021.PubMed/NCBI View Article : Google Scholar
|
|
3
|
GBD 2017 Disease and Injury Incidence and
Prevalence Collaborators. Global, regional, and national incidence,
prevalence, and years lived with disability for 354 diseases and
injuries for 195 countries and territories, 1990-2017: A systematic
analysis for the Global Burden of Disease Study 2017. Lancet.
392:1789–858. 2018.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Sun X, Zhen X, Hu X, Li Y, Gu S, Gu Y and
Dong H: Osteoarthritis in the Middle-Aged and Elderly in China:
Prevalence and influencing factors. Int J Environ Res Public
Health. 16(4701)2019.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Hawker GA: Osteoarthritis is a serious
disease. Clin Exp Rheumatol. 37 (Suppl 120):S3–S6. 2019.PubMed/NCBI
|
|
6
|
Bindu S, Mazumder S and Bandyopadhyay U:
Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A
current perspective. Biochem Pharmacol. 180(114147)2020.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Palukuru UP, McGoverin CM and Pleshko N:
Assessment of hyaline cartilage matrix composition using near
infrared spectroscopy. Matrix Biol. 38:3–11. 2014.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Yan X, Kononenko NL, Bruel A, Thomsen JS
and Poy MN: Neuronal cell adhesion molecule 1 regulates leptin
sensitivity and bone mass. Calcif Tissue Int. 102:329–336.
2018.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Huang W, Cheng C, Shan WS, Ding ZF, Liu
FE, Lu W, He W, Xu JG and Yin ZS: Knockdown of SGK1 alleviates the
IL-1β-induced chondrocyte anabolic and catabolic imbalance by
activating FoxO1-mediated autophagy in human chondrocytes. FEBS J.
287:94–107. 2020.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Zheng L, Zhang Z, Sheng P and Mobasheri A:
The role of metabolism in chondrocyte dysfunction and the
progression of osteoarthritis. Ageing Res Rev.
66(101249)2021.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Chomchalao P, Pongcharoen S,
Sutheerawattananonda M and Tiyaboonchai W: Fibroin and fibroin
blended three-dimensional scaffolds for rat chondrocyte culture.
Biomed Eng Online. 12(28)2013.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Yunus MHM, Nordin A and Kamal H:
Pathophysiological perspective of osteoarthritis. Medicina
(Kaunas). 56(614)2020.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Hwang HS and Kim HA: Chondrocyte apoptosis
in the pathogenesis of osteoarthritis. Int J Mol Sci.
16:26035–26054. 2015.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Krishnan Y and Grodzinsky AJ: Cartilage
diseases. Matrix Biol. 71-72:51–69. 2018.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Jen J and Wang YC: Zinc finger proteins in
cancer progression. J Biomed Sci. 23(53)2016.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Ye Q, Liu J and Xie K: Zinc finger
proteins and regulation of the hallmarks of cancer. Histol
Histopathol. 34:1097–109. 2019.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Liu TM, Lee EH, Lim B and Shyh-Chang N:
Concise review: Balancing stem cell self-renewal and
differentiation with PLZF. Stem Cells. 34:277–287. 2016.PubMed/NCBI View Article : Google Scholar
|
|
18
|
Seidel K, Kirsch S, Lucht K, Zaade D,
Reinemund J, Schmitz J, Klare S, Li Y, Schefe JH, Schmerbach K, et
al: The promyelocytic leukemia zinc finger (PLZF) protein exerts
neuroprotective effects in neuronal cells and is dysregulated in
experimental stroke. Brain Pathol. 21:31–43. 2011.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Onizuka S, Iwata T, Park SJ, Nakai K,
Yamato M, Okano T and Izumi Y: ZBTB16 as a downstream target gene
of osterix regulates osteoblastogenesis of human multipotent
mesenchymal stromal cells. J Cell Biochem. 117:2423–2434.
2016.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Alvarez-Garcia O, Fisch KM, Wineinger NE,
Akagi R, Saito M, Sasho T, Su AI and Lotz MK: Increased DNA
methylation and reduced expression of transcription factors in
human osteoarthritis cartilage. Arthritis Rheumatol. 68:1876–1886.
2016.PubMed/NCBI View Article : Google Scholar
|
|
21
|
Naito M, Vongsa S, Tsukune N, Ohashi A and
Takahashi T: Promyelocytic leukemia zinc finger mediates
glucocorticoid-induced cell cycle arrest in the chondroprogenitor
cell line ATDC5. Mol Cell Endocrinol. 417:114–123. 2015.PubMed/NCBI View Article : Google Scholar
|
|
22
|
Gurevich VV and Gurevich EV: GPCR
signaling regulation: The role of GRKs and arrestins. Front
Pharmacol. 10(125)2019.PubMed/NCBI View Article : Google Scholar
|
|
23
|
Guccione M, Ettari R, Taliani S, Da
Settimo F, Zappalà M and Grasso S: G-Protein-coupled receptor
Kinase 2 (GRK2) inhibitors: Current trends and future perspectives.
J Med Chemistry. 59:9277–9294. 2016.PubMed/NCBI View Article : Google Scholar
|
|
24
|
Kang JH, Toita R, Kawano T, Murata M and
Asai D: Design of substrates and inhibitors of G protein-coupled
receptor kinase 2 (GRK2) based on its phosphorylation reaction.
Amino Acids. 52:863–870. 2020.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Carlson EL, Karuppagounder V, Pinamont WJ,
Yoshioka NK, Ahmad A, Schott EM, Le Bleu HK, Zuscik MJ, Elbarbary
RA and Kamal F: Paroxetine-mediated GRK2 inhibition is a
disease-modifying treatment for osteoarthritis. Sci Transl Med.
13(eaau8491)2021.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Xu WB, Kotheeranurak V, Zhang HL, Feng JY,
Liu JW, Chen CM, Lin GX and Rui G: Identification of the
circRNA-miRNA-mRNA regulatory network in osteoarthritis using
bioinformatics analysis. Front Genet. 13(994163)2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Jia Z and Wei QJ: CircRNA-MSR regulates
LPS-induced C28/I2 chondrocyte injury through miR-643/MAP2K6
signaling pathway. Cartilage. 13 (2_suppl):785S–795S.
2021.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408.
2001.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Wang Y, Chen LY and Liu-Bryan R:
Mitochondrial biogenesis, activity, and DNA isolation in
chondrocytes. Methods Mol Biol. 2245:195–213. 2021.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Caron MM, Emans PJ, Coolsen MM, Voss L,
Surtel DA, Cremers A, van Rhijn LW and Welting TJ:
Redifferentiation of dedifferentiated human articular chondrocytes:
Comparison of 2D and 3D cultures. Osteoarthritis Cartilage.
20:1170–1178. 2012.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Nguyen QT, Jacobsen TD and Chahine NO:
Effects of inflammation on multiscale biomechanical properties of
cartilaginous cells and tissues. ACS Biomater Sci Eng. 3:2644–2656.
2017.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Luo X, Wang J, Wei X, Wang S and Wang A:
Knockdown of lncRNA MFI2-AS1 inhibits lipopolysaccharide-induced
osteoarthritis progression by miR-130a-3p/TCF4. Life Sci.
240(117019)2020.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Lee YM, Son E, Kim SH, Kim OS and Kim DS:
Anti-inflammatory and anti-osteoarthritis effect of Mollugo
pentaphylla extract. Pharm Biol. 57:74–81. 2019.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Hu Y, Gui Z, Zhou Y, Xia L, Lin K and Xu
Y: Quercetin alleviates rat osteoarthritis by inhibiting
inflammation and apoptosis of chondrocytes, modulating synovial
macrophages polarization to M2 macrophages. Free Radic Biol Med.
145:146–160. 2019.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Drevet S, Gavazzi G, Grange L, Dupuy C and
Lardy B: Reactive oxygen species and NADPH oxidase 4 involvement in
osteoarthritis. Exp Gerontol. 111:107–117. 2018.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Salucci S, Falcieri E and Battistelli M:
Chondrocyte death involvement in osteoarthritis. Cell Tissue Res.
389:159–170. 2022.PubMed/NCBI View Article : Google Scholar
|
|
37
|
Chen CH, Chen TH, Wu MY, Chou TC, Chen JR,
Wei MJ, Lee SL, Hong LY, Zheng CM, Chiu IJ, et al: Far-infrared
protects vascular endothelial cells from advanced glycation end
products-induced injury via PLZF-mediated autophagy in diabetic
mice. Sci Rep. 7(40442)2017.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Rahmati M, Nalesso G, Mobasheri A and
Mozafari M: Aging and osteoarthritis: Central role of the
extracellular matrix. Ageing Res Rev. 40:20–30. 2017.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Mehana EE, Khafaga AF and El-Blehi SS: The
role of matrix metalloproteinases in osteoarthritis pathogenesis:
An updated review. Life Sci. 234(116786)2019.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Yang CY, Chanalaris A and Troeberg L:
ADAMTS and ADAM metalloproteinases in osteoarthritis-looking beyond
the ‘usual suspects’. Osteoarthritis Cartilage. 25:1000–1009.
2017.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Wang C, Gao Y, Zhang Z, Chi Q, Liu Y, Yang
L and Xu K: Safflower yellow alleviates osteoarthritis and prevents
inflammation by inhibiting PGE2 release and regulating
NF-κB/SIRT1/AMPK signaling pathways. Phytomedicine.
78(153305)2020.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Fréchette I, Darsigny M, Brochu-Gaudreau
K, Jones C and Boudreau F: The Promyelocytic Leukemia Zinc Finger
(PLZF ) gene is a novel transcriptional target of the
CCAAT-displacement-protein (CUX1) repressor. FEBS J. 277:4241–4253.
2010.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Labbaye C, Spinello I, Quaranta MT, Pelosi
E, Pasquini L, Petrucci E, Biffoni M, Nuzzolo ER, Billi M, Foà R,
et al: A three-step pathway comprising PLZF/miR-146a/CXCR4 controls
megakaryopoiesis. Nat Cell Biol. 10:788–801. 2008.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Cheng H, Guo P, Su T, Jiang C, Zhu Z, Wei
W, Zhang L and Wang Q: G protein-coupled receptor kinase type 2 and
β-arrestin2: Key players in immune cell functions and inflammation.
Cell Signal. 95(110337)2022.PubMed/NCBI View Article : Google Scholar
|